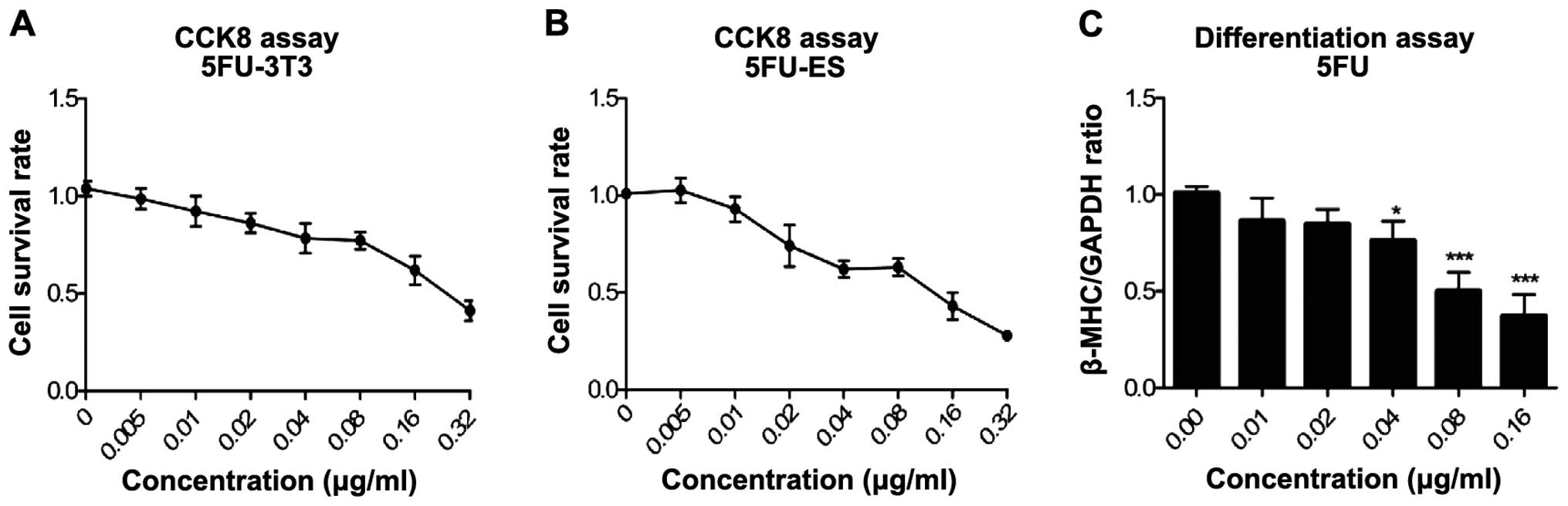
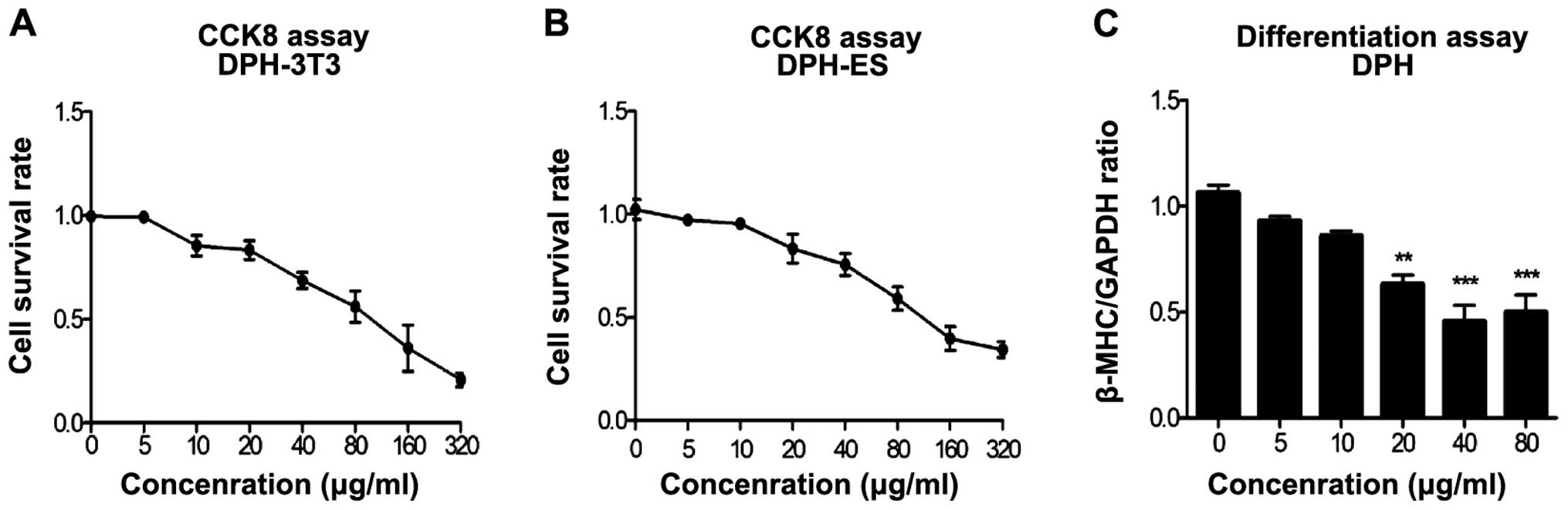
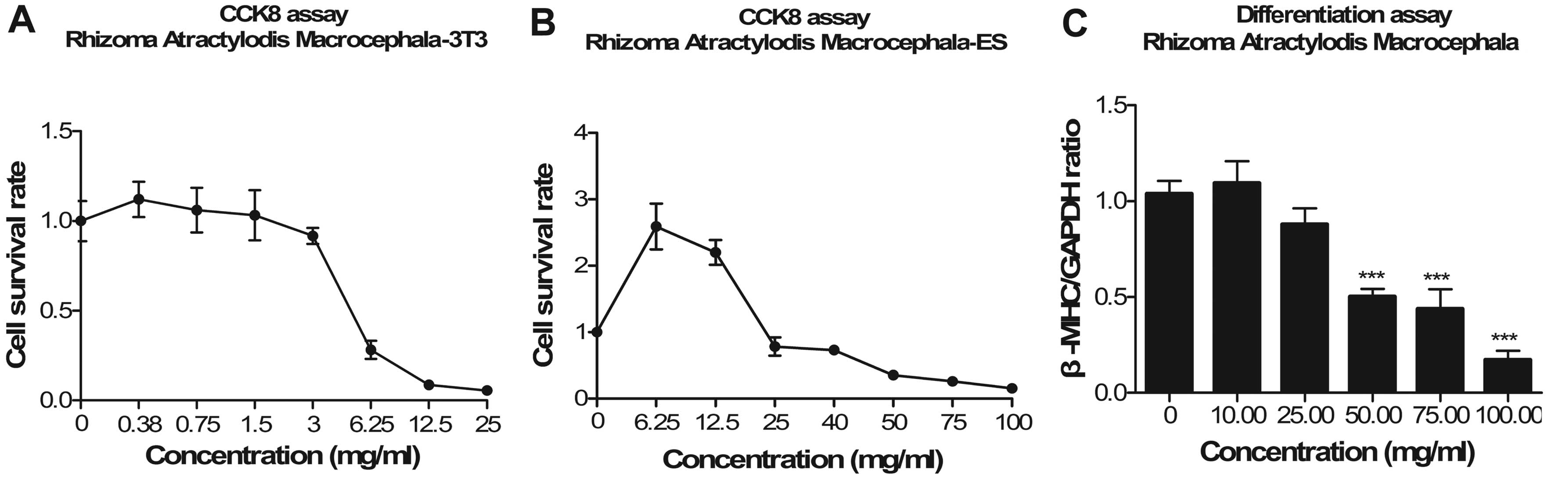
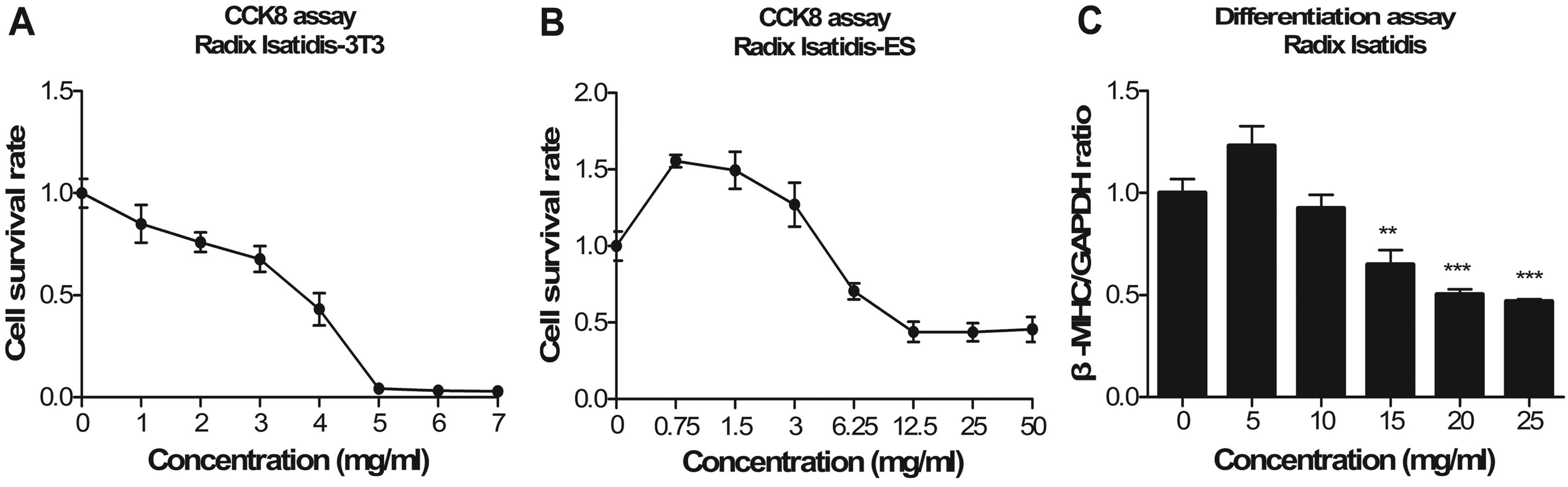
|
1
|
Lu F, Cao M, Wu B, et al: Urinary
metabonomics study on toxicity biomarker discovery in rats treated
with Xanthii Fructus. J Ethnopharmacol. 149:311–320. 2013.
View Article : Google Scholar : PubMed/NCBI
|
|
2
|
Lu F, Gu Q, Wu R and Xu J: A
structure-similarity-based software for the cardiovascular toxicity
prediction of traditional Chinese medicine. Bioinformation.
8:110–113. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
3
|
Zhang ZH, Zhao YY, Cheng XL, et al:
General toxicity of Pinellia ternata (Thunb) Berit in rat: a
metabonomic method for profiling of serum metabolic changes. J
Ethnopharmacol. 149:303–310. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
