Introduction
Hepatic oval cells (HOCs) are thought to possess
self-renewal ability and a bipotential capacity for
differentiation, which allows them to differentiate into
hepatocytes and cholangiocytes (1). The differentiation of HOCs has been
reported to be regulated by various signaling pathways, including
Notch, Wnt, transforming growth factor-β, bone morphogenetic
protein, hepatocyte growth factor and fibroblast growth factor
signaling pathways (2).
Macroautophagy, which is also referred to as
autophagy, is a highly conserved and important metabolic process in
eukaryotic cells. Autophagy has been characterized as an essential
process associated with cellular homeostasis. Under stress or
starvation conditions, unnecessary or dysfunctional cellular
components are degraded and recycled through autophagy, thus
promoting cell survival and growth (3). Autophagy, which is initiated by the
microenvironment or cytokines, serves a role in various
physiological and pathological processes, including cancer,
metabolic disease, neurodegenerative disorders, cell growth, cell
death, ontogeny, and cell differentiation and self-renewal
(4). However, how autophagy
contributes to proliferation and differentiation of HOCs remains to
be elucidated.
Mitogen-activated protein kinases (MAPKs) are a
family of serine/threonine kinases, which function as broad
intracellular signal mediators of cell survival, proliferation,
motility, apoptosis and differentiation (5). In addition, MAPKs are essential for
stem cell self-renewal and commitment to lineage differentiation
(6). Extracellular
signal-regulated kinase (ERK) 1/2 is required for osteogenic
mesenchymal stem cell differentiation and skeletal development
(7). It has previously been
reported that MAPK/ERK serves an important role in the
proliferation of HOCs (8).
Furthermore, induction of autophagy is associated with epidermal
growth factor-induced MAPK phosphorylation. However, the effects of
autophagy on the MAPK/ERK signaling pathways in HOC proliferation
and biliary differentiation are currently not well understood.
Therefore, to investigate these effects, as well as the underlying
molecular mechanisms, the present study investigated the effects of
autophagy on HOC proliferation, apoptosis and differentiation into
cholangiocytes, and the role of the MAPK/ERK signaling pathway in
these processes.
Materials and methods
Cell lines, culture and drug
treatment
The WB-F344 HOC line was purchased from the Cell
Bank of the Type Culture Collection of the Chinese Academy of
Sciences (Shanghai, China). This cell line possesses stem cell
features and has been reported previously (9). WB-F344 cells were cultured in a 25
cm3 flask with RPMI-1640 medium, supplemented with 10%
(v/v) fetal bovine serum (FBS) (both from (Gibco; Thermo Fisher
Scientific, Inc., Waltham, MA, USA), 100 µg/ml streptomycin
(Sigma-Aldrich; Merck KGaA, Darmstadt, Germany) and 100 U/ml
penicillin (Gibco; Thermo Fisher Scientific, Inc.) in a humidified
incubator containing 5% CO2 at 37°C. The culture medium
was changed every second day and cells were passaged 1:3 using
0.05% trypsin plus 0.02% EDTA. To regulate autophagy in WB-F344
cells, chloroquine (Chlo; 100 mg) and rapamycin (Rapa; 1.0 mg/l)
(both from Sigma-Aldrich; Merck KGaA, Darmstadt, Germany) were
administered to inhibit or induce autophagy, respectively (10). Briefly, WB-F344 cells were seeded
in 6-well culture plates at 3×105 cells/well and were
cultured overnight. The following day, the cells were treated with
or without Rapa (40 nM) or Chlo (50 µM) for 24 h at 37°C. For the
following experiments, Rapa and Chlo were dissolved in dimethyl
sulfoxide (DMSO; Sigma-Aldrich; Merck KGaA). Control cells were
treated with an equal amount of DMSO used for drug treatment [not
exceeding 0.1% (v/v)]. Morphology of WB-F344 cells was evaluated by
a light microscope (Olympus CX21; Olympus Corporation, Tokyo,
Japan) after Rapa or Chlo treatment.
Biliary differentiation of hepatic
progenitor cells
WB-F344 cells had been used as a good model to study
the mechanism of biliary differentiation (11). To induce biliary differentiation,
WB-F344 cells which were seeded in 6-well culture plates at a
density of 5×105 cells/well were treated with 3.75 mM
sodium butyrate (SB; Sigma-Aldrich; Merck KGaA) (11). The medium was changed every 2 days.
The days of differentiation were numbered consecutively beginning
at the first day of the SB treatment (0 day) to the last day (3
day).
Determination of cell viability
Cell viability was detected using the Cell Counting
Kit (CCK)-8 assay. WB-F344 cells were plated in 96-well plates with
three duplicate wells in each group. When the cells had grown to
70–80% confluency, the cells were treated with or without Rapa (30
and 700 nM) or Chlo (20–500 µM). The CCK-8 solution (10 µl) at a
1:10 dilution in FBS-free RPMI-1640 medium (100 µl) was added to
each well, and the plates were incubated for 2 h at 37°C.
Absorbance was measured at 450 nm using a microplate reader
(Molecular Devices, LLC, Sunnyvale, CA, USA). The mean optical
density (OD) of three wells in the indicated groups was used to
calculate the inhibition rate of the cells as follows: Inhibition
rate = (ODcontrol group - ODtreatment
group)/(ODcontrol group - ODblank
group) × 100%. The experiment was performed in
quadruplicate.
Immunofluorescence staining
After treatment, 3×106 cells in 6-well
plates, were fixed with 4% paraformaldehyde and incubated with CK19
(cat. no. 3092, 1:200; Cell Signaling Technology, Leiden, the
Netherlands) overnight at 4°C. And then incubated with Alexa Fluor
488-labeled secondary antibodies (cat. no. A-11034, 1:200;
Molecular Probes; Thermo Fisher Scientific, Inc.) for 1 h at 37°C.
DAPI (Sigma-Aldrich; Merck KGaA) was used to stain the nuclei. Five
fields from each well were taken at magnification, ×200.
