Introduction
Pulmonary hypertension (PH) is a complex and
life-threatening type of lung disease, which is defined by a mean
pulmonary arterial pressure of ≥25 mmHg at rest as measured by
right heart catheterization (1).
Recently, PH was clinically classified into five groups: Pulmonary
arterial hypertension, PH due to left heart disease, PH due to lung
diseases and/or hypoxia, chronic thromboembolic PH, and PH with
unclear and/or multifactorial mechanisms (2). Hypoxia-induced PH, which is the
focus of the present study, results from hypoxic pulmonary
vasoconstriction, polycythemia and pulmonary arterial remodeling
(3). Pathological changes in
patients with hypoxia-induced PH manifest as dysfunction of smooth
muscle cells, fibroblasts and pulmonary artery endothelial cells,
which are associated with progressive obliteration of the pulmonary
arteries (4,5). Several pathways participate in the
complex process of hypoxia-induced PH; however, the underlying
mechanism is not well-defined. Furthermore, the current treatment
strategies available are limited and the prognosis is poor for
patients with PH (6). Thus, novel
strategies for the diagnosis and treatment of patients with
hypoxia-induced PH are urgently required.
Circular RNAs (circRNAs) are a novel class of the
non-coding RNA family that feature a covalently closed loop
structure without a 5′-terminal cap or 3′-terminal poly(A) tail
(7). Initially, circRNAs were
considered to be functionless byproducts of the normal splicing
processing. With the advent of next-generation sequencing, it was
revealed that circRNAs were widely distributed stable entities in
various cell types and were expressed in species-, tissue-,
disease- and developmental stage-specific manners (8,9).
To date, the functions of circRNAs have not been fully elucidated.
The most widely studied function of circRNAs is their ability to
function as miRNA sponges. The representative circRNA, ciRS-7, has
>70 conventional binding sites, which bind to miR-7 to decrease
its expression levels and further increase the levels of
miR-7-targeted transcripts (10).
CircRNA located in the nucleus may participate in the regulation of
transcription and alternative splicing (11). Furthermore, a number of circRNAs
have been demonstrated to be translated into proteins. Legnini
et al (12) firstly
demonstrated that circ-ZNF609 could be translated into a small
protein that specifically controlled the myoblast proliferation.
These unique features of circRNAs have brought a new perspective to
our understanding of the biological mechanisms underlying various
diseases, including cardiovascular diseases, cancer, neurological
disorders and diabetes, and the possibility of novel diagnostic
biomarkers and innovative strategies for treatment (13–16). However, the role of circRNAs in
hypoxia-induced PH remains unknown.
In the present study, a murine model of
hypoxia-induced PH was established. To the best of our knowledge,
circRNA microarray analysis was used for the first time to
determine the differential expression profile of circRNAs in the
lungs of mice with hypoxia-induced PH compared with the control
mice. A total of 12 candidate circRNAs were selected for further
validation by reverse transcription-quantitative polymerase chain
reaction (RT-qPCR). Bioinformatics analyses identified the
potential functions of the candidate circRNAs in the mechanism of
hypoxia-induced PH. These candidate circRNAs have the potential to
function as diagnostic biomarkers and targets for the treatment of
patients with the disease.
Materials and methods
Animal models
A total of 20 male C57BL/6 mice (6–8 weeks old,
weighing 17–20 g) were obtained from JSJ Laboratory Animal Co.,
Ltd. (Shanghai, China). The experimental protocols involving the
animals were approved by the Institutional Animal Care and Use
Committee of Huadong Hospital, Fudan University (Shanghai, China).
All mice were housed in room temperature and relative humidity of
~60% environment with a 12-h light-dark cycle, and fed with normal
chow and water. Following acclimatization for 1 week, mice were
randomly assigned into two groups, including the hypoxia-PH group
(n=10) and the control group (n=10). The hypoxia-PH mice were
exposed to hypoxia (10% O2) in a normobaric chamber for
21 days (17). The control mice
were housed in room air. The feeding conditions were the same for
both groups.
PH measurements
The right ventricular systolic pressure (RVSP) and
right ventricular hypertrophy (RVH) were considered to be
indicators of PH. At the end of the treatment period, mice were
anesthetized with 20% urethane (0.4 ml/100 g, intraperitoneal), and
then a catheter was inserted into the right ventricle (RV) via the
right jugular vein. RVSP was measured by a gauge pressure
transducer (RM-6000; Nihon Kohden Corporation, Tokyo, Japan).
Animals were then sacrificed and the hearts were collected. The RV
was dissected from the left ventricle (LV) and septum, and the two
ventricles were then weighed. The RVH was determined based on the
ratio of the RV weight compared with the weight of the LV and
septum [calculated as RV/(LV+S)]. Additionally, lung tissues were
harvested and stored at −80°C for subsequent circRNA microarray
analysis.
CircRNA microarray analysis
Lung samples from the hypoxia-PH group were randomly
distributed into three subgroups for further analysis. The two
subgroups had three lung samples from the hypoxia-PH group and the
other subgroup has four lung samples from the hypoxia-PH group. The
same grouping strategy was applied to the control group. Total RNA
from each subgroup was isolated using TRIzol reagent (Invitrogen;
Thermo Fisher Scientific, Inc., Waltham, MA, USA) according to the
manufacturer’s protocol. The RNA concentration was quantified using
a NanoDrop spectrophotometer (ND-1000; Thermo Fisher Scientific,
Inc., Wilmington, DE, USA), while RNA integrity was determined
using gel electrophoresis. Following RNA quality control,
fluorescence labeling and microarray hybridization were performed
according to the manufacturer’s protocol for the Arraystar Super
RNA Labeling kit (Arraystar, Inc., Rockville, MD, USA). Briefly,
linear RNAs were eliminated using RNase R (Epicentre; Illumina,
Inc., San Diego, CA, USA), and the enriched circRNAs were
subsequently amplified and transcribed into fluorescent
complementary RNA (cRNA) by employing a random priming method
(Arraystar Super RNA Labeling kit; Arraystar, Inc.). Labeled cRNA
was then purified using the RNeasy Mini kit (Qiagen GmbH, Hilden,
Germany). Labeled cRNA quality and quantity were determined using
the NanoDrop ND-1000 spectrophotometer. Subsequently, the labeled
cRNA was fragmented and hybridized onto an Arraystar mouse circRNA
microarray (6x7K; Arraystar, Inc.). Subsequent to washing with gene
expression wash buffer 1 (Agilent Technologies, Inc., Santa Clara,
CA, USA) for 1 min at 37°C and then gene expression wash buffer 2
(Agilent Technologies, Inc.) for 1 min at 37°C, the hybridized
arrays were scanned using an Axon GenePix 4000B microarray scanner
(Molecular Devices, LLC, Sunnyvale, CA, USA).
CircRNA data analysis
GenePix Pro 6.0 software (Axon; Molecular Devices,
LLC) was used to analyze the scanned images of circRNA microarray
in order to extract the raw data. Normalization and data processing
were accomplished using the R software package (version 3.4.1; R
Foundation Inc., Vienna, Austria). The statistical significance of
the expression of circRNAs between the two groups was estimated by
fold-change filtering and an unpaired t-test. To increase the
number of circRNAs detected, the screening criterion for
differentially expressed circRNAs was a fold-change of ≥1.5 and a
P-value of <0.05. Hierarchical cluster analyses, box plot,
volcano plot and scatter plot were used to display the differential
expression of circRNAs between the two groups.
