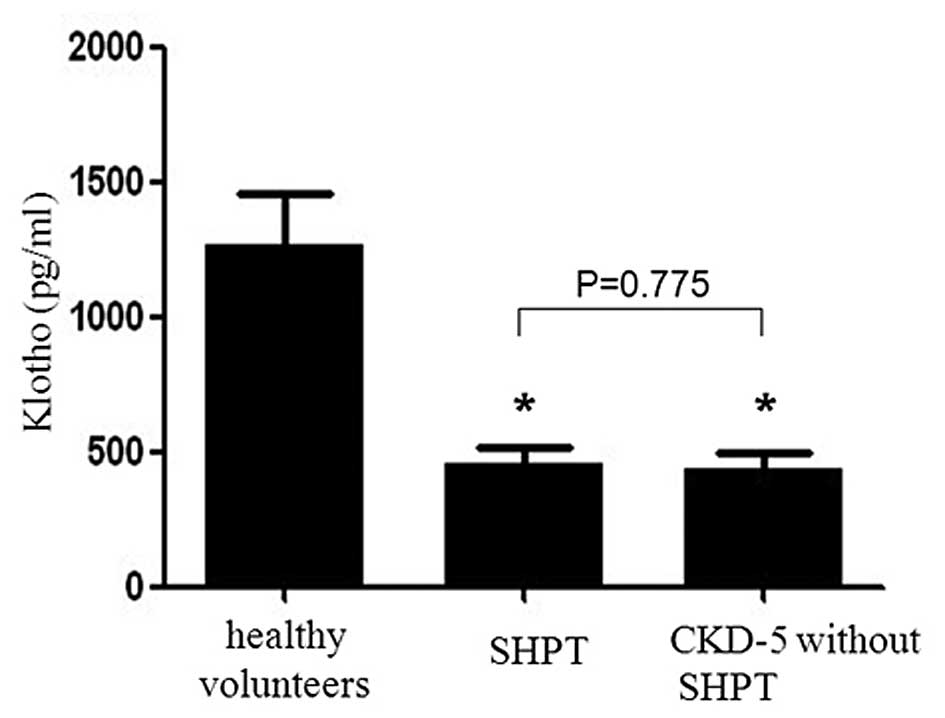
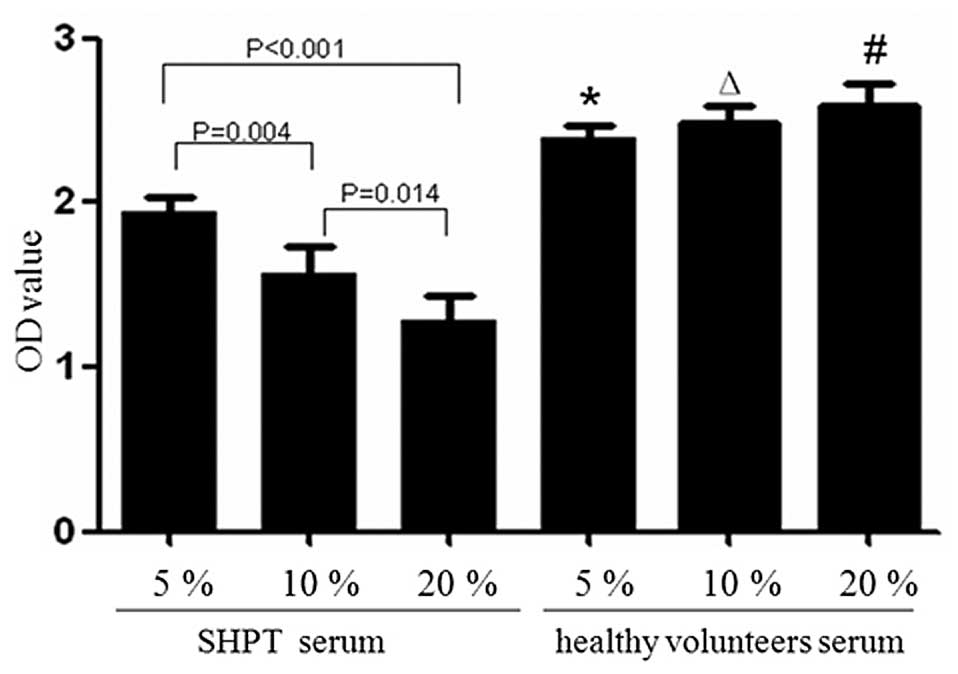
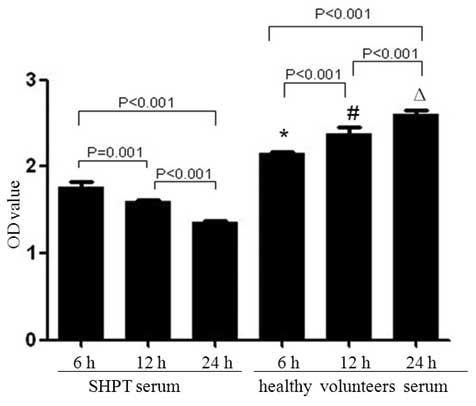
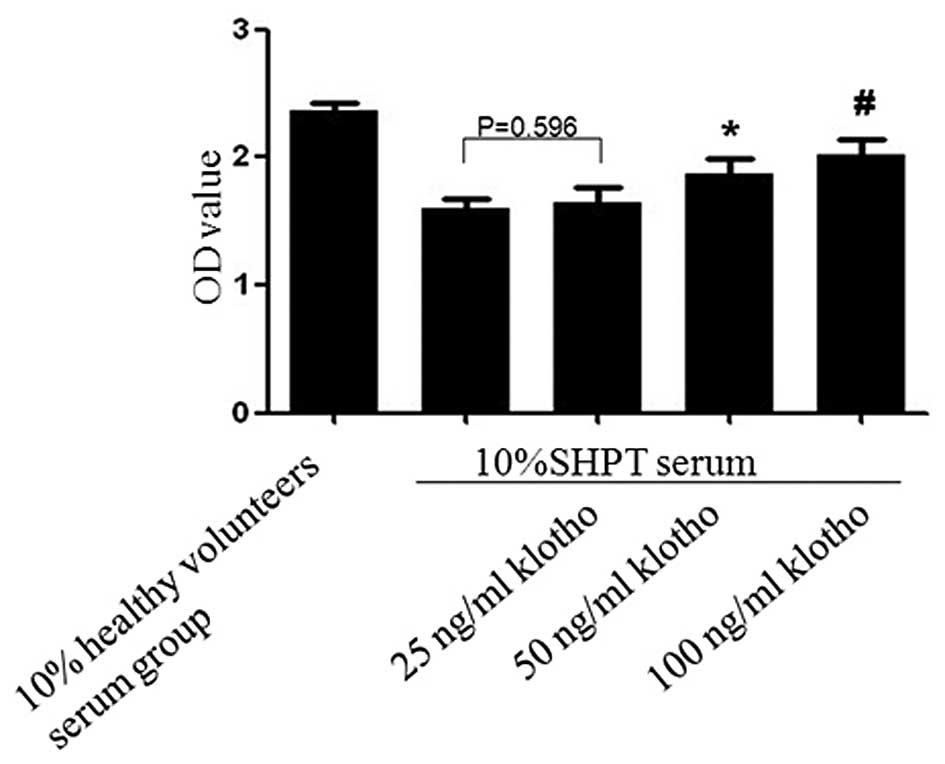
|
1
|
Foley RN, Parfrey PS and Sarnak MJ:
Clinical epidemiology of cardiovascular disease in chronic renal
disease. Am J Kidney Dis. 32(5 Suppl 3): S122–S129. 1998.
View Article : Google Scholar
|
|
2
|
Jourde-chiche N, Dou L, Cerini C, et al:
Vascular incompetence in dialysis patients-protein-bound uremic
toxins and endothelial dysfunction. Semin Dial. 24:327–337. 2011.