Fluorescence intensity was evaluated by using a confocal microscope
(Leica TCS SP2; Leica Microsystems, GmbH, Wetzlar, Germany).
Flow cytometry to determine the
effects of autophagy on apoptosis
To analyze cell cycle progression and to assess
apoptosis, flow cytometric analysis was used to identify
sub-G1 cells/apoptotic cells and to measure the
percentage of sub-G1 cells after propidium iodide (PI)
staining in hypotonic buffer as previously described (12). The cells were collected at 24 h
after drug treatment and apoptotic cells were analyzed by flow
cytometry using a fluorescein isothiocyanate (FITC)-Annexin V
Apoptosis Detection kit (cat. no. 556547; BD Biosciences, Franklin
Lakes, NJ, USA). Briefly, following trypsinization and three washes
in PBS, cells were resuspended in 500 µl 1X Annexin V binding
buffer, 5 µl FITC-Annexin V and 5 µl PI, and were incubated in the
dark for 15 min at 37°C after gentle mixing. Following the addition
of 200 µl 1X Annexin V binding buffer, apoptosis was analyzed by
flow cytometry (BD FACSCanto II; BD Biosciences, San Jose, CA, USA)
within 1 h. For cell cycle analysis after 24 h, after drug
treatment cells were washed with cold PBS for three times and fixed
in 70% ethyl alcohol at 4°C overnight. Cells were then treated with
10 µg/ml RNase and were stained with 50 µg/ml PI for 30 min at room
temperature in the dark. The cell cycle was then measured by BD
FACSCanto II and the cell cycle distribution was analyzed by ModFit
LT v3.3 software (BD Biosciences).
Western blot analysis
Western blot analysis was performed as described
previously (12). Briefly,
following cell lysis and protein extraction with
radioimmunoprecipitation assay lysis buffer (Beyotime Institute of
Biotechnology, Haimen, China), protein concentration was measured
using the bicinchoninic acid protein assay reagent. The samples (50
µg) were then denatured in sample buffer which containing SDS and
the proteins were separated by 10–12% SDS-PAGE gels. The proteins
were then electroblotted onto polyvinylidene difluoride membranes.
After blocking with 5% skimmed milk for 1 h at room temperature,
the blots were incubated with rabbit anti-microtubule-associated
proteins 1A/1B light chain 3B (LC3B; cat. no. 4108, 1:1,000),
rabbit anti-p62 (cat. no. 5114, 1:1,000), rabbit anti-cytokeratin
19 (CK19; cat. no. 13092, 1:1,000), and rabbit anti-cleaved PARP
(cat. no. 9545, 1:1,000) (all from CST Biological Reagents Co.,
Ltd., Shanghai, China), rabbit anti-Bcl-2 (cat. no. ab59348,
1:1,000) or anti-GAPDH (CST Biological Reagents CO., Ltd., cat. no.
5174, 1:1,000) antibodies (1:2,000) overnight at 4°C. Membranes
were also incubated with anti-phosphorylated (p)-p38 MAPK
(Thr180/Tyr182; cat. no. 9215, 1:1,000) and p-p44/42 MAPK (ERK1/2;
cat. no. 41370, 1:1,000) (both from CST Biological Reagents Co.,
Ltd.) antibodies (Cell Signaling Technology, Inc., Danvers, MA,
USA) at a 1:1,000 dilutions. Blots were stripped and re-probed with
anti-p38 MAPK (cat. no. 9212l; Abcam, Cambridge, MA, USA) and
anti-p44/42 MAPK (ERK1/2; cat. no. 4695, 1:1,000; CST Biological
Reagents Co., Ltd.) antibodies at a 1:2,000 dilution. After three
washes with Tris-buffered saline containing 0.05% Tween-20,
membranes were incubated with horseradish peroxidase-conjugated
secondary antibodies (cat. no. GHPA002, 1:2,000; Xi'an Guanyu
Bio-Tech Co., Ltd., Xi'an, China) for 1 h at room temperature. The
membranes were washed a further three times for 5 min. Blots were
visualized using the Pierce Enhanced Chemiluminescence Western
Blotting Substrate (Thermo Fisher Scientific, Inc.) followed by
autoradiography. Grey value analysis was performed using Image Lab
software (version 5.2.1; Bio-Rad Laboratories, Inc., Hercules, CA,
USA). To ensure equal loading, membranes were probed with an
anti-GAPDH antibody.
Statistical analysis
All experiments were repeated three times. Data are
presented as the mean ± standard error and were analyzed using SPSS
13.0 software (SPSS Inc., Chicago, IL, USA). Data were
statistically analyzed by one-way analysis of variance followed by
Dunnett's test for multiple comparisons. P<0.05 was considered
to indicate a statistically significant difference.
