Introduction
Colorectal cancer is one of the most common cancer
types worldwide and the second most lethal cancer in the USA
(1); its incidence has been
increasing in Asian countries, including South Korea, and it has
become a severe public health problem worldwide (2–4).
Although surgery remains the most effective treatment for patients
with colorectal cancer, the majority of patients experience relapse
within 5 years following complete surgical resection (5–7).
Given the heterogeneous properties of colorectal cancer tumors,
patients with colorectal cancer have a poor overall survival in
response to treatment (8,9). In addition, similar histopathological
tumors may elicit considerably different clinical courses (10,11).
Therefore, for improved treatment and management of patients with
colorectal cancer, knowledge of each cancer property that is
associated with the differential responses to drug treatment is
imperative for predicting patient outcomes and developing novel
therapeutic targets may be beneficial.
Resveratrol (3,5,4′-trihydroxystilbene; RSV) is a
polyphe-nolic compound that naturally occurs in grapes, peanuts and
berries (12). Considerable
research has been conducted to determine the benefits of RSV
against various human cancer types (12–16).
RSV has anti-proliferative properties against numerous cancer
types, including those of the liver, breast, prostate, lung and
colorectum (13,17–22).
RSV induces apoptosis and inhibits cell growth, cell cycle
progression, migration and invasion (19,22).
RSV has antitumor effects in colorectal cancer by inhibiting
various carcinogenic processes (21,23–26).
Gong et al (27)
demonstrated that RSV inhibited colorectal cancer proliferation by
suppressing cyclooxygenase-2 (COX-2) expression. Karimi Dermani
et al (28) reported that
RSV inhibited proliferation, invasion and epithelial-mesenchymal
transition by increasing miR-200c expression in HCT-116 colorectal
cancer cells. Furthermore, sirtuin 1 is required for RSV-mediated
chemopreventive effects in colorectal cancer cells (29). Hence, RSV serves an important role
in colorectal cancer. However, the precise molecular mechanism
underlying the effect of RSV on colorectal cancer has not been
clearly understood.
Tristetraprolin (TTP) is an AU-rich element
(Res)-binding protein that regulates mRNA stability (30) and is a key protein involved in
regulating cytokine expression (31). Decreased TTP expression is observed
in patients with colorectal, lung, breast and pancreatic cancer,
and TTP dysfunction serves as an important indicator of cancer
development (32–37). TTP suppresses the growth of human
colon cancer cells by regulating vascular endothelial growth factor
expression (38). Recent studies
have addressed the association between RSV and TTP in cancer. Ryu
et al (39) reported that
RSV induced apoptosis by activating TTP in glioma cells, whereas
RSV inhibited MCF-7 cell proliferation by upregulating TTP
(31). However, the precise
association between TTP and RSV in colorectal cancer has not been
completely understood. The present study reports for the first
time, to the best of our knowledge, that RSV induced apoptosis in
colorectal cancer cells by activating TTP. RSV-induced TTP
expression inhibited baculoviral IAP repeat containing 3 (cIAP2),
large tumor suppressor kinase 2 (LATS2), E2F transcription factor 1
(E2F1), and lin-28 homolog A (Lin28) and MDM2 proto-oncogene (MDM2)
mRNAs by binding to their 3′ untranslated regions (3′UTRs).
Furthermore, RSV-induced TTP upregulation inhibited the cell
proliferation and metastasis of human colorectal cells. The results
of the present study may increase understanding of the antitumor
effects of RSV and suggest future applications for RSV as an
anticancer agent in clinical cancer therapy.
Materials and methods
Cell lines and reagents
Colorectal cancer cells (HCT116 and SNU81) were
purchased from the Korean Cell Line Bank (Seoul, Korea) and
maintained in RPMI-1640 medium supplemented with 10% fetal bovine
serum (FBS) and 1% penicillin-streptomycin (all Capricorn
Scientific GmbH, Ebsdorfergrund, Germany) at 37°C in a humidified
5% CO2 atmosphere. RSV was purchased from Sigma-Aldrich
(Merck KGaA, Darmstadt, Germany; cat. no. 274666) and suspended in
dimethyl sulfoxide (DMSO; cat. no. D2650; Sigma-Aldrich; Merck
KGaA). Colorectal cancer cells were treated with RSV in medium
containing 2.5% FBS for 24 h at 37°C.
Plasmid construction and luciferase
assay
The pGL3/TTPp-1411 promoter construct (40) and variable target gene of TTP in
the psiCHECK2 luciferase reporter constructs [psiCHECK2/cIAP2
(41), E2F1 (42), LATS2 (43), Lin28 (44) and MDM2 3′UTR constructs] were as
previously described. Cells were transfected with various types of
plasmid constructs using iN-fect™ in vitro transfection
reagent (Intron Biotechnology, Inc., Seongnam, Korea). For the
luciferase assays, HCT116 and SNU81 cells were seeded
1×105 cells/well in a 12-well plate and transfected with
the constructs (500 ng) for 24 h, and treated with RSV (5, 10 and
20 µM) for 24 h. Transfected cells were lysed with a lysis
buffer (Promega Corporation, Madison, WI, USA) and mixed with the
luciferase assay reagent (Promega Corporation). The
chemiluminescent signal was measured using the Wallac Victor 1420
multilabel counter (PerkinElmer Inc., Waltham, MA, USA). In each
sample, the firefly luciferase activity was normalized to the
Renilla luciferase activity. All luciferase assays reported
herein represent at least three independent experiments, each
comprising three wells per transfection.
SDS-PAGE and immunoblotting
The total protein was extracted from HCT116 and
SNU81 cells using ice-cold radio-immunoprecipitation assay buffer
[50 mM Tris HCl, pH 7.4; 150 mM NaCl; 1 mM EDTA; 1% (v/v) Triton
X-100; 0.1% (w/v) SDS] and a protease inhibitor cocktail (Roche
Diagnostics GmbH, Mannheim, Germany). Protein concentrations were
determined using the bicinchoninic acid assay, according to the
manufacturer's protocol (Thermo Fisher Scientific, Inc., Waltham,
MA, USA). The protein samples (10 µg) were loaded onto a 10%
SDS-PAGE gel, and transferred onto Hybond-P membranes (GE
Healthcare Bio-Sciences, Pittsburgh, PA, USA). Membranes were
blocked using 5% skim milk for 1 h at room temperature and probed
using appropriate dilutions of rabbit anti-human TTP (cat. no.
