Introduction
Stem cells from apical papilla (SCAP) are a type of
multipotent mesenchymal stem cells, which can be isolated from the
apical papilla of immature teeth (1,2).
SCAP possess the potential to differentiate into odontoblasts,
which may contribute to dentin-pulp complex formation. SCAP also
exhibit clear proliferative activity (3–5). Due
to the clear osteo-/odontogenic potential and proliferative
capacity, SCAP are regarded as valuable seed cells, and may be used
in dentin regeneration.
SCAP have specific phenotypic markers, including
STRO-1, CD146 and CD24 (3). STRO-1
and CD146 are specific markers of mesenchymal stem cells (3,6).
STRO-1- and CD146-positive SCAP have previously been revealed to
exhibit osteo-/odontogenic potential (7). CD24, which had not been detected in
dental pulp stem cells (DPSCs) (3), has been reported to be a specific
marker of SCAP (2,7).
Certain cytokines and growth factors participate in
regulation of proliferation, differentiation and mineralization of
SCAP, including bone morphogenetic protein (BMP) and vascular
endothelial growth factor (VEGF) (8–12). A
previous study has indicated that BMP2 gene transfection is an
effective method to enhance the osteo-/odontogenic differentiation
capacity of DPSCs by increasing osteo-/odontogenic gene and protein
expression levels (12). Another
previous study reported that lentiviral-mediated BMP2 gene
transfection was effective in improving the odontogenic
differentiation capacity of human SCAP in vitro (11). VEGF is also known to exhibit
osteogenetic activity (13–15),
and has been widely used in the regulation of proliferation and
osteo-/odontogenic differentiation in stem cells derived from
dental tissue (16–18).
The application of BMP2 or VEGF individually has
been confirmed to be effective in improving osteo-/odontogenic
differentiation of certain stem cells, however, simultaneous
delivery of BMP2 and VEGF resulted in markedly improved results for
osteogenesis (19–22). However, one study contradictorily
reported that VEGF inhibited the expression of BMP2 and
osteogenesis in rat bone marrow mesenchymal stem cells (BMMSCs)
(23). The effects of BMP2 and
VEGF gene co-transfection on the osteo-/odontogenic differentiation
of SCAP require further elucidation. The aim of the current study
was to analyze osteo-/odontogenic differentiation and the
mineralization characteristics of human SCAP with
lentiviral-mediated BMP2 and VEGF gene co-transfection.
Materials and methods
Isolation and identification of SCAP
Human SCAP were isolated from an extracted immature
mandibular third molar of a male patient aged 18 years old by a
method used in a previous study (11). All protocols were reviewed and
approved by the Ethics Committee of Guanghua School and Hospital of
Stomatology, Sun Yat-sen University (Guangzhou, China). The
osteogenic and adipogenic differentiation capacities of SCAP were
identified by Alizarin Red staining and Oil Red O staining (Cyagen
Biosciences, Inc., Guangzhou, China), respectively (11). The typical phenotypes, including
STRO-1/Alexa Fluor 647-Allophycocyanin (BioLegend, Inc., San Diego,
CA, USA), CD146/Phycoerythrin (BD Pharmingen, San Diego, CA, USA),
CD24/Fluorescein Isothiocyanate (FITC) (BD Pharmingen) and
CD45/FITC (BD Pharmingen), using the 2nd passage of SCAP were
assessed using a FACSCalibur flow cytometer (BD Biosciences,
Franklin Lakes, NJ, USA).
Construction of lentivirus plasmid and
cell infection
Human BMP2 and VEGF gene primers were designed and
amplified as described previously (11,16).
The oligonucleotides were combined into the lentiviral vector
pCDH-CMV-MCS-EF1-copGFP (pCDH; System Biosciences, Mountain View,
CA, USA) to build the recombinant plasmids, pCDH-BMP2 and
pCDH-VEGF. The recombinant plasmids and packaging plasmids were
then co-transfected into 293FT cells (Cyagen Biosciences, Inc.).
The media of the 293FT cells containing lentivirus was collected 48
h after transfection, and then were purified by ultracentrifugation
at 1,000 × g, 37°C for 10 min, and the supernatant was subsequently
filtered through a 0.2-µm syringe filter (EMD Millipore,
Billerica, MA, USA). The 4th passage SCAP were infected with the
cell supernatants which contained lentiviral constructs
(multiplicity of infection =70) to obtain SCAP-BMP2 and SCAP-VEGF.
Furthermore, SCAP-BMP2-VEGF were generated by infecting SCAP with
cell supernatants which contained lentiviral constructs expressing
BMP2 and VEGF, sequentially. Serving as the control, a blank vector
transfected with SCAP [SCAP-green fluorescent protein (GFP)] was
constructed. The expression of BMP2 and VEGF in the four groups of
cells was analyzed by reverse transcription-quantitative polymerase
chain reaction (RT-qPCR) and western blot analysis four days
subsequent to transfection.
Cell proliferation Cell Counting Kit 8
(CCK8) assay
Four groups of cells, SCAP-GFP, SCAP-BMP2, SCAP-VEGF
and SCAP-BMP2-VEGF, were seeded into 96-well plates at a density of
2×103 cells/well and were cultured in α-minimum
essential medium (MEM) with 15% fetal bovine serum (FBS) (Cyagen
Biosciences, Inc.). The proliferation rate of the cells was
analyzed using the CCK8 assay kit (Dojindo Molecular Technologies,
Inc., Kumamoto, Japan) on the 1st, 2nd, 4th and 8th days subsequent
to gene transfection.
RT-qPCR
Total RNA was extracted from SCAP-GFP, SCAP-BMP2,
SCAP-VEGF and SCAP-BMP2-VEGF using TRIzol (Invitrogen; Thermo
Fisher Scientific, Inc., Waltham, MA, USA) on days 1, 4, 8 and 16
subsequent to transfection. The synthesis of complementary DNA
(cDNA) was conducted using the RevertAid First Strand cDNA
Synthesis kit (Thermo Fisher Scientific, Inc.), according to the
manufacturer's protocol. For each sample 2 µg RNA was used
to synthesize the cDNA. The RT-qPCR reaction was conducted using iQ
SYBR Green Supermix (BioRad Laboratories, Inc., Hercules, CA, USA)
and regulated by the spectrofluorimetric iQ5 Thermal iCycler
(BioRad Laboratories, Inc.). For PCR amplification, specific
primers were used, as indicated in Table I. Initial denaturation was
conducted at 95°C for 3 min. This was succeeded by 30 cycles as
follows: Denaturation, 95°C for 15 sec; annealing, 55°C for 30 sec;
extension, 72°C for 1 min. This was followed by a final extension
at 72°C for 7 min, prior to storage at 4°C for 10 min. The mRNA
expression of VEGF (GenBank accession no. MIM 612369), BMP2
(GenBank accession no. MIM 612369) and four osteo-/odontogenic
genes, including alkaline phosphatase (ALP; GenBank accession no.