RT-qPCR validation
The top 12 candidate dysregulated circRNAs
(including six upregulated circRNAs and six downregulated circRNAs)
were selected for validation by RT-qPCR. Briefly, total RNA was
extracted from the two groups using TRIzol reagent (Thermo Fisher
Scientific, Inc.). The concentration of total RNA was measured by a
NanoVue Plus spectrophotometer (GE Healthcare, Inc., Chicago, IN,
USA) and total RNA (1 µg) was reverse transcribed into cDNA
using a PrimeScript™ RT Reagent kit (Takara Bio, Inc., Otsu, Japan)
according to the manufacturer’s protocol. The RT reaction was
conducted at 37°C for 15 min and 85°C for 5 sec. Next, the RT-qPCR
reaction was performed using SYBR® Premix Ex Taq™
(Takara Bio, Inc.) using a Bio-Rad Real-Time PCR system (Bio-Rad
Laboratories, Inc., Hercules, CA, USA). The amplification reaction
involved initial denaturation at 95°C for 1 min, and 40 cycles of
denaturation at 95°C for 5 sec and 60°C for 30 sec. The primers for
the circRNAs were designed in divergent orientation, and the primer
sequence pairs are presented in Table
I. The relative level of circRNA expression for each target
gene is shown as the 2−ΔΔCQ value (18).
 | Table IPrimers of validated circRNAs. |
Table I
Primers of validated circRNAs.
| Name | Forward | Reverse |
|---|
| GAPDH |
5′-GTTGTCTCCTGCGACTTCA-3′ |
5′-GCCCCTCCTGTTATTATGG-3′ |
|
mmu_circRNA_010744 |
5′-GGCTTACACCTCAACTACCATCC-3′ |
5′-CCCAGCATTCCGAAGAACC-3′ |
|
mmu_circRNA_014597 |
5′-CTTCAATGATTTTCACCTCCAG-3′ |
5′-AGCGGTCATCCATGCTTATAT-3′ |
|
mmu_circRNA_008379 |
5′-CTGTCCCAACTGTAAAGAAGGTG-3′ |
5′-CATCGGTTTGGTGCTCCTC-3′ |
|
mmu_circRNA_000267 |
5′-GGCACCCAACCTCATCTTC-3′ |
5′-TTCGCACTTCTCATAGTCACTCA-3′ |
|
mmu_circRNA_015666 |
5′-ACGCCCAGGATTATGAAGAGG-3′ |
5′-GGTACTGTGGTGAGTCTGGACGA-3′ |
|
mmu_circRNA_013176 |
5′-CCCCACTGAACAGGAAGAAGA-3′ |
5′-ACGATACAAGCACCCAGATAGG-3′ |
|
mmu_circRNA_016128 |
5′-CTTCTGGTTCCTGGAGTTGG-3′ |
5′-AGTTCTGGGATGCTTGCTTTA-3′ |
|
mmu_circRNA_018351 |
5′-GCTCAACCTCATCCAGCACG-3′ |
5′-GCGAGTCACAGCCTTCCAT-3′ |
|
mmu_circRNA_004592 |
5′-TGTGGTTGGCAAGGATGTC-3′ |
5′-CGCATGGGAACTGTAGTAAGAT-3′ |
|
mmu_circRNA_018701 |
5′-GAAGATAGTGCCTTTCAGCCATAC-3′ |
5′-CACTCCCGTTTATCCCACTCA-3′ |
|
mmu_circRNA_016636 |
5′-GGAAAGGAGCAAACACGAAGG-3′ |
5′-TGTCAGCGGCAAGTCATCG-3′ |
|
mmu_circRNA_010120 |
5′-GCAGTACGCCGTCATCAGTG-3′ |
5′-GGAAGGCAGCAGACAAGAGC-3′ |
Bioinformatics analysis
CircRNAs contain miRNA response elements (MREs) and
function as miRNA sponges, which regulate miRNA and downstream gene
expression through a competing endogenous RNA (ceRNA) mechanism
(19). Putative
circRNA-miRNA-mRNA networks were generated using various
bioinformatics tools. The circRNA-miRNA interaction was predicted
using the miRNA target prediction software (Arraystar, Inc.), which
is based on the TargetScan (20)
and miRanda (21). The top five
circRNA-bound miRNAs were identified based on the prediction of
miRNA binding sites. To ensure the efficiency and accuracy of the
analysis, circRNAs were selected (fold-change ≥2 and P<0.05) and
their target genes were predicted using miRWalk 2.0, which
integrates miRWalk, RNA22, miRanda and TargetScan (22). The top 10 circRNA target genes for
each miRNA were identified and used to construct circRNA-miRNA-mRNA
networks using Cytoscape software (version 3.5.1; The Cytoscape
Consortium, San Diego, CA, USA). The functional roles of the
circRNA-target genes were further analyzed using Gene Ontology (GO;
geneontology.org) and were classified into three
categories, including biological process (BP), molecular function
(MF) and cellular component (CC). In addition, the biological
pathways of circRNA-target genes were identified using Kyoto
Encyclopedia of Genes and Genomes (KEGG; www.kegg.jp). The GO and KEGG analyses were performed
using DIANA-miRPath, version 3.0 (23).
Statistical analysis
Validated data from RT-qPCR are expressed as the
mean ± standard error of the mean. Differences between the groups
were assessed by SPSS statistical software version 19.0 (IBM Corp.,
Armonk, NY, USA) using one-way analysis of variance. P<0.05 was
considered to indicate a statistically significant difference.
Results
Expression profile of circRNAs
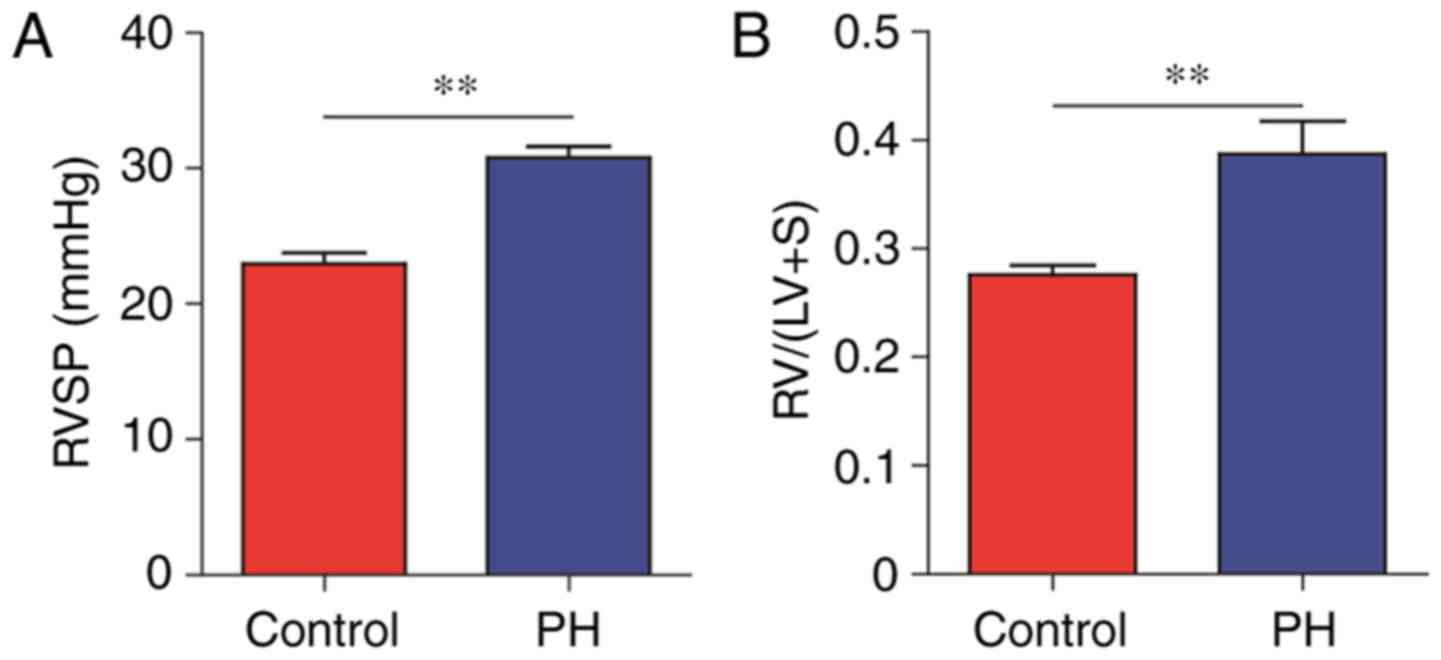
The murine model of hypoxia-induced PH was evaluated
by measuring the RVSP and RVH (24). When compared with the control
group, the hypoxia-induced PH group had significantly increased
levels of RVSP (30.77 vs. 22.96 mmHg, P<0.01) and RVH (0.39 vs.