View Article : Google Scholar : PubMed/NCBI
|
|
3
|
Van Ballegooijen AJ, Reinders I, Visser M,
et al: Parathyroid hormone and cardiovascular disease events: A
systematic review and meta-analysis of prospective studies. Am
Heart J. 165:655–664. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
4
|
Kuro-o M, Matsumura Y, Aizawa H, et al:
Mutation of the mouse klotho gene leads to a syndrome resembling
aging. Nature. 3909:45–51. 1997. View
Article : Google Scholar
|
|
5
|
Kamemori M, Ohyama Y, Kurabayashi M, et
al: Expression of klotho protein in the inner ear. Hear Res.
171:103–110. 2002. View Article : Google Scholar : PubMed/NCBI
|
|
6
|
Nagai T, Yamada K, Kim HC, et al:
Cognition impairment in the genetic model of aging klotho gene
mutant mice: a role of oxidative stress. FASEB J. 17:50–52.
2003.
|
|
7
|
Anamizu Y, kawaguchi H, Seichi A, et al:
Klotho insufficiency causes decrease of ribosomal RNA gene
transcription activity, cytoplasmic RNA and rough ER in the spinal
anterior horn cells. Acta Neuropathol. 109:457–466. 2005.
View Article : Google Scholar : PubMed/NCBI
|
|
8
|
Hu MC, Shi M, Zhang J, et al: Klotho
deficiency causes vascular calcification in chronic kidney disease.
J Am Soc Nephrol. 22:124–136. 2011. View Article : Google Scholar :
|
|
9
|
Rakugi H, Matsukawa N, Ishikawa K, et al:
Anti-oxidative effect of klotho on endothelial cells through cAMP
activation. Endocrine. 31:82–87. 2007. View Article : Google Scholar : PubMed/NCBI
|
|
10
|
Carracedo J, Buendia P, Merino A, et al:
klotho modulates the stress response in human senescent endothelial
cells. Mech Ageing Dev. 133:647–654. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
11
|
Yang K, Nie L, Huang Y, et al: Ameliration
of uremic toxin indoxyl sulfate-induced endothelial cell
dysfunction by klotho protein. Toxicol Lett. 215:77–83. 2012.
View Article : Google Scholar : PubMed/NCBI
|
|
12
|
Komaba H and Fukagawa M: FGF23-parathyroid
interaction: implications in chronic kidney disease. Kidney Int.
77:292–298. 2010. View Article : Google Scholar
|
|
13
|
Linder A, Charra B and Sherrard DJ:
Accelerated atherosclerosis in prolonged maintenance hemodialysis.
N Eng J Med. 290:697–701. 1974. View Article : Google Scholar
|
|
14
|
Eberhardt RT, Forgione MA, Cap A, et al:
Endothelial dysfunc dysfunction in a murine model of mild
hyperhomocyst(e)inemia. J Clin Invest. 106:483–491. 2000.
View Article : Google Scholar : PubMed/NCBI
|
|
15
|
Raff AC, Meyer TW and Hostetter TH: New
insights into uremic toxicity. Curr Opin Nephrol Hypertens.
17:560–565. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
16
|
Bhuriva R, Li S, Chen SC, et al: Plasma
parathyroid hormone level and prevalent cardiovascular disease in
CKD stages 3 and 4: an analysis from the Kidney Early Evaluation
Program. Am J Kidney Dis. 5(Suppl 4): 3–10. 2009. View Article : Google Scholar
|
|
17
|
de Groot K, Bahlmann FH, Sowa J, et al:
Uremia causes endo-thelial progenitor cell deficiency. Kidney Int.
66:641–646. 2004. View Article : Google Scholar : PubMed/NCBI
|
|
18
|
Choi JH, Kim KL, Huh W, et al: Decreased
number and impaired angiogenic function of endothelial progenitor
cells in patients with chronic renal failure. Arterioscler Thromb
Vasc Biol. 24:1246–1252. 2004. View Article : Google Scholar : PubMed/NCBI
|
|
19
|
Carmeliet P, Moons L, Stassen JM, et al:
Vascular wound healing and neointima formation induced by
perivascular electric injury in mice. Am J Patho. 150:761–776.