MIM 612369), osteocalcin (OCN; GenBank accession no. MIM 612369),
dentin sialophos-phoprotein (DSPP; GenBank accession no. MIM
612369) and dentin matrix acidic phosphoprotein gene 1 (DMP1;
GenBank accession no. MIM 612369), were analyzed by RT-qPCR.
Glyceraldehyde 3-phosphate dehydrogenase (GAPDH) was selected as
the reference gene (Table I).
 | Table IReverse transcription-quantitative
polymerase chain reaction primers. |
Table I
Reverse transcription-quantitative
polymerase chain reaction primers.
| Gene | Primer
sequence | Product size
(bp) |
|---|
| BMP2 | F:
5′-CACTGTGCGCAGCTTCC-3′ | 107 |
| R:
5′-CCTCCGTGGGGATAGAACTT-3′ | |
| VEGF | F:
5′-CTACCTCCACCATGCCAAGT-3′ | 104 |
| R:
5′-AGCTGCGCTGATAGACATCC-3′ | |
| ALP | F:
5′-CTATCCTGGCTCCGTGCTC-3′ | 100 |
| R:
5′-GCTGGCAGTGGTCAGATGTT-3′ | |
| OCN | F:
5′-CTCACACTCCTCGCCCTATT-3′ | 107 |
| R:
5′-TTGGACACAAAGGCTGCAC-3′ | |
| DSPP | F:
5′-GCCACTTTCAGTCTTCAAAGAGA-3′ | 130 |
| R:
5′-GCCCAAATGCAAAAATATGTAA-3′ | |
| DMP1 | F:
5′-AAAATTCTTTGTGAACTACGGAGG-3′ | 94 |
| R:
5′-GAGCACAGGATAATCCCCAA-3′ | |
| GAPDH | F:
5′-AAGGTGAAGGTCGGAGTCAA-3′ | 108 |
| R:
5′-AATGAAGGGGTCATTGATGG-3′ | |
Western blotting
The protein extracts from the four groups of cells
were separated on 12% sodium dodecyl sulfate-polyacrylamide gels
(Beyotime Institute of Biotechnology, Haimen, China) and
transferred onto polyvinylidene difluoride membranes (Thermo Fisher
Scientific, Inc.) at 200 mA for 2 h. The membranes were blocked
with 5% non-fat milk for 2 h, and were incubated overnight at 4°C
with the following primary antibodies: Rabbit polyclonal anti-VEGF
(1:500; cat. no. ab46154; Abcam, Cambridge, MA, USA); rabbit
polyclonal anti-BMP2 (1:500; cat. no. ab14933; Abcam) and rabbit
polyclonal anti-dentin sialoprotein (DSP; 1:500; cat. no. sc-33586;
Santa Cruz Biotechnology, Inc., Santa Cruz, CA, USA). The membranes
were subsequently incubated with a horseradish
peroxidase-conjugated goat anti-rabbit antibody (1:20,000; cat. no.
ab97051; Abcam) at 37°C for 2 h. Rabbit polyclonal anti-GAPDH
(1:2,500; cat. no. ab9485; Abcam) was selected as the internal
control. The resultant bands were visualized by the
Chemiluminescence Western Blotting Detection system (EMD Millipore)
on X-ray films (Kodak, Rochester, NY, USA).
ALP staining
The transfected cells were seeded into 6-well plates
at an initial density of 5×104 cells/well and were
cultured to 70% confluence in 2 ml α-MEM containing 15% FBS. The
ALP Staining kit (Jiancheng Biotech. Co., Ltd., Nanjing, China) was
used to stain mineralized granules on day 16 subsequent to
transfection (11). The number of
mineralized nodules was counted three times using an Axio Observer
Z1 microscope (Zeiss AG, Oberkochen, Germany) at a magnification of
×200.
Statistical analysis
The data are presented as the mean ± standard
deviation, and were evaluated by one-way analysis of variance to
analyze the differences between the four groups of cells. SPSS
software, version 16.0 (SPSS, Inc., Chicago, IL, USA) was used for
statistical analysis. P<0.05 was considered to indicate a
statistically significant difference.
Results
Morphology, multipotent differentiation
and phenotypes of SCAP
The kidney-shaped papillae, which had a dense and
pink appearance, were separated from the third molars (Fig. 1A). The isolated primary SCAP
presented with a short spindle-like appearance and formed classic
cell colonies subsequent to eight days culture (Fig. 1B). Alizarin Red staining
illustrated that SCAP formed abundant mineralized nodules 32 days
subsequent to osteogenic induction (Fig. 1C), and Oil Red O staining indicated
that SCAP formed lipid droplets 16 days following adipogenic
induction (Fig. 1D). The freshly
isolated SCAP exhibited typical forward scatter/side scatter
characteristics (Fig. 1E), similar
to those reported previously (4,7). The
presence of the phenotypic markers, including STRO-1, CD146 and
CD24, were observed to be 23.1%, 89.0% and 22.2%, respectively in
the isolated cells (Fig.
1F–H).