Results
Pharmacological regulation of
autophagic activity in WB-F344 cells
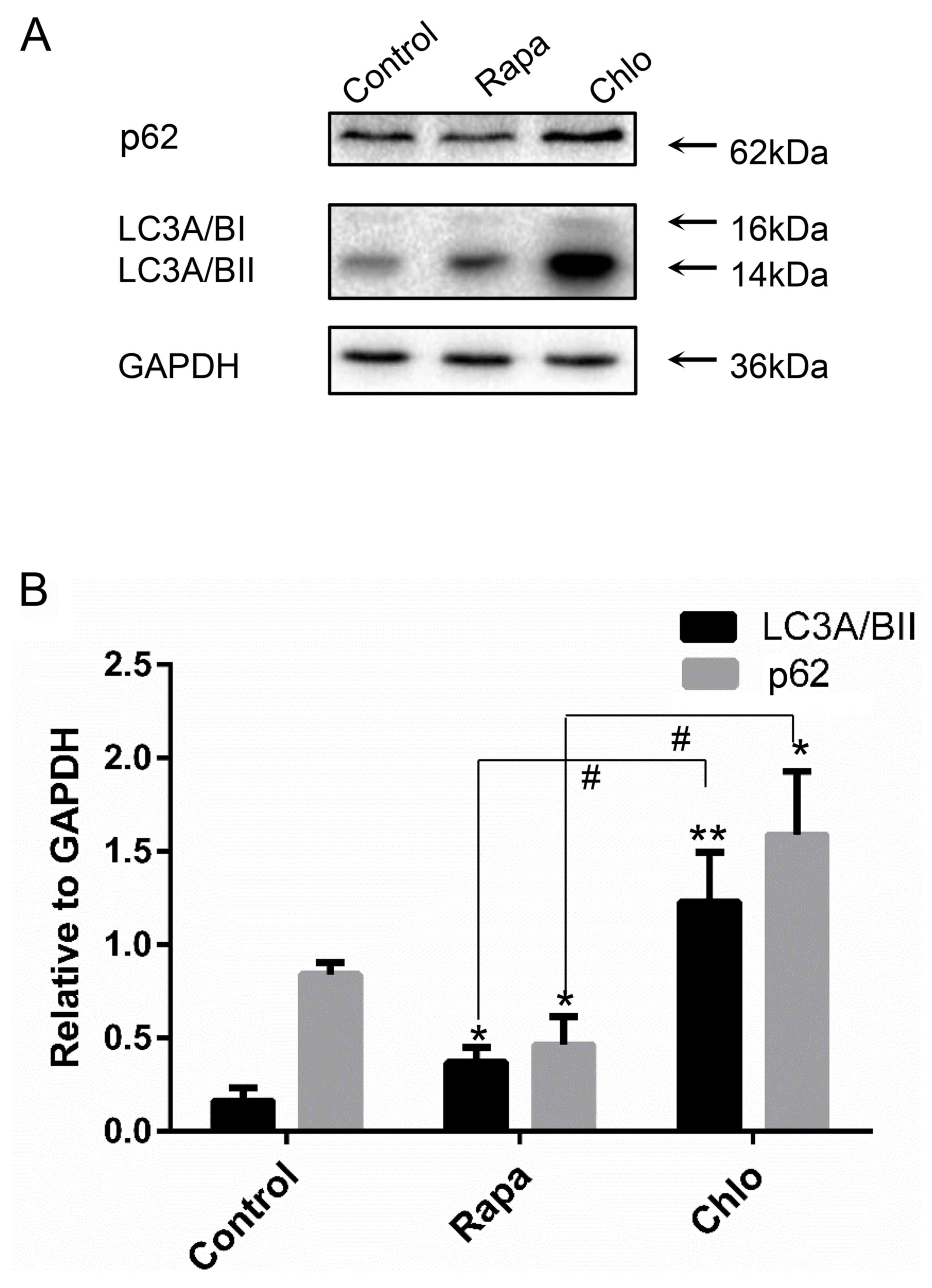
The present study detected the autophagic activity
of WB-F344 cells following treatment with Chlo or Rapa for 24 h.
Treatment with Rapa, which is known to induce autophagy, for 24 h
promoted the ratio of LC3A/BII and LC3A/BI and the degradation of
p62/sequestome 1, which is a selective substrate of autophagy.
Conversely, treatment with the autophagy inhibitor Chlo
significantly enhanced LC3A/BII and p62 expression (Fig. 1A and B). These results indicated
that Rapa was able to enhance autophagy, whereas Chlo inhibited
autophagy in WB-F344 cells.
Inhibition of autophagy decreases
WB-F344 cell proliferation and induces WB-F344 cell apoptosis
Proliferation of WB-F344 cells was measured 24 h
after Rapa or Chlo treatment. As presented in Fig. 2A and B, Chlo significantly
decreased the proliferation of WB-F344 cells (P<0.05). The
anti-proliferative effects of Chlo on WB-F344 cells were verified
using the CCK-8 assay, which can be used to examine cell
proliferation. When the cells were treated with 20–500 µmol/l Chlo
for 24 h the proliferation of WB-F344 cells was inhibited in a
time- and dose-dependent manner (Fig.
2B). However, Rapa, which induces autophagy, had no significant
effect on viability (Fig. 2B). To
determine whether cell cycle progression was affected by Rapa or
Chlo treatment, a cell cycle analysis was conducted using flow
cytometry. Rapa (0.4%±0.8) did not alter cell cycle progression,
which is consistent with the findings of the CCK-8 assay. However,
following treatment with 50 µM Chlo (2.8%±1.2), subG1
cell cycle arrest was increased compare with the control group
(0.5%±0.9, P<0.05; Fig. 2C).
These findings indicated that Chlo induces apoptosis of WB-F344
cells. In order to confirm the apoptotic effects of Chlo, cellular
apoptosis was determined by Annexin V-PI staining and flow
cytometry. Flow cytometric analysis demonstrated that treatment
with the autophagy inducer Rapa slightly reduced the apoptotic rate
of WB-F344 cells (3.4%±0.6); however, this was not significantly
different compared with the control group. (5.4%±0.7; Fig. 2D). Following treatment with the
autophagy inhibitor Chlo, the apoptotic rate of WB-F344 cells
(16.6%±1.1) was significantly enhanced compared with the control
group (P<0.05; 2D). In Fig. 2E and
F, in the Rapa group, Rapa increased the expression of BCL-2
and decreased the expression of cleaved PARP (P<0.01). However,
the results of chlo were the opposite of that of rapa (P<0.05).
These results indicated that autophagy inhibits apoptosis, whereas
inhibition of autophagy promotes apoptosis of WB-F344 cells. But in
Fig. 2D. this was not
significantly different compared with the control group. We
believed that autophagy can reduce apoptosis, but autophagy can
also promote cell death. Therefore, the apoptosis of Rapa group was
not obvious compared with the control group.