T5327; Sigma-Aldrich; Merck KGaA; 1:3,000) and anti-β-actin (cat.
no. A2228, Sigma-Aldrich; Merck KGaA; 1:3,000) antibodies overnight
at 4°C. Secondary antibody rabbit-IgG (cat. no. ADI-SAB-300; Enzo
Life Sciences, Inc., Farmingdale, NY, USA; 1:5,000) was probed for
90 min at room temperature. Immunoreactivity was determined using
an enhanced chemiluminescence detection system (GE Healthcare
Bio-Sciences). Films were exposed at multiple time points to ensure
that the images were not saturated. ImageJ (v.1.51j8; National
Institutes of Health, Bethesda, MD, USA) was used to analyze the
blot images.
Reverse transcription-quantitative
polymerase chain reaction (RT-qPCR)
RNA isolation was performed using TRIzol reagent
(Thermo Fisher Scientific, Inc.) and was synthesized to cDNA using
Moloney murine leukemia virus reverse transcriptase kit (cat. no.
3201; Beams Biotechnology, Seongnam, Korea) and Oligo-dT primer
(cat. no. 79237, Qiagen, Hilden, Germany), according to
manufacturer's protocol for 60 min at 37°C. For the PCR (5
µl), the cDNA product, PCR master mix (cat. no. RT500;
Enzynomics, Inc., Daejeon, Korea), and a Bio-Rad system (CFX96
Optics Module; Bio-Rad Laboratories, Inc., Hercules, CA, USA) were
used while monitoring, in real-time, the increase in the
fluorescence of the SYBR Green dye (cat. RT500, Enzynomics Co.,
Ltd., Daejeon, Korea). The specificity of each primer pair was
confirmed by melting curve analysis (45,46).
The PCR primer pairs used were as follows: TTP, CGCTACAAGACTGAGCTAT
and GAGGTAGAACTT GTGACAGA; β-actin, CCCTGGAGAAGAGCTACGAG and
AGGTAGTTTCGTGGATGCCA. PCR cycling conditions were 94°C for 10 min
to activate the DNA polymerase, followed by 40 cycles of 94°C for
15 sec, 60°C for 30 sec, and 72°C for 30 sec.
MTT assay
For the MTT assay, 1×104 cells/well were
seeded in 96-well culture plates with complete RPMI-1640 medium.
The cells were subsequently incubated with different RSV
concentrations (10, 20 or 50 µM) for up to 72 h. The culture
medium was removed, and the cells were incubated with MTT (5 mg/ml;
Sigma-Aldrich; Merck KGaA) for 90 min at 37°C. Following incubation
for 90 min, the MTT solution was removed, and the formazan product
was extracted and diluted with DMSO (cat. no. D2650; Sigma-Aldrich;
Merck KGaA) with gentle agitation for 15 min. The absorbance was
measured using the VICTOR3 Multilabel Reader 1420 (PerkinElmer) at
490 nm. Three independent experiments were performed in four
duplicated wells.
Migration and invasion assay
The effect of RSV on the invasive properties of
HCT116 and SNU81 cells was assessed using Boyden chambers (Neuro
Probe, Inc., Gaithersburg, MD, USA) that were precoated with
Matrigel (BD Biosciences, Franklin Lakes, NJ, USA) and incubated
for 1 h at room temperature. The bottom wells were filled with 28
µl medium with 2% serum. In total, 1×105 cells/56
µl were seeded into the upper compartment and incubated for
24 h at 37°C and 5% CO2. Following incubation for 24 h,
the cells attached to the upper surface of the filter were removed
using a cotton swab, and those attached to the lower surface were
stained using Diff-Quik reagents (Sysmex Corporation, Kobe, Japan)
for 3 min at room temperature and counted (five fields/well). The
invasion percentage was expressed as the percentage of invading
cells through the Matrigel. A representative graph of six
independent experiments is presented. Images from each well were
immediately captured using the Axiovert 40 CFL inverted
fluorescence microscope (Carl Zeiss AG, Oberkochen, Germany) in
five random fields at ×40 magnification. For the migration assay,
the membrane was pre-coated with collagen (cat. no. C7661;
Sigma-Aldrich; Merck KGaA) and 10% acetic acid for 1 h at room
temperature.
Clonogenic assay
HCT116 and SNU81 cells were seeded into 12-well
plates at 1×104 cells/well and incubated for 24 h at
37°C and 5% CO2. The cells were subsequently treated
with RSV in a dose-dependent manner and incubated for 10 days at
37°C and 5% CO2. Fresh medium containing RSV (5, 10 and
20 µM) was added on the 3rd day. On the 10th day, the medium
was removed from the plates, and the cells were washed once with
PBS. The colonies were fixed and stained with methanol (25% v/v)
that contained crystal violet (0.05% w/v) for 30 min at room
temperature. Thereafter, the residual staining solution was
removed, and the plates were washed with water. When the plates had
been rinsed three times with PBS and air-dried, the colonies were
counted using ImageJ.
siRNA transfection and treatments
TTP small interfering RNA (siRNA; cat. no. sc-36760)
or control siRNA (cat. no. sc-37007; both Santa Cruz Biotechnology,
Inc., Dallas, TX, USA) was transfected (100 nM) into HCT116 and
SNU81 cells using Lipofectamine® 2000 (Invitrogen;
Thermo Fisher Scientific, Inc.), according to the manufacturer's
protocol. After 24 h, cells were treated with RSV (20 µM)
for 24 h.
Microarray
The mirVana™ miRNA isolation labeling kit (Ambion;
Thermo Fisher Scientific, Inc.) was used for isolating total RNA
from the HCT116 cells at 90% confluence, according to the
manufacturer's protocol. The Illumina Total Prep RNA amplification
kit (Illumina, Inc., San Diego, CA, USA) was used for hybridization
with biotin-labeled cRNA. Illumina Human-12 BeadChip V.4 microarray
(Illumina, Inc.) was used for hybridizing samples, and data were
extracted using the Genome Studio (Illumina, Inc.). R software
(R-3.50; http://crans.us.r-project.org) was used for data
analysis, and the Cluster 3.0 (Eisen Lab; University of California
Berkeley, Berkeley, CA, USA) and Treeview (Eisen Lab; University of
California Berkeley) software packages were used for generating the
gene expression heat map.