0.28, P<0.01; Fig. 1A and B).
Subsequent to obtaining the aforementioned measurements, the lungs
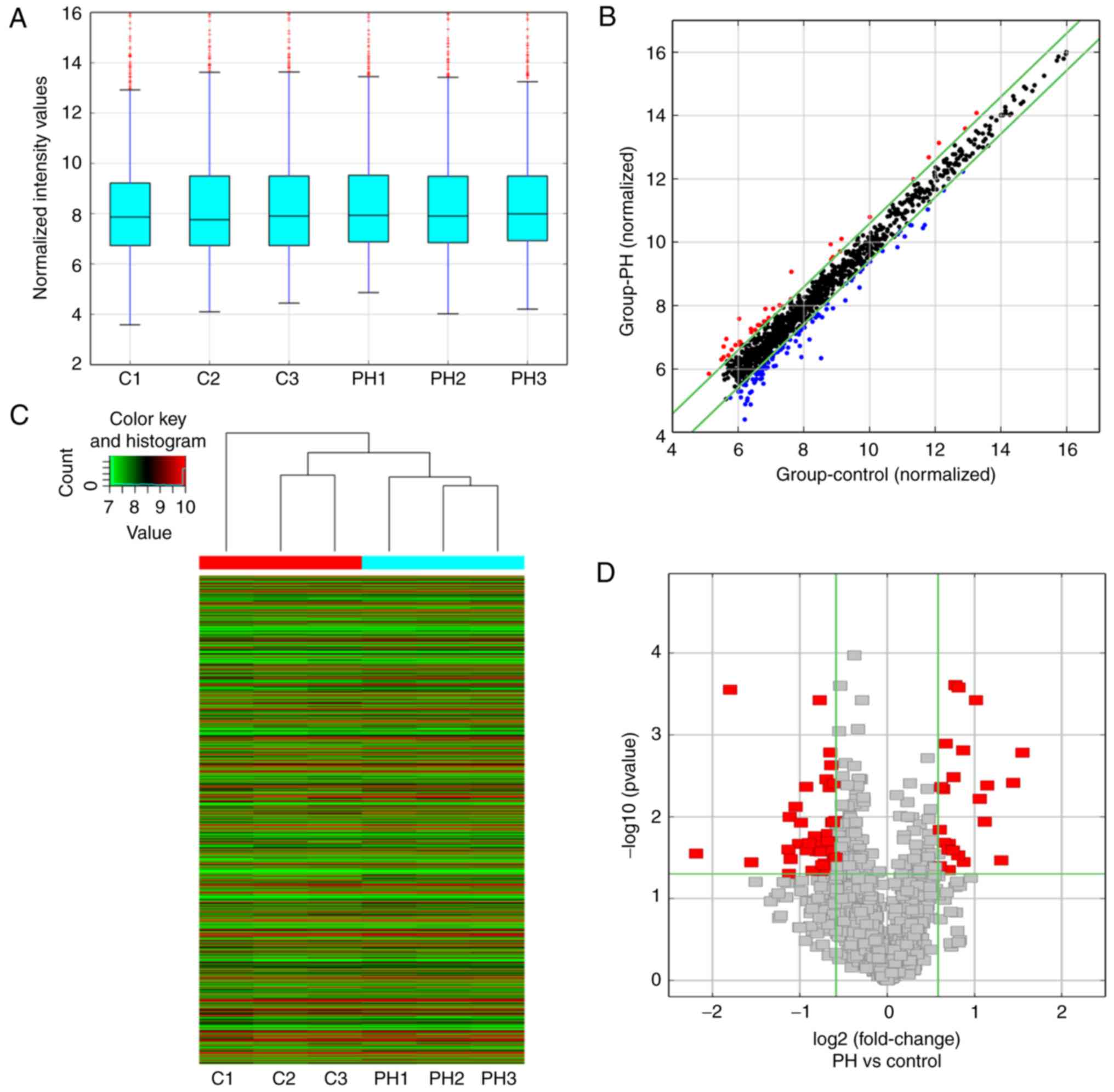
were harvested for circRNA microarray detection. The box plot in
Fig. 2A demonstrates that there
was no abnormal distribution of circRNA expression in the six
samples after normalization. Next, a scatter plot was used to
assess the expression variation of circRNAs between the hypoxia-PH
and control groups (Fig. 2B). The
hierarchical cluster analysis revealed the differential expression
profile of circRNAs in these two groups (Fig. 2C). A volcano plot was then
constructed using fold-change values and P-values in order to
visualize the differential expression of circRNAs between the two
groups (Fig. 2D). In total, 64
differentially expressed circRNAs were identified between the
hypoxia-PH and control groups, of which 23 circRNAs were
upregulated and 41 circRNAs were downregulated. The top 20
upregulated and top 20 downregulated circRNAs are listed in
Table II.
 | Table IITop 20 upregulated and top 20
downregulated circRNAs in mice with hypoxia-induced pulmonary
hypertension. |
Table II
Top 20 upregulated and top 20
downregulated circRNAs in mice with hypoxia-induced pulmonary
hypertension.
| CircRNA | Fold change | P-value | Regulation | CircRNA type | Chromosome | Gene symbol |
|---|
|
mmu_circRNA_004592 | 2.925894 | 0.001652 | Up | Exonic | X | Fam120c |
|
mmu_circRNA_018701 | 2.717783 | 0.003849 | Up | Exonic | 10 | Jmjd1c |
|
mmu_circRNA_010744 | 2.469385 | 0.03412 | Up | Exonic | 1 | Sntg1 |
|
mmu_circRNA_015666 | 2.211677 | 0.004139 | Up | Exonic | 4 | Ikbkap |
|
mmu_circRNA_016128 | 2.168588 | 0.011483 | Up | Exonic | 7 | Nars2 |
|
mmu_circRNA_016636 | 2.085027 | 0.006036 | Up | Exonic | 12 | Ttc7b |
|
mmu_circRNA_002378 | 2.02187 | 0.000377 | Up | Intronic | 2 | Kif5c |
|
mmu_circRNA_011174 | 1.834095 | 0.035723 | Up | Intragenic | 17 | XLOC_010800 |
|
mmu_circRNA_004941 | 1.82896 | 0.001544 | Up | Intragenic | 13 | Cdc14b |
|
mmu_circRNA_015350 | 1.759724 | 0.000263 | Up | Exonic | 1 | Ncoa2 |
|
mmu_circRNA_011414 | 1.756762 | 0.029613 | Up | Exonic | 18 | Tcf4 |
|
mmu_circRNA_008988 | 1.717354 | 0.000246 | Up | Exonic | 4 | Unc13b |
|
mmu_circRNA_011332 | 1.691847 | 0.003271 | Up | Intragenic | 8 | Large |
|
mmu_circRNA_014444 | 1.684291 | 0.025843 | Up | Exonic | 4 | Haus6 |
|
mmu_circRNA_006839 | 1.632387 | 0.044651 | Up | Intronic | 17 | Crim1 |
|
mmu_circRNA_007240 | 1.632184 | 0.021443 | Up | Exonic | 5 | Bre |
|
mmu_circRNA_017590 | 1.627719 | 0.025193 | Up | Exonic | 6 | Ccdc132 |
|
mmu_circRNA_013053 | 1.59081 | 0.001276 | Up | Exonic | 6 | Gmcl1 |
|
mmu_circRNA_000113 | 1.566056 | 0.02078 | Up | Antisense | 12 | Rian |
|
mmu_circRNA_012851 | 1.561305 | 0.004609 | Up | Exonic | 2 | Arhgap21 |
|
mmu_circRNA_010120 | 4.557204 | 0.028091 | Down | Exonic | 14 | Cryl1 |
|
mmu_circRNA_018351 | 3.479684 | 0.00028 | Down | Exonic | 8 | Zfhx3 |
|
mmu_circRNA_013176 | 2.940519 | 0.036055 | Down | Exonic | 4 | Frmd3 |
|
mmu_circRNA_012978 | 2.189961 | 0.025108 | Down | Intronic | 17 | Tsc2 |
|
mmu_circRNA_007835 | 2.181447 | 0.049532 | Down | Intragenic | 13 | Cdc14b |
|
mmu_circRNA_008379 | 2.167655 | 0.010006 | Down | Exonic | 2 | Sp3 |
|
mmu_circRNA_001170 | 2.150695 | 0.032906 | Down | Intragenic | 10 | Hsp90b1 |
|
mmu_circRNA_000267 | 2.068873 | 0.007548 | Down | Exonic | 3 | Plekho1 |
|
mmu_circRNA_014597 | 2.014597 | 0.021489 | Down | Exonic | 1 | Kifap3 |
|
mmu_circRNA_003780 | 1.983159 | 0.011809 | Down | Exonic | 13 | Cdyl |
|
mmu_circRNA_005086 | 1.901804 | 0.004295 | Down | Intragenic | 10 | Tbpl1 |
|
mmu_circRNA_010464 | 1.894022 | 0.025737 | Down | Exonic | 6 | Ezh2 |
|
mmu_circRNA_009705 | 1.859579 | 0.020918 | Down | Exonic | 7 | Mettl9 |
|
mmu_circRNA_001362 | 1.809006 | 0.045429 | Down | Exonic | 5 | Gm15800 |
|
mmu_circRNA_018468 | 1.776092 | 0.017304 | Down | Exonic | 18 | Arhgap26 |
|
mmu_circRNA_006562 | 1.752095 | 0.026256 | Down | Intragenic | 10 | Erbb3 |
|
