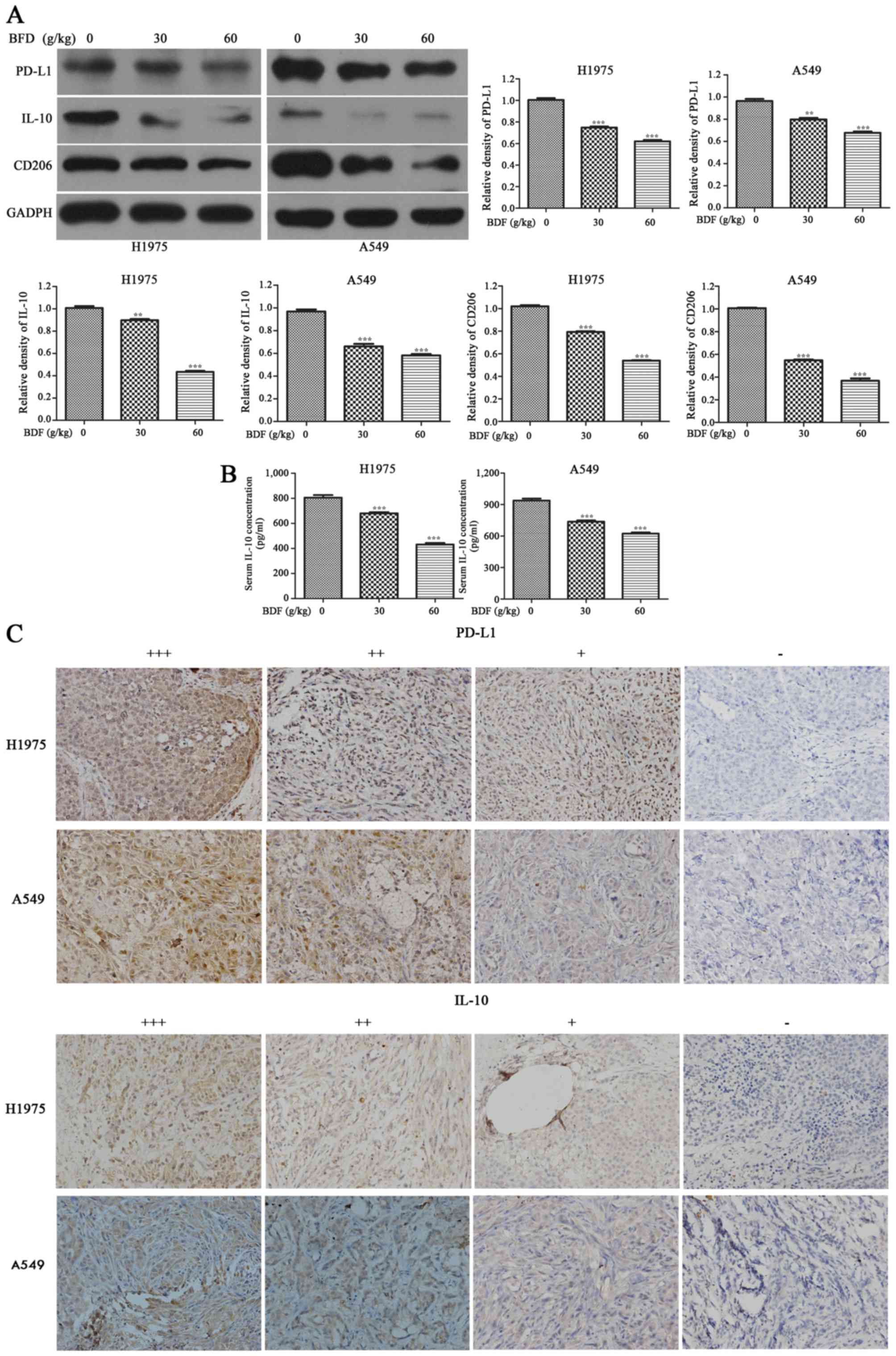
|
1
|
Torre LA, Bray F, Siegel RL, Ferlay J,
Lortet-Tieulent J and Jemal A: Global cancer statistics, 2012. CA
Cancer J Clin. 65:87–108. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
2
|
Howlader N, Noone AM, Krapcho M, Miler D,
Bishop K, Altekruse SF, Kosary CL, Yu M, Ruhl J, Tatalovich Z, et
al: SEER Cancer Statistics Review, 1975–2013. National Cancer
Institute; Bethesda, MD: http://seer.cancer.gov/csr/1975_2013/urisimpleseer.cancer.gov/csr/1975_2013/(based
on November 2015 SEER data submission, posted to the SEER web site,
April 2016).