Statistics
GraphPad Prism 5.0 (GraphPad Software, Inc., La
Jolla, CA, USA) was used for all statistical analyses. Data are
presented as the mean ± standard deviation. Comparisons among the
groups were performed by paired Student's t-test and a two-way
analysis of variance with Duncan's multiple range test. P<0.05
was considered to indicate a statistically significant
difference.
Results
RSV suppresses colon cancer cell
proliferation
To investigate the effect of RSV on colorectal
cancer progression, MTT and clonogenic assays were performed. The
MTT assay demonstrated that treatment with RSV had a dose-dependent
inhibitory effect on HCT116 and SNU81 cell viability (Fig. 1A). The clonogenic assay
additionally demonstrated that treatment with RSV significantly
inhibited HCT116 and SNU81 cell proliferation in a dose-dependent
manner (Fig. 1B). Therefore, RSV
inhibited the progression and proliferation of colorectal cancer
cells.
RSV suppresses the invasive and migratory
effects of colon cancer cells
The effect of RSV on the migration and invasion of
colorectal cancer cells was also investigated. RSV-treated HCT116
and SNU81 cells were assessed using Matrigel invasion and collagen
migration assays. RSV significantly inhibited the invasive ability
of colorectal cancer cells in a dose-dependent manner (Fig. 2A). The migration assay also
confirmed that RSV significantly inhibited the migratory ability of
colorectal cancer cells in dose-dependent manner (Fig. 2B). Therefore, these results
suggested that RSV inhibited the migration and invasion of
colorectal cancer cells.
Microarray data reveal an RSV-induced
increase in TTP gene expression
To investigate the effect of RSV on gene expression
levels in colorectal cancer cells, the microarray experiment was
performed. RSV administration significantly regulated several genes
associated with inflammation, proliferation, cell death,
angiogenesis and metastasis (Fig.
3). Increased ZFP36 (TTP) gene expression was observed in
RSV-treated HCT116 cells. In addition, RSV decreased the expression
of the oncogenes Myc proto-oncogene (Myc), KRAS proto-oncogene
GTPase (KRAS) and Fos proto-oncogene AP-1 transcription factor
subunit (FOS), and the downstream target genes of TTP, including
E2F1. Therefore, RSV-induced TTP upregulation may inhibit the
growth and metastasis of colorectal cancer cells.
 | Figure 3Microarray heat map demonstrating the
effect of RSV on genes associated with inflammation, proliferation,
cell death, angiogenesis and metastasis in HCT116 cells. Data are
presented in matrix format with rows indicating the individual gene
and columns indicating each sample. Red and green represent
upregulated and downregulated gene expression levels, respectively,
as indicated in the scale bar (log 2 transformed). RSV,
resveratrol; Cont, control; ZFP36, ZFP36 ring finger protein; TNF,
tumor necrosis factor; IL, interleukin; MIP, major intrinsic
protein of lens fiber; PTGS, prostaglandin-endoperoxide synthase;
CCND1, cyclin D1; MYC, Myc proto-oncogene; KRAS, KRAS
proto-oncogene GTPase; FOS, Fos proto-oncogene AP-1 transcription
factor subunit; E2F transcription factor 1; AHRR, aryl-hydrocarbon
receptor repressor; BCL2, apoptosis regulator Bcl-2; TRAF2, TNF
receptor associated factor 2; VEGF, vascular endothelial growth
factor; NOS1, nitric oxide synthase 1; LIN28, lin-28 homolog A;
LIN28B, lin-28 homolog B. |
RSV induces the mRNA and protein
expression of TTP
The present study further determined the endogenous
mRNA and protein expression levels of TTP in HCT116 and SNU81 cells
using RT-qPCR and western blot analysis (Fig. 4A). Endogenous mRNA and protein
expression levels of TTP were increased in HCT116 and SNU81 cells
(Fig. 4A). In addition, siTTP
decreased the mRNA and protein levels of TTP in HCT116 and SNU81
cells (Fig. 4A). To determine
whether RSV regulated TTP expression in colorectal cancer cells,
HCT116 and SNU81 cells were treated with 0, 10, 20 and 50 µM
RSV. RSV significantly increased the mRNA and protein expression
levels of TTP in HCT116 and SNU81 cells in a dose-dependent manner
(Fig. 4B). In particular, >20
µM RSV significantly increased TTP expression in colorectal
cancer cells (Fig. 4B). To further
test whether RSV regulates TTP expression, HCT116 and SNU81 cells
were transfected with scramble RNA or siTTP (100 nM). It was
observed that RSV restored TTP mRNA expression in HCT116 and SNU81
cells whose TTP expression was reduced by siTTP (Fig. 4C). Treatment with RSV reversed the
inhibition of TTP expression. In addition, siRNA-induced TTP
inhibition attenuated the effects of RSV on the cell growth of
HCT116 and SNU81 cells (Fig. 4D).
Therefore, these data indicate that TTP mediated the apoptotic
effects of RSV in colorectal cancer cells and that RSV induced TTP
expression in colorectal cancer cells.
 | Figure 4RSV induces TTP expression in
colorectal cancer cells. (A) Endogenous mRNA and protein expression
levels of TTP in colon cancer cells (HCT116 and SNU81).
*P<0.05. (B) RSV induced the mRNA and protein
expression of TTP in a dose-dependent manner in HCT116 and SNU81
cells. β-actin was detected as the loading control for RT-qPCR and
western blotting. The relative protein expression level was
calculated as a ratio of the control (DMSO), using ImageJ software
to detect the intensity of the protein band. **P<0.01
vs. DMSO. (C) RSV restored TTP expression in cell lines whose TTP
expression was reduced by TTP siRNA. HCT116 and SNU81 cells were
transfected with scRNA or TTP siRNA (100 nM). After 24 h, DMSO or
RSV (20 µM) was added to the cells, followed by RNA
extraction after 24 h. β-actin was detected as the loading control
for RT-qPCR. Each bar represents the mean ± SD of three independent
experiments. **P<0.01. (D) Cell survival was assessed
using the MTT assay. Cell viability relative to that of the control
is expressed as the mean ± SD of three independent experiments.