mmu_circRNA_002959 | 1.727222 | 0.020858 | Down | Exonic | 8 | Phkb |
|
mmu_circRNA_005155 | 1.710652 | 0.000376 | Down | Exonic | 16 | Robo1 |
|
mmu_circRNA_000270 | 1.694113 | 0.026716 | Down | Exonic | 2 | Fam188a |
|
mmu_circRNA_009674 | 1.687043 | 0.046010 | Down | Exonic | 5 | Rnf4 |
Validation of circRNAs expression
In order to validate the results of the circRNA
microarray, 12 differentially expressed circRNAs (including six
upregulated and six downregulated circRNAs) were selected for
RT-qPCR analysis on the basis of the fold-change and P-values. The
six upregulated circRNAs were as follows: mmu_circRNA_004592, m
mu_ci rcR NA_018701, m mu_ci rcR NA_01074 4, m mu_ci rcR NA_015666,
m mu_ci rcR NA_016128 and mmu_circRNA_016636. The six downregulated
circRNAs were as follows: mmu_circRNA_010120, m mu_ci rcR
NA_018351, m mu_ci rcR NA_013176, mmu_circRNA_008379,
mmu_circRNA_000267 and mmu_circRNA_014597. Analysis by RT-qPCR
revealed that five of the upregulated circRNAs and two of the
down-regulated circRNAs, exhibited changes in their expression
levels that were consistent with the results of the circRNA
microarray. More specifically, mmu_circRNA_004592,
mmu_circRNA_018701, mmu_circRNA_010744, mmu_circRNA_015666 and
mmu_circRNA_016636 were found to be significantly upregulated in
the hypoxia-PH group compared with their levels in the control
group (Fig. 3A–E), while
mmu_circRNA_018351 and mmu_circRNA_008379 were significantly
downregulated compared with their levels in the control group
(Fig. 3F and G).
Identification of circRNA-targeting
miRNAs
CircRNAs typically have multiple MREs to bind
miRNAs, which reduces the expression of miRNAs and indirectly
upregulates the expression of miRNA target genes (7). Thus, putative circRNA-miRNA
interactions were predicted according to the complementary matching
sequence using the TargetScan and miRanda tools. All circRNAs
identified in the present study were found to have miRNA binding
sites. The top five miRNA binding sites for dysregulated circRNAs
were predicted (data not shown). Table III lists the seven validated
circRNAs and their top five miRNA binding sites. The top five
potential miRNA binding sites for the most significantly
upregulated circRNA (namely mmu_circRNA_004592) were
mmu-miR-742-3p, mmu-miR-6373, mmu-miR-880-5p, mmu-miR-298-3p and
mmu-miR-152-3p. In addition, the top five potential miRNA binding
sites for the most significantly downregulated circRNA (namely
mmu_circRNA_018351) were mmu-miR-6992-3p, mmu-miR-5133,
mmu-miR-6936-5p, mmu-miR-6938-5p and mmu-miR-7015-3p.
 | Table IIITop five miRNA binding sites for
validated circRNAs. |
Table III
Top five miRNA binding sites for
validated circRNAs.
| CircRNA | MRE1 | MRE2 | MRE3 | MRE4 | MRE5 |
|---|
|
mmu_circRNA_016636 |
mmu-miR-7239-3p | mmu-miR-6408 | mmu-miR-877-5p | mmu-miR-149-3p |
mmu-miR-7047-5p |
|
mmu_circRNA_015666 | mmu-miR-1903 |
mmu-miR-7008-3p |
mmu-miR-103-2-5p |
mmu-miR-103-1-5p | mmu-miR-679-5p |
|
mmu_circRNA_010744 | mmu-miR-8113 |
mmu-miR-7012-5p |
mmu-miR-7674-5p | mmu-miR-432 | mmu-miR-6350 |
|
mmu_circRNA_018701 | mmu-miR-653-5p | mmu-miR-17-3p | mmu-miR-6394 |
mmu-miR-6948-3p |
mmu-miR-148b-3p |
|
mmu_circRNA_004592 | mmu-miR-742-3p | mmu-miR-6373 | mmu-miR-880-5p | mmu-miR-298-3p | mmu-miR-152-3p |
|
mmu_circRNA_008379 |
mmu-miR-6997-5p | mmu-miR-378b |
mmu-miR-7233-3p | mmu-miR-6359 | mmu-miR-1967 |
|
mmu_circRNA_018351 | mmu-miR-877-3p | mmu-miR-1903 | mmu-miR-667-5p | mmu-miR-207 | mmu-miR-665-5p |
Construction of circRNA-miRNA-mRNA
networks
The function of circRNA as an miRNA sponge is the
main biological role of circRNA in various diseases. Thus,
circRNA-miRNA-mRNA network analysis was used to predict the
potential function of differentially expressed circRNAs in
hypoxia-induced PH. The two most significant circRNAs
(mmu_circRNA_004592 and mmu_circRNA_018351) were selected for ceRNA
analysis. The circRNA-targeting genes were predicted by integrating
four miRNA-target prediction tools, including miRWalk, RNA22,
miRanda and TargetScan.