|
|
3
|
Miller KD, Siegel RL, Lin CC, Mariotto AB,
Kramer JL, Rowland JH, Stein KD, Alteri R and Jemal A: Cancer
treatment and survivorship statistics, 2016. CA Cancer J Clin.
66:271–289. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
4
|
Gardiner RE, Jahangeer S, Forde P, Ariffin
AB, Bird B, Soden D and Hinchion J: Low immunogenicity in non-small
cell lung cancer; do new developments and novel treatments have a
role? Cancer Metastasis Rev. 34:129–144. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
5
|
Geissmann F, Manz MG, Jung S, Sieweke MH,
Merad M and Ley K: Development of monocytes, macrophages, and
dendritic cells. Science. 327:656–661. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
6
|
Solinas G, Germano G, Mantovani A and
Allavena P: Tumor-associated macrophages (TAM) as major players of
the cancer-related inflammation. J Leukoc Biol. 86:1065–1073. 2009.
View Article : Google Scholar : PubMed/NCBI
|
|
7
|
Taniguchi K, Hikiji H, Okinaga T,
Hashidate-Yoshida T, Shindou H, Ariyoshi W, Shimizu T, Tominaga K
and Nishihara T: Essential role of lysophosphatidylcholine
acyltransferase 3 in the induction of macrophage polarization in
PMA-treated U937 cells. J Cell Biochem. 116:2840–2848. 2015.
View Article : Google Scholar : PubMed/NCBI
|
|
8
|
Coffelt SB, Hughes R and Lewis CE:
Tumor-associated macrophages: Effectors of angiogenesis and tumor
progression. Biochim Biophys Acta. 1796:11–18. 2009.PubMed/NCBI
|
|
9
|
Zwadlo G, Voegeli R, Schulze Osthoff K and
Sorg C: A monoclonal antibody to a novel differentiation antigen on
human macrophages associated with the down-regulatory phase of the
inflammatory process. Exp Cell Biol. 55:295–304. 1987.PubMed/NCBI
|
|
10
|
Lee JH, Lee GT, Woo SH, Ha YS, Kwon SJ,
Kim WJ and Kim IY: BMP-6 in renal cell carcinoma promotes tumor
proliferation through IL-10-dependent M2 polarization of
tumor-associated macrophages. Cancer Res. 73:3604–3614. 2013.
View Article : Google Scholar : PubMed/NCBI
|
|
11
|
Liu C-Y, Xu J-Y, Shi X-Y, Huang W, Ruan
T-Y, Xie P and Ding J-L: M2-polarized tumor-associated macrophages
promoted epithelial-mesenchymal transition in pancreatic cancer
cells, partially through TLR4/IL-10 signaling pathway. Lab Invest.
