Introduction
Lipid dysregulation serves a critical role in the
progression of cardiovascular diseases (1), metabolic syndrome (2) and non-alcoholic fatty liver disease
(3). These disorders pose major
public health concerns, and are associated with family burden and a
high socioeconomic cost (2).
Currently available lipid-lowering agents used in the treatment of
hyperlipidemia include statins and fibrates; however, these agents
have been associated with serious adverse effects, including
gastrointestinal disturbances, severe muscle damage and
hepatotoxicity (4). Therefore,
natural products and herbal medicines with improved safety profiles
have garnered attention for the treatment of lipid disorders.
The capitula of Coreopsis tinctoria, also
known as snow chrysanthemum, have been used in the form of a
tea-like beverage for the prevention of cardiovascular disorders,
diarrhea and diabetes in traditional Chinese medicine (5). Coreopsis tinctoria has been
revealed to contain high concentrations of flavonoids (6), and it has been reported to exert
anti-inflammatory effects (5), to
promote pancreatic cell recovery (7,8) and
to regulate lipid metabolism in hyperlipidemic mice (9). However, the main active compounds of
Coreopsis tinctoria, as well as their exact pharmacologic
effects on hyperlipidemia, have yet to be elucidated.
An increasing body of evidence has demonstrated that
oxidative stress is a key trigger in the progression of
hyperlipidemia (1,10). Lipids are thought to be among the
most sensitive biological molecules in terms of reactive oxygen
species (ROS) susceptibility (11). In addition, lipid peroxidation is
known to disturb the integrity of cellular membranes, leading to
leakage of cytoplasmic enzymes, which in turn causes cell death and
cell death ultimately drives disease progression (11,12).
A previous study has demonstrated that flavonoids have the capacity
for anti-oxidative activities by reducing the production of ROS and
preventing lipid peroxidation, which may be associated with
alleviated hyperlipidemia (13).
The aim of the present study was to identify the
main active compounds of Coreopsis tinctoria, to evaluate
their antihyperlipidemic properties in vivo, and to
investigate the molecular mechanisms underlying their effects on
lipid regulation in vitro.
Materials and methods
Materials
Commercially available analytical reagents were
purchased from Shanghai Aladdin Bio-Chem Technology Co., Ltd.
(Shanghai, China). Dulbecco's modified Eagle's medium (DMEM), fetal
bovine serum (FBS), trypsin and penicillin-streptomycin-glutamine
were obtained from Beyotime Institute of Biotechnology (Haimen,
China). Dimethylsulfoxide (for MTT assay),
2,2-diphenyl-picrylhydrazyl (DPPH), thiobarbituric acid (TBA),
bovine serum albumin (BSA), MTT, 2′,7′-dichlorofluorescein
diacetate (DCFH-DA) and mouse monoclonal anti-GAPDH antibody
(1:10,000; cat no. G8795) were purchased from Sigma-Aldrich; Merck
KGaA (Darmstadt, Germany). Rabbit monoclonal antifatty acid
synthase (FAS; 1:1,000; cat no. 3180S) and rabbit monoclonal
anti-protein disulfide-isomerase A3 precursor (ERp57; 1:1,000; cat
no. 2881S) antibodies were purchased from Cell Signaling
Technology, Inc. (Danvers, MA, USA). Horseradish
peroxide-conjugated goat antimouse immunoglobulin (Ig)G (1:5,000;
cat no. sc-2005) and goat anti-rabbit IgG (1:5,000; cat no.
sc-2004) were purchased from Santa Cruz Biotechnology, Inc.
(Dallas, TX, USA).
Preparation and analysis of snow
chrysanthemum aqueous extract and its main compounds
Snow chrysanthemum, the capitulum of Coreopsis
tinctoria, was collected in the Uighur Autonomous Region of
Xinjiang Province in September 2012, and was identified by
Professor Yu-Hai Guo (China Agricultural University, Beijing,
China). A voucher specimen (cat no. 201209) was preserved in the
herbarium of the Laboratory of Ethnopharmacology of West China
Hospital, West China Medical School of Sichuan University (Sichuan,
China). Air-dried snow chrysanthemum (100 g) was ground into a
powder and decocted with distilled water (0.8 l) by heating reflux
extraction at 98°C for 3 h. Subsequently, the snow chrysanthemum
aqueous extract (SCAE) was dried until water content was <10%.
The total flavonoid content in SCAE was assessed using a
colorimetric method, as previously described (14).
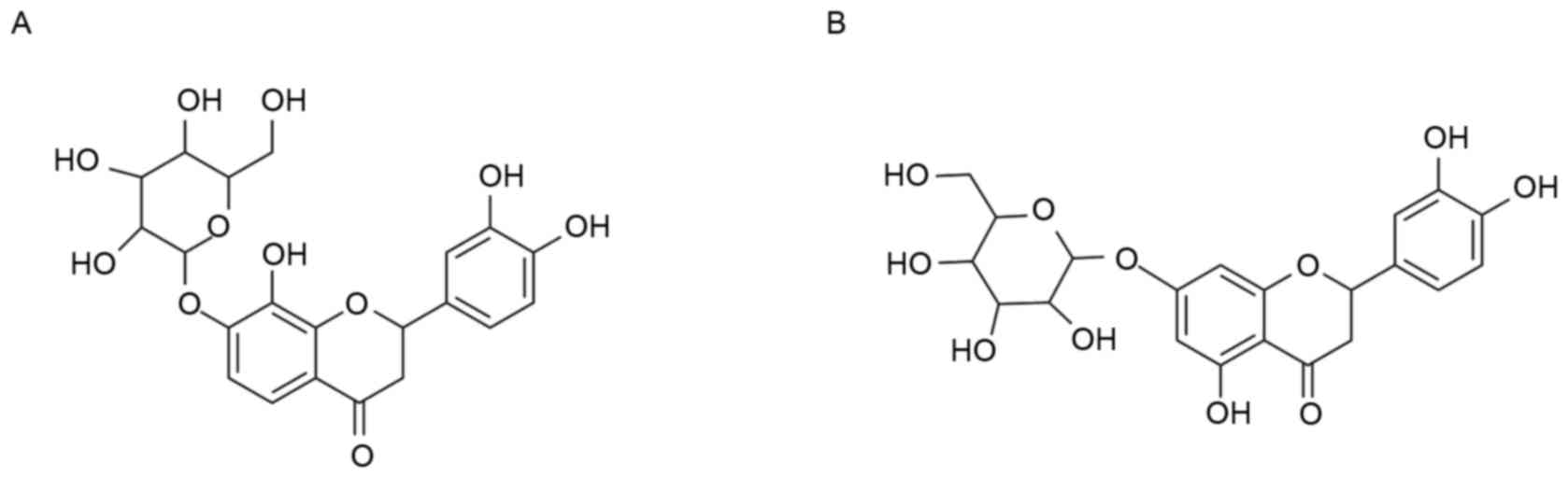
Its main compounds flavanomarein (compound 1) and
eriodictyol 7-O-β-D glucopyranoside (compound 2) were isolated and
purified using preparative high-performance liquid chromatography
(HPLC; Shimadzu Corporation, Kyoto, Japan). Preparative HPLC was
carried out on a SHIMADZU LC-6AD instrument with an SPD-20A
detector, using a YMC-Pack ODS-A column (250×20 mm; 5 µm; YMC Co.,
Ltd., Kyoto, Japan). The dried powders (5 kg) of Coreopsis
tinctoria were extracted three times successively with water
and 70% ethyl alcohol to obtain the crude extract. The extract was
subjected to polyamide resin (Chongqing Change Chemical Co., Ltd.,
Chongqing, China) column chromatography eluted with water, 30%
ethyl alcohol and 70% ethyl alcohol to give three fractions (A-C,
respectively). Fraction B was chromatographed over a Sephadex LH-20
column (GE Healthcare Bio-Sciences, Uppsala, Sweden) eluted with
50% methanol to give six fractions 1 to 6. Compound 1 was obtained
and further purified by ecrystallization with 100% methanol from
fraction 6. Compound 2 was obtained by Prep. HPLC (Shimadazu,
YMC-Pack ODS-A; 5 µm, 250×20 mm; Shimadzu Corporation) from
fraction 3. The mobile phase was acetonitrile (18%; solvent B):
water (82%; solvent A), and the flow rate was 6 ml/min. Compounds 1
and 2 were identified by 1H NMR (600 MHz) and
13C NMR (150 MHz) run on AV II spectrometer (Bruker
Corporation, Ettlingen, Germany).