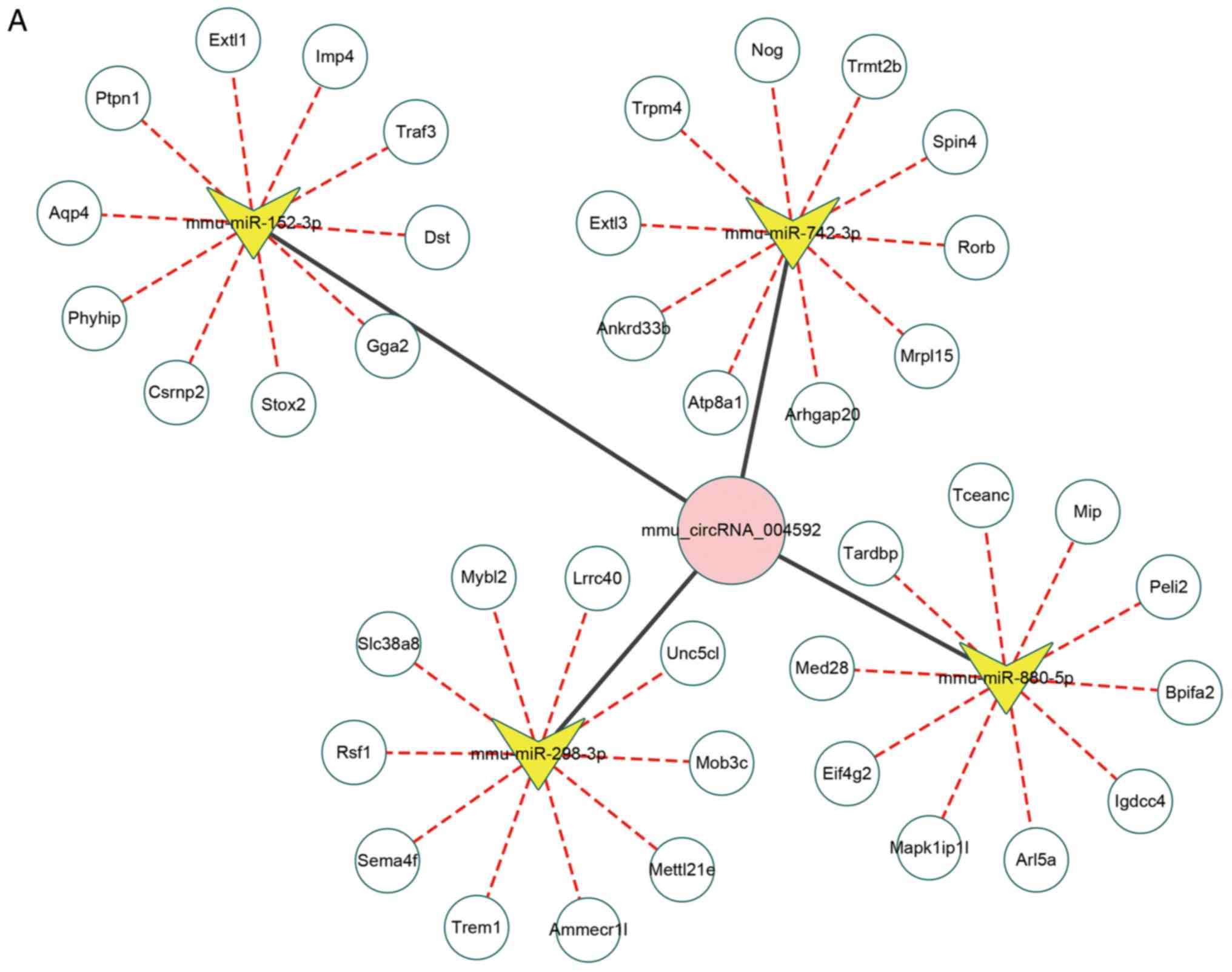
For mmu_circRNA_004592, there was no predicted data
for one of the five miRNAs targets (mmu-miR-6373), and therefore
this target does not appear in the analysis shown in Fig. 4A. For mmu_circRNA_004592, the
circRNA-miRNA-mRNA network was constructed (Fig. 4A). The upregulated
mmu_circRNA_004592 was predicted to inhibit the expression level of
mmu-miR-742-3p, mmu-miR-880-5p, mmu-miR-298-3p and mmu-miR-152-3p,
and further promote the expression of miRNA-targeted genes. The top
ten miRNA-targeted genes are presented in Fig. 4A. The sequence analysis of MREs of
mmu_circRNA_004592 was presented in Fig. 4B. The 2D structure demonstrated
the MRE sequence, the target miRNA seed type and the 3′ pairing
sequence for mmu_circRNA_004592. Similarly, the circRNA-miRNA-mRNA
network for mmu_circRNA_018351 was constructed (Fig. 5A). The downregulated
mmu_circRNA_018351 was predicted to increase the expression level
of mmu-miR-6992-3p, mmu-miR-5133, mmu-miR-6936-5p, mmu-miR-6938-5p
and mmu-miR-7015-3p, and further reduce the expression of
miRNA-targeted genes. The top ten miRNA-targeted genes are
presented in Fig. 5A. The
sequence analysis of MREs of mmu_circRNA_018351 identified the MRE
sequence, the target miRNA seed type and the 3′ pairing sequence
for mmu_circRNA_004592 in 2D sequence (Fig. 5B).
GO and KEGG analyses for
circRNA-targeting genes
CircRNAs with a fold-change of ≥2 and a P-value of
<0.05 were selected from the dysregulated circRNAs in the
hypoxia-induced PH group, and the role of these circRNAs was
further investigated by GO and KEGG analyses. For the target genes
of the upregulated circRNAs, the top 10 enriched GO terms in the BP
and CC categories, as well as the total eight GO terms in the MF
category were identified. The top three enriched GO terms were
biological processes, cellular nitrogen compound metabolic
processes and anatomical structure development in BP; cellular
component, cell and intracellular in CC; molecular function, ion
binding and enzyme binding in MF (Fig. 6A). For the target genes of the
downregulated circRNAs, the top 10 enriched GO terms in the BP and
CC categories, as well as the total five GO terms in the MF
category were displayed. The top three enriched were biological
processes, anatomical structure development and cellular nitrogen
compound metabolic process in BP; cellular component, cell and
intracellular in CC; molecular function, ion binding and nucleic
acid binding transcription factor activity in MF (Fig. 6B).
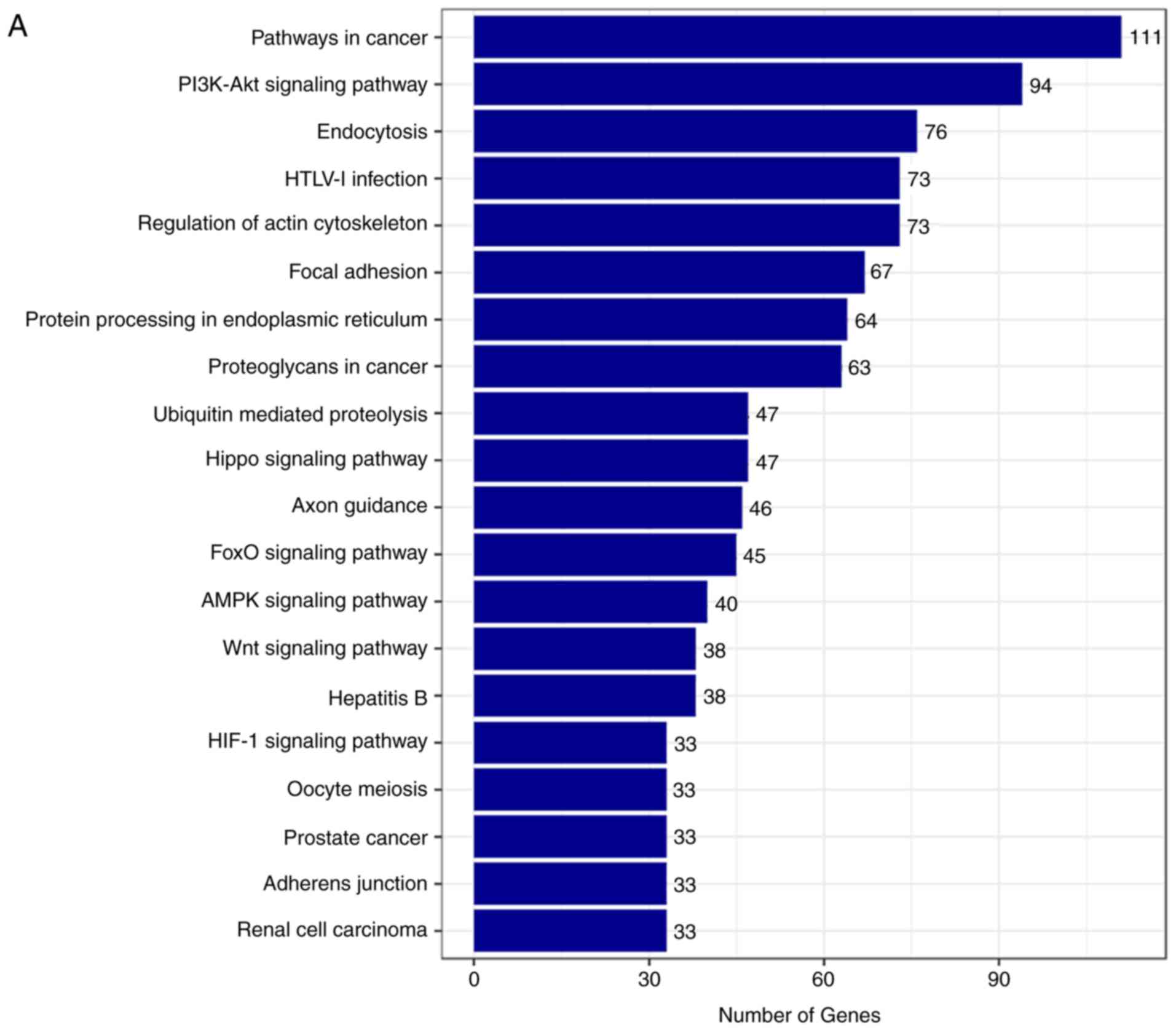
The top 20 KEGG pathways were also identified for
the target genes of the upregulated and downregulated circRNAs,
respectively (Fig. 7A–D). The top
five enriched pathways for the target genes of the upregulated
circRNAs included pathways in cancer, the PI3K-Akt signaling
pathway, endocytosis, HTLV-I infection and regulation of actin
cytoskeleton (Fig. 7A and B).