93:844–854. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
12
|
Wang R, Zhang J, Chen S, Lu M, Luo X, Yao
S, Liu S, Qin Y and Chen H: Tumor-associated macrophages provide a
suitable microenvironment for non-small lung cancer invasion and
progression. Lung Cancer. 74:188–196. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
13
|
Tjiu JW, Chen JS, Shun CT, Lin SJ, Liao
YH, Chu CY, Tsai TF, Chiu HC, Dai YS, Inoue H, et al:
Tumor-associated macrophage-induced invasion and angiogenesis of
human basal cell carcinoma cells by cyclooxygenase-2 induction. J
Invest Dermatol. 129:1016–1025. 2009. View Article : Google Scholar
|
|
14
|
Li CC, Hou YC, Yeh CL and Yeh SL: Effects
of eicosapentaenoic acid and docosahexaenoic acid on prostate
cancer cell migration and invasion induced by tumor-associated
macrophages. PLoS One. 9:e996302014. View Article : Google Scholar : PubMed/NCBI
|
|
15
|
Condeelis J and Pollard JW: Macrophages:
Obligate partners for tumor cell migration, invasion, and
metastasis. Cell. 124:263–266. 2006. View Article : Google Scholar : PubMed/NCBI
|
|
16
|
Kurahara H, Shinchi H, Mataki Y, Maemura
K, Noma H, Kubo F, Sakoda M, Ueno S, Natsugoe S and Takao S:
Significance of M2-polarized tumor-associated macrophage in
pancreatic cancer. J Surg Res. 167:e211–e219. 2011. View Article : Google Scholar
|
|
17
|
Mellor AL and Munn DH: Creating immune
privilege: Active local suppression that benefits friends, but
protects foes. Nat Rev Immunol. 8:74–80. 2008. View Article : Google Scholar
|
|
18
|
Fan Q-M, Jing Y-Y, Yu GF, Kou XR, Ye F,
Gao L, Li R, Zhao QD, Yang Y, Lu ZH, et al: Tumor-associated
macrophages promote cancer stem cell-like properties via
transforming growth factor-beta1-induced epithelial-mesenchymal
transition in hepatocellular carcinoma. Cancer Lett. 352:160–168.
2014. View Article : Google Scholar : PubMed/NCBI
|
|
19
|
Pollard JW: Tumour-educated macrophages
promote tumour progression and metastasis. Nat Rev Cancer. 4:71–78.
2004. View Article : Google Scholar : PubMed/NCBI
|
|
20
|
Lin EY, Nguyen AV, Russell RG and Pollard
JW: Colony-stimulating factor 1 promotes progression of mammary
tumors to malignancy. J Exp Med. 193:727–740. 2001. View Article : Google Scholar : PubMed/NCBI
|
|
21
|
Laoui D, Van Overmeire E, De Baetselier P,
Van Ginderachter JA and Raes G: Functional relationship between
tumor-associated macrophages and macrophage colony-stimulating
factor as contributors to cancer progression. Front Immunol.
5:4892014. View Article : Google Scholar : PubMed/NCBI
|
|
22
|
Robinson SC, Scott KA, Wilson JL, Thompson
RG, Proudfoot AE and Balkwill FR: A chemokine receptor antagonist
inhibits experimental breast tumor growth. Cancer Res.
63:8360–8365. 2003.PubMed/NCBI
|
|
23
|
Whitehurst B, Flister MJ, Bagaitkar J,
Volk L, Bivens CM, Pickett B, Castro-Rivera E, Brekken RA, Gerard
RD and Ran S: Anti-VEGF-A therapy reduces lymphatic vessel density
and expression of VEGFR-3 in an orthotopic breast tumor model. Int
J Cancer. 121:2181–2191. 2007. View Article : Google Scholar : PubMed/NCBI
|
|
24
|
Dineen SP, Lynn KD, Holloway SE, Miller
AF, Sullivan JP, Shames DS, Beck AW, Barnett CC, Fleming JB and
Brekken RA: Vascular endothelial growth factor receptor 2 mediates
macrophage infiltration into orthotopic pancreatic tumors in mice.
Cancer Res. 68:4340–4346. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
25
|
Rauh MJ, Sly LM, Kalesnikoff J, Hughes MR,
Cao LP, Lam V and Krystal G: The role of SHIP1 in macrophage
programming and activation. Biochem Soc Trans. 32:785–788. 2004.