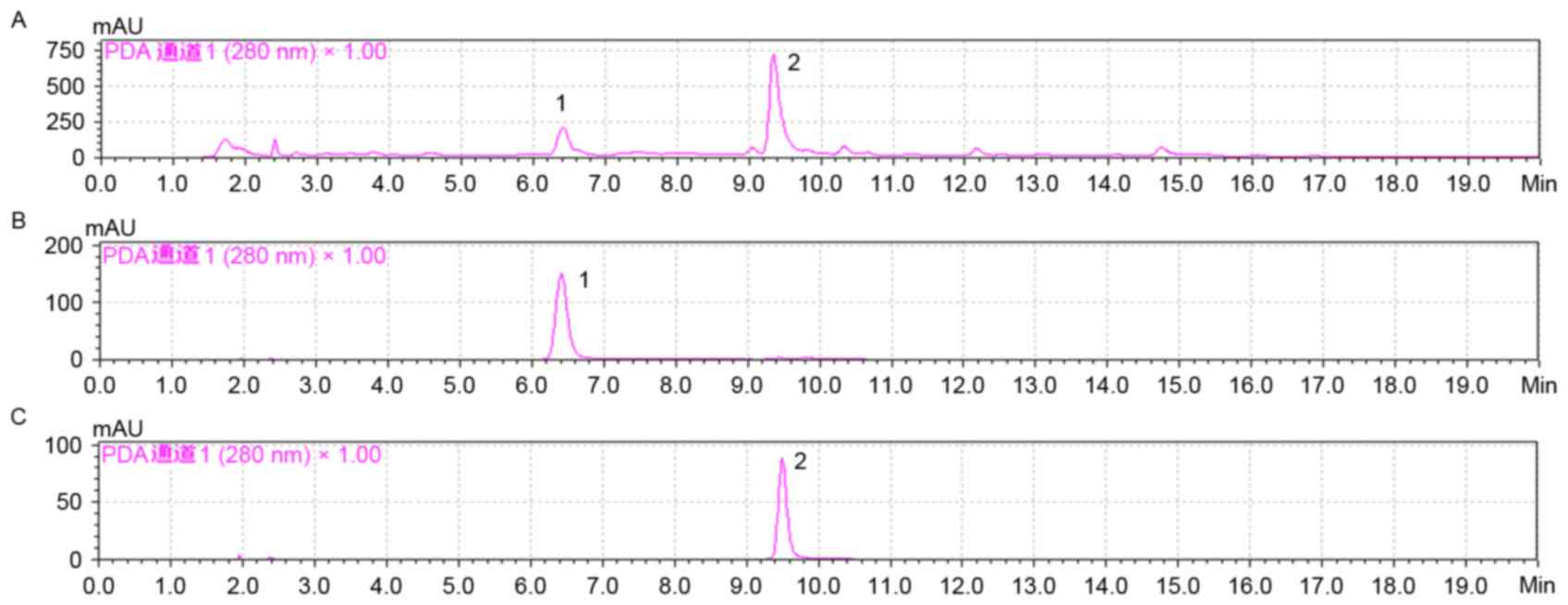
HPLC profiles of SCAE, compound 1 and 2, were
analyzed using a reverse column (LC-20A, Inertsil® ODS-SP; 4.6×150
nm; 3.5 µm; Shimadzu Corporation). Equal quantities (20 µl) of
SCAE, compounds 1 and 2, were used for analysis. They were eluted
at a 1 ml/min flow rate with solvent A, water with 0.1% formic
acid, and solvent B (acetonitrile with 0.1% formic acid) at 280 nm.
The gradient started from 15% B for the first 5 min, then to 65% by
15 min, and finally to 100% by 20 min at 22°C.
Animals
The animal experiments were approved by the Ethics
Committee of the Institutional Animal Care and Treatment Committee
of Sichuan University (permit no. 2014002B; Chengdu, China). Male
Kunming mice (weight, 18–22 g; age, 4–6 weeks) were provided by the
Chengdu Dashuo Experimental Animal Co, Ltd. (Chengdu, China). The
mice were housed in controlled temperature (22±1°C) and humidity
(55±5%) conditions, under a 12/12 h light/dark cycle with free
access to food and water.
Animal experiments
The mice were divided into the following 3 groups
(n=10 mice/group): Groups I, II and III. Mice in group I were
maintained on a normal pellet diet, whereas mice in groups II and
III were maintained on a high-fat diet for the induction of
hyperlipidemia, which consisted of the following: Normal diet
supplemented with 10% cholesterol, 10% lard, 2% sodium deoxycholic
acid and 0.1% propylthiouracil. Following 21 days, group II were
treated with 0.5% sodium carboxymethyl cellulose (vehicle). Group
III received SCAE (60 mg/kg; compounds 1 and 2). Treatments were
given orally twice a day for 42 days. At the end of the study, the
mice were sacrificed, and blood, liver and kidney tissue samples
were collected for biochemical analysis. Serum was separated by
centrifugation at 1,000 × g for 15 min at 4°C, then assays of total
cholesterol (TC), triglyceride (TG), low-density
lipoprotein-cholesterol (LDL-C), glutathione peroxidase (GSH-Px)
and nitric oxide synthase (NOS) levels were performed. Liver
samples were homogenized (10%, w/v) in cold saline, then
centrifuged at 1,000 × g for 15 min at 4°C. The supernatant was
used for assaying the superoxide dismutase (SOD) and
malondialdehyde (MDA) levels. Kidney samples were homogenized (10%,
w/v) in cold saline and centrifuged at 1,000 × g for 10 min at 4°C
for the lipid peroxidation assay. Protein concentration was
determined using a bicinchoninic acid (BCA) protein assay kit
(Beyotime Institute of Biotechnology). The commercially available
kits used for these measurements included: TC assay kit (cat no.
A111-1), TG assay kit (cat no. A110-1), LDL-C assay kit (cat no.
