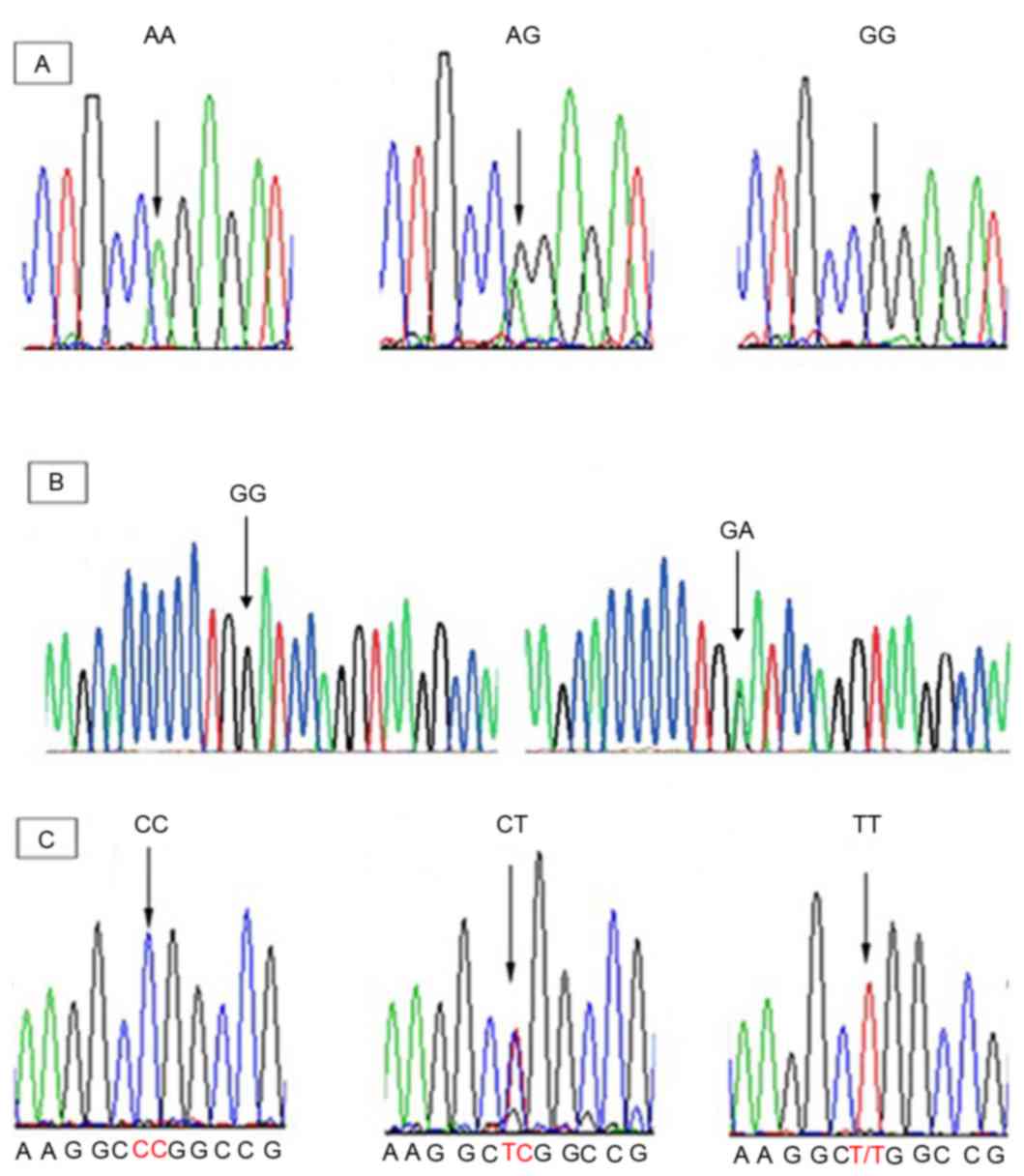
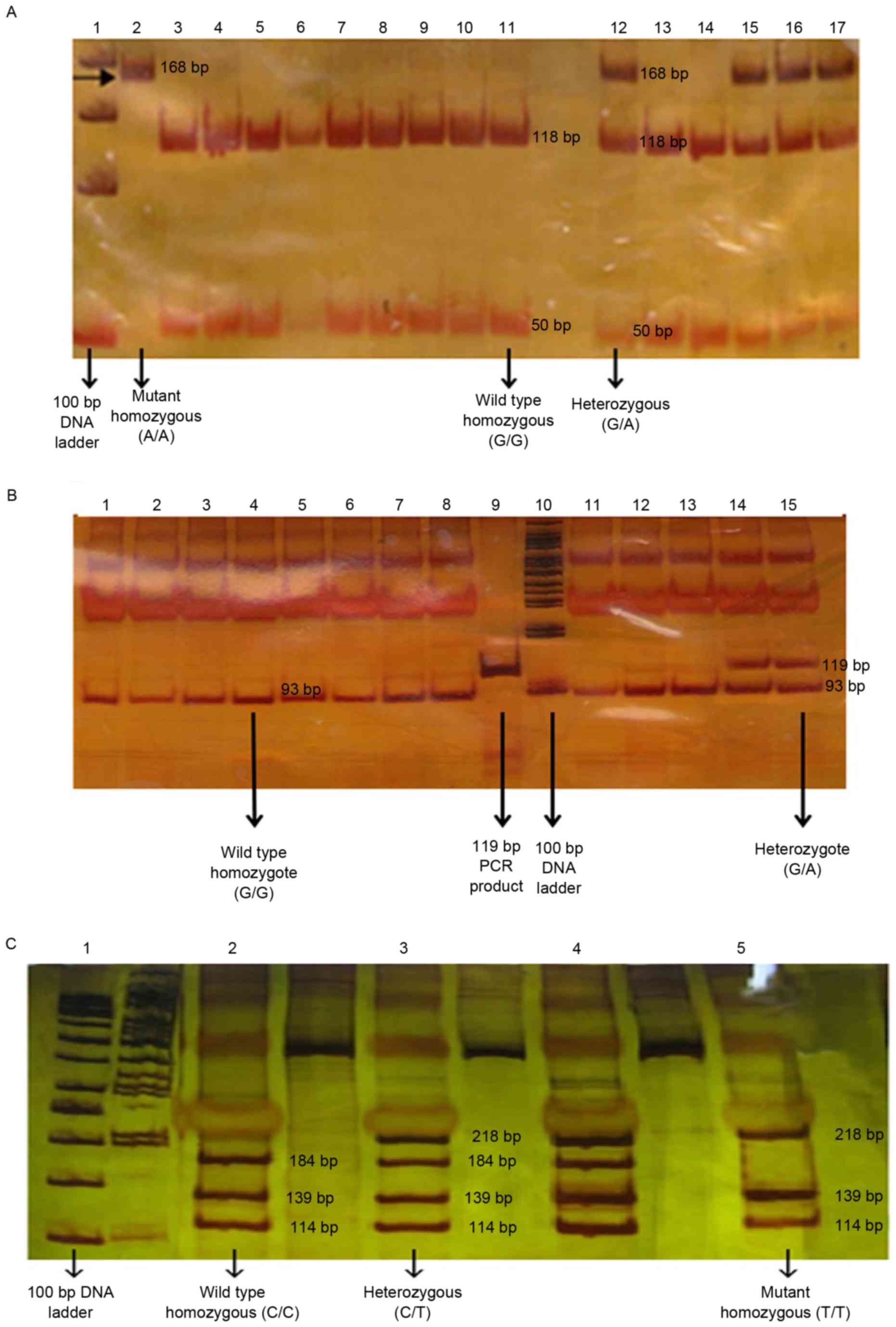