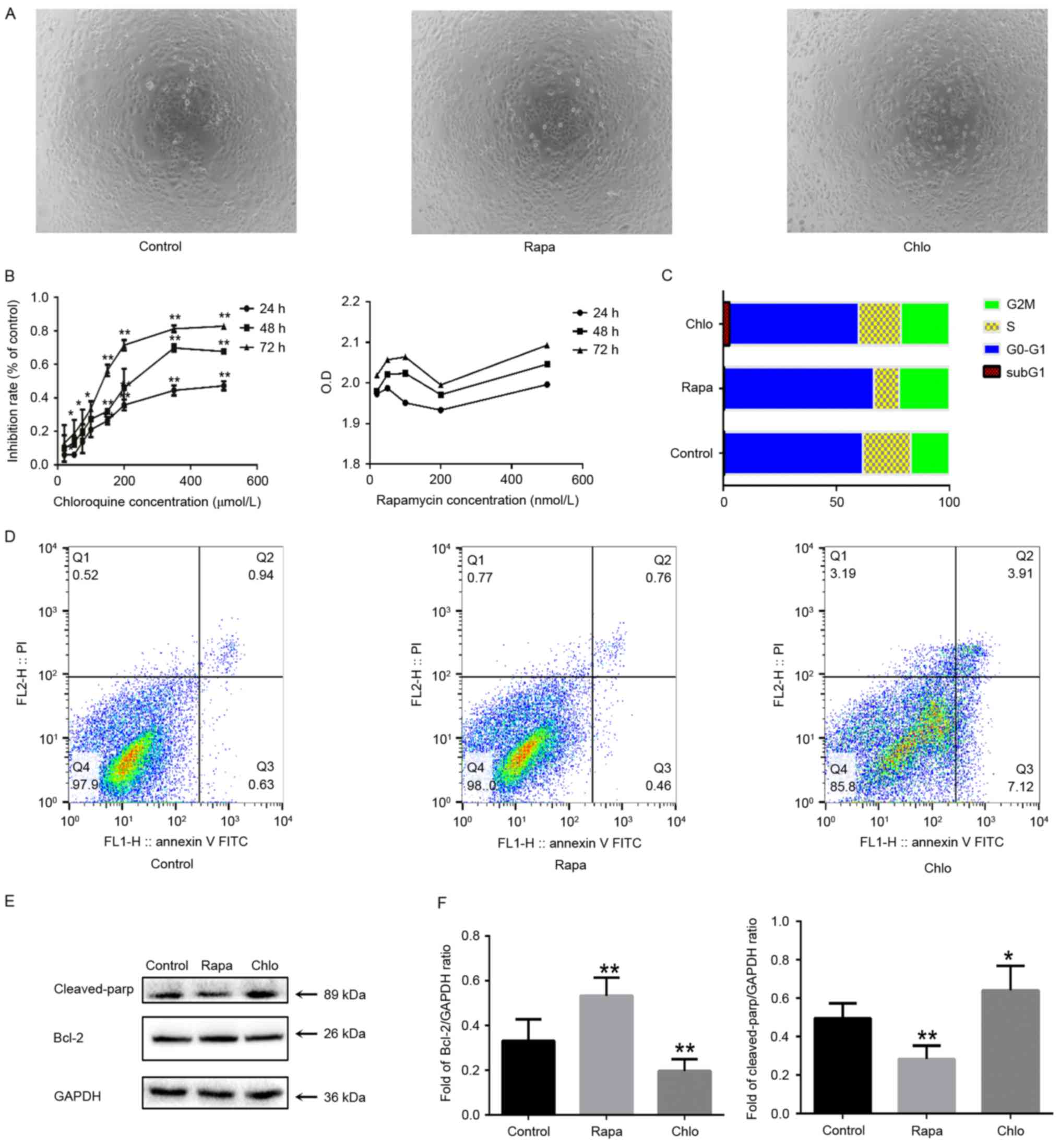 | Figure 2.Chlo decreases the proliferation of
WB-F344 cells and induces apoptosis. (A) Morphology of WB-F344
cells 24 h after Rapa or Chlo treatment (magnification, ×40). (B)
Chlo (20–500 µM) significantly decreased the proliferation of
WB-F344 cells, as determined by Cell Counting Kit-8 assay. Data are
presented as the mean ± standard deviation (n=3). *P<0.05,
**P<0.01 vs. the control group. (C) Cell cycle analysis
indicated that 50 µM Chlo increased cell cycle arrest at SubG1,
which may induce apoptosis. (D) WB-F344 cells were analyzed by
Annexin V and PI staining followed by flow cytometry. (E and F)
Western blot analysis of the expression levels of cleaved PARP and
Bcl-2 in WB-F344 cells. *P<0.05, **P<0.01 vs. the control
group. Bcl-2, B-cell lymphoma 2; Chlo, chloroquine; FITC,
fluorescein isothiocyanate; OD, optical density; PARP,
poly(ADP-ribose) polymerase; PI, propidium iodide; Rapa,
rapamycin. |
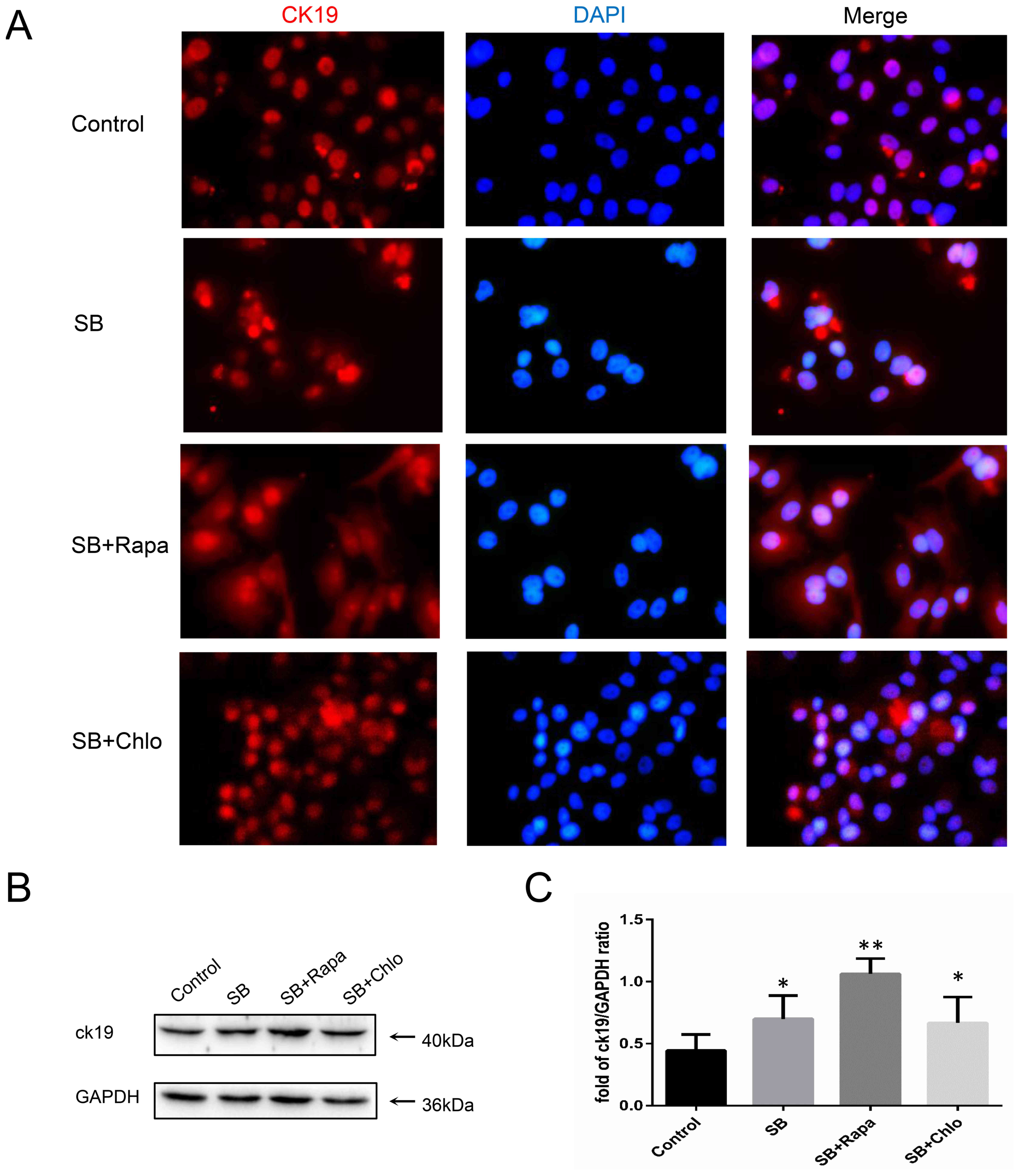
Autophagy enhances the biliary
differentiation of WB-F344 cells
Since autophagy can promote proliferation of WB-F344
cells, the present study aimed to determine whether autophagy
exerts an effect on the differentiation of WB-F344 cells.
Therefore, the effects of Rapa and Chlo were determined on the
biliary differentiation of WB-F344 cells. WB-F344 cells were
co-treated with sodium butyrate (SB) and Rapa or Chlo for 3 days.
As presented in Fig. 3A, when