***P<0.001. TTP, tristetraprolin; RSV, resveratrol;
RT-qPCR, reverse transcription-quantitative polymerase chain
reaction; si, small interfering; DMSO, dimethyl sulfoxide; ns, not
significant; SD, standard deviation; sc, scramble. |
RSV controls the downstream target gene
of TTP
The present study further assessed whether RSV
increased TTP promoter activity in a reporter assay. HCT116 and
SNU81 cells were transiently transfected with a pGL3/hTTPp-1411
construct, followed by treatment with RSV. Following treatment with
RSV for 24 h, the luciferase activity was measured, revealing
significantly increased TTP promoter activity in HCT116 and SNU81
cells (Fig. 5A). Given a previous
finding (41–44) that TTP reduced cIAP2, E2F1, LATS2
and Lin28 expression, and inhibited cancer cell growth, the present
study subsequently investigated whether RSV regulates the promoter
activities of cIAP2, E2F1, LATS2 and Lin28, which bind with TTP in
colorectal cancer cells. HCT116 and SNU81 cells were transfected
with luciferase reporter constructs that incorporated the 3′UTRs of
cIAP2, E2F1, LATS2 and Lin28 mRNA (psiCHECK2/cIAP2, psiCHECK2/E2F1,
psiCHECK2/LATS2 and psiCHECK2/Lin28). Each transfected cell was
treated with RSV (20 µM), and the luciferase activity was
measured 24 h post-treatment. Consistent with previous studies, the
induction of TTP expression reduced the luciferase activities of
cIAP2, E2F1, LATS2 and Lin28 in HCT116 and SNU81 cells (Fig. 5B). RSV enhanced the inhibitory
activity of TTP on target genes in HCT116 and SNU81 cells (Fig. 5B). In addition, siTTP increased
mRNA and protein levels of E2F1 in HCT116 and SNU81 cells (Fig. 5C). Therefore, our results indicated
that RSV induces TTP expression and its target gene mRNA-decaying
activity in colorectal cancer cells.
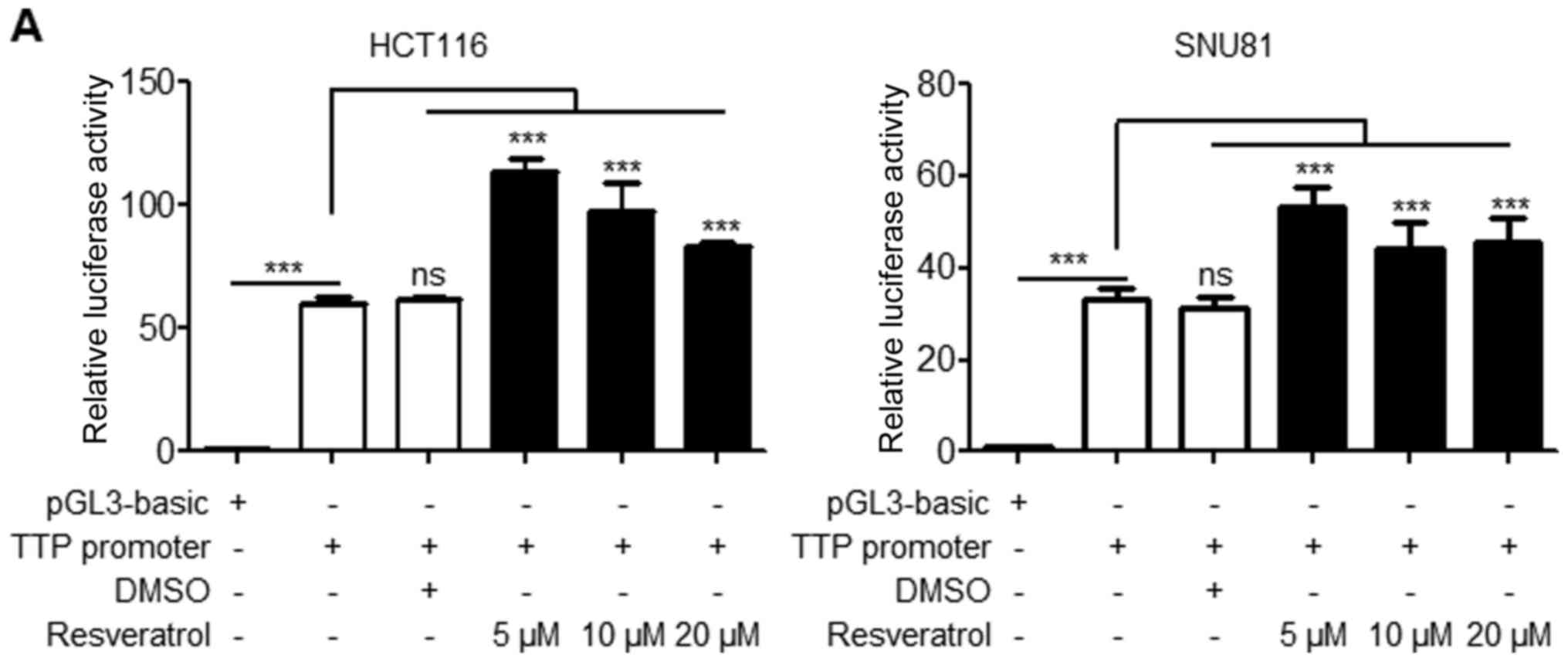 | Figure 5RSV controls the downstream target
genes of TTP. (A) HCT116 and SNU81 cells were transfected with
pGL3/TTPp-1411 containing the TTP promoter (−1343 to +68) for 24 h
and treated with RSV at increasing doses for 24 h. Following
treatment with RSV for 24 h, the luciferase activity was measured.
The expression levels obtained from pGL3-basic (empty
vector)-transfected cells without treatment with RSV were set to 1.
Each bar represents the mean ± SD of three independent experiments.