1997.
|
|
20
|
Goligorsky MS, Yasuda K and Ratliff B:
Dysfunctional endothelial progenitor cells in chronic kidney
disease. J Am Soc Nephrol. 21:911–919. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
21
|
Matsumura Y, Aizawa H, Nakamura T, et al:
Identification of the human klotho gene and its two transcripts
encoding membrance and secreted klotho protein. Biochem Biophys Res
Commun. 242:626–630. 1998. View Article : Google Scholar : PubMed/NCBI
|
|
22
|
Shiraki-lida T, Aizawa H, Matsumura Y, et
al: Structure of the mouse klotho gene and its two transcripts
encoding membrane and secreted protein. FEBS Lett. 424:6–10.
1998.
|
|
23
|
Tohyma O, Imura A, Iwano A, et al: Klotho
is novel beta-glucuronidase capable of hydrolyzing steroid
beta-glucuronidase. J Biol Chem. 279:9777–9784. 2004. View Article : Google Scholar
|
|
24
|
Kachiwala SJ, Harris SE, Wright AF, et al:
Genetic influences on oxidative stress and their association with
normal cognitive aging. Neurosci Lett. 386:116–120. 2005.
View Article : Google Scholar : PubMed/NCBI
|
|
25
|
Yamamoto M, Clark JD, Pastor JV, et al:
Regulation of oxidative stress by the anti-aging hormone klotho. J
Biol Chem. 280:38029–38034. 2005. View Article : Google Scholar : PubMed/NCBI
|
|
26
|
Jie YU, Deng M, Zhao J, et al: Decreased
expression of klotho gene in uremic atherosclerosis in
apolipoprotein E-deficient mice. Biochem Biophys Res Commun.
391:261–266. 2010. View Article : Google Scholar
|
|
27
|
Kooman JP, Broers NJ, Uswat L, et al: Out
of control: accelerated aging in uremia. Nephrol Dial Transplant.
28:48–54. 2013. View Article : Google Scholar
|
|
28
|
Zou MH, Shi C and Cohen RA: Oxidation of
the zinc-thiolate complex and uncoupling of endothelial nitric
oxide synthase by peroxynitrite. J Clin Invest. 109:817–826. 2002.
View Article : Google Scholar : PubMed/NCBI
|
|
29
|
Moncada S and Higgs EA: Nitric oxide and
the vascular endothelium. Handb Exp Pharmacol. 176(Pt 1): 213–254.
2006.PubMed/NCBI
|
|
30
|
Lubos E, Handy DE and Loscalzo J: Role of
oxidative stress and nitric oxide in atherothrombosis. Front
Biosci. 13:5323–5344. 2008. View
Article : Google Scholar : PubMed/NCBI
|
|
31
|
Lawrence MC, Jivan A, Shao C, et al: The
roles of MAPKs in disease. Cell Res. 18:436–442. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
32
|
Cross TG, Scheel-Toellner D, Henriquez NV,
et al: Serine/threonine protein kinases and apoptosis. Exp Cell
Res. 256:34–41. 2000. View Article : Google Scholar : PubMed/NCBI
|
|
33
|
Pearson G, Robinson F, Beers Gibson T, et
al: Mitogenactivated protein (MAP) kinase pathways: regulation and
physiological functions. Endocr Rev. 22:153–183. 2001.PubMed/NCBI
|
|
34
|
Cobb MH: MAP kinase pathways. Prog Biophys
Mol Biol. 71:479–500. 1999. View Article : Google Scholar : PubMed/NCBI
|
|
35
|
Xia Z, Dickens M, Raingeaud J, et al:
Opposing effects of ERK and JNK-p38 MAP kinases on apoptosis.
Science. 270:1326–1331. 1995. View Article : Google Scholar : PubMed/NCBI
|
|
36
|
Steelman LS, Bertrand FE and McCubrey JA:
The complexity of PTEN: mutation, marker and potential target for
therapeutic intervention. Expert Opin Ther Targets. 8:537–550.
2004. View Article : Google Scholar : PubMed/NCBI
|
|
37
|
Steelman LS, Pohnert SC, Shelton JG, et
al: JAK/STAT, Raf/MEK/ERK, PI3K/Akt and BCR-ABL in cell cycle
progression and leukemogenesis. Leukemia. 18:189–218. 2004.
View Article : Google Scholar : PubMed/NCBI
|