A113-1), GSH-Px assay kit (Colorimetric method; cat no. A005),
Total NOS assay kit (cat no. A014-2), Total (T-) SOD assay kit
(Hydroxylamine method; cat no. A001-1) and MDA assay kit (TBA
method; cat no. A003-1; (all from Nanjing Jiancheng Bioengineering
Institute, Nanjing, China) kits, according to the manufacturers'
protocol. High-density lipoprotein cholesterol (HDL-C) levels were
calculated according to the following formula: HDL-C = TC-[(1/5xTG)
+ LDL-C].
Antioxidant assays
The putative free radical-scavenging properties of
SCAE were investigated using DPPH, as previously described
(15,16). Various concentrations of compounds
1 and 2 (0, 10, 20, 40, 80 and 160 µmol/l), were added to 500
µmol/l alcoholic DPPH solution. A total of 500 µmol/l alcoholic
DPPH solution, without compounds 1 and 2, was used as the control.
Following incubation for 30 min in the dark at room temperature,
the absorbance of each sample was measured at 517 nm.
Lipid peroxidation was assessed using the TBA
method, as previously described (17). Briefly, mouse liver and kidney
samples were homogenized (10%, w/v) in cold saline and centrifuged
at 1,000 × g for 15 min at 4°C. Then, the liver and kidney tissue
homogenates (100 µl, 10%) were mixed with 100 µl compounds 1 or 2
(10, 20, 40, 80 and 160 µmol/l), and ferrous sulfate (8 µl, 70
mmol/l) was added to each mixture. The mixtures were incubated for
30 min at 37°C. Subsequently, 300 µl 20% acetic acid and 300 µl
0.8% TBA in 1.1% sodium dodecyl sulfate was added, and the final
mixtures were incubated at 95°C for 60 min. Following cooling, the
mixtures were centrifuged at 5,000 × g for 10 min at 4°C and their
absorbance was measured at 532 nm (17).
The IC50 value denotes the effective
concentration of compounds 1 or 2 used to reduce 50% of available
DPPH radicals or inhibit 50% of liver and kidney lipid
peroxidation. The IC50 value of compounds 1 and 2 was
calculated using SPSS software version 19.0 (IBM Corp., Armonk, NY,
USA).
Cell culture and viability assay
The human HepG2 hepatocellular carcinoma cell line
was obtained from the Cell Bank of the Shanghai Institute of
Biochemistry and Cell Biology, Chinese Academy of Sciences (cat no.
TCHu72; Shanghai, China). Cells were cultured at 37°C in DMEM
supplemented with 10% FBS, 100 U/ml penicillin and 100 µg/ml
streptomycin, as previously described (18). HepG2 cells (5×103 cells/well) were
seeded in each well of 96-well plates (Costar; Corning
Incorporated, Corning, NY, USA) and cultured for 24 h at 37°C.
Cells were then incubated with compounds 1 or 2 (0, 1, 5, 25, 125
or 625 µmol/l) at 37°C for 24 h. Cells without treatment with
compounds 1 and 2 were used as the controls. Cell viability was
assessed using an MTT assay, as previously described (18).
Cell lipid accumulation assays
HepG2 cells (4×104 cells/well) were incubated in a
6-well plate (Costar; Corning Incorporated) for 24 h at 37°C. HepG2
cells cultured to 75% confluence were exposed to 1 mmol/l free
fatty acids (FFAs) for 24 h to assess hepatic lipid accumulation
and lipid peroxidation. The FFA mixture contained 1 mmol/l oleate
(cat no. O7501) and 1 mmol/l palmitate (cat no. P9767) (both from
Sigma-Aldrich; Merck KGaA) at a ratio of 2:1, and was diluted in
the culture medium to obtain the desired final concentration (1
mmol/l). In addition, the FFAs mixture contained BSA (10% w/v;
Sigma-Aldrich; Merck KGaA), as previously described (18). HepG2 cells, cultured to 75%
confluence, were treated with either DMEM containing BSA (10% w/v;
Sigma-Aldrich; Merck KGaA) as a control, or HepG2 cells, cultured
to 75% confluence, were treated with 1 mmol/l FFAs alone or
together with compounds 1 or 2 (25 µmol/l). A total of 24 h
following treatment at 37°C, cells were stained using Oil Red O to
assess intracellular lipid droplet accumulation, as previously
described by Cui et al (19).
To further investigate the effects of compounds 1
and 2 on intracellular lipid levels, HepG2 cells at 75% confluence
were treated for 24 h as aforementioned. FFA-containing medium was
removed and the cells were washed twice with PBS. The cells from
the various treatment groups were lysed in 1% Triton-X-100 (cat no.
T8787; Sigma-Aldrich; Merck KGaA) for 30 min on ice. The cell
lysates were determined using a BCA protein assay kit (Beyotime
Institute of Biotechnology) and were diluted in 1% Triton-X-100 to
obtain the final concentration of 5 mgprot/ml, then prepared for TG
level assessments using the Triglyceride Quantification
Colorimetric/Fluorometric kit (BioVision, Inc., Milpitas, CA, USA),
according to the manufacturer's protocol.
Cell lipid peroxidation assay
To further evaluate the effects of compounds 1 and 2
on intracellular lipid peroxidation, HepG2 cells at 75% confluence
were treated with compounds 1 or 2 (25 µmol/l), together with 1
mmol/l FFAs for 24 h. Cell lysates were obtained as aforementioned
using 1% Triton-X-100 to assess lipid peroxidation via measuring
MDA levels, using a commercially available MDA kit (cat no. A003-4;
Nanjing Jiancheng Bioengineering Institute), according to the
manufacturer's protocol.
Intracellular ROS production
HepG2 cells (1×104 cells/well) were incubated in a
24-well plate (Costar; Corning Incorporated) for 24 h at 37°C.
HepG2 cells at 75% confluence were plated in 24-well plates and
were treated with 1 mmol/l FFAs alone or together with 25 µmol/l
compound 2 for 24 h. Subsequently, cells were incubated with 10
µmol/l membrane-permeable oxidation-sensitive fluorescent dye
DCFH-DA (cat no. D6883; Sigma-Aldrich; Merck KGaA) for 20 min at
37°C. Stained cells were observed under an Eclipse Ti laser
scanning confocal microscope (Nikon Corporation, Tokyo, Japan) and
photomicrographs were captured. In addition, HepG2 cells were
treated with 1 mmol/l FFAs alone or together with compound 2 (25
µmol/l) for 24 h in a black opaque 96-well microplate (Corning
Incorporated). Subsequently, cells were incubated with 10 µmol/l
DCFH-DA for 20 min at 37°C. During this process, DCFH-DA is cleaved
and oxidized to green fluorescent 2′-7-′-dichlorofluorescein via
ROS mediation (DCF; excitation/emission, 488/530 nm), the level of
which was measured using the Synergy™ Mx microplate reader (BioTek
Instruments, Inc., Winooski, VT, USA).