***P<0.001. (B) RSV induced the mRNA-decaying
activity of TTP. HCT116 and SNU81 cells were transfected with
psiCHECK2-cIAP2, LATS2, Lin28 and E2F1 3′UTRs. At 24 h
post-transfection, cells were treated with 20 µM RSV for 24
h, followed by determination of the luciferase activity. The
Renilla luciferase activity was normalized to the firefly
luciferase activity. The luciferase activity obtained from
psiCHECK2-cIAP2, LATS2, Lin28, and E2F1 3′UTR-transfected and
DMSO-treated cells was set to 1. Each bar represents the mean ±
standard deviation of three independent experiments.
***P<0.001 vs. respective control. (C) TTP inhibits
E2F1 expression in colorectal cancer cells. siTTP increased the
mRNA and protein expression levels of E2F1 in HCT116 and SNU81
cells. Each bar represents the mean ± standard deviation of three
independent experiments. *P<0.05 vs. respective
control. TTP, tristetraprolin; RSV, resveratrol; DMSO, dimethyl
sulfoxide; ns, not significant; SD, standard deviation; cIAP2,
baculoviral IAP repeat containing 3; LATS2, large tumor suppressor
kinase 2; Lin28, lin-28 homolog A; E2F1, E2F transcription factor
1;si, small interfering; sc, scramble. |
Discussion
Despite the fact that RSV has been studied in
various human cancers, its biological effects on colorectal cancer
have not been fully elucidated. The present study aimed to
investigate the effects of RSV in human colorectal cancer cells and
to elucidate its effect on regulating TTP expression. Although TTP
serves key roles in cancer cells, TTP regulation by RSV in
colorectal cancer cells is yet to be clearly investigated. The
present study demonstrated that RSV inhibited cell proliferation
and invasion/metastasis by activating TTP in human colorectal
cancer cells.
Phytochemicals are promising therapeutic agents for
cancer treatment. RSV, a natural compound occurring in grapes,
peanuts and berries, has anticancer properties against a number of
types of cancer, including colorectal cancer (28,47,48).
RSV induces cellular apoptosis, and decreases migration and
invasion, by regulating a number of mechanisms in colorectal cancer
cells (28,29,47,48).
Du et al (47) accordingly
reported that the inhibitory effect of RSV on colorectal cancer
cells was mediated by the hedgehog/GLI family zinc finger 1
signaling pathways. Feng et al (48) demonstrated that treatment with RSV
inhibits proliferation and induces apoptosis in human colon cancer
cells. Furthermore, Buhrmann et al (29) demonstrated that RSV suppresses the
tumorigenesis of colorectal cancer cells by targeting sirtuin 1 and
suppressing nuclear factor-κB activation. In addition, RSV has been
comprehensively investigated with respect to its role in cell cycle
regulation in colorectal cancer. RSV upregulated cyclin dependent
kinase inhibitor 1A, causing cell-cycle arrest at the
G0/G1 and G2/M phases, and the
activation of the caspase-dependent cyclin/cyclin dependent kinase
pathway in colon cancer cells (49,50).
Therefore, RSV appears to have antitumor properties in colorectal
cancer cells by targeting various signaling pathways. Consistent
with previous studies, the present results indicated that RSV
inhibited the viability of HCT116 and SNU81 cells in a dose- and
time-dependent manner. RSV also significantly inhibited the colony
formation of HCT116 and SNU81 cells in a dose-dependent manner.
These observations are in accordance with previous studies
(28,29,47,48)
that demonstrated that RSV negatively regulates the growth of
colorectal cancer cells. In addition, RSV significantly reduced the
invasion and metastasis of colorectal cancer cells (HCT116 and
SNU81 cells) in a dose-dependent manner. These results regarding
RSV regulation of cell mobility and adhesion, and the inhibition of
invasion and metastasis in colorectal cancer cells, were consistent
with those of previous studies (28,29,47,48).
An increasing number of studies have validated the
importance of TTP expression in human cancer (51–54).
The loss of TTP gene expression has been reported in various cancer
types (51,53), and reduced TTP gene expression has
been demonstrated to lead to cancer development (32,33,55).
Therefore, TTP overexpression may be a novel strategy for cancer
prevention. TTP also serves an important role in a number of cancer
types, including colon cancer. TTP inhibits IL-23 expression
(56) and downregulates vascular
endothelial growth factor (VEGF) and COX-2 expression in human
colon cancer (38,57). Accordingly, TTP expression may be
controlled via the aforementioned signaling pathways. Therefore,
examining whether RSV regulates TTP expression in colorectal cancer
cells is essential. To investigate the association between RSV and
TTP gene expression in colorectal cancer cells (HCT116), gene
expression profiling was performed. The microarray experiment
demonstrated that RSV significantly increased TTP expression.
Furthermore, RSV regulated genes associated with inflammation, cell
proliferation, cell death, angiogenesis and metastasis, and
suppressed Myc, KRAS, and FOS gene expression. The microarray
experiment also indicated that RSV suppressed E2F1, a downstream
target gene of TTP. Consistent with the microarray data, the
RT-qPCR data indicated that RSV increased TTP expression in HCT116
and SNU81 cells in a dose-dependent manner. Similar results were
also found with respect to the western blot analysis, wherein RSV
administration dose-dependently increased TTP protein expression.
Furthermore, RSV restored TTP mRNA expression following TTP
silencing in HCT116 and SNU81 cells. siRNA-induced TTP inhibition
attenuated the effects of RSV on cell growth. These results
suggested that TTP may be involved in the effect of RSV on the
inhibition of human colorectal cancer cell growth. RSV inhibited
HCT116 and SNU81 cell proliferation by upregulating TTP. The
present observations are in accordance with earlier studies wherein
RSV increased TTP expression in glioma and breast cancer cells
(31,39). RSV was demonstrated to inhibit
MCF-7 cell proliferation by upregulating TTP via COX-2 and VEGF
downregulation, and inducible nitric oxide synthase upregulation
(31). Furthermore, RSV increased
TTP expression to induce glioma cell apoptosis in U87MG human
glioma cells (39). These results
suggested that RSV upregulates TTP expression in colorectal cancer
cells.