Furthermore, the top five enriched pathways for the target genes of
the downregulated circRNAs involved pathways in cancer, the
PI3K-Akt signaling pathway, regulation of actin cytoskeleton,
proteoglycans in cancer, and the MAPK signaling pathway (Fig. 7C and D).
Discussion
To the best of our knowledge, the present study is
the first to perform circRNA microarray analysis to detect the
differential expression profile of circRNAs in the lungs of mice
with hypoxia-induced PH. In total, 23 significantly upregulated and
41 significantly downregulated circRNAs were identified. Of these,
12 differentially expressed circRNAs were selected for further
validation using RT-qPCR. Bioinformatics tools were used to
construct the circRNA-miRNA-mRNA networks, and the putative
functions and pathways were obtained using GO and KEGG enrichment
analyses. The results from circRNA-miRNA-mRNA networks analysis and
GO and KEGG analysis suggested that dysregulated circRNAs may serve
key roles in the pathogenesis of hypoxia-induced PH.
Pulmonary vascular remodeling is a key feature of
hypoxia-induced PH with associated dysfunction of endothelial
cells, smooth muscle cells and fibroblasts (4). Recently, novel molecular mechanisms
of PH have been identified with particular advances in ‘omics’
approaches (25). Several types
of noncoding RNAs, such as long noncoding RNAs and miRNAs,
participate in the pathological process of hypoxia-induced PH, and
certain of these are considered to be diagnostic or therapeutic
targets (26,27). CircRNAs are novel members of the
non-coding RNA family and serve an important role in the
development of several pulmonary diseases. The biological functions
of circRNAs are varied; however, their function as miRNA sponges is
likely to be the primary mechanism of action in a number of
pulmonary diseases, including non-small cell lung cancer (28,29), pulmonary tuberculosis (30), acute respiratory distress syndrome
(31) and chronic thromboembolic
PH (32). However, to the best of
our knowledge, no previous study to date has investigated the
expression of circRNAs in hypoxia-induced PH. Thus, to further
elucidate the molecular RNA-based mechanism of PH, the circRNA
expression profile in hypoxia-induced PH was determined in the
present study, using microarray analysis and bioinformatics to
identify several circRNAs that are potentially important in the
development of hypoxia-induced PH.
Based on the circRNA target prediction analysis, all
of the differentially expressed circRNAs that were identified
contained MREs to sponge different miRNAs. Of these, the two best
candidate circRNAs were selected, namely mmu_circRNA_004592 and
mmu_circRNA_018351, and their circRNA-miRNA-mRNA networks were
constructed using bioinformatics tools. The upregulated
mmu_circRNA_004592 was predicted to negatively regulate the
function of mmu-miR-742-3p, mmu-miR-6373, mmu-miR-880-5p,
mmu-miR-298-3p and mmu-miR-152-3p. Although there is no direct
evidence to show the role of these miRNAs in hypoxia-induced PH, Wu
et al (33) have
previously reported that hypoxia significantly inhibited the
expression of miR-152 in human umbilical vein endothelial cells and
that miR-152 targeted ADAM17 to inhibit cell migration and
proliferation. Furthermore, several studies have suggested that
miR-152 is a tumor suppressor (34). miR-152 has been reported to
inhibit cancer cell proliferation, invasion and migration by
targeting Wnt1 (35), PIK3CA
(36), DNMT1 (37) and PTEN (38). Additionally, Gu et al
(39) identified that miR-152
promoted senescence in stem cells from human dental pulp by
targeting SIRT7 expression. Since mmu_circRNA_004592 expression was
upregulated in the present study, it can be hypothesized that
miR-152 is downregulated, which promotes proliferation, and
inhibits senescence and apoptosis of endothelial cells, smooth
muscle cells and fibroblasts in the pulmonary artery. These actions
may further raise the pressure in the pulmonary vessels.
The current study also demonstrated that
mmu_circRNA_018351 was downregulated and had potential binding
sites for mmu-miR-877-3p, mmu-miR-1903, mmu-miR-667-5p, mmu-miR-207
and mmu-miR-665-5p. Of these, miR-207 serves a protective role
against cell apoptosis following isch-emic stroke (40). In addition, miR-665 regulates
insulin-like growth factor 2 to protect from sevoflurane
anesthesia-induced cognitive dysfunction through inhibiting
apoptosis (41). Based on the
bioinformatics analysis of the present study, it is hypothesized
that miR-207 and miR-665 are upregulated as a consequence of the
downregulated mmu_circRNA_018351 expression to repress the
apoptosis of endothelial cells, smooth muscle cells and fibroblasts
in hypoxia-induced PH.
Function and pathway analyses for the target genes
of the circRNAs were performed to provide further insight into the
mechanism of hypoxia-induced PH. Among the enriched GO terms in BP,
the cellular nitrogen compound metabolic process was identified.
Several studies have demonstrated that the level of nitric oxide
(NO) is decreased in patients with PH (42). NO is an important vasodilator for
pulmonary vessels, and a decrease in the levels of NO could
increase the vascular resistance and vascular pressure (43). Although current therapy targeting
the NO pathway improves the clinical outcome of patients with PH,
the role of the NO pathway in the pathogenesis of pulmonary
vascular remodeling in PH has not yet been fully elucidated
(44). Thus, it is proposed that
the circRNAs identified in the present study contribute to the
pathophysiological effect of NO in PH and are potential clinical
biomarkers.
Bioinformatics analysis also identified several
hypoxia and vascular remodeling-associated pathways. The PI3K-Akt
signaling pathway serves an important role in hypoxia-induced PH
through promoting the proliferation of endothelial cells and smooth
muscle cells (45,46). Furthermore, the Hippo, Wnt, AMPK
and HIF-1 signaling pathways, which were identified in KEGG pathway
analysis in the present study, have been reported to have a close
association with the development of hypoxia-induced PH (47,48). These findings suggest that
circRNAs may be key modulators in the molecular mechanism of
hypoxia-induced PH and that, in the future, specific circRNAs could
be tested as diagnostic and therapeutic targets of hypoxia-induced
PH.