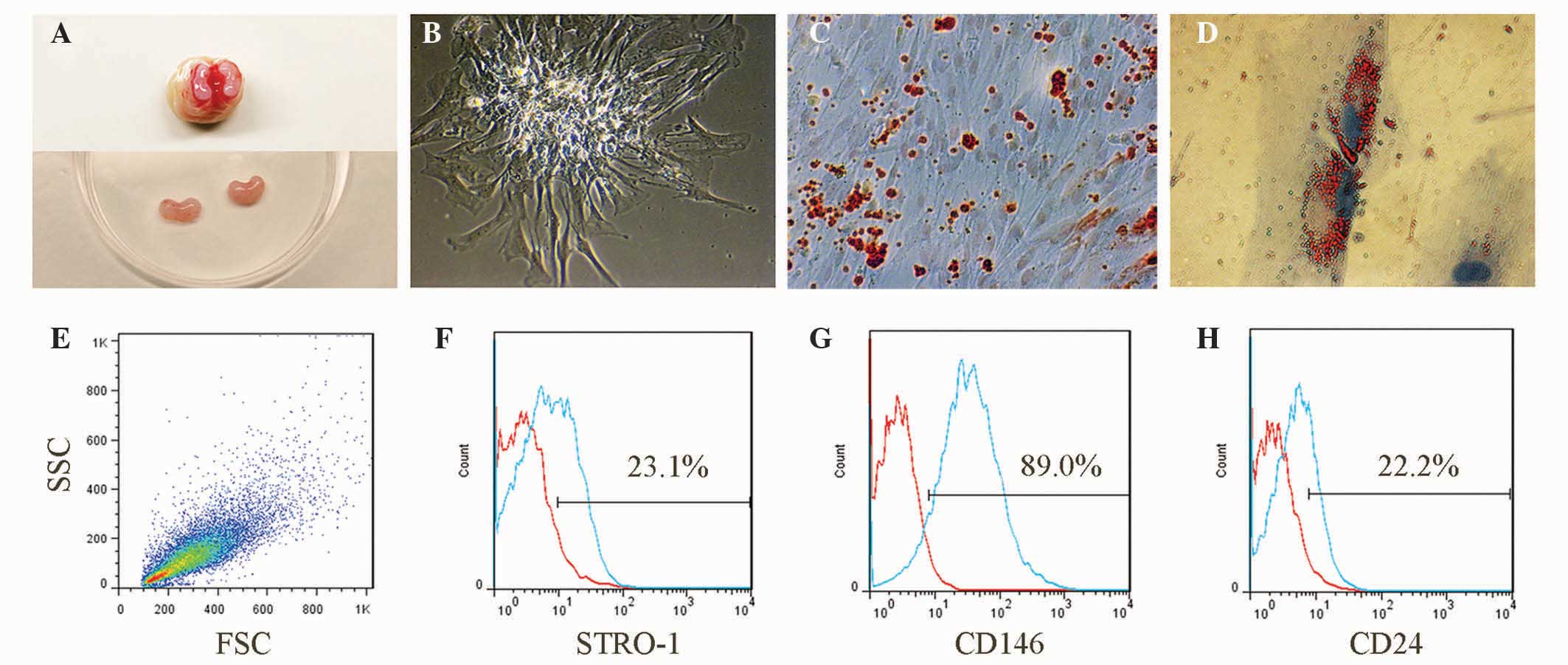 | Figure 1Isolation and identification of SCAP.
(A) Apical papillae, which were separated from the apical part of
third molars, exhibited with a pink and kidney-shaped appearance.
(B) The primary SCAP formed classical cell colonies eight days
subsequent to isolation (original magnification, ×200). (C)
Alizarin red-positive mineralized deposits were observed following
32 days osteogenic induction (original magnification, ×100). (D)
Oil Red-positive lipid droplets were observed following 16 days
adipogenic induction (original magnification, ×800). (E) The
isolated cells from the apical papilla had the typical FSC/SSC
features of SCAP. Representative flow cytometry results
demonstrated that the isolated cells had positive expression of (F)
STRO-1, (G) CD146 and (H) CD24, which were consistent with the
phenotypic characteristics of SCAP. SCAP, stem cells from apical
papilla; FSC, forward scatter; SSC, side scatter. |
Overexpression VEGF and BMP2 in SCAP
A total of four days subsequent to transfection, the
mRNA expression levels of VEGF in SCAP-GFP, SCAP-BMP2, SCAP-VEGF
and SCAP-BMP2-VEGF were 1.00±0.07, 1.32±0.09, 11.14±0.72 and
9.03±0.18, respectively. The relative mRNA expression levels of
BMP2 in SCAP-GFP, SCAP-BMP2, SCAP-VEGF and SCAP-BMP2-VEGF were
1.00±0.12, 6.79±0.10, 3.11±0.05 and 5.94±0.12, respectively
(Fig. 2A). The relative VEGF
protein expression levels, which were quantified by measuring the
grayscale of the western blotting bands in SCAP-GFP, SCAP-BMP2,
SCAP-VEGF and SCAP-BMP2-VEGF, were 1.00±0.10, 1.09±0.09, 2.79±0.26
and 3.03±0.15, respectively. The relative BMP2 protein expression
levels were 1.00±0.04, 3.32±0.09, 2.67±0.10 and 3.83±0.15,
respectively (Fig. 2B and C). The
mRNA and protein expression levels of VEGF and BMP2 were observed
to be significantly enhanced in SCAP-BMP2-VEGF compared with
SCAP-GFP (P<0.05) (Fig. 2A–C).