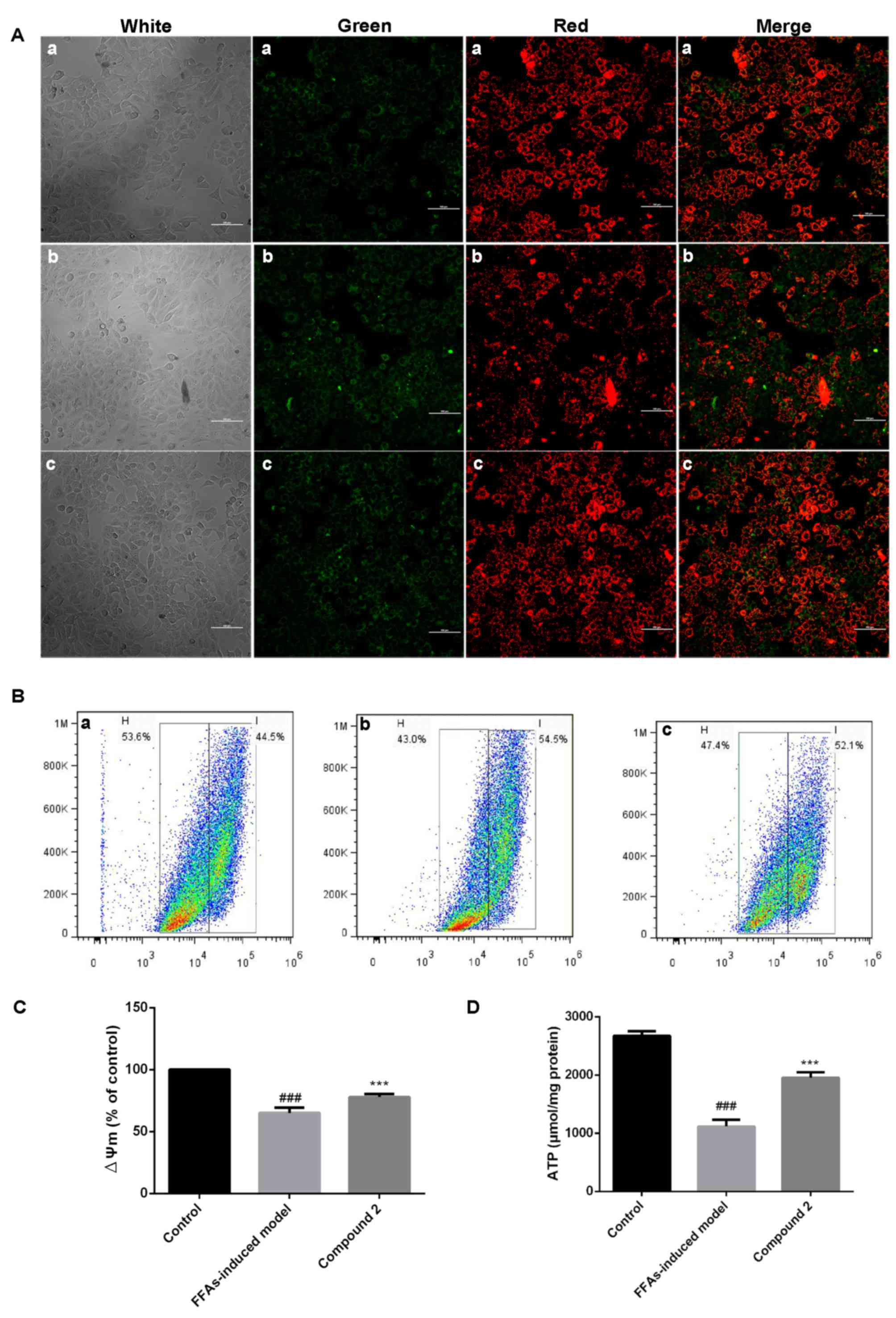
Mitochondrial membrane potential (∆Ψm)
analysis
HepG2 cells (1×104 cells/well) were incubated in a
24-well plate (Costar; Corning Incorporated) for 24 h at 37°C.
HepG2 cells at 75% confluence were treated with 1 mmol/l FFAs alone
or together with 25 µmol/l compound 2 for 24 h. Cells were stained
with 5 µg/ml JC-1 dye, as a ∆Ψm indicator, for 15 min (20), and then observed under an Eclipse
Ti laser scanning confocal microscope (Nikon Corporation). In
addition, HepG2 cells (4×104 cells/well) were plated in 6-well
plates for 24 h at 37°C, then treated with 1 mmol/l FFAs alone or
together with compound 2 (25 µmol/l) for 24 h at 37°C. Cells were
harvested by trypsinization, stained with 5 µg/ml JC-1 dye (cat no.
M34152; Thermo Fisher Scientific, Inc., Waltham, MA, USA) without
cell fixation for 15 min at 37°C, then washed twice with ice-cold
PBS and resuspended in 0.5 ml ice-cold FBS-free DMEM. The intensity
of fluorescence was determined using a MoFlo Cytomation, Modular
flow cytometer (Dako; Agilent Technologies, Inc., Santa Clara, CA,
USA) and the data were analyzed with Summit software version 4.3
(Cytomation, Inc., Fort Collins, CO, USA).
Intracellular adenosine triphosphate
(ATP) levels
HepG2 cells (4×104 cells/well) were incubated in a
6-well plate for 24 h at 37°C. HepG2 cells were then treated with 1
mmol/l FFAs alone or together with compound 2 (25 µmol/l) for 24 h.
Subsequently, cells were lysed using an ATP assay kit (cat no.
A22026; Invitrogen; Thermo Fisher Scientific, Inc.) according to
the manufacturer's instructions, centrifuged at 12,000 × g for 5
min at 4°C, and the supernatants were collected. Protein
concentration was determined using a BCA protein assay kit
(Beyotime Institute of Biotechnology) and cells were transferred to
a black opaque 96-well microplate (Corning Incorporated). Cellular
ATP levels were also assessed using the ATP assay kit (Invitrogen;
Thermo Fisher Scientific, Inc.) with the Synergy™ Mx microplate
reader (BioTek Instruments, Inc.), according to the manufacturer's
protocol (21).