There are a number of limitations in the present
study. Firstly, the differentially expressed circRNAs were
identified only in a mouse model with hypoxia-induced PH and should
be further validated in patients with PH. Secondly, the functions
of the differentially expressed circRNAs were predicted using
bioinformatics tools, and there were no further studies to
demonstrate the role of candidate circRNAs in the pathological
process of hypoxia-induced PH. Thirdly, the circRNA-miRNA-mRNA
networks were constructed in the current study according to the
premise that the majority of circRNAs function as miRNA sponges.
However, the molecular functions of >99.9% of identified
circRNAs remain unknown, and numerous other functions, such as
translation and regulation of transcription and alternative
splicing, have been reported (49). Thus, in the future, functional
experiments should be conducted to elucidate the roles of these
circRNAs in hypoxia-induced PH.
In conclusion, the present study is the first to
profile circRNA expression in hypoxia-induced PH. In total, 23
significantly upregulated and 41 significantly downregulated
circRNAs were identified. The circRNA-miRNA interactions were also
predicted and circRNA-miRNA-mRNA networks were constructed for two
leading candidate circRNAs. Furthermore, GO and pathway analyses
revealed the potential role of the target genes of circRNAs in
hypoxia-induced PH. The findings suggest that circRNAs serve a key
role in the pathological process of hypoxia-induced PH. Thus,
candidate circRNAs are potential targets for treatment and
potential biomarkers for diagnosis of patients with hypoxia-induced
PH.
Acknowledgments
Not applicable.
Funding
The study was supported by the State Key Basic
Research Program Project (grant no. 2015CB553404), the National
Natural Science Foundation of China (grant nos. 81770055, 81500026,
81570028 and 81600056), the Key Grant (grant nos. 81630001 and
81490533), the National Natural Science Foundation of China for
Young Researcher (grant no. 81600056), the Shanghai Science and
Technology Committee Grant (grant nos. 15DZ1930600, 15DZ193060 and
16ZR1405700) and the Shanghai Municipal Commission of Health and
Family Planning (grant no. 201540370).
Availability of data and materials
All data generated or analyzed during this study are
included in this published article.
Authors’ contributions
SJX, HH and YLS designed and supervised the study.
JW, MCZ and JZW made the mouse model. LLW and HYG contributed to
data collection. CCC and XDT analyzed and interpreted the data. JW
and MCZ drew the figures and wrote the manuscript. BK and YLS
provided solutions for data analysis and edited the manuscript. All
authors read and approved the final article.
Ethics approval and consent to
participate
This study was approved by the Institutional Animal
Care and Use Committee of Huadong Hospital, Fudan University,
Shanghai, China.
Patient consent for publication
Not applicable.
Competing interests
The authors declare that they have no competing
interests.
References
|
1
|
Thompson AAR and Lawrie A: Targeting
vascular remodeling to treat pulmonary arterial hypertension.
Trends Mol Med. 23:31–45. 2017. View Article : Google Scholar
|
|
2
|
Task Force for Diagnosis and Treatment of
Pulmonary Hypertension of European Society of Cardiology (ESC);
European Respiratory Society (ERS); International Society of Heart
and Lung Transplantation (ISHLT); Galiè N, Hoeper MM, Humbert M,
Torbicki A, Vachiery JL, Barbera JA, Beghetti M, Corris P, Gaine S,
Gibbs JS, et al: Guidelines for the diagnosis and treatment of
pulmonary hypertension. Eur Heart J. 32:2493–2537. 2009.
|
|
3
|
Naeije R and Dedobbeleer C: Pulmonary
hypertension and the right ventricle in hypoxia. Exp Physiol.
98:1247–1256. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
4
|
Ghofrani HA, Voswinckel R, Reichenberger
F, Weissmann N, Schermuly RT, Seeger W and Grimminger F: Hypoxia-
and non-hypoxia-related pulmonary hypertension-established and new
therapies. Cardiovasc Res. 72:30–40. 2006. View Article : Google Scholar : PubMed/NCBI
|
|
5
|
Kylhammar D and Rådegran G: The principal
pathways involved in the in vivo modulation of hypoxic pulmonary
vasoconstriction, pulmonary arterial remodelling and pulmonary
hypertension. Acta Physiol. 219:728–756. 2017. View Article : Google Scholar
|
|
6
|
McLaughlin VV: Looking to the future: A
new decade of pulmonary arterial hypertension therapy. Eur Respir
Rev. 20:262–269. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
7
|
Liu L, Wang J, Khanabdali R, Kalionis B,
Tai X and Xia S: Circular RNAs: Isolation, characterization and
their potential role in diseases. RNA Biol. 14:1715–1721. 2017.
View Article : Google Scholar : PubMed/NCBI
|
|
8
|
Salzman J, Gawad C, Wang PL, Lacayo N and
Brown PO: Circular RNAs are the predominant transcript isoform from
hundreds of human genes in diverse cell types. PLoS One.
7:e307332012. View Article : Google Scholar : PubMed/NCBI
|
|
9
|
Liu J, Liu T, Wang X and He A: Circles
reshaping the RNA world: From waste to treasure. Mol Cancer.
16:582017. View Article : Google Scholar : PubMed/NCBI
|
|
10
|
Hansen TB, Jensen TI, Clausen BH, Bramsen
JB, Finsen B, Damgaard CK and Kjems J: Natural RNA circles function
as efficient microRNA sponges. Nature. 495:384–388. 2013.
View Article : Google Scholar : PubMed/NCBI
|
|
11
|
Han B, Chao J and Yao H: Circular RNA and
its mechanisms in disease: From the bench to the clinic. Pharmacol
Ther. 187:31–44. 2018. View Article : Google Scholar : PubMed/NCBI
|
|
12
|
Legnini I, Di Timoteo G, Rossi F, Morlando
M, Briganti F, Sthandier O, Fatica A, Santini T, Andronache A, Wade
M, et al: Circ-ZNF609 is a circular RNA that can be translated and
functions in myogenesis. Mol Cell. 66:22–37. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
13
|
Kristensen LS, Hansen TB, Veno MT and
Kjems J: Circular RNAs in cancer: Opportunities and challenges in
the field. Oncogene. 37:555–565. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
14
|
Fan X, Weng X, Zhao Y, Chen W, Gan T and
Xu D: Circular RNAs in cardiovascular disease: An overview. Biomed
Res Int. 2017:51357812017. View Article : Google Scholar : PubMed/NCBI
|
|
15
|
Floris G, Zhang L, Follesa P and Sun T:
Regulatory role of circular RNAs and neurological disorders. Mol
Neurobiol. 54:5156–5165. 2017. View Article : Google Scholar
|
|
16
|
Shan K, Liu C, Liu BH, Chen X, Dong R, Liu
X, Zhang YY, Liu B, Zhang SJ, Wang JJ, et al: Circular noncoding
RNA HIPK3 mediates retinal vascular dysfunction in diabetes
mellitus. Circulation. 136:1629–1642. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
17
|
Ciuclan L, Bonneau O, Hussey M, Duggan N,
Holmes AM, Good R, Stringer R, Jones P, Morrell NW, Jarai G, et al:
A novel murine model of severe pulmonary arterial hypertension. Am
J Respir Crit Care Med. 184:1171–1182. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
18
|
Livak KJ and Schmittgen TD: Analysis of
relative gene expression data using real-time quantitative PCR and
the 2−ΔΔCT method. Methods.
25:402–408. 2001. View Article : Google Scholar
|
|
19
|
Wang Y, Mo Y, Gong Z, Yang X, Yang M,
Zhang S, Xiong F, Xiang B, Zhou M, Liao Q, et al: Circular RNAs in
human cancer. Mol Cancer. 16:252017. View Article : Google Scholar : PubMed/NCBI
|
|
20
|
Enright AJ, John B, Gaul U, Tuschl T,
Sander C and Marks DS: MicroRNA targets iDrosophila. Genome Biol.
5:R12003. View Article : Google Scholar
|
|
21
|
Pasquinelli AE: MicroRNAs and their
targets: Recognition, regulation and an emerging reciprocal
relationship. Nat Rev Genet. 13:271–282. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
22
|
Dweep H and Gretz N: miRWalk2.0: A
comprehensive atlas of microRNA-target interactions. Nat Methods.