WB-F344 cells were treated with SB, CK19 was slightly expressed in
the cytoplasm; however, CK19 protein expression was significantly
increased in the Rapa group compared with the control group
(P<0.01). Furthermore, the results of the western blot analysis
were consistent with the findings of immunofluorescence; upon
biliary differentiation, the expression of CK19 was increased in
the Rapa-treated group compared with in the control group
(P<0.01; Fig. 3B and C). These
results indicated that activation of autophagy promotes the biliary
differentiation of WB-F344 cells.
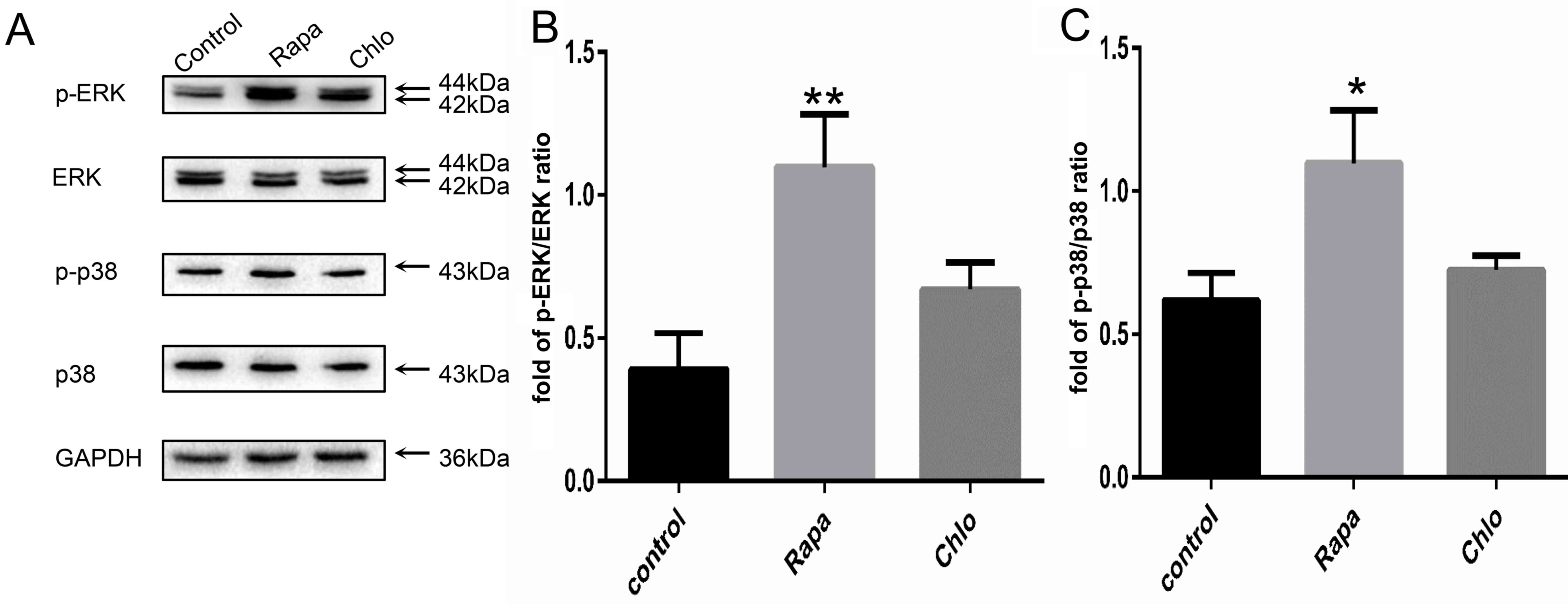
Effects of autophagy on the MAPK/ERK
signaling pathway in WB-F344 cells
To determine the effects of MAPK/ERK signaling on
autophagy-mediated enhancement of biliary differentiation of
WB-F344 cells, western blotting was used to detect the expression
levels of MAPK/ERK after modulation of autophagy. The results
demonstrated that treatment with the autophagy inducer Rapa
increased the phosphorylation of ERK1/2 and p38 (Fig. 4A) compared with in the control
group. Semi-quantification of the blots indicated that the levels
of p-ERK1/2 and p-p38 were increased ~2-fold following Rapa
treatment compared with the control group (Fig. 4B and C). However, the expression
levels of p-ERK1/2 and p-p38 were not significantly altered in the
Chlo group. These results suggested that autophagy may promote
differentiation of WB-F344 cells by upregulating the MAPK/ERK
signaling pathway.