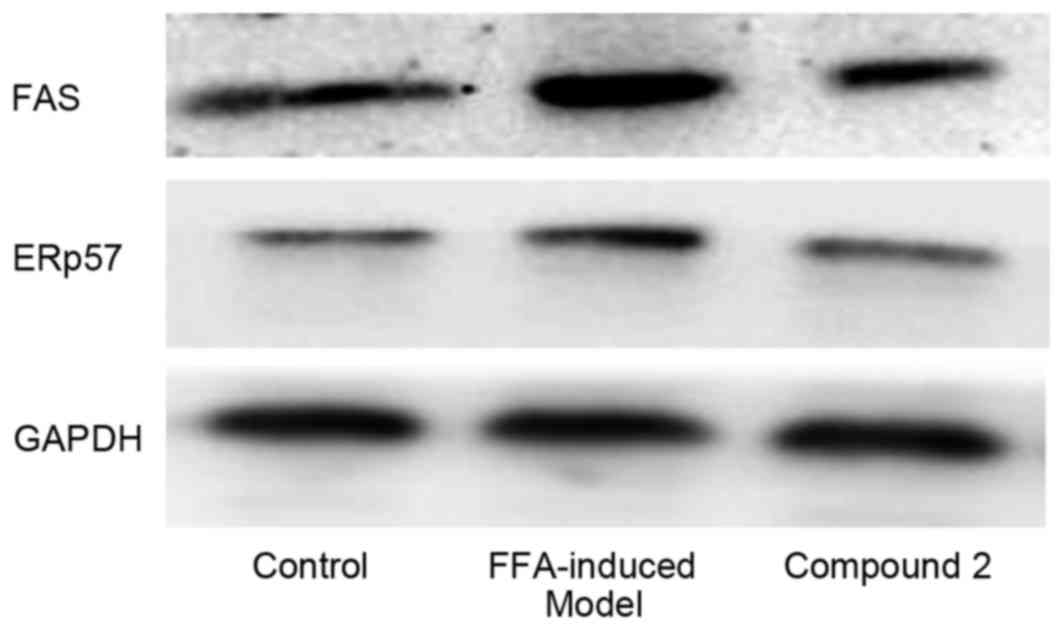
Western blot analysis
HepG2 cells were treated with 1 mmol/l FFAs alone or
together with compound 2 (25 µmol/l) for 24 h. Cells were lysed
using radioimmunoprecipitation assay lysis buffer (Beyotime
Institute of Biotechnology) containing 1 mmol/l phenylmethane
sulfonylfluoride for 20 min on ice. Subsequently, cell lysates were
centrifuged at 12,000 × g for 10 min at 4°C. The protein
concentration was determined using a BCA protein assay kit
(Beyotime Institute of Biotechnology). Equal amounts (40 µg) of
extracted protein samples were separated by 15% SDS-PAGE and
transferred onto a polyvinylidene fluoride membrane (EMD Millipore,
Billerica, MA, USA). The membrane was blocked with 5% non-fat milk
for 1 h at room temperature (~22°C), and then incubated with
anti-GAPDH, anti-ERp57 and anti-FAS primary antibodies at 4°C
overnight. Following washing three times with TBST (TBS containing
0.1% Tween-20; cat no. P0231; Beyotime Institute of Biotechnology),
the membranes were incubated with horseradish peroxidase-conjugated
goat anti-mouse and anti-rabbit IgG secondary antibodies at room
temperature for 2 h. Protein bands were visualized by enhanced
chemiluminescence using SuperSignal™ West Pico Chemiluminescent
Substrate (Thermo Fisher Scientific, Inc.).
Statistical analysis
The statistical significance of the differences
between groups was assessed using one-way analysis of variance
followed by a post hoc Scheffé's test for multiple comparisons.
Data are expressed as the mean ± standard deviation of three
repeated experiments. P<0.05 was considered to indicate a
statistically significant difference. Statistical analysis was
performed using SPSS software version 19.0 (IBM Corp.).
Results
Antihyperlipidemic effects of
SCAE
The present results demonstrated that SCAE (60
mg/kg) significantly decreased the serum levels of TC, TG and LDL-C
by ~26, 33 and 28%, respectively, whereas it increased the serum
levels of HDL-C by >2-fold, compared with the high-fat diet
group (P<0.05; Table I). In
addition, treatment with SCAE (60 mg/kg) resulted in a significant
increase in hepatic SOD and serum GSH-Px concentrations
(P<0.05), as well as a significant decrease in hepatic MDA
levels (P<0.05) in hyperlipidemic mice maintained on a high-fat
diet (Table II).
 | Table I.Lipid-lowering effects of SCAE on
high-fat diet-induced hyperlipidemic mice. |
Table I.
Lipid-lowering effects of SCAE on
high-fat diet-induced hyperlipidemic mice.
| Group | TC (mmol/l) | TG (mmol/l) | LDL-C (mmol/l) | HDL-C (mmol/l) |
|---|
| I | 2.01±0.51 | 0.73±0.37 | 0.46±0.11 | 1.5±0.13 |
| II |
3.98±0.78a |
1.66±0.61a |
0.99±0.13a |
0.41±0.18a |
| III |
2.96±0.61b |
1.11±0.42b |
0.71±0.12c |
0.82±0.18c |
 | Table II.Antioxidative effects of SCAE on
high-fat diet-induced hyperlipidemic mice. |
Table II.
Antioxidative effects of SCAE on
high-fat diet-induced hyperlipidemic mice.
| Group | Serum GSH-Px
(U/ml) | Serum NOS
(U/ml) | Liver SOD
(U/mgprot) | Liver MDA
(nmol/mgprot) |
|---|
| I | 1,162.76±81.33 | 22.54±2.21 | 61.31±2.85 | 2.18±0.42 |
| II |
776.74±42.10a |
19.33±2.03b |
43.22±2.35a |
4.59±0.61a |
| III |
991.22±22.53c | 21.05±1.43 |
55.9±2.89c |
1.94±0.37c |
The main compounds of SCAE were isolated using HPLC
and were identified as compound 1 and compound 2 by comparing the
NMR results with previous reports (22,23)
(Figs. 1 and 2). The antioxidative properties of
compounds 1 and 2 were assessed using free radical-scavenging DPPH
and lipid peroxidation TBA assays. Compound 2 was revealed to exert
more potent antioxidative effects compared with compound 1
