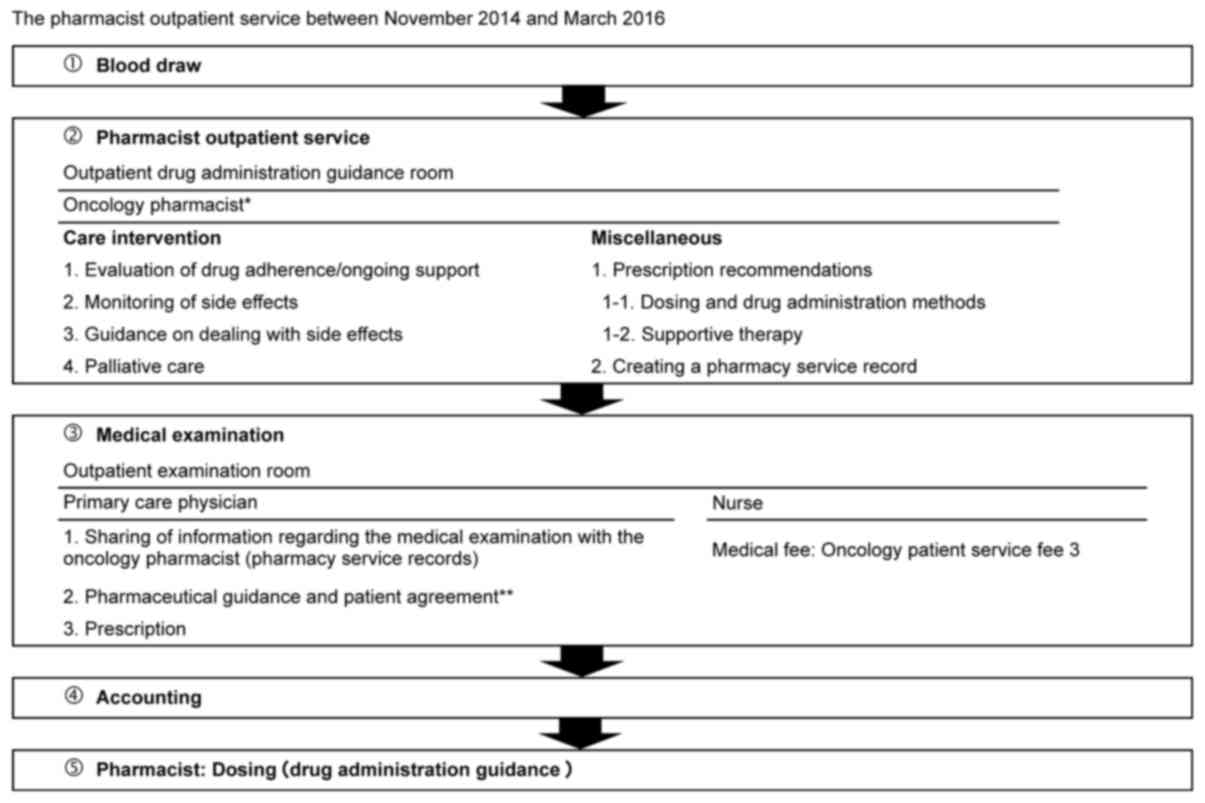
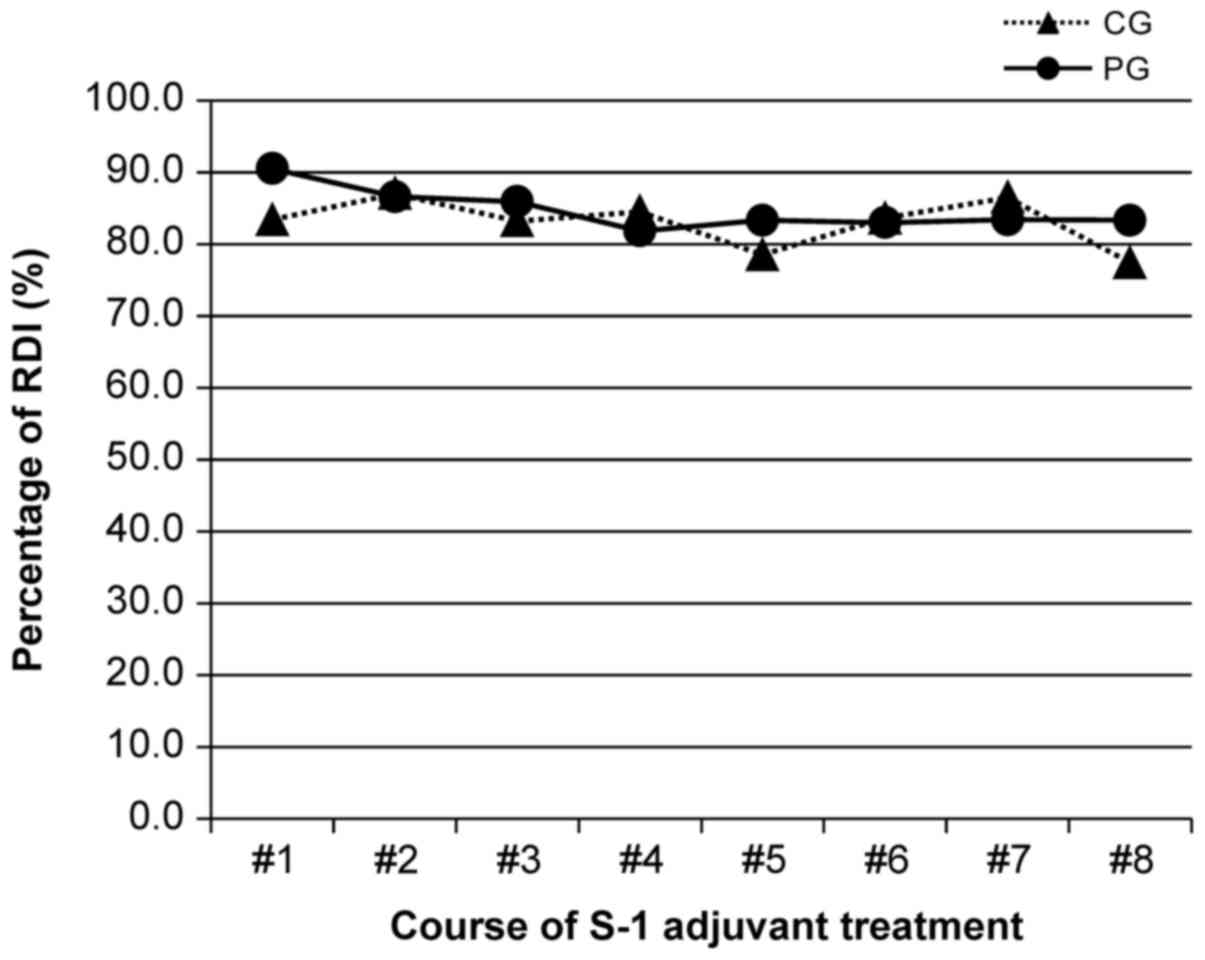
|
1
|
Sakuramoto S, Sasako M, Yamaguchi T,
Kinoshita T, Fujii M, Nashimoto A, Furukawa H, Nakajima T, Ohashi
Y, Imamura H, et al: Adjuvant chemotherapy for gastric cancer with
S-1, an oral fluoropyrimidine. N Engl J Med. 357:1810–1820. 2007.
View Article : Google Scholar : PubMed/NCBI
|
|
2
|
Kimura M, Usami E, Yoshimura T, Yasuda T,
Kaneoka Y, Teramachi H, Sugiyama T and Tsuchiya T: Pharmaceutical
care for patients undergoing S-1 plus cisplatin therapy for
unresectable recurrent gastric cancer. J Pharm Pract. 26:409–414.