Discussion
Autophagy has been demonstrated to protect cells
from stress conditions, including hypoxia, and to serve an
important role in self-renewal, proliferation, differentiation and
apoptosis of stem cells (13–16).
In addition, autophagy can efficiently and rapidly downregulate
enzymes, transcription factors, adhesion molecules and certain
secreted proteins (17,18), which are important for stem cell
proliferation and differentiation (19,20).
Autophagy mediates the degradation of cellular components,
including proteins and organelles, in lysosomes (21), and contributes to cellular
homeostasis, quality control and maintenance of energetic balance,
which are essential for cell proliferation (15). In epidermal stem cells, mesenchymal
stem cells, hematopoietic stem cells and dermal stem cells, basal
autophagy levels are higher (22–26).
After the induction of differentiation, autophagic levels are
decreased. Nevertheless, autophagy is increased during the
differentiation of embryonic stem cells, neural stem cells and
cardiac stem cells (27–32). It has also been reported that
autophagy serves a crucial role in regulating the stemness of
hepatic progenitor cells (HPCs) (33). High autophagic levels are detected
in the majority of the stem cells when they differentiate into
other cells, which is essential for the maintenance of cellular
stemness and homeostasis. There are many studies of the role of
pathways on autophagy (34,35).
Therefore, the present study investigated the effects of autophagy
on proliferation and differentiation of HOCs.
Stem cells undergo self-renewal and have the
potential to differentiate into various cell populations. It has
been established that a bipotential cell type, which has stem
cell-like properties, exists in the adult liver and can
differentiate into hepatic cells and cholangiocytes, these cells
are referred to as HOCs or HPCs (36). HOCs are activated in reaction to
various types of chronic liver disease (37).
It has been reported that autophagy regulates
biliary differentiation of HPCs via the Notch1 signaling pathway
(38). However, the role of
autophagy in the proliferation, apoptosis and differentiation of
WB-F344 cells remains to be elucidated. In the present study, Chlo
was used to inhibit autophagy in WB-F344 cells, and was
demonstrated to increase the SubG1 population of cells
and promote apoptosis, whereas treatment with Rapa exerted the
opposite effects, thus suggesting that inhibition of autophagy
inhibits WB-F344 cell proliferation. These results are consistent
with previous studies, which reported that inhibition of autophagy
suppresses proliferation and increases the population of apoptotic
cells (39,40). Although autophagy and apoptosis are
mutually inhibitory (41), in the
present study, autophagy appeared to exert anti-apoptotic
effects.
The present study also aimed to determine the
effects of autophagy on HOC-cholangiocyte differentiation, the
results confirmed that SB induced WB-F344 cell differentiation into
a cholangiocyte phenotype and revealed that the expression of the
biliary marker, CK19, was significantly increased in response to
Rapa-induced autophagy. These findings suggested that autophagy may
be involved in WB-F344 cell differentiation. Conversely, a previous
study by Zeng et al (38)
demonstrated that activation of autophagy may suppress the biliary
differentiation of WB-F344 cells.
Further investigation suggested that Rapa-induced
activation of autophagy may stimulate the MAPK/ERK signaling
pathway. These results are consistent with those of previous
studies, which indicated that activation of the MAPK/ERK pathway
can be induced by Rapa (42,43),
and may promote HOC proliferation (8). Therefore, the ERK signaling pathway
may be considered an important regulator for the proliferation and
differentiation of WB-F344 cells (8). These findings suggested that
activation of MAPK/ERK signaling induced by autophagy is essential
for cell differentiation.
HOCs are clinically significant, as they are
potentially useful for cell and/or gene therapy for the treatment
of metabolic liver diseases (37).
However, the role of autophagy in WB-F344 cells involves
alterations in signaling pathways, such as in Akt and Hedgehog, and
the mechanisms are still highly heterogeneous (34,35).
The present study, demonstrated that inhibition of autophagy can
inhibit HOCs proliferation and autophagy promoting HOCs
differentiation. In addition, MAPK/ERK pathway is likely to be
involved. Future studies are required to explore how autophagy
affects the MAPK/ERK pathway during liver regeneration, and to
reveal the potential mechanisms underlying HOC proliferation and
differentiation, thus providing subclinical strategies for
HOC-mediated liver regeneration or the inhibition of HOC-mediated
tumorigenesis.
Acknowledgements
The present study was supported by grants from the
Zhejiang Province Natural Science Foundation of China (grant no.
LY12H03006).