(Table III).
 | Table III.IC50 of the antioxidative
capabilities of SCAE compounds 1 and 2 in vitro, and in
liver and kidney samples isolated from mice. |
Table III.
IC50 of the antioxidative
capabilities of SCAE compounds 1 and 2 in vitro, and in
liver and kidney samples isolated from mice.
|
|
IC50 |
|---|
|
|
|
|---|
| Assay | Compound 1
(µmol/l) | Compound 2
(µmol/l) |
|---|
| DPPH |
44.12±1.18 | 27.02±1.40 |
| TBA (liver) |
61.61±1.68 | 43.22±2.92 |
| TBA (kidney) | 140.97±9.11 | 59.97±3.30 |
Effects of compounds 1 and 2 on lipid
accumulation in HepG2 cells
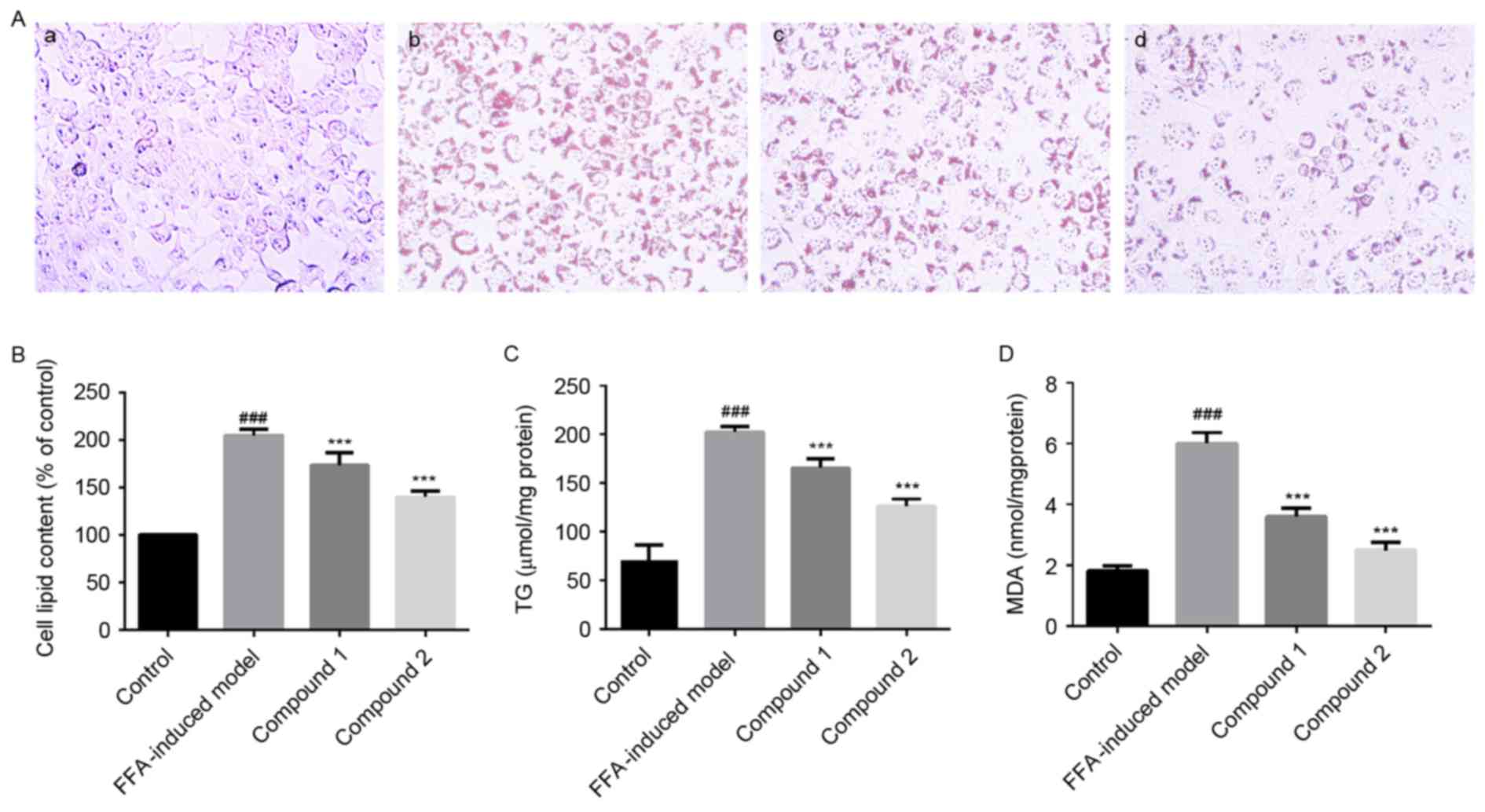
Following treatment of HepG2 cells with compounds 1
and 2, no detectable morphological changes and toxicity were
observed (data not shown). Treatment with compounds 1 and 2 (25
µmol/l) was demonstrated to significantly reduce lipid accumulation
in FFA-treated HepG2 cells (Fig. 3A
and B). In addition, compounds 1 and 2 significantly suppressed
the FFA-induced elevation in hepatocellular TG levels to 81 and
62%, respectively (P<0.001; Fig.
3C).
Effects of compounds 1 and 2 on lipid
peroxidation in HepG2 cells
As presented in Fig.
3D, lipid peroxidation was significantly enhanced in HepG2
cells exposed to 1 mmol/l FFAs compared with control cells.
However, treatment with compounds 1 and 2 was revealed to
significantly inhibit hepatic lipid peroxidation (P<0.001).
Notably, compound 2 appeared to exert more potent effects on
hepatic lipid accumulation and peroxidation compared with compound
1, thus suggesting that compound 2 may be characterized by higher
biological activity.
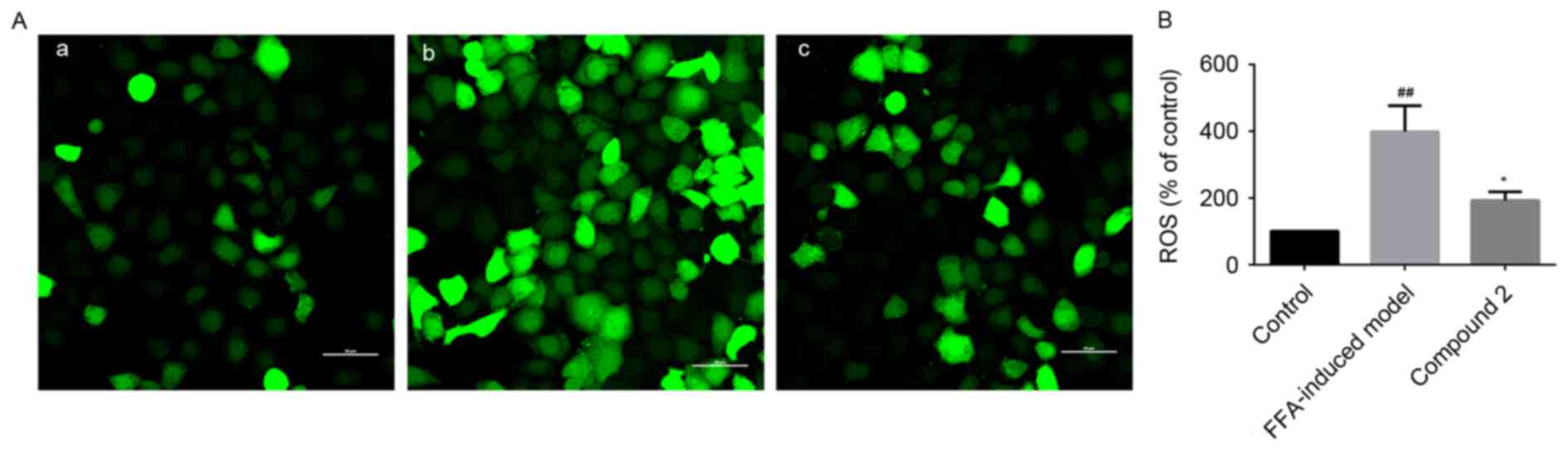
Effects of compound 2 on ROS
production in HepG2 cells
HepG2 cells exposed to FFAs exhibited increased
intracellular ROS production, as demonstrated by the increased
ROS-mediated oxidation of the acetate moieties of DCFH-DA to green
DCF. DCF fluorescence intensity was revealed to be increased by
4-fold in HepG2 cells exposed to FFAs compared with control cells.
Notably, compound 2 was demonstrated to significantly suppress the
FFA-induced increase in hepatic ROS generation (Fig. 4).
Effects of compound 2 on ∆Ψm
As presented in Fig.
5A, HepG2 cells exposed to FFAs demonstrated decreased ∆Ψm,
whereas treatment with compound 2 was revealed to reverse the
FFA-induced ∆Ψm decrease. Flow cytometric analysis also
demonstrated that HepG2 cells exposed to FFAs exhibited a
significant decrease (35%) in ∆Ψm, which was significantly
attenuated following treatment with compound 2 (Fig. 5B and C).
Effects of compound 2 on intracellular
ATP levels
Following exposure of HepG2 cells to FFAs,
intracellular ATP levels were significantly decreased, whereas
treatment with compound 2 was revealed to counter act the
FFA-induced decrease in ATP levels (Fig. 5D). These findings suggested that
compound 2 may ameliorate hepatic lipid accumulation due to its
protective effects on mitochondrial function, exerted through the
reduction in ROS production and the regulation of ∆Ψm and ATP
production.
Effects of compound 2 on the
expression of lipogenesis-associated proteins
As presented in Fig.
6, following exposure to FFAs for 24 h, the expression levels
of the lipogenesis-associated proteins ERp57 and FAS appeared to be
upregulated. Compound 2 was demonstrated to markedly attenuate the
FFA-induced upregulation in ERp57 and FAS expression. These results
suggested that compound 2 may suppress hepatic lipid accumulation
through the suppression of lipogenesis, via downregulating the
expression of proteins involved in lipogenesis, including ERp57 and
FAS.