Given that TTP gene silencing triggers cancer
development, it may be hypothesized that the loss of TTP function
in cancer cells may induce transcriptional silencing through TTP
promoter regulation. The present study indicated that RSV increased
the mRNA and protein expression levels of TTP in human colorectal
cancer cells. Further studies regarding whether RSV regulates TTP
promoter activity in colorectal cancer cells demonstrated that RSV
significantly increased TTP promoter activity in HCT116 and SNU81
cells. Furthermore, RSV significantly inhibited the promoter
activities of cIAP2, E2F1, LATS2 and Lin28, which are downstream
target genes of TTP in HCT116 and SNU81 cells. RSV enhanced the TTP
inhibitory activity in HCT116 and SNU81 cells by negatively
regulating cIAP2, E2F1, LATS2 and Lin28 expression. These findings
are in accordance with earlier studies wherein TTP overexpression
suppressed the stability of E2F1 and Lin28 mRNA (42,57),
and controlled the stability of cIAP2 and LATS2 mRNA by binding to
the 3′UTR of cIAP2 mRNA or promoting let-7 biogenesis (38,43),
demonstrating that cIAP2, E2F1, LATS2, and Lin28 may be
physiological targets of TTP. In addition, in agreement with
previous studies, it was demonstrated that siTTP significantly
increased the mRNA and protein expression levels of E2F1 in HCT116
and SNU81 cells. Likewise, other studies have reported that TTP
inhibits the expression of LIN28, cIAP and LATS2 in human cancer
cells (40,41,43,44).
Therefore, it may be inferred that RSV suppresses the viability of
colorectal cancer cells by regulating the stability of LIN28, cIAP
and LATS2 mRNA, mediated via TTP regulation. In conclusion, the
results of the present study suggested RSV inhibits the
proliferation and invasion/metastasis of colorectal cancer cells by
upregulating TTP, which is associated with the downregulation of
TTP target genes, including cIAP2, E2F1, LATS2 and Lin28.
Acknowledgments
Not applicable.
References
|
1
|
Arnold M, Sierra MS, Laversanne M,
Soerjomataram I, Jemal A and Bray F: Global patterns and trends in
colorectal cancer incidence and mortality. Gut. 66:683–691. 2017.
View Article : Google Scholar
|
|
2
|
Pan R, Zhu M, Yu C, Lv J, Guo Y, Bian Z,
Yang L, Chen Y, Hu Z, Chen Z, et al China Kadoorie Biobank
Collaborative Group: Cancer incidence and mortality: A cohort study
in China, 2008-2013. Int J Cancer. 141:1315–1323. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
3
|
Chen W, Zheng R, Zhang S, Zeng H, Xia C,
Zuo T, Yang Z, Zou X and He J: Cancer incidence and mortality in
China, 2013. Cancer Lett. 401:63–71. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
4
|
Shin A, Jung KW and Won YJ: Colorectal
cancer mortality in Hong Kong of China, Japan, South Korea, and
Singapore. World J Gastroenterol. 19:979–983. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
5
|
Fitzgerald TL, Biswas T, O'Brien K, Zervos
EE and Wong JH: Neoadjuvant radiotherapy for rectal cancer:
Adherence to evidence-based guidelines in clinical practice. World
J Surg. 37:639–645. 2013. View Article : Google Scholar
|
|
6
|
Murray DM, Katz ML, Post DM, Pennell ML,
Young GS, Tatum CM and Paskett ED: Enhancing cancer screening in
primary care: Rationale, design, analysis plan, and recruitment
results. Contemp Clin Trials. 34:356–363. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
7
|
Halabi WJ, Kang CY, Jafari MD, Nguyen VQ,
Carmichael JC, Mills S, Stamos MJ and Pigazzi A: Robotic-assisted
colorectal surgery in the United States: A nationwide analysis of
trends and outcomes. World J Surg. 37:2782–2790. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
8
|
Lu YW and Wang KH: Research progress on
genetic heterogeneity between primary and paired metastatic
colorectal cancer. Yi Chuan. 39:482–490. 2017.PubMed/NCBI
|
|
9
|
Kopetz S, Chang GJ, Overman MJ, Eng C,
Sargent DJ, Larson DW, Grothey A, Vauthey JN, Nagorney DM and
McWilliams RR: Improved survival in metastatic colorectal cancer is
associated with adoption of hepatic resection and improved
chemotherapy. J Clin Oncol. 27:3677–3683. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
10
|
Loree JM and Kopetz S: Recent developments
in the treatment of metastatic colorectal cancer. Ther Adv Med
Oncol. 9:551–564. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
11
|
Midgley RS, Yanagisawa Y and Kerr DJ:
Evolution of nonsurgical therapy for colorectal cancer. Nat Clin
Pract Gastroenterol Hepatol. 6:108–120. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
12
|
McCubrey JA, Lertpiriyapong K, Steelman
LS, Abrams SL, Yang LV, Murata RM, Rosalen PL, Scalisi A, Neri LM,
Cocco L, et al: Effects of resveratrol, curcumin, berberine and
other nutra-ceuticals on aging, cancer development, cancer stem
cells and microRNAs. Aging (Albany NY). 9:1477–1536. 2017.
|
|
13
|
de Lima E Silva TC, da Silveira LTR,
Fragoso MF, da Silva FRM, Martinez MF, Zapaterini JR, Diniz OHG,
Scarano WR and Barbisan LF: Maternal resveratrol treatment reduces
the risk of mammary carcinogenesis in female offspring prenatally
exposure to 2,3,7,8-tetrachlorodibenzo-p-dioxin. Horm Cancer.
8:286–297. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
14
|
Perez-Vizcaino F and Fraga CG: Research
trends in flavonoids and health. Arch Biochem Biophys. 646:107–112.
2018. View Article : Google Scholar : PubMed/NCBI
|
|
15
|
Darwish MA, Abo-Youssef AM, Khalaf MM,
Abo-Saif AA, Saleh IG and Abdelghany TM: Resveratrol influences
platinum pharmacokinetics: A novel mechanism in protection against
cisplatin-induced nephrotoxicity. Toxicol Lett. 290:73–82. 2018.