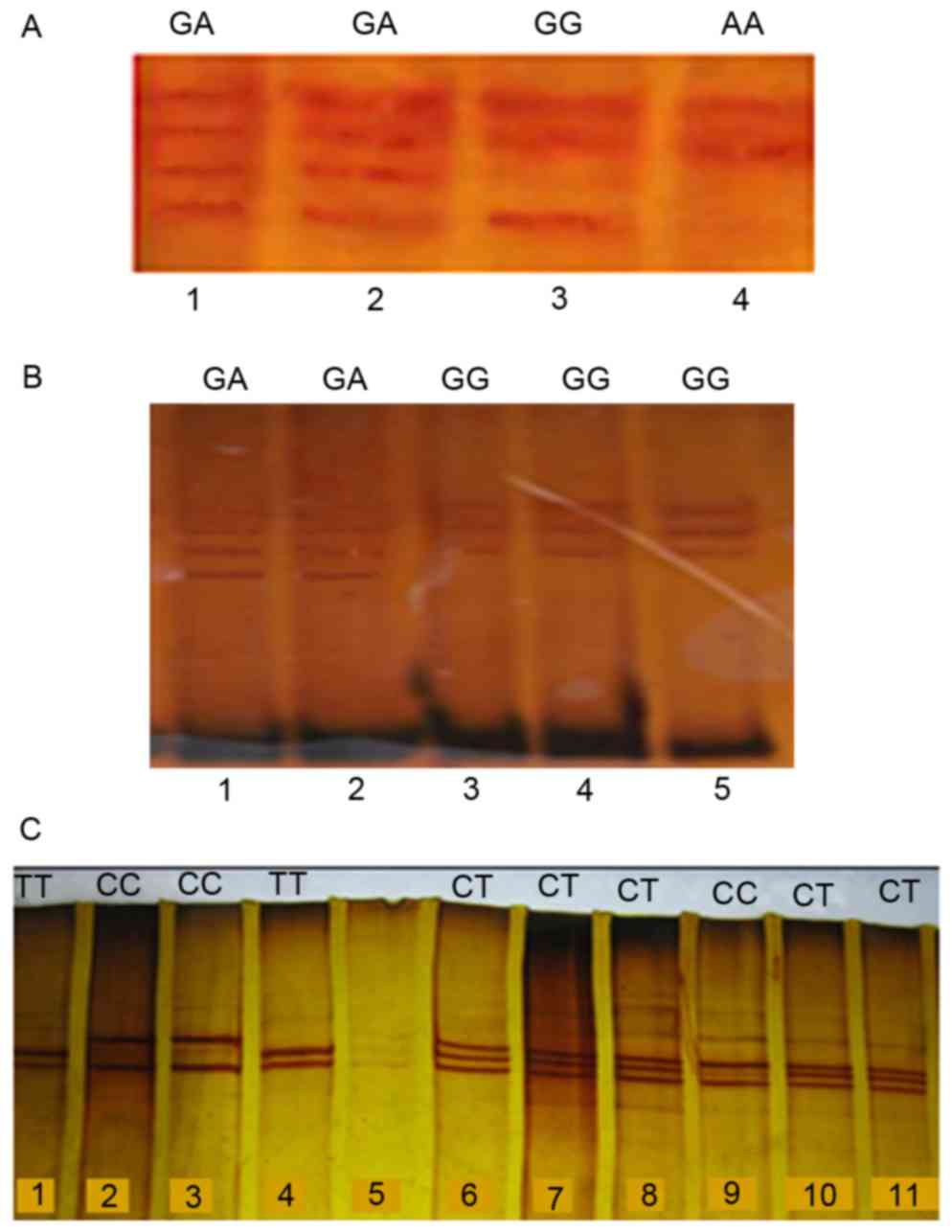