2013. View Article : Google Scholar : PubMed/NCBI
|
|
3
|
Kimura M, Morihata K, Ito D, Iwai M, Okada
K, Usami E, Nakao T, Yoshimura T and Yasuda T: Continuous
administration and safety of S-1 in adjuvant chemotherapy for
gastric cancer. Gan To Kagaku Ryoho. 37:829–834. 2010.(In
Japanese). PubMed/NCBI
|
|
4
|
Iwai M, Kimura M, Yoshimura T and Yasuda
T: The necessity of adverse effect monitoring in patients using
TS-1: A survey of reasons for treatment withdrawal or switching. J
Jpn Soc Hosp Pharm. 44:1386–1388. 2008.(In Japanese).
|
|
5
|
Kimura M, Usami E, Iwai M, Nakao T,
Yoshimura T, Mori H, Sugiyama T and Teramachi H: Oral anticancer
agent medication adherence by outpatients. Oncol Lett. 8:2318–2324.
2014.PubMed/NCBI
|
|
6
|
Kimura M, Nakashima K, Usami E, Iwai M,
Nakao T, Yoshimura T, Mori H and Teramachi H: Adherence and
awareness of the therapeutic intent of oral anticancer agents in an
outpatient setting. Oncol Lett. 9:2341–2346. 2015.PubMed/NCBI
|
|
7
|
Kawabata R, Imamura H, Kishimoto T,
Hachino Y, Yasui Y, Fujino M, Fujii C, Fukunaga M, Ohzato H and
Furukawa H: Examination of factors influencing continuity of S-1
adjuvant chemotherapy for gastric cancer patients. Gan To Kagaku
Ryoho. 39:1205–1208. 2012.(In Japanese). PubMed/NCBI
|
|
8
|
Imamura M, Nakura H and Takemoto C:
Evaluation of usefulness of pharmaceutical outpatient clinic for
cancer patients. Jpn J Pharm Health Care Sci. 36:85–98. 2010.(In
Japanese). View Article : Google Scholar
|
|
9
|
Nakashima K, Mano Y, Ohuchi K, Sato D,
Iwata K, Higuchi A, Ebara K, Kato Y, Hirosawa I, Tjima M, et al:
Role of pharmaceutical outpatient clinic in cancer patients and
evaluation. Jpn J Pharm Health Care Sci. 38:599–608. 2012.(In
Japanese). View Article : Google Scholar
|
|
10
|
Aimono Y, Nemoto M, Sato W, Saito Y,
Aoyama Y, Joko F, Maruyama T and Kamoshida T: Examination of the
usefulness of the pharmacists' outpatient clinic for treatment with
oral molecule-targeting drugs. Gan To Kagaku Ryoho. 40:901–905.
2013.(In Japanese). PubMed/NCBI
|
|
11
|
MacLeod A, Branch A, Cassidy J, McDonald
A, Mohammed N and MacDonald L: A nurse-/pharmacy-led capecitabine
clinic for colorectal cancer: Results of a prospective audit and
retrospective survey of patient experiences. Eur J Oncol Nurs.
11:247–254. 2007. View Article : Google Scholar : PubMed/NCBI
|
|
12
|
Wong SF, Bounthavong M, Nguyen C,
Bechtoldt K and Hernandez E: Implementation and preliminary
outcomes of a comprehensive oral chemotherapy management clinic. Am
J Health Syst Pharm. 71:960–965. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
13
|
Simons S, Ringsdorf S, Braun M, Mey UJ,
Schwindt PF, Ko YD, Schmidt-Wolf I, Kuhn W and Jaehde U: Enhancing
adherence to capecitabine chemotherapy by means of
multidisciplinary pharmaceutical care. Support Care Cancer.
19:1009–1018. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
14
|
Arakawa-Todo M, Yoshizawa T, Zennami K,
Nishikawa G, Kato Y, Kobayashi I, Kajikawa K, Yamada Y, Matsuura K,
Tsukiyama I, et al: Management of adverse events in patients with
metastatic renal cell carcinoma treated with sunitinib and clinical
outcomes. Anticancer Res. 33:5043–5050. 2013.PubMed/NCBI
|
|
15
|
Shah NN, Casella E, Capozzi D, McGettigan
S, Gangadhar TC, Schuchter L and Myers JS: Improving the safety of
oral chemotherapy at an academic medical center. J Oncol Pract.