View Article : Google Scholar : PubMed/NCBI
|
|
16
|
Kiskova T, Demeckova V, Jendzelovska Z,
Kiktava M, Venglovska K, Bohmdorfer M, Jager W and Thalhammer T:
Nocturnal resveratrol administration inhibits chemically induced
breast cancer formation in rats. J Physiol Pharmacol. 68:867–875.
2017.
|
|
17
|
Guthrie AR, Chow HS and Martinez JA:
Effects of resveratrol on drug- and carcinogen-metabolizing
enzymes, implications for cancer prevention. Pharmacol Res
Perspect. 5:e002942017. View
Article : Google Scholar : PubMed/NCBI
|
|
18
|
Bartolacci C, Andreani C, Amici A and
Marchini C: Walking a tightrope: A perspective of resveratrol
effects on breast cancer. Curr Protein Pept Sci. 19:311–322. 2018.
View Article : Google Scholar
|
|
19
|
Rauf A, Imran M, Butt MS, Nadeem M, Peters
DG and Mubarak MS: Resveratrol as an anti-cancer agent: A review.
Crit Rev Food Sci Nutr. 58:1428–1447. 2018. View Article : Google Scholar
|
|
20
|
Feng Y, Zhou J and Jiang Y: Resveratrol in
lung cancer- a systematic review. J BUON. 21:950–953.
2016.PubMed/NCBI
|
|
21
|
Yin TF, Wang M, Qing Y, Lin YM and Wu D:
Research progress on chemopreventive effects of phytochemicals on
colorectal cancer and their mechanisms. World J Gastroenterol.
22:7058–7068. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
22
|
Li C, Xu X, Tao Z, Sun C and Pan Y:
Resveratrol derivatives: An updated patent review (2012–2015).
Expert Opin Ther Pat. 1–12. 2016.
|
|
23
|
Li YH, Niu YB, Sun Y, Zhang F, Liu CX, Fan
L and Mei QB: Role of phytochemicals in colorectal cancer
prevention. World J Gastroenterol. 21:9262–9272. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
24
|
Fajardo AM and Piazza GA: Chemoprevention
in gastrointestinal physiology and disease. Anti-inflammatory
approaches for colorectal cancer chemoprevention. Am J Physiol
Gastrointest Liver Physiol. 309:G59–G70. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
25
|
Carter LG, D'Orazio JA and Pearson KJ:
Resveratrol and cancer: Focus on in vivo evidence. Endocr Relat
Cancer. 21:R209–R225. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
26
|
Juan ME, Alfaras I and Planas JM:
Colorectal cancer chemoprevention by trans-resveratrol. Pharmacol
Res. 65:584–591. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
27
|
Gong WH, Zhao N, Zhang ZM, Zhang YX, Yan L
and Li JB: The inhibitory effect of resveratrol on COX-2 expression
in human colorectal cancer: A promising therapeutic strategy. Eur
Rev Med Pharmacol Sci. 21:1136–1143. 2017.PubMed/NCBI
|
|
28
|
Karimi Dermani F, Saidijam M, Amini R,
Mahdavinezhad A, Heydari K and Najafi R: Resveratrol inhibits
proliferation, invasion, and epithelial-mesenchymal transition by
increasing miR-200c expression in HCT-116 colorectal cancer cells.
J Cell Biochem. 118:1547–1555. 2017. View Article : Google Scholar
|
|
29
|
Buhrmann C, Shayan P, Popper B, Goel A and
Shakibaei M: Sirt1 is required for resveratrol-mediated
chemopreventive effects in colorectal cancer cells. Nutrients.
8:1452016. View Article : Google Scholar : PubMed/NCBI
|
|
30
|
Baou M, Jewell A and Murphy JJ: TIS11
family proteins and their roles in posttranscriptional gene
regulation. J Biomed Biotechnol. 2009:6345202009. View Article : Google Scholar : PubMed/NCBI
|
|
31
|
Li C, Tang C and He G: Tristetraprolin: A
novel mediator of the anticancer properties of resveratrol. Genet
Mol Res. 15:152016.
|
|
32
|
Deng K, Wang H, Shan T, Chen Y, Zhou H,
Zhao Q and Xia J: Tristetraprolin inhibits gastric cancer
progression through suppression of IL-33. Sci Rep. 6:245052016.
View Article : Google Scholar : PubMed/NCBI
|
|
33
|
Pandiri I, Chen Y, Joe Y, Kim HJ, Park J,
Chung HT and Park JW: Tristetraprolin mediates the
anti-proliferative effects of metformin in breast cancer cells.
Breast Cancer Res Treat. 156:57–64. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
34
|
Wei ZR, Liang C, Feng D, Cheng YJ, Wang
WM, Yang DJ, Wang YX and Cai QP: Low tristetraprolin expression
promotes cell proliferation and predicts poor patients outcome in
pancreatic cancer. Oncotarget. 7:17737–17750. 2016.PubMed/NCBI
|
|
35
|
Xu L, Ning H, Gu L, Wang Q, Lu W, Peng H,
Cui W, Ying B, Ross CR, Wilson GM, et al: Tristetraprolin induces
cell cycle arrest in breast tumor cells through targeting
AP-1/c-Jun and NF-κB pathway. Oncotarget. 6:41679–41691. 2015.
View Article : Google Scholar : PubMed/NCBI
|
|
36
|
Sobolewski C, Sanduja S, Blanco FF, Hu L
and Dixon DA: Histone deacetylase inhibitors activate
tristetraprolin expression through induction of early growth
response protein 1 (EGR1) in colorectal cancer cells. Biomolecules.
5:2035–2055. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
37
|
Kang S, Min A, Im SA, Song SH, Kim SG, Kim
HA, Kim HJ, Oh DY, Jong HS, Kim TY, et al: TGF-β suppresses COX-2
expression by tristetraprolin-mediated RNA destabilization in A549
human lung cancer cells. Cancer Res Treat. 47:101–109. 2015.