Notably, the current results demonstrated that VEGF gene
transfection significantly enhanced the BMP2 expression of SCAP
(P<0.05).
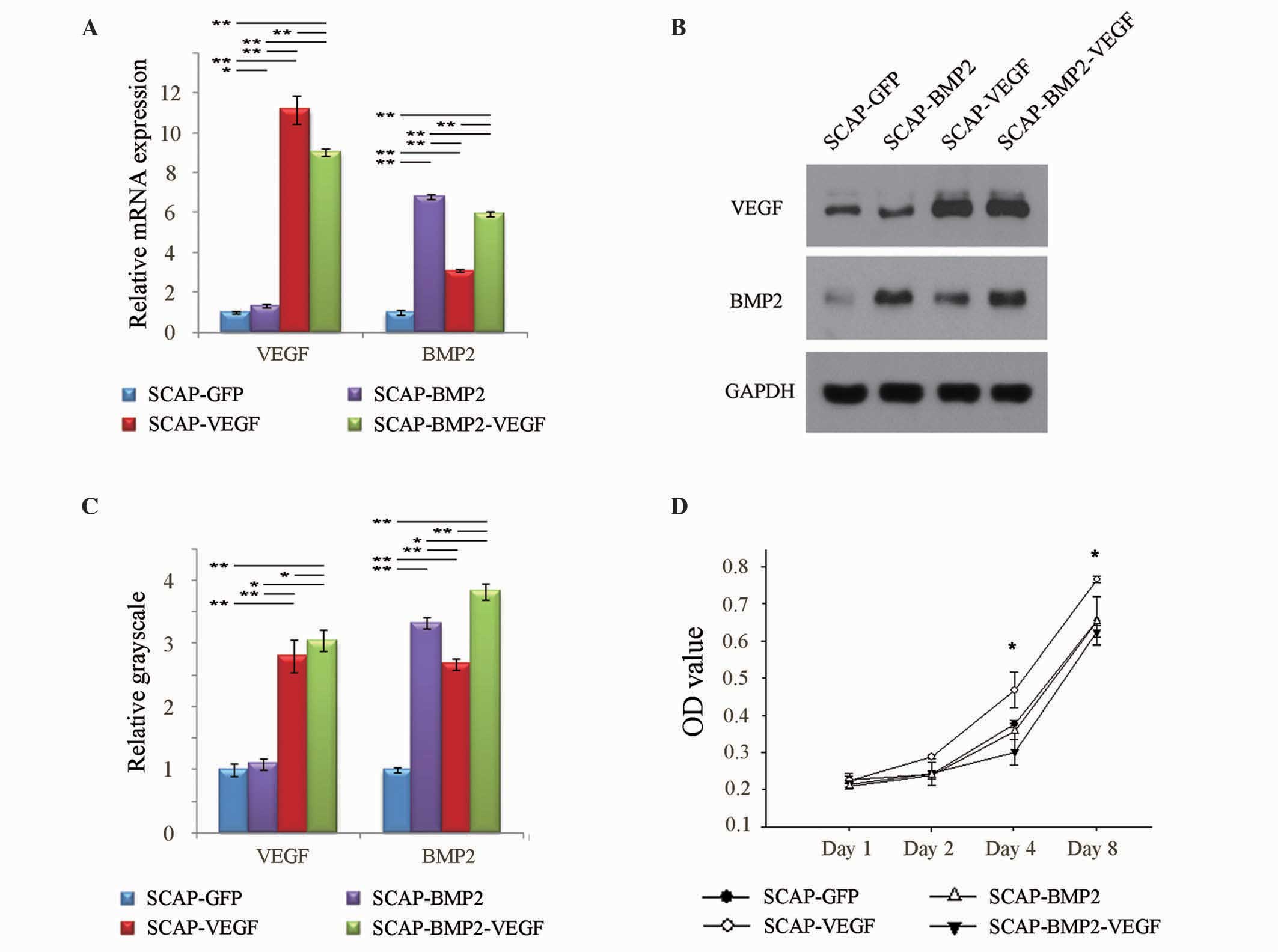 | Figure 2Transfection efficiency and
proliferation status. (A) A total of four days subsequent to
lentivirus-mediated gene transfection, the relative mRNA expression
of VEGF was significantly (P<0.05) enhanced in SCAP-BMP2,
SCAP-VEGF and SCAP-BMP2-VEGF compared with SCAP-GFP. In addition,
the relative mRNA expression levels of BMP2 were significantly
(P<0.05) increased in SCAP-BMP2, SCAP-VEGF and SCAP-BMP2-VEGF
compared with SCAP-GFP. (B and C) Western blot analysis confirmed
that the VEGF peptide expression was significantly (P<0.01)
enhanced in SCAP-VEGF and SCAP-BMP2-VEGF compared with SCAP-GFP,
and the BMP2 peptide expression was significantly (P<0.01)
enhanced in SCAP-BMP2, SCAP-VEGF and SCAP-BMP2-VEGF compared with
SCAP-GFP. (D) SCAP-VEGF transfection resulted in significantly
(P<0.05) increased OD values, and SCAP-BMP2-VEGF transfection
resulted in the lowest OD values on the 4th and 8th days subsequent
to gene transfection. *P<0.05,
**P<0.01. VEGF, vascular endothelial growth factor;
SCAP, stem cells from apical papilla; BMP2, bone morphogenetic
protein 2; GFP, green fluorescent protein; OD, optical density;
GAPDH, glyceraldehyde 3-phosphate dehydrogenase. |
VEGF and BMP2 control the proliferation
of SCAP
On days 4 and 8 subsequent to lentivirus-mediated
gene transfection, SCAP-VEGF exhibited increased proliferation when
compared with the other three groups of cells (P<0.05), while
SCAP-BMP2-VEGF exhibited reduced proliferation (Fig. 2D).
VEGF and BMP2 gene co-transfection
increased the expression of osteo-/odontogenic gene and protein
expression of SCAP
The mRNA expression of osteo-/odontogenic genes in
SCAP-BMP2-VEGF, including ALP, OCN, DSPP and DMP1, were observed to
increase, reaching peak levels of expression on day 16. In