4
|
Chuang CH, Hsieh WS, Guo YL, et al:
Chinese herbal medicines used in pregnancy: a population-based
survey in Taiwan. Pharmacoepidemiol Drug Saf. 16:464–468. 2007.
View Article : Google Scholar
|
|
5
|
Nordeng H and Havnen GC: Use of herbal
drugs in pregnancy: a survey among 400 Norwegian women.
Pharmacoepidemiol Drug Saf. 13:371–380. 2004. View Article : Google Scholar : PubMed/NCBI
|
|
6
|
Xu CL, Zhao YF, Shang XY and Niu WN: The
effects of supplementing diets with Atractylodes macrocephala Koidz
rhizomes on growth performance and immune function in piglets. J
Animal Feed Sci. 21:302–312. 2012.
|
|
7
|
Jin C, Zhang PJ, Bao CQ, et al: Protective
effects of Atractylodes macrocephala polysaccharide on liver
ischemia-reperfusion injury and its possible mechanism in rats. Am
J Chin Med. 39:489–502. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
8
|
Zheng HZ, Dong ZH and Yu J: Modern Study
of Traditional. Chin Med J. 328–334. 1997.
|
|
9
|
Xiao PG and Lian WY: The Illustrated
Medicinal Plants of China. Shanghai Education Press; Shanghai:
1998
|
|
10
|
Zhou BN: Some progress on the chemistry of
natural bioactive terpenoids from Chinese medicinal plants. Mem
Inst Oswaldo Cruz. 86(Suppl 2): 219–226. 1991. View Article : Google Scholar : PubMed/NCBI
|
|
11
|
Lee MY, Park BY, Kwon OK, et al:
Anti-inflammatory activity of (−)-aptosimon isolated from Daphne
genkwa in RAW264.7 cells. Int Immunopharmacol. 9:878–885. 2009.
View Article : Google Scholar : PubMed/NCBI
|
|
12
|
Zhao Y, Yuan S, Li A, Zhang B and Wang Z:
Effects of processing on toxicity and pharmacological action of
Flos Genkwa. Zhongguo Zhong Yao Za Zhi. 23:344–347. 382–343.