View Article : Google Scholar : PubMed/NCBI
|
|
26
|
Torisu-Itakura H, Lee JH, Huynh Y, Ye X,
Essner R and Morton DL: Monocyte-derived IL-10 expression predicts
prognosis of stage IV melanoma patients. J Immunother. 30:831–838.
2007. View Article : Google Scholar : PubMed/NCBI
|
|
27
|
Kuang DM, Zhao Q, Peng C, Xu J, Zhang JP,
Wu C and Zheng L: Activated monocytes in peritumoral stroma of
hepatocellular carcinoma foster immune privilege and disease
progression through PD-L1. J Exp Med. 206:1327–1337. 2009.
View Article : Google Scholar : PubMed/NCBI
|
|
28
|
Xia Y, Wang Y, Yang J and Song K: Study on
effects of Bufei Decoction on α-SMA in lung tissue of pulmonary
fibrosis rats. Zhonghua Zhongyiyao Xuekan. 23:1916–1918. 2014.In
Chinese.
|
|
29
|
Zhang H, Xia YL, Yang J and Song K: Study
on effects of Bufei Decoction on MMP-9 and TIMP-1 in lung tissue of
pulmonary fibrosis rats. Zhonghua Zhongyiyao Xuekan. 29:2673–2679.
2011.
|
|
30
|
Du X, Du X, Zhang K, Wang J, Guo G and
Wang R: The clinical trials to observe the effect of Astragalus
injection combine with chemotherapy in patients with malignant
tumor. Shanghai Med Pharm J. 2:162010.
|
|
31
|
Chiu PY, Lam PY, Yan CW and Ko KM:
Schisandrin B protects against solar irradiation-induced oxidative
injury in BJ human fibroblasts. Fitoterapia. 82:682–691. 2011.
View Article : Google Scholar : PubMed/NCBI
|
|
32
|
Chi D and Huang Q: The effect of Fructus
schisandrae B element to vascular endothelial growth factor in
human glioma. PhD thesis. Southern Medical University; Guangzhou:
2008, In Chinese.
|
|
33
|
Reagan-Shaw S, Nihal M and Ahmad N: Dose
translation from animal to human studies revisited. FASEB J.
22:659–661. 2008. View Article : Google Scholar
|
|
34
|
Geng L, Huang D, Liu J, Qian Y, Deng J, Li
D, Hu Z, Zhang J, Jiang G and Zheng S: B7-H1 up-regulated
expression in human pancreatic carcinoma tissue associates with
tumor progression. J Cancer Res Clin Oncol. 134:1021–1027. 2008.
View Article : Google Scholar : PubMed/NCBI
|
|
35
|
Ostuni R, Kratochvill F, Murray PJ and
Natoli G: Macrophages and cancer: From mechanisms to therapeutic
implications. Trends Immunol. 36:229–239. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
36
|
Chen C, Shen Y, Qu QX, Chen XQ, Zhang XG
and Huang JA: Induced expression of B7-H3 on the lung cancer cells
and macrophages suppresses T-cell mediating anti-tumor immune
response. Exp Cell Res. 319:96–102. 2013. View Article : Google Scholar
|
|
37
|
Lei H, Ju DW, Yu Y, Tao Q, Chen G, Gu S,
Hamada H and Cao X: Induction of potent antitumor response by
vaccination with tumor lysate-pulsed macrophages engineered to
secrete macrophage colony-stimulating factor and interferon-γ. Gene
Ther. 7:707–713. 2000. View Article : Google Scholar : PubMed/NCBI
|
|
38
|
Salazar-Onfray F: Interleukin-10: A
cytokine used by tumors to escape immunosurveillance. Med Oncol.