addition, the expression of ALP, OCN, DSPP and DMP1 in
SCAP-BMP2-VEGF was greater than in the other groups at the majority
of the time points measured (P<0.05) (Fig. 3). Consistent with the mRNA
expression results, western blotting indicated that the expression
of DSP in SCAP-BMP2-VEGF was significantly increased when compared
with the other three groups of cells on days 1, 4, 8 and 16
(P<0.01), and the expression increased over time.
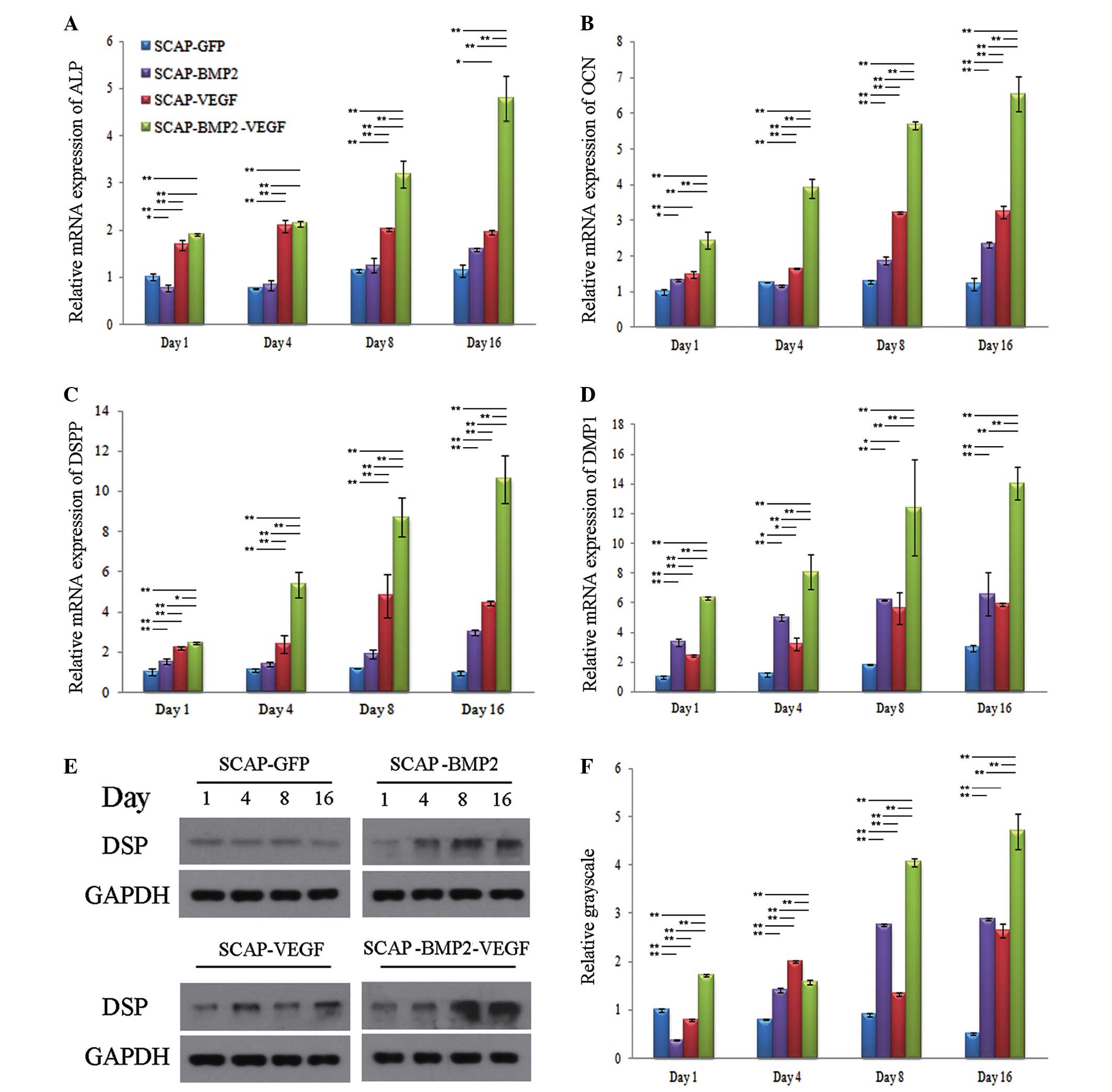 | Figure 3Osteo-/odontogenic differentiation
gene mRNA and protein expression levels. The relative mRNA
expression levels of (A) ALP, (B) OCN, (C) DSPP and (D) DMP1 in
SCAP-BMP2-VEGF was observed to be significantly greater than the
other three groups at the majority of the time points (P<0.05),
and increases in expression were time-dependent. (E and F) The DSP
expression in SCAP-BMP2-VEGF, detected by relative grayscale, was
significantly stronger than the other three groups at the majority
of the time points (P<0.05), and increases in expression were
broadly time-dependent. *P<0.05,
**P<0.01. ALP, alkaline phosphatase; OCN,
osteocalcin; DSPP, dentin sialophosphoprotein; DMP1, dentin matrix
acidic phosphoprotein gene 1; SCAP, stem cells from apical papilla;
BMP2, bone morphogenetic protein 2; VEGF, vascular endothelial
growth factor; DSP, dentin sialoprotein; GAPDH, glyceraldehyde
3-phosphate dehydrogenase. |
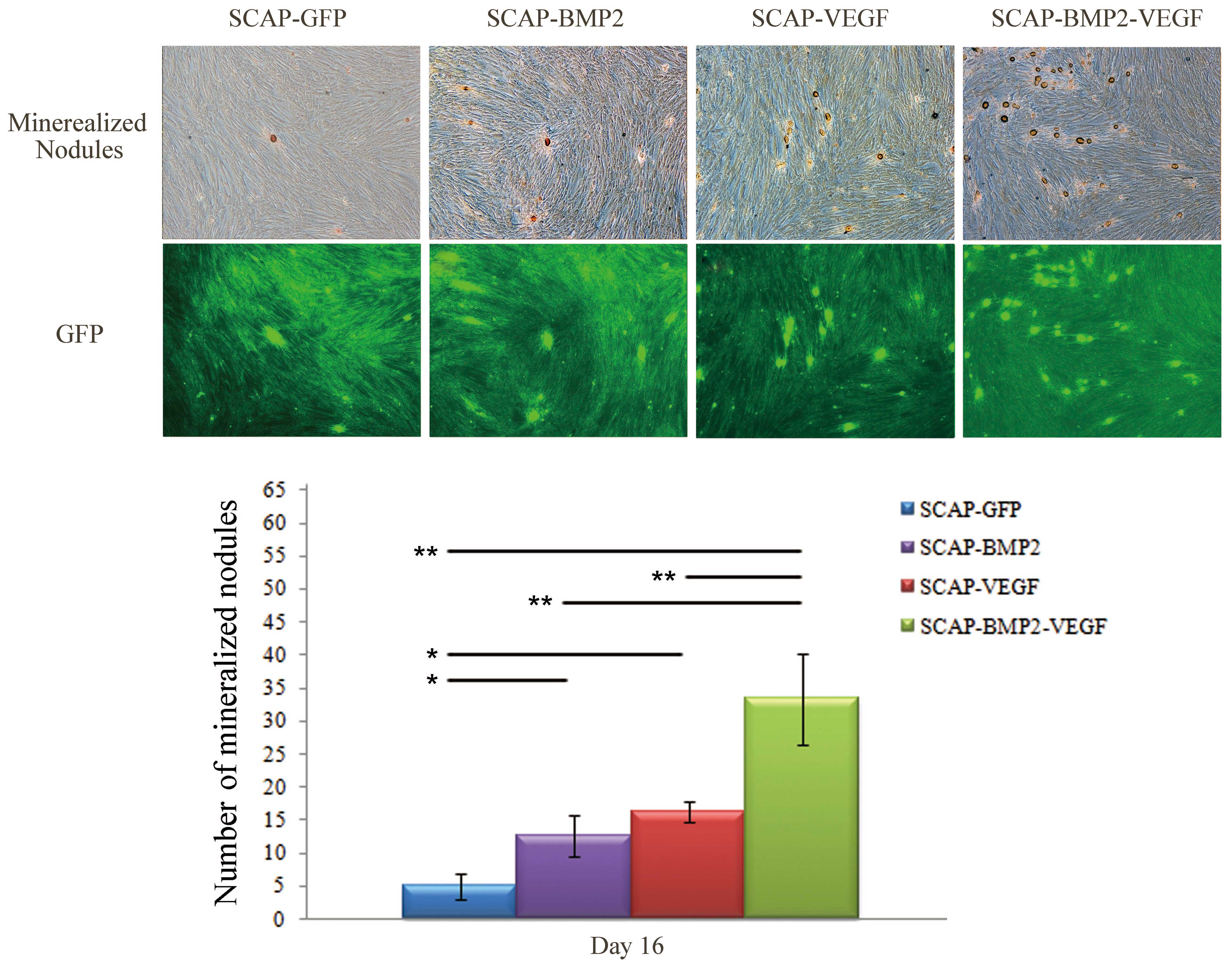
Mineralization nodule formation of
gene-transfected SCAP
The mineralized nodules, which have a strong
positive expression of ALP, were stained with a golden color
(Fig. 4). The number of
mineralized nodules in the SCAP-BMP2-VEGF group was significantly
greater than in the remaining three groups (P<0.01). In
addition, the number of mineralized nodules in the SCAP-BMP2 and
SCAP-VEGF groups was observed to be significantly greater than that
of SCAP-GFP (P<0.01).