12:e71–e77. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
16
|
Kimura M, Go M, Iwai M, Usami E, Teramachi
H and Yoshimura T: Evaluation of the role and usefulness of a
pharmacist outpatient service for patients undergoing monotherapy
with oral anti-cancer agents. J Oncol Pharm Pract. Jun
20–2016.(Epub ahead of print). View Article : Google Scholar
|
|
17
|
US Department Of Health And Human
Services: Common terminology criteria for adverse events (CTCAE)
version 4.0. United States, National Cancer Institute, .
2009.http://www.acrin.org/Portals/0/Administration/Regulatory/CTCAE_4.02_2009-09-15_QuickReference_5X7.pdf
|
|
18
|
Matsumoto H, Hirai T, Hirabayashi Y,
Murakami H, Higashida M, Kawabe Y, Fuchimoto M, Fujikura H, Hato S,
Urakami A, et al: Pharmacokinetics of 5-FU after S-1 oral
administration for adjuvant chemotherapy in gastric cancer
patients. Gan To Kagaku Ryoho. 34:869–873. 2007.(In Japanese).
PubMed/NCBI
|
|
19
|
van Groeningen CJ, Pinedo HM, Heddes J,
Kok RM, de Jong AP, Wattel E, Peters GJ and Lankelma J:
Pharmacokinetics of 5-fluouracil assessed with a sensitive mass
spectrometric method in patient on a dose escalation schedule.
Cancer Res. 48:6956–6961. 1988.PubMed/NCBI
|
|
20
|
Findlay MP, Raynaud F, Cunningham D,
Iveson A, Collins DJ and Leach MO: Measurement of plasma
5-fluorouracil by high-performance liquid chromatography with
comparison of results to tissue drug levels observed using in vivo
19F magnetic resonance spectroscopy in patients on a protracted
venous infusion with or without interferon-alpha. Ann Oncol.
7:47–53. 1996. View Article : Google Scholar : PubMed/NCBI
|
|
21
|
Aslani A, Smith RC, Allen BJ, Pavlakis N
and Levi JA: The predictive value of body protein for
chemotherapy-induced toxicity. Cancer. 88:796–803. 2000. View Article : Google Scholar : PubMed/NCBI
|
|
22
|
Gusella M, Toso S, Ferrazzi E, Ferrari M
and Padrini R: Relationships between body composition parameters
and fluorouracil pharmacokinetics. Br J Clin Pharmacol. 54:131–139.
2002. View Article : Google Scholar : PubMed/NCBI
|
|
23
|
Prado CM, Baracos VE, McCargar LJ,
Mourtzakis M, Mulder KE, Reiman T, Butts CA, Scarfe AG and Sawyer
MB: Body composition as an independent determinant of
5-fluorouracil-based chemotherapy toxicity. Clin Cancer Res.
13:3264–3268. 2007. View Article : Google Scholar : PubMed/NCBI
|
|
24
|
Prado CM, Baracos VE, McCargar LJ, Reiman
T, Mourtzakis M, Tonkin K, Mackey JR, Koski S, Pituskin E and
Sawyer MB: Sarcopenia as a determinant of chemotherapy toxicity and
time to tumor progression in metastatic breast cancer patients
receiving capecitabine treatment. Clin Cancer Res. 15:2920–2926.
2009. View Article : Google Scholar : PubMed/NCBI
|
|
25
|
Tatematsu M, Kuroki R, Hata T, Hata T and
Muro K: Outcomes of support for ambulatory cancer patients through
a collaborative alliance of doctors, nurses, hospital pharmacists
and community pharmacists. Jpn J Pharm Palliat Care Sei. 7:13–19.
2014.(In Japanese).
|
|
26
|
Kishimoto T, Imamura H, Kawabata R, Kimura
Y, Fukunaga M and Ohzato H: Feasibility and outcome of S-1 adjuvant
chemotherapy for patients with gastric cancer treated based on the
liaison-clinical pathway. Gan To Kagaku Ryoho. 40:489–492. 2013.(In
Japanese). PubMed/NCBI
|