Glossary
Abbreviations
Abbreviations:
|
CK19
|
cytokeratin 19
|
|
HOC
|
hepatic oval cell
|
|
Rapa
|
rapamycin
|
|
Chlo
|
chloroquine
|
|
SB
|
sodium butyrate
|
|
MAPK
|
mitogen-activated protein kinase
|
References
|
1
|
Miyajima A, Tanaka M and Itoh T:
Stem/progenitor cells in liver development, homeostasis,
regeneration, and reprogramming. Cell Stem Cell. 14:561–574. 2014.
View Article : Google Scholar : PubMed/NCBI
|
|
2
|
Mishra L, Banker T, Murray J, Byers S,
Thenappan A, He AR, Shetty K, Johnson L and Reddy EP: Liver stem
cells and hepatocellular carcinoma. Hepatology. 49:318–329. 2009.
View Article : Google Scholar : PubMed/NCBI
|
|
3
|
Lapierre LR, Gelino S, Meléndez A and
Hansen M: Autophagy and lipid metabolism coordinately modulate life
span in germline-less C. elegans. Curr Biol. 21:1507–1514. 2011.
View Article : Google Scholar : PubMed/NCBI
|
|
4
|
Choi AM, Ryter SW and Levine B: Autophagy
in human health and disease. N Engl J Med. 368:651–662. 2013.
View Article : Google Scholar : PubMed/NCBI
|
|
5
|
Chang L and Karin M: Mammalian MAP kinase
signalling cascades. Nature. 410:37–40. 2001. View Article : Google Scholar : PubMed/NCBI
|
|
6
|
Kunath T, Saba-El-Leil MK, Almousailleakh
M, Wray J, Meloche S and Smith A: FGF stimulation of the Erk1/2
signalling cascade triggers transition of pluripotent embryonic
stem cells from self-renewal to lineage commitment. Development.
134:2895–2902. 2007. View Article : Google Scholar : PubMed/NCBI
|
|
7
|
Ge C, Xiao G, Jiang D and Franceschi RT:
Critical role of the extracellular signal-regulated kinase-MAPK
pathway in osteoblast differentiation and skeletal development. J
Cell Biol. 176:709–718. 2007. View Article : Google Scholar : PubMed/NCBI
|
|
8
|
Wang HY, Yang SL, Liang HF and Li CH: HBx
protein promotes oval cell proliferation by up-regulation of cyclin
D1 via activation of the MEK/ERK and PI3K/Akt pathways. Int J Mol
Sci. 15:3507–3518. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
9
|
Oh SH, Hatch HM and Petersen BE: Hepatic
oval ‘stem’ cell in liver regeneration. Semin Cell Dev Biol.
13:405–409. 2002. View Article : Google Scholar : PubMed/NCBI
|
|
10
|
Zhang Q, Yang YJ, Wang H, Dong QT, Wang
TJ, Qian HY and Xu H: Autophagy activation: A novel mechanism of
atorvastatin to protect mesenchymal stem cells from hypoxia and
serum deprivation via AMP-activated protein kinase/mammalian target
of rapamycin pathway. Stem Cells Dev. 21:1321–1332. 2012.
View Article : Google Scholar : PubMed/NCBI
|
|
11
|
Couchie D, Holic N, Chobert MN, Corlu A
and Laperche Y: In vitro differentiation of WB-F344 rat liver
epithelial cells into the biliary lineage. Differentiation.
69:209–215. 2002. View Article : Google Scholar : PubMed/NCBI
|
|
12
|
She M, Pan I, Sun L and Yeung SC:
Enhancement of manumycin A-induced apoptosis by methoxyamine in
myeloid leukemia cells. Leukemia. 19:595–602. 2005. View Article : Google Scholar : PubMed/NCBI
|
|
13
|
Mizushima N and Levine B: Autophagy in
mammalian development and differentiation. Nat Cell Biol.
12:823–830. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
14
|
Phadwal K, Watson AS and Simon AK:
Tightrope act: Autophagy in stem cell renewal, differentiation,
proliferation, and aging. Cell Mol Life Sci. 70:89–103. 2013.
View Article : Google Scholar : PubMed/NCBI
|
|
15
|
Mizushima N, Levine B, Cuervo AM and
Klionsky DJ: Autophagy fights disease through cellular
self-digestion. Nature. 451:1069–1075. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
16
|
Guan JL, Simon AK, Prescott M, Menendez
JA, Liu F, Wang F, Wang C, Wolvetang E, Vazquez-Martin A and Zhang
J: Autophagy in stem cells. Autophagy. 9:830–849. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
17
|
Zirin J and Perrimon N: Drosophila as a
model system to study autophagy. Semin Immunopathol. 32:363–372.
2010. View Article : Google Scholar : PubMed/NCBI
|
|
18
|
Rubinsztein DC, Gestwicki JE, Murphy LO
and Klionsky DJ: Potential therapeutic applications of autophagy.
Nat Rev Drug Discov. 6:304–312. 2007. View
Article : Google Scholar : PubMed/NCBI
|
|
19
|
Coller HA, Sang L and Roberts JM: A new
description of cellular quiescence. PLoS Biol. 4:e832006.
View Article : Google Scholar : PubMed/NCBI
|
|
20
|
Mortensen M, Watson AS and Simon AK: Lack
of autophagy in the hematopoietic system leads to loss of
hematopoietic stem cell function and dysregulated myeloid
proliferation. Autophagy. 7:1069–1070. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
21
|
Yang Z and Klionsky DJ: Eaten alive: A
history of macroautophagy. Nat Cell Biol. 12:814–822. 2010.
View Article : Google Scholar : PubMed/NCBI
|
|
22
|
Liu F, Lee JY, Wei H, Tanabe O, Engel JD,
Morrison SJ and Guan JL: FIP200 is required for the cell-autonomous
maintenance of fetal hematopoietic stem cells. Blood.
116:4806–4814. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
23
|
Mortensen M, Soilleux EJ, Djordjevic G,
Tripp R, Lutteropp M, Sadighi-Akha E, Stranks AJ, Glanville J,
Knight S, Jacobsen SE, et al: The autophagy protein Atg7 is
essential for hematopoietic stem cell maintenance. J Exp Med.