Discussion
SCAP are multipotent stem cells, which can
differentiate into osteoblasts, odontoblasts and adipocytes
(1–3). The current study confirmed the
osteogenic and adipogenic differentiation capacities of SCAP. In
agreement with previous studies (1–3,7,11,24),
significant mineralized deposits and lipid droplets were formed by
SCAP following osteogenic and adipogenic induction. Furthermore,
the isolated cells from apical papilla exhibited the specific
phenotypic characteristics of SCAP, including the presence of
STRO-1, CD146 and CD24 (3).
Previous studies have indicated that the positive percentages in
the 1st passage of SCAP were approximately 20–30% (3,7),
47–84% (4) and 3–15% (7) for STRO-1, CD146 and CD24,
respectively. In the present study, the representative expression
of STRO-1, CD146 and CD24 of the 2nd passage SCAP was positive, and
these results are consistent with previous studies (3,4,7).
Even though SCAP has the capacity to differentiate
into osteoblasts and odontoblasts, and can form mineralized tissues
in vivo (9,11,25,26),
the mechanisms of osteo-/odontogenic differentiation promotion
remain to be fully elucidated. VEGF and BMP2, which exhibited
specific expression in the proliferation and differentiation of
osteoblasts, are regarded as important regulators of osteogenesis
(27,28). In a normal bone healing model, VEGF
was previously observed to exhibit maximal expression a number of
days earlier than for BMP2 (29),
indicating the sequence of vascular bed establishment and bone
formation. In dentin development, BMP2 and VEGF were detected
during the differentiation and maturation process of odontoblasts
(30–33). Previous studies have reported that
gene transfection of BMP2 or VEGF genes alone was effective in
activating the osteo-/odontogenic potential of DPSCs by increasing
the expression levels of osteo-/odontogenic genes, including ALP,
OCN, COLI, bone sialoprotein, DSP and DMP1 (12,16).
ALP, detected in the process of mineralization, was regarded as an
early signal of osteo-/odontogenic differentiation of stem cells
derived from dental tissues (24,34,35).
OCN is typically recognized as a signal of osteo-/odontogenic
differentiation at a later stage in the process (4). DMP1 serves an essential role in the
maturation of ameloblasts, osteoblasts and odontoblasts, in
addition to the progression of mineralization in certain stem cells
(36). DSP, the amino-terminal
part of DSPP, has been reported to be an important protein during
the process of odontogenic differentiation (37). In the current study, the
osteo-/odontogenic gene and protein expression levels were observed
to be significantly upregulated in SCAP-BMP2 and SCAP-VEGF when
compared with SCAP-GFP at the majority of the time points. This
indicated that BMP2 or VEGF gene transfection alone aided in
enhancing the osteo-/odontogenic differentiation of SCAP.
Previous studies have used combined delivery of BMP2
and VEGF in order to improve osteogenic differentiation of
mesenchymal stem cells (20,22,38–43),
osteoprogenitor cells (19,44)
and osteoblasts (45). When
pre-osteoblasts were induced by BMP2, the expression of VEGF was
observed to increase (46).
Similarly, VEGF gene transfection enhanced the expression of VEGF
in the bone marrow stromal cells, in addition to marginally
increasing the expression of BMP2 (41). However, a previous study contracted
this, stating that VEGF transfection inhibits the expression of
BMP2 in mesenchymal stem cells (23). In the current study, VEGF gene
transfection was observed to enhance the expression of VEGF, in
addition to significantly increasing the expression of BMP2 in
SCAP. These results indicated that VEGF may promote the
osteo-/odontogenic differentiation of SCAP by acting cooperatively
with BMP2. In addition, a previous study demonstrated that VEGF and
BMP2 were able to promote bone regeneration by facilitating stem
cell homing (39). A previous
study indicated that BMP2- and VEGF-co-transfected BMMSCs resulted
in the peak ALP expression on day 7 (22). However, SCAP-BMP2-VEGF demonstrated
an increased expression of ALP over time in the current study. The
results of current study supported the hypothesis that BMP2 and
VEGF gene co-transfection was more effective in improving
osteo-/odontogenic differentiation of SCAP than single gene
transfection.
A previous study indicated that VEGF increased the
rate of proliferation in rat osteoblasts in a dose-dependent
manner, while no significant alterations were observed with BMP2
(45). The results of the present
study are consistent with this previous study (45), observing that the VEGF-transfected
SCAP group exhibited marginally increased proliferation compared
with the other groups on days 4 and 8 subsequent to transfection.
The lowest levels of proliferation were observed in the BMP2 and
VEGF co-transfection group on days 4 and 8, thus the BMP2 and VEGF
had a synergistic effect on the proliferation of SCAP.
In summary, human dental papilla stem cells were
successfully transfected with the BMP2 and VEGF genes. The
SCAP-BMP2-VEGF group exhibited increased expression levels of
osteo-/odontogenic differentiation-associated genes and protein,
and increased mineralization deposits than the SCAP-BMP2, SCAP-VEGF
and SCAP-GFP groups in vitro. These results suggest that the
co-transfection of homologous BMP2 and VEGF genes is an effective
strategy to improve the osteo-/odontogenic differentiation of SCAP
in vitro.
References
|
1
|
Huang GT, Sonoyama W, Liu Y, Liu H, Wang S
and Shi S: The hidden treasure in apical papilla: The potential
role in pulp/dentin regeneration and bioroot engineering. J Endod.