12:6972015. View Article : Google Scholar : PubMed/NCBI
|
|
23
|
Vlachos IS, Zagganas K, Paraskevopoulou
MD, Georgakilas G, Karagkouni D, Vergoulis T, Dalamagas T and
Hatzigeorgiou AG: DIANA-miRPath v3.0: Deciphering microRNA function
with experimental support. Nucleic Acids Res. 43:W460–W466. 2015.
View Article : Google Scholar : PubMed/NCBI
|
|
24
|
Wang W, Liu J, Ma A, Miao R, Jin Y, Zhang
H, Xu K, Wang C and Wang J: mTORC1 is involved in hypoxia-induced
pulmonary hypertension through the activation of Notch3. J Cell
Physiol. 229:2117–2125. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
25
|
Kan M, Shumyatcher M and Himes BE: Using
omics approaches to understand pulmonary diseases. Respir Res.
18:1492017. View Article : Google Scholar : PubMed/NCBI
|
|
26
|
Bienertova-Vasku J, Novak J and Vasku A:
MicroRNAs in pulmonary arterial hypertension: Pathogenesis,
diagnosis and treatment. J Am Soc Hypertens. 9:221–234. 2015.
View Article : Google Scholar : PubMed/NCBI
|
|
27
|
Deng L, Bradshaw AC and Baker AH: Role of
noncoding RNA in vascular remodelling. Curr Opin Lipidol.
27:439–448. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
28
|
Yao JT, Zhao SH, Liu QP, Lv MQ, Zhou DX,
Liao ZJ and Nan KJ: Overexpression of CircRNA_100876 in non-small
cell lung cancer and its prognostic value. Pathol Res Pract.
213:453–456. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
29
|
Zhu X, Wang X, Wei S, Chen Y, Chen Y, Fan
X, Han S and Wu G: hsa_circ_0013958: A circular RNA and potential
novel biomarker for lung adenocarcinoma. FEBS J. 284:2170–2182.
2017. View Article : Google Scholar : PubMed/NCBI
|
|
30
|
Zhuang ZG, Zhang JA, Luo HL, Liu GB, Lu
YB, Ge NH, Zheng BY, Li RX, Chen C, Wang X, et al: The circular RNA
of peripheral blood mononuclear cells: Hsa_circ_0005836 as a new
diagnostic biomarker and therapeutic target of active pulmonary
tuberculosis. Mol Immunol. 90:264–272. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
31
|
Wan QQ, Wu D and Ye QF: The expression
profiles of circRNAs in lung tissues from rats with
lipopolysaccharide-induced acute respiratory distress syndrome: A
microarray study. Biochem Biophys Res Commun. 493:684–689. 2017.
View Article : Google Scholar : PubMed/NCBI
|
|
32
|
Miao R, Wang Y, Wan J, Leng D, Gong J, Li
J, Liang Y, Zhai Z and Yang Y: Microarray expression profile of
circular RNAs in chronic thromboembolic pulmonary hypertension.
Medicine. 96:e73542017. View Article : Google Scholar : PubMed/NCBI
|
|
33
|
Wu Y, Huang A, Li T, Su X, Ding H, Li H,
Qin X, Hou L, Zhao Q, Ge X, et al: MiR-152 reduces human umbilical
vein endothelial cell proliferation and migration by targeting
ADAM17. Febs Lett. 588:2063–2069. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
34
|
Liu X, Li J, Qin F and Dai S: miR-152 as a
tumor suppressor microRNA: Target recognition and regulation in
cancer. Oncol Lett. 11:3911–3916. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
35
|
Nie L, Zhao YB, Pan JL, Lei Y, Liu M, Long
Y, Zhang JH, Hu Y, Xu MQ, Yuan DZ and Yue LM: Progesterone-induced
miR-152 inhibits the proliferation of endometrial epithelial cells
by down-regulating WNT-1. Reprod Sci. 24:1444–1453. 2017.
View Article : Google Scholar : PubMed/NCBI
|
|
36
|
Ge S, Wang D, Kong Q, Gao W and Sun J:
Function of miR-152 as a tumor suppressor in human breast cancer by
targeting PIK3CA. Oncol Res. 25:1363–1371. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
37
|
Sun J, Tian X, Zhang J, Huang Y, Lin X,
Chen L and Zhang S: Regulation of human glioma cell apoptosis and
invasion by miR-152-3p through targeting DNMT1 and regulating NF2:
MiR-152-3p regulate glioma cell apoptosis and invasion. J Exp Clin
Cancer Res. 36:1002017. View Article : Google Scholar :
|
|
38
|
Wang S, Wang L, Dou L, Guo J, Fang W, Li
M, Meng X, Man Y, Shen T, Huang X and Li J: MicroRNA 152 regulates
hepatic glycogenesis by targeting PTEN. FEBS J. 283:1935–1946.
2016. View Article : Google Scholar : PubMed/NCBI
|
|
39
|
Gu S, Ran S, Liu B and Liang J: miR-152
induces human dental pulp stem cell senescence by inhibiting SIRT7
expression. FEBS Lett. 590:1123–1131. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
40
|
Tao J, Liu W, Shang G, Zheng Y, Huang J,
Lin R and Chen L: MiR-207/352 regulate lysosomal-associated
membrane proteins and enzymes following ischemic stroke.
Neuroscience. 305:1–14. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
41
|
Lu X, Lv S, Mi Y, Wang L and Wang G:
Neuroprotective effect of miR-665 against sevoflurane
anesthesia-induced cognitive dysfunction in rats through PI3K/Akt
signaling pathway by targeting insulin-like growth factor 2. Am J
Transl Res. 9:1344–1356. 2017.PubMed/NCBI
|
|
42
|
Sehgal PB and Mukhopadhyay S: Pulmonary
arterial hypertension: A disease of tethers, SNAREs and SNAPs. Am J
Physiol Heart Circ Physiol. 293:H77–H85. 2007. View Article : Google Scholar : PubMed/NCBI
|
|
43
|
Fulton D, Li X, Bordan Z, Haigh S, Bentley
A, Chen F and Barman SA: Reactive oxygen and nitrogen species in
the development of pulmonary hypertension. Antioxidants. 6:E542017.
View Article : Google Scholar : PubMed/NCBI
|
|
44
|
Thenappan T and Weir EK: The nitric oxide
pathway-A potential target for precision medicine in pulmonary
arterial hypertension. Am J Cardiol. 120:S69–S70. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
45
|
Zhang C, Ma C, Yao H, Zhang L, Yu X, Liu
Y, Shen T, Zhang L, Zhang F, Chen X and Zhu D: 12-Lipoxygenase and
12-hydroxyeicosatetraenoic acid regulate hypoxic angiogenesis and
survival of pulmonary artery endothelial cells via PI3K/Akt
pathway. Am J Physiol Lung Cell Mol Physiol. 314:L606–L616. 2018.
View Article : Google Scholar
|
|
46
|
Xia XD, Lee J, Khan S, Ye L, Li Y and Dong
L: Suppression of phosphatidylinositol 3-kinase/Akt signaling
attenuates hypoxia-induced pulmonary hypertension through the
downregulation of lysyl oxidase. DNA Cell Biol. 35:599–606. 2016.
View Article : Google Scholar : PubMed/NCBI
|
|
47
|
Lim CS, Kiriakidis S, Sandison A, Paleolog
EM and Davies AH: Hypoxia-inducible factor pathway and diseases of
the vascular wall. J Vasc Surg. 58:219–230. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
48
|
Awad KS, West JD, de Jesus PV and MacLean
M: Novel signaling pathways in pulmonary arterial hypertension
(2015 Grover conference series). Pulm Circ. 6:285–294. 2016.
View Article : Google Scholar : PubMed/NCBI
|
|
49
|
Wilusz JE: A 360° view of circular RNAs:
From biogenesis to functions. WIREs RNA. e14782018. View Article : Google Scholar
|