16:86–94. 1999. View Article : Google Scholar : PubMed/NCBI
|
|
39
|
Wang R, Lu M, Zhang J, Chen S, Luo X, Qin
Y and Chen H: Increased IL-10 mRNA expression in tumor-associated
macrophage correlated with late stage of lung cancer. J Exp Clin
Cancer Res. 30:622011. View Article : Google Scholar : PubMed/NCBI
|
|
40
|
Lee SJ, Jang BC, Lee SW, Yang YI, Suh SI,
Park YM, Oh S, Shin JG, Yao S, Chen L, et al: Interferon regulatory
factor-1 is prerequisite to the constitutive expression and
IFN-gamma-induced upregulation of B7-H1 (CD274). FEBS Lett.
580:755–762. 2006. View Article : Google Scholar : PubMed/NCBI
|
|
41
|
Dong H, Strome SE, Salomao DR, Tamura H,
Hirano F, Flies DB, Roche PC, Lu J, Zhu G, Tamada K, et al:
Tumor-associated B7-H1 promotes T-cell apoptosis: A potential
mechanism of immune evasion. Nat Med. 8:793–800. 2002. View Article : Google Scholar : PubMed/NCBI
|
|
42
|
Barber DL, Wherry EJ, Masopust D, Zhu B,
Allison JP, Sharpe AH, Freeman GJ and Ahmed R: Restoring function
in exhausted CD8 T cells during chronic viral infection. Nature.
439:682–687. 2006. View Article : Google Scholar
|
|
43
|
Chen J, Li G, Meng H, Fan Y, Song Y, Wang
S, Zhu F, Guo C, Zhang L and Shi Y: Upregulation of B7-H1
expression is associated with macrophage infiltration in
hepatocellular carcinomas. Cancer Immunol Immunother. 61:101–108.
2012. View Article : Google Scholar
|
|
44
|
Bloch O, Crane CA, Kaur R, Safaee M,
Rutkowski MJ and Parsa AT: Gliomas promote immunosuppression
through induction of B7-H1 expression in tumor-associated
macrophages. Clin Cancer Res. 19:3165–3175. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
45
|
Velcheti V, Schalper KA, Carvajal DE,
Anagnostou VK, Syrigos KN, Sznol M, Herbst RS, Gettinger SN, Chen
LP and Rimm DL: Programmed death ligand-1 expression in non-small
cell lung cancer. Lab Invest. 94:107–116. 2014. View Article : Google Scholar
|
|
46
|
Chanmee T, Ontong P, Konno K and Itano N:
Tumor-associated macrophages as major players in the tumor
microenvironment. Cancers (Basel). 6:1670–1690. 2014. View Article : Google Scholar
|
|
47
|
Hatanaka H, Abe Y, Kamiya T, Morino F,
Nagata J, Tokunaga T, Oshika Y, Suemizu H, Kijima H, Tsuchida T, et
al: Clinical implications of interleukin (IL)-10 induced by
non-small-cell lung cancer. Ann Oncol. 11:815–819. 2000. View Article : Google Scholar : PubMed/NCBI
|
|
48
|
Jin M, Zhao K, Huang Q and Shang P:
Structural features and biological activities of the
polysaccharides from Astragalus membranaceus. Int J Biol Macromol.
64:257–266. 2014. View Article : Google Scholar
|
|
49
|
Li W, Sun YN, Yan XT, Yang SY, Song SB,
Lee YM and Kim YH: NF-κB inhibitory activity of sucrose fatty acid
esters and related constituents from Astragalus membranaceus. J
Agric Food Chem. 61:7081–7088. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
50
|
Chen X, May B, Di YM, Zhang AL, Lu C, Xue
CC and Lin L: Oral Chinese herbal medicine combined with
pharmacotherapy for stable COPD: A systematic review of effect on
BODE index and six minute walk test. PLoS One. 9:e918302014.
View Article : Google Scholar : PubMed/NCBI
|
|
51
|
Wang T, Sun Y, Jin L, Xu Y, Wang L, Ren T
and Wang K: Enhancement of non-specific immune response in sea
cucumber (Apostichopus japonicus) by Astragalus membranaceus and
its polysaccharides. Fish Shellfish Immunol. 27:757–762. 2009.
View Article : Google Scholar : PubMed/NCBI
|