34:645–651. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
2
|
Sonoyama W, Liu Y, Fang D, Yamaza T, Seo
BM, Zhang C, Liu H, Gronthos S, Wang CY, Wang S and Shi S:
Mesenchymal stem cell-mediated functional tooth regeneration in
swine. PloS One. 1:e792006. View Article : Google Scholar : PubMed/NCBI
|
|
3
|
Huang GT, Gronthos S and Shi S:
Mesenchymal stem cells derived from dental tissues vs. those from
other sources: Their biology and role in regenerative medicine. J
Dent Res. 88:792–806. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
4
|
Bakopoulou A, Leyhausen G, Volk J,
Tsiftsoglou A, Garefis P, Koidis P and Geurtsen W: Comparative
analysis of in vitro osteo/odontogenic differentiation potential of
human dental pulp stem cells (DPSCs) and stem cells from the apical
papilla (SCAP). Arch Oral Biol. 56:709–721. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
5
|
Tziafas D and Kodonas K: Differentiation
potential of dental papilla, dental pulp and apical papilla
progenitor cells. J Endod. 36:781–789. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
6
|
Lin CS, Xin ZC, Dai J and Lue TF: Commonly
used mesenchymal stem cell markers and tracking labels: Limitations
and challenges. Histol Histopathol. 28:1109–1116. 2013.PubMed/NCBI
|
|
7
|
Bakopoulou A, Leyhausen G, Volk J, Koidis
P and Geurtsen W: Comparative characterization of STRO-1
(neg)/CD146 (pos) and STRO-1 (pos)/CD146 (pos) apical papilla stem
cells enriched with flow cytometry. Arch Oral Biol. 58:1556–1568.
2013. View Article : Google Scholar : PubMed/NCBI
|
|
8
|
Friedlander LT, Cullinan MP and Love RM:
Dental stem cells and their potential role in apexogenesis and
apexification. Int Endod J. 42:955–962. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
9
|
Wang J, Zhang H, Zhang W, Huang E, Wang N,
Wu N, Wen S, Chen X, Liao Z, Deng F, et al: Bone morphogenetic
protein-9 effectively induces osteo/odontoblastic differentiation
of the reversibly immortalized stem cells of dental apical papilla.
Stem Cells Dev. 23:1405–1416. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
10
|
Lin ZM, Qin W, Zhang NH, Xiao L and Ling
JQ: Adenovirus-mediated recombinant human bone morphogenetic
protein-7 expression promotes differentiation of human dental pulp
cells. J Endod. 33:930–935. 2007. View Article : Google Scholar : PubMed/NCBI
|
|
11
|
Zhang W, Zhang X, Ling J, Liu W, Ma J and
Zheng J: Proliferation and odontogenic differentiation of BMP2
genetransfected stem cells from human tooth apical papilla: An in
vitro study. Int J Mol Med. 34:1004–1012. 2014.PubMed/NCBI
|
|
12
|
Yang X, van der Kraan PM, Bian Z, Fan M,
Walboomers XF and Jansen JA: Mineralized tissue formation by
BMP2-transfected pulp stem cells. J Dent Res. 88:1020–1025. 2009.
View Article : Google Scholar : PubMed/NCBI
|
|
13
|
Hiltunen MO, Ruuskanen M, Huuskonen J,
Mähönen AJ, Ahonen M, Rutanen J, Kosma VM, Mahonen A, Kröger H and
Ylä-Herttuala S: Adenovirus-mediated VEGF-A gene transfer induces
bone formation in vivo. FASEB J. 17:1147–1149. 2003.PubMed/NCBI
|
|
14
|
Jacobsen KA, Al-Aql ZS, Wan C, Fitch JL,
Stapleton SN, Mason ZD, Cole RM, Gilbert SR, Clemens TL, Morgan EF,
et al: Bone formation during distraction osteogenesis is dependent
on both VEGFR1 and VEGFR2 signaling. J Bone Miner Res. 23:596–609.
2008. View Article : Google Scholar : PubMed/NCBI
|
|
15
|
Dvorak HF: Angiogenesis: Update 2005. J
Thromb Haemost. 3:1835–1842. 2005. View Article : Google Scholar : PubMed/NCBI
|
|
16
|
Zhang W, Liu W, Ling J, Lin Z, Gao Y, Mao
X and Jian Y: Odontogenic differentiation of vascular endothelial
growth factor-transfected human dental pulp stem cells in vitro.
Mol Med Rep. 10:1899–1906. 2014.PubMed/NCBI
|
|
17
|
Lee JH, Um S, Jang JH and Seo BM: Effects
of VEGF and FGF-2 on proliferation and differentiation of human
periodontal ligament stem cells. Cell Tissue Res. 348:475–484.
2012. View Article : Google Scholar : PubMed/NCBI
|
|
18
|
Matsushita K, Motani R, Sakuta T,
Yamaguchi N, Koga T, Matsuo K, Nagaoka S, Abeyama K, Maruyama I and
Torii M: The role of vascular endothelial growth factor in human
dental pulp cells: Induction of chemotaxis, proliferation and
differentiation and activation of the AP-1-dependent signaling
pathway. J Dent Res. 79:1596–1603. 2000. View Article : Google Scholar : PubMed/NCBI
|
|
19
|
Kanczler JM, Ginty PJ, White L, Clarke NM,
Howdle SM, Shakesheff KM and Oreffo RO: The effect of the delivery
of vascular endothelial growth factor and bone morphogenic
protein-2 to osteoprogenitor cell populations on bone formation.
Biomaterials. 31:1242–1250. 2010. View Article : Google Scholar
|
|
20
|
Kumar S, Wan C, Ramaswamy G, Clemens TL
and Ponnazhagan S: Mesenchymal stem cells expressing osteogenic and
angiogenic factors synergistically enhance bone formation in a
mouse model of segmental bone defect. Mol Ther. 18:1026–1034. 2010.
View Article : Google Scholar : PubMed/NCBI
|
|
21
|
Lin CY, Chang YH, Lin KJ, Yen TC, Tai CL,
Chen CY, Lo WH, Hsiao IT and Hu YC: The healing of critical-sized
femoral segmental bone defects in rabbits using
baculovirus-engineered mesenchymal stem cells. Biomaterials.
31:3222–3230. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
22
|
Lin Z, Wang JS, Lin L, Zhang J, Liu Y,
Shuai M and Li Q: Effects of BMP2 and VEGF165 on the osteogenic
differentiation of rat bone marrow-derived mesenchymal stem cells.
Exp Ther Med. 7:625–629. 2014.PubMed/NCBI
|
|
23
|
Schönmeyr BH, Soares M, Avraham T, Clavin
NW, Gewalli F and Mehrara BJ: Vascular endothelial growth factor
inhibits bone morphogenetic protein 2 expression in rat mesenchymal
stem cells. Tissue Eng Part A. 16:653–662. 2010. View Article : Google Scholar :
|
|
24
|
Chen K, Xiong H, Huang Y and Liu C:
Comparative analysis of in vitro periodontal characteristics of
stem cells from apical papilla (SCAP) and periodontal ligament stem
cells (PDLSCs). Arch Oral Biol. 58:997–1006. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
25
|
Wang J, Liu B, Gu S and Liang J: Effects
of Wnt/β-catenin signalling on proliferation and differentiation of
apical papilla stem cells. Cell Prolif. 45:121–131. 2012.