Discussion
Hyperlipidemia has been identified as an important
risk factor for the development of atherosclerosis (1) and acute necrotic pancreatitis
(24). Coreopsis tinctoria
is a herbal medicine used to regulate lipid metabolism in
traditional Chinese medicine (9).
However, the exact pharmacological effects of Coreopsis
tinctoria, as well as the main active compounds and the
molecular mechanisms responsible for these effects, have yet to be
elucidated. In the present study, SCAE was demonstrated to decrease
serum lipid levels in a mouse model of hyperlipidemia, possibly due
to its antioxidative properties. Its main active compounds,
compounds 1 and 2, were revealed to decrease lipid accumulation in
HepG2 cells, possibly through the reduction of oxidative stress,
the protection of mitochondrial function and the suppression of
lipogenesis.
Administration of a high-fat diet has been reported
to increase fat and cholesterol intake, decrease the β-oxidation of
fatty acids and accelerate TG synthesis in rats, resulting in
increased TC and TG levels in the bloodstream (25). The present results suggested that
the flavonoid-rich SCAE may attenuate lipid disorders and regulate
TG levels. The liver is primarily responsible for lipid synthesis,
metabolism and transportation (13,26),
and hyperlipidemia has been reported to increase hepatic lipid
content, thus enhancing ROS generation and lipid peroxidation
(27). A previous study revealed
that flavonoids may attenuate hyperlipidemia, possibly due to their
potent antioxidative effects (25). The present study suggested that
SCAE may enhance the endogenous antioxidative defense mechanisms of
hepatocytes, thus ameliorating hyperlipidemia, due to its high
flavonoid content and potent antioxidative properties.
In the present study, compounds 1 and 2 were the
main compounds isolated from SCAE. Treatment of HepG2 cells with
FFAs leads to increased lipid accumulation, TG synthesis and lipid
peroxidation, and has been used to evaluate the effects of putative
lipid-lowering agents on lipid accumulation and lipid peroxidation
in vitro (18,28). The present results suggested that
compound 2 may be characterized by more potent lipid-lowering
capabilities compared with compound 1. In addition, compound 2
appeared to exert stronger antioxidative effects, as demonstrated
by its greater capabilities for scavenging free radicals and
inhibiting lipid peroxidation compared with compound 1. These
results suggested that compound 2 may be the main bioactive
compound of SCAE responsible for its lipid-lowering effects, due to
its potent antioxidative capabilities.
Mitochondria have been identified as the center of
cellular lipid metabolism and one of the main sources of
intracellular ROS generation (29,30).
Excessive fatty acid metabolism has been associated with increased
ROS generation, as well as decreased activity of antioxidant
enzymes, ultimately resulting in mitochondrial damage (3,10,25).
Malfunctioning mitochondria release higher quantities of ROS
(30), thus resulting in a vicious
cycle of mitochondrial dysfunction, decreased mitochondrial fatty
acid β-oxidation and increased TG synthesis. The present results
demonstrated that compound 2 counteracted the FFA-induced increase
in intracellular ROS production. Furthermore, it was revealed to
prevent the FFA-induced collapse of the ∆Ψm and the decrease in
cellular ATP levels, thus suggesting that compound 2 may protect
mitochondrial function.
The endoplasmic reticulum (ER) is known to serve a
central role in de novo lipogenesis. Oxidative and ER stress
have been reported to occur simultaneously or successively, and ER
stress has been associated with hepatic lipid accumulation
(31). The ER-associated protein
ERp57 has been revealed to be upregulated during FFA-induced
cellular steatosis, whereas its knockdown significantly reduced
lipid accumulation in steatotic cells (31). FAS has been identified as a key
enzyme during lipogenesis, as it catalyzes the terminal steps in
de novo fatty acid synthesis (32). The present results demonstrated
that compound 2 downregulated the protein expression levels of
ERp57 and FAS in FFA-treated HepG2 cells. These results suggested
that compound 2 may prevent de novo lipogenesis, via
suppressing the expression of ERp57 and FAS in hepatocytes.
In conclusion, the present study suggested that
compound 2 may be the main active compound of Coreopsis
tinctoria, responsible for its lipid-regulating effects.
Furthermore, compound 2 was demonstrated to enhance the endogenous
antioxidative defense mechanisms of hepatocytes and to protect
mitochondria against oxidative damage. In addition, its effects on
ER stress reduction and the inhibition of de novo
lipogenesis may be involved in the molecular mechanisms underlying
the lipid-lowering effects of SCAE. The present results suggested
that compound 2 may have potential for the development of novel
therapeutic strategies for the treatment of patients with
hyperlipidemia.
Acknowledgements
The present study was supported by the National
Natural Science Foundation of China (grant no. 81673710).
References
|
1
|
Abliz A, Aji Q, Abdusalam E, Sun X,
Abdurahman A, Zhou W, Moore N and Umar A: Effect of Cydonia oblonga
Mill. leaf extract on serum lipids and liver function in a rat
model of hyperlipidaemia. J Ethnopharmacol. 151:970–974. 2014.
View Article : Google Scholar : PubMed/NCBI
|
|
2
|
Cignarella A, Bellosta S, Corsini A and
Bolego C: Hypolipidemic therapy for the metabolic syndrome.
Pharmacol Res. 53:492–500. 2006. View Article : Google Scholar : PubMed/NCBI
|
|
3
|
Luedde T, Kaplowitz N and Schwabe RF: Cell
death and cell death responses in liver disease: Mechanisms and
clinical relevance. Gastroenterology. 147:765–783. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
4
|
Irudayaraj SS, Sunil C, Duraipandiyan V
and Ignacimuthu S: In vitro antioxidant and antihyperlipidemic
activities of Toddaliaasiatica (L) Lam. Leaves in Triton
WR-1339 and high fat diet induced hyperlipidemic rats. Food Chem
Toxicol. 60:135–140. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
5
|
Zhang Y, Shi S, Zhao M, Chai X and Tu P:
Coreosides A-D, C14-polyacetylene glycosides from the capitula of
Coreopsis tinctoria and its anti-inflammatory activity against
COX-2. Fitoterapia. 87:93–97. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
6
|
Zhang Y, Mourboul A and Li ZY: Research
advance in medicinal plants from genus Coreopsis. Zhongguo Zhong
Yao Za Zhi. 38:2633–2638. 2013.(In Chinese). PubMed/NCBI
|
|
7
|
Dias T, Bronze MR, Houghton PJ,
Mota-Filipe H and Paulo A: The flavonoid-rich fraction of Coreopsis
tinctoria promotes glucose tolerance regain through pancreatic
function recovery in streptozotocin-induced glucose-intolerant
rats. J Ethnopharmacol. 132:483–490. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
8
|
Dias T, Liu B, Jones P, Houghton PJ,
Mota-Filipe H and Paulo A: Cytoprotective effect of Coreopsis
tinctoria extracts and flavonoids on tBHP and cytokine-induced cell
injury in pancreatic MIN6 cells. J Ethnopharmacol. 139:485–492.