View Article : Google Scholar
|
|
38
|
Lee HH, Son YJ, Lee WH, Park YW, Chae SW,
Cho WJ, Kim YM, Choi HJ, Choi DH, Jung SW, et al: Tristetraprolin
regulates expression of VEGF and tumorigenesis in human colon
cancer. Int J Cancer. 126:1817–1827. 2010. View Article : Google Scholar
|
|
39
|
Ryu J, Yoon NA, Seong H, Jeong JY, Kang S,
Park N, Choi J, Lee DH, Roh GS, Kim HJ, et al: Resveratrol induces
glioma cell apoptosis through activation of tristetraprolin. Mol
Cells. 38:991–997. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
40
|
Lee JY, Kim HJ, Yoon NA, Lee WH, Min YJ,
Ko BK, Lee BJ, Lee A, Cha HJ, Cho WJ, et al: Tumor suppressor p53
plays a key role in induction of both tristetraprolin and let-7 in
human cancer cells. Nucleic Acids Res. 41:5614–5625. 2013.
View Article : Google Scholar : PubMed/NCBI
|
|
41
|
Kim CW, Kim HK, Vo MT, Lee HH, Kim HJ, Min
YJ, Cho WJ and Park JW: Tristetraprolin controls the stability of
cIAP2 mRNA through binding to the 3′UTR of cIAP2 mRNA. Biochem
Biophys Res Commun. 400:46–52. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
42
|
Lee HH, Lee SR and Leem SH:
Tristetraprolin regulates prostate cancer cell growth through
suppression of E2F1. J Microbiol Biotechnol. 24:287–294. 2014.
View Article : Google Scholar
|
|
43
|
Lee HH, Vo MT, Kim HJ, Lee UH, Kim CW, Kim
HK, Ko MS, Lee WH, Cha SJ, Min YJ, et al: Stability of the LATS2
tumor suppressor gene is regulated by tristetraprolin. J Biol Chem.
285:17329–17337. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
44
|
Kim CW, Vo MT, Kim HK, Lee HH, Yoon NA,
Lee BJ, Min YJ, Joo WD, Cha HJ, Park JW, et al: Ectopic
over-expression of tristet-raprolin in human cancer cells promotes
biogenesis of let-7 by down-regulation of Lin28. Nucleic Acids Res.
40:3856–3869. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
45
|
Li W, Matsuoka M, Kai M, Thapa P, Khadge
S, Hagge DA, Brennan PJ and Vissa V: Real-time PCR and
high-resolution melt analysis for rapid detection of Mycobacterium
leprae drug resistance mutations and strain types. J Clin
Microbiol. 50:742–753. 2012. View Article : Google Scholar :
|
|
46
|
Ashrafi R, Bruneaux M, Sundberg LR,
Pulkkinen K and Ketola T: Application of high resolution melting
assay (HRM) to study temperature-dependent intraspecific
competition in a pathogenic bacterium. Sci Rep. 7:9802017.
View Article : Google Scholar : PubMed/NCBI
|
|
47
|
Du Z, Zhou F, Jia Z, Zheng B, Han S, Cheng
J, Zhu G and Huang P: The hedgehog/Gli-1 signaling pathways is
involved in the inhibitory effect of resveratrol on human
colorectal cancer HCT116 cells. Iran J Basic Med Sci. 19:1171–1176.
2016.PubMed/NCBI
|
|
48
|
Feng M, Zhong LX, Zhan ZY, Huang ZH and
Xiong JP: Resveratrol treatment inhibits proliferation of and
induces apoptosis in human colon cancer cells. Med Sci Monit.
22:1101–1108. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
49
|
Ali I and Braun DP: Resveratrol enhances
mitomycin C-mediated suppression of human colorectal cancer cell
proliferation by up- regulation of p21WAF1/CIP1. Anticancer Res.
34:5439–5446. 2014.PubMed/NCBI
|
|
50
|
Liu B, Zhou Z, Zhou W, Liu J, Zhang Q, Xia
J, Liu J, Chen N, Li M and Zhu R: Resveratrol inhibits
proliferation in human colorectal carcinoma cells by inducing
G1/S-phase cell cycle arrest and apoptosis through
caspase/cyclin-CDK pathways. Mol Med Rep. 10:1697–1702. 2014.
View Article : Google Scholar : PubMed/NCBI
|
|
51
|
Guo J, Qu H, Chen Y and Xia J: The role of
RNA-binding protein tristetraprolin in cancer and immunity. Med
Oncol. 34:1962017. View Article : Google Scholar : PubMed/NCBI
|
|
52
|
Guo J, Wang H, Jiang S, Xia J and Jin S:
The Cross-talk between tristetraprolin and cytokines in cancer.
Anticancer Agents Med Chem. 17:1477–1486. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
53
|
Wang Q, Ning H, Peng H, Wei L, Hou R, Hoft
DF and Liu J: Tristetraprolin inhibits macrophage IL-27-induced
activation of antitumour cytotoxic T cell responses. Nat Commun.
8:8672017. View Article : Google Scholar : PubMed/NCBI
|
|
54
|
Vo MT, Choi SH, Lee JH, Hong CH, Kim JS,
Lee UH, Chung HM, Lee BJ, Park JW and Cho WJ: Tristetraprolin
inhibits mitochondrial function through suppression of α-Synuclein
expression in cancer cells. Oncotarget. 8:41903–41920. 2017.
View Article : Google Scholar : PubMed/NCBI
|
|
55
|
Zeng B, Zhu D, Su Z, Li Z and Yu Z:
Tristetraprolin exerts tumor suppressive functions on the
tumorigenesis of glioma by targeting IL-13. Int Immunopharmacol.
39:63–70. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
56
|
Lee HH, Yang SS, Vo MT, Cho WJ, Lee BJ,
Leem SH, Lee SH, Cha HJ and Park JW: Tristetraprolin down-regulates
IL-23 expression in colon cancer cells. Mol Cells. 36:571–576.
2013. View Article : Google Scholar : PubMed/NCBI
|
|
57
|
Cha HJ, Lee HH, Chae SW, Cho WJ, Kim YM,
Choi HJ, Choi DH, Jung SW, Min YJ, Lee BJ, et al: Tristetraprolin
downregulates the expression of both VEGF and COX-2 in human colon
cancer. Hepatogastroenterology. 58:790–795. 2011.PubMed/NCBI
|



