1998.In Chinese.
|
|
13
|
Hong JY, Chung HJ, Lee HJ, Park HJ and Lee
SK: Growth inhibition of human lung cancer cells via
down-regulation of epidermal growth factor receptor signaling by
yuanhuadine, a daphnane diterpene from Daphne genkwa. J Nat Prod.
74:2102–2108. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
14
|
Goel RK, Prabha T, Kumar MM, Dorababu M,
Prakash and Singh G: Teratogenicity of Asparagus racemosus Willd
root, a herbal medicine. Indian J Exp Biol. 44:570–573.
2006.PubMed/NCBI
|
|
15
|
Moallem SA, Ahmadi A, Moshafi M and
Taghavi MM: Teratogenic effects of HESA-A, a natural anticancer
product from Iran, in mice. Hum Exp Toxicol. 30:851–859. 2011.
View Article : Google Scholar
|
|
16
|
Chen CC and Chan WH: Impact effects of
puerarin on mouse embryonic development. Reprod Toxicol.
28:530–535. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
17
|
de Jong E, Barenys M, Hermsen SA, et al:
Comparison of the mouse Embryonic Stem cell Test, the rat Whole
Embryo Culture and the Zebrafish Embryotoxicity Test as alternative
methods for developmental toxicity testing of six 1,2,4-triazoles.
Toxicol Appl Pharmacol. 253:103–111. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
18
|
Spielmann H and Liebsch M: Lessons learned
from validation of in vitro toxicity test: from failure to
acceptance into regulatory practice. Toxicol In Vitro. 15:585–590.
2001. View Article : Google Scholar : PubMed/NCBI
|
|
19
|
Bremer S, Worth AP, Paparella M, et al:
Establishment of an in vitro reporter gene assay for developmental
cardiac toxicity. Toxicology In Vitro. 15:215–223. 2001. View Article : Google Scholar : PubMed/NCBI
|
|
20
|
Genschow E, Scholz G, Brown N, et al:
Development of prediction models for three in vitro embryotoxicity
tests in an ECVAM validation study. In Vitr Mol Toxicol. 13:51–66.
2000.PubMed/NCBI
|
|
21
|
Whitlow S, Bürgin H and Clemann N: The
embryonic stem cell test for the early selection of pharmaceutical
compounds. ALTEX. 24:3–7. 2007.PubMed/NCBI
|
|
22
|
Paquette JA, Kumpf SW, Streck RD, Thomson
JJ, Chapin RE and Stedman DB: Assessment of the Embryonic Stem Cell
Test and application and use in the pharmaceutical industry. Birth
Defects Res B Dev Reprod Toxicol. 83:104–111. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
23
|
Kong D, Xing L, Liu R, et al: Individual
and combined developmental toxicity assessment of bisphenol A and
genistein using the embryonic stem cell test in vitro. Food Chem
Toxicol. 60:497–505. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
24
|
Zhang W, Song DR, Wang YN and Zhu Z:
Evaluation of embryotoxicity of baicalin based on embryonic stem
cell test system. Chin J Pharmacol Toxicol. 26:864–869. 2012.
|
|
25
|
Spielmann H and Liebsch M: Validation
successes: chemicals. Altern Lab Anim. 30(Suppl 2): 33–40.
2002.
|
|
26
|
Zhao AS, Sun YL and Zhang LS: Safety
assessment of Baizhu. Chin J Public Health. 1:43–45. 2006.
|
|
27
|
Wang HY, Ma YF and Zhou WY: On IRPS
toxicity test and effects on immune system. Journal of Mianyang
Normal University. 31:52012.
|
|
28
|
Pang Z, Tang J, Zhu W and Lu Y: Genetic
toxicity of water boiled juice of Isatic Tinctoria L. in mice.
Academic Journal of Guangzhou Medical College. 28:41–42. 2000.
|
|
29
|
Qiu SH, Tang WB, Li FY, Xiao JR and Chen
BY: Experimental study of the acute toxicity on common-used bitter
and cold medicines. Central South Pharmacy. 2:37–38. 2004.
|
|
30
|
Pasqual M, Lauer C, Moyna P and Henriques
JA: Genotoxicity of isoquinoline alkaloid berberine in prokaryotic
and eukaryotic organisms. Mutat Res. 286:243–152. 1993. View Article : Google Scholar : PubMed/NCBI
|