2012. View Article : Google Scholar : PubMed/NCBI
|
|
9
|
Li YL, Chen X, Xue J, Liu J, Chen X and
Wulasihan M: Flavonoids furom Coreopsis tinctoria adjust lipid
metabolism in hyperlipidemia animals by down-regulating adipose
differentiation-related protein. Lipids Health Dis. 13:1932014.
View Article : Google Scholar : PubMed/NCBI
|
|
10
|
Gong G, Qin Y, Huang W, Zhou S, Wu X, Yang
X, Zhao Y and Li D: Protective effects of diosgenin in the
hyperlipidemic rat model and in human vascular endothelial cells
against hydrogen peroxide-induced apoptosis. Chem-Biol Interact.
184:366–375. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
11
|
El-Demerdash FM and Nasr HM: Antioxidant
effect of selenium on lipid peroxidation, hyperlipidemia and
biochemical parameters in rats exposed to diazinon. J Trace Elem
Med Bio. 28:89–93. 2014. View Article : Google Scholar
|
|
12
|
Miri R, Saadati H, Ardi P and Firuzi O:
Alterations in oxidative stress biomarkers associated with mild
hyperlipidemia and smoking. Food Chem Toxicol. 50:920–926. 2012.
View Article : Google Scholar : PubMed/NCBI
|
|
13
|
Zarzecki MS, Araujo SM, Bortolotto VC, de
Paula MT, Jesse RJ and Prigol M: Hypolipidemic action of chrysin on
Triton WR-1339-induced hyperlipidemia in female C57BL/6 mice.
Toxicol Rep. 1:200–208. 2014. View Article : Google Scholar
|
|
14
|
Kim JS, Kang OJ and Gweon OC: Comparison
of phenolic acids and flavonoids in black garlic at different
thermal processing steps. J Funct Foods. 5:80–86. 2013. View Article : Google Scholar
|
|
15
|
Gerhäuser C, Klimo K, Heiss E, Neumann I,
Gamal-Eldeen A, Knauft J, Liu GY, Sitthimonchai S and Frank N:
Mechanism-based in vitro screening of potential cancer
chemopreventive agents. Mutat Res 523–524. 163–172. 2003.
View Article : Google Scholar
|
|
16
|
Hajiaghaalipour F, Kanthimathi MS, Sanusi
J and Rajarajeswaran J: White tea (Camellia sinensis) inhibits
proliferation of the colon cancer cell line, HT-29, activates
caspases and protects DNA of normal cells against oxidative damage.
Food Chem. 169:401–410. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
17
|
Grespan R, Aguiar RP, Giubilei FN, Fuso
RR, Damião MJ, Silva EL, Mikcha JG, Hernandes L, Amado C Bersani
and Cuman RK: Hepatoprotective effect of pretreatment with Thymus
vulgar is Essential Oil in experimental model of
acetaminophen-induced injury. Evid Based Complement Alternat Med.
2014:9541362014. View Article : Google Scholar : PubMed/NCBI
|
|
18
|
Seo MS, Hong SW, Yeon SH, Kim YM, Um KA,
Kim JH, Kim HJ, Chang KC and Park SW: Magnolia officinalis
attenuates free fatty acid-induced lipogenesis via AMPK
phosphorylation in hepatocytes. J Ethnopharmacol. 157:140–148.
2014. View Article : Google Scholar : PubMed/NCBI
|
|
19
|
Cui W, Chen SL and Hu KQ: Quantification
and mechanisms of oleic acid-induced steatosis in HepG2 cells. Am J
Transl Res. 2:95–104. 2010.PubMed/NCBI
|
|
20
|
Sung DK, Chang YS, Kang S, Song HY, Park
WS and Lee BH: Comparative evaluation of hypoxic-ischemic brain
injury by flow cytometric analysis of mitochondrial membrane
potential with JC-1 in neonatal rats. J Neurosci Methods.
193:232–238. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
21
|
Gyamfi D, Everitt HE, Tewfik I, Clemens DL
and Patel VB: Hepatic mitochondrial dysfunction induced by fatty
acids and ethanol. Free Radic Biol Med. 53:2131–2145. 2012.
View Article : Google Scholar : PubMed/NCBI
|
|
22
|
Pan J, Zhang S, Yan L, Tai J, Xiao Q, Zou
K, Zhou Y and Wu J: Separation of flavanone enantiomers and
flavanone glucoside diastereomers from Balanophora involucrata
Hook. F. by capillary electrophoresis and reversed-phase
high-performance liquid chromatography on a C18 column. J
Chromatogr A. 1185:117–129. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
23
|
Zhang Y, Shi S, Zhao M and Tu P: Novel
chalcone from Coreopsis tinctoria Nutt. Biochem Systemat Ecol.
34:766–769. 2006. View Article : Google Scholar
|
|
24
|
Czakó L, Szabolcs A, Vajda A, Csáti S,
Venglovecz V, Rakonczay Z Jr, Hegyi P, Tiszlavicz L, Csont T, Pósa
A, et al: Hyperlipidemia induced by a cholesterol-rich diet
aggravates necrotizing pancreatitis in rats. Eur J Pharmacol.
572:74–81. 2007. View Article : Google Scholar : PubMed/NCBI
|
|
25
|
Feng LJ, Yu CH, Ying KJ, Hua J and Dai XY:
Hypolipidemic and antioxidant effects of total flavonoids of
Perilla frutescens leaves in hyperlipidemia rats induced by
high-fat diet. Food Res Int. 44:404–409. 2011. View Article : Google Scholar
|
|
26
|
Neuschwander-Tetri BA: Hepatic
lipotoxicity and the pathogenesis of nonalcoholic steatohepatitis:
The central role of nontriglyceride fatty acid metabolites.
Hepatology. 52:774–788. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
27
|
Zhao Y, Peng L, Lu W, Wang Y, Huang X,
Gong C, He L, Hong J, Wu S and Jin X: Effect of Eclipta prostrata
on lipid metabolism in hyperlipidemic animals. Exp Gerontol.
62:37–44. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
28
|
Wu X, Zhang L, Gurley E, Studer E, Shang
J, Wang T, Wang C, Yan M, Jiang Z, Hylemon PB, et al: Prevention of
free fatty acid-induced hepatic lipotoxicity by
18beta-glycyrrhetinic acid through lysosomal and mitochondrial
pathways. Hepatology. 47:1905–1915. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
29
|
Schönfeld P, Więckowski MR, Lebiedzińska M
and Wojtczak L: Mitochondrial fatty acid oxidation and oxidative
stress: Lack of reverse electron transfer-associated production of
reactive oxygen species. Biochim Biophys Acta. 1797:929–938. 2010.
View Article : Google Scholar : PubMed/NCBI
|
|
30
|
Schönfeld P and Wojtczak L: Fatty acids as
modulators of the cellular production of reactive oxygen species.
Free Radic Biol Med. 45:231–241. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
31
|
Wang H, Chan PK, Pan SY, Kwon KH, Ye Y,
Chu JH, Fong WF, Tsui WM and Yu ZL: ERp57 is up-regulated in free
fatty acids-induced steatotic L-02 cells and human nonalcoholic
fatty livers. J Cell Biochem. 110:1447–1456. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
32
|
Menendez JA and Lupu R: Fatty acid
synthase and the lipogenic phenotype in cancer pathogenesis. Nat
Rev Cancer. 7:763–777. 2007. View
Article : Google Scholar : PubMed/NCBI
|