View Article : Google Scholar : PubMed/NCBI
|
|
26
|
Qu B, Liu O, Fang X, Zhang H, Wang Y, Quan
H, Zhang J, Zhou J, Zuo J, Tang J and Tang Z: Distal-less homeobox
2 promotes the osteogenic differentiation potential of stem cells
from apical papilla. Cell Tissue Res. 357:133–143. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
27
|
Dai J and Rabie AB: VEGF: An essential
mediator of both angiogenesis and endochondral ossification. J Dent
Res. 86:937–950. 2007. View Article : Google Scholar : PubMed/NCBI
|
|
28
|
Ryoo HM, Lee MH and Kim YJ: Critical
molecular switches involved in BMP-2-induced osteogenic
differentiation of mesenchymal cells. Gene. 366:51–57. 2006.
View Article : Google Scholar
|
|
29
|
Uchida S, Sakai A, Kudo H, Otomo H,
Watanuki M, Tanaka M, Nagashima M and Nakamura T: Vascular
endothelial growth factor is expressed along with its receptors
during the healing process of bone and bone marrow after drill-hole
injury in rats. Bone. 32:491–501. 2003. View Article : Google Scholar : PubMed/NCBI
|
|
30
|
Casagrande L, Demarco FF, Zhang Z, Araujo
FB, Shi S and Nör JE: Dentin-derived BMP-2 and odontoblast
differentiation. J Dent Res. 89:603–608. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
31
|
Mastrangelo F, Piccirilli M, Dolci M, Teté
S, Speranza L, Patruno A, Gizzi F, Felaco M, Artese L and De Lutiis
MA: Vascular endothelial growth factor (VEGF) in human tooth germ
center. Int J Immunopathol Pharmacol. 18:587–594. 2005.PubMed/NCBI
|
|
32
|
Scheven BA, Man J, Millard JL, Cooper PR,
Lea SC, Walmsley AD and Smith AJ: VEGF and odontoblast-like cells:
Stimulation by low frequency ultrasound. Arch Oral Biol.
54:185–191. 2009. View Article : Google Scholar
|
|
33
|
Virtej A, Løes S, Iden O, Bletsa A and
Berggreen E: Vascular endothelial growth factors signalling in
normal human dental pulp: A study of gene and protein expression.
Eur J Oral Sci. 121:92–100. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
34
|
Wu J, Huang GT, He W, Wang P, Tong Z, Jia
Q, Dong L, Niu Z and Ni L: Basic fibroblast growth factor enhances
stemness of human stem cells from the apical papilla. J Endod.
38:614–622. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
35
|
Choi MH, Noh WC, Park JW, Lee JM and Suh
JY: Gene expression pattern during osteogenic differentiation of
human periodontal ligament cells in vitro. J Periodontal Implant
Sci. 41:167–175. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
36
|
MacDougall M, Gu TT and Simmons D: Dentin
matrix protein-1, a candidate gene for dentinogenesis imperfecta.
Connect Tissue Res. 35:267–272. 1996. View Article : Google Scholar : PubMed/NCBI
|
|
37
|
Lee SY, Kim SY, Park SH, Kim JJ, Jang JH
and Kim EC: Effects of recombinant dentin sialoprotein in dental
pulp cells. J Dent Res. 91:407–412. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
38
|
Zhang W, Zhu C, Ye D, Xu L, Zhang X, Wu Q,
Zhang X, Kaplan DL and Jiang X: Porous silk scaffolds for delivery
of growth factors and stem cells to enhance bone regeneration. PloS
One. 9:e1023712014. View Article : Google Scholar : PubMed/NCBI
|
|
39
|
Zhang W, Zhu C, Wu Y, Ye D, Wang S, Zou D,
Zhang X, Kaplan DL and Jiang X: VEGF and BMP-2 promote bone
regeneration by facilitating bone marrow stem cell homing and
differentiation. Eur Cell Mater. 27:1–11; discussion 11–12. 2014.
View Article : Google Scholar : PubMed/NCBI
|
|
40
|
Bai Y, Li P, Yin G, Huang Z, Liao X, Chen
X and Yao Y: BMP-2, VEGF and bFGF synergistically promote the
osteogenic differentiation of rat bone marrow-derived mesenchymal
stem cells. Biotechnol Lett. 35:301–308. 2013. View Article : Google Scholar
|
|
41
|
Xiao C, Zhou H, Liu G, Zhang P, Fu Y, Gu
P, Hou H, Tang T and Fan X: Bone marrow stromal cells with a
combined expression of BMP-2 and VEGF-165 enhanced bone
regeneration. Biomed Mater. 6:0150132011. View Article : Google Scholar : PubMed/NCBI
|
|
42
|
Tian XB, Sun L, Yang SH, Fu RY, Wang L, Lu
TS, Zhang YK and Fu DH: Ectopic osteogenesis of mouse bone marrow
stromal cells transfected with BMP 2/VEGF (165) genes in vivo.
Orthop Surg. 1:322–325. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
43
|
Hou H, Zhang X, Tang T, Dai K and Ge R:
Enhancement of bone formation by genetically-engineered bone marrow
stromal cells expressing BMP-2, VEGF and angiopoietin-1. Biotechnol
Lett. 31:1183–1189. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
44
|
Behr B, Sorkin M, Lehnhardt M, Renda A,
Longaker MT and Quarto N: A comparative analysis of the osteogenic
effects of BMP-2, FGF-2 and VEGFA in a calvarial defect model.
Tissue Eng Part A. 18:1079–1086. 2012. View Article : Google Scholar :
|
|
45
|
Li P, Bai Y, Yin G, Pu X, Huang Z, Liao X,
Chen X and Yao Y: Synergistic and sequential effects of BMP-2, bFGF
and VEGF on osteogenic differentiation of rat osteoblasts. J Bone
Miner Metab. 32:627–635. 2014. View Article : Google Scholar
|
|
46
|
Akeel S, El-Awady A, Hussein K, El-Refaey
M, Elsalanty M, Sharawy M and Al-Shabrawey M: Recombinant bone
morphogenetic protein-2 induces up-regulation of vascular
endothelial growth factor and interleukin 6 in human
pre-osteoblasts: Role of reactive oxygen species. Arch Oral Biol.
57:445–452. 2012. View Article : Google Scholar
|