208:455–467. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
24
|
Warr MR, Binnewies M, Flach J, Reynaud D,
Garg T, Malhotra R, Debnath J and Passegué E: FOXO3A directs a
protective autophagy program in haematopoietic stem cells. Nature.
494:323–327. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
25
|
Oliver L, Hue E, Priault M and Vallette
FM: Basal autophagy decreased during the differentiation of human
adult mesenchymal stem cells. Stem Cells Dev. 21:2779–2788. 2012.
View Article : Google Scholar : PubMed/NCBI
|
|
26
|
Salemi S, Yousefi S, Constantinescu MA,
Fey MF and Simon HU: Autophagy is required for self-renewal and
differentiation of adult human stem cells. Cell Res. 22:432–435.
2012. View Article : Google Scholar : PubMed/NCBI
|
|
27
|
Vázquez P, Arroba AI, Cecconi F, de la
Rosa EJ, Boya P and de Pablo F: Atg5 and Ambra1 differentially
modulate neurogenesis in neural stem cells. Autophagy. 8:187–199.
2012. View Article : Google Scholar : PubMed/NCBI
|
|
28
|
Wang S, Li B, Qiao H, Lv X, Liang Q, Shi
Z, Xia W, Ji F and Jiao J: Autophagy-related gene Atg5 is essential
for astrocyte differentiation in the developing mouse cortex. EMBO
Rep. 15:1053–1061. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
29
|
Wang C, Liang CC, Bian ZC, Zhu Y and Guan
JL: FIP200 is required for maintenance and differentiation of
postnatal neural stem cells. Nat Neurosci. 16:532–542. 2013.
View Article : Google Scholar : PubMed/NCBI
|
|
30
|
Zhang J, Liu J, Huang Y, Chang JY, Liu L,
McKeehan WL, Martin JF and Wang F: FRS2α-mediated FGF signals
suppress premature differentiation of cardiac stem cells through
regulating autophagy activity. Circ Res. 110:e29–e39. 2012.
View Article : Google Scholar : PubMed/NCBI
|
|
31
|
Mizushima N, Yamamoto A, Hatano M,
Kobayashi Y, Kabeya Y, Suzuki K, Tokuhisa T, Ohsumi Y and Yoshimori
T: Dissection of autophagosome formation using Apg5-deficient mouse
embryonic stem cells. J Cell Biol. 152:657–668. 2001. View Article : Google Scholar : PubMed/NCBI
|
|
32
|
Tra T, Gong L, Kao LP, Li XL, Grandela C,
Devenish RJ, Wolvetang E and Prescott M: Autophagy in human
embryonic stem cells. PLoS One. 6:e274852011. View Article : Google Scholar : PubMed/NCBI
|
|
33
|
Cheng Y, Wang B, Zhou H, Dang S, Jin M,
Shi Y, Hao L, Yang Z and Zhang Y: Autophagy is required for the
maintenance of liver progenitor cell functionality. Cell Physiol
Biochem. 36:1163–1174. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
34
|
Heras-Sandoval D, Pérez-Rojas JM,
Hernández-Damián J and Pedraza-Chaverri J: The role of
PI3K/AKT/mTOR pathway in the modulation of autophagy and the
clearance of protein aggregates in neurodegeneration. Cell Signal.
26:2694–2701. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
35
|
Milla LA, González-Ramírez CN and Palma V:
Sonic Hedgehog in cancer stem cells: A novel link with autophagy.
Biol Res. 45:223–230. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
36
|
Tsao MS, Smith JD, Nelson KG and Grisham
JW: A diploid epithelial cell line from normal adult rat liver with
phenotypic properties of ‘oval’ cells. Exp Cell Res. 154:38–52.
1984. View Article : Google Scholar : PubMed/NCBI
|
|
37
|
Sun C, Jin XL and Xiao JC: Oval cells in
hepatitis B virus-positive and hepatitis C virus-positive liver
cirrhosis: Histological and ultrastructural study. Histopathology.
48:546–555. 2006. View Article : Google Scholar : PubMed/NCBI
|
|
38
|
Zeng J, Jing Y, Shi R, Pan X, Lai F, Liu
W, Li R, Gao L, Hou X, Wu M and Wei L: Autophagy regulates biliary
differentiation of hepatic progenitor cells through Notch1
signaling pathway. Cell Cycle. 15:1602–1610. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
39
|
Fukuda T, Oda K, Wada-Hiraike O, Sone K,
Inaba K, Ikeda Y, Miyasaka A, Kashiyama T, Tanikawa M, Arimoto T,
et al: The anti-malarial chloroquine suppresses proliferation and
overcomes cisplatin resistance of endometrial cancer cells via
autophagy inhibition. Gynecol Oncol. 137:538–545. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
40
|
Yuan H, He M, Cheng F, Bai R, da Silva SR,
Aguiar RC and Gao SJ: Tenovin-6 inhibits proliferation and survival
of diffuse large B-cell lymphoma cells by blocking autophagy.
Oncotarget. 8:14912–14924. 2017.PubMed/NCBI
|
|
41
|
Mariño G, Niso-Santano M, Baehrecke EH and
Kroemer G: Self-consumption: The interplay of autophagy and
apoptosis. Nat Rev Mol Cell Biol. 15:81–94. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
42
|
Gu J, Hu W, Song ZP, Chen YG, Zhang DD and
Wang CQ: Rapamycin inhibits cardiac hypertrophy by promoting
autophagy via the MEK/ERK/Beclin-1 pathway. Front Physiol.
7:1042016. View Article : Google Scholar : PubMed/NCBI
|
|
43
|
Martinez-Lopez N, Athonvarangkul D,
Mishall P, Sahu S and Singh R: Autophagy proteins regulate ERK
phosphorylation. Nat Commun. 4:27992013. View Article : Google Scholar : PubMed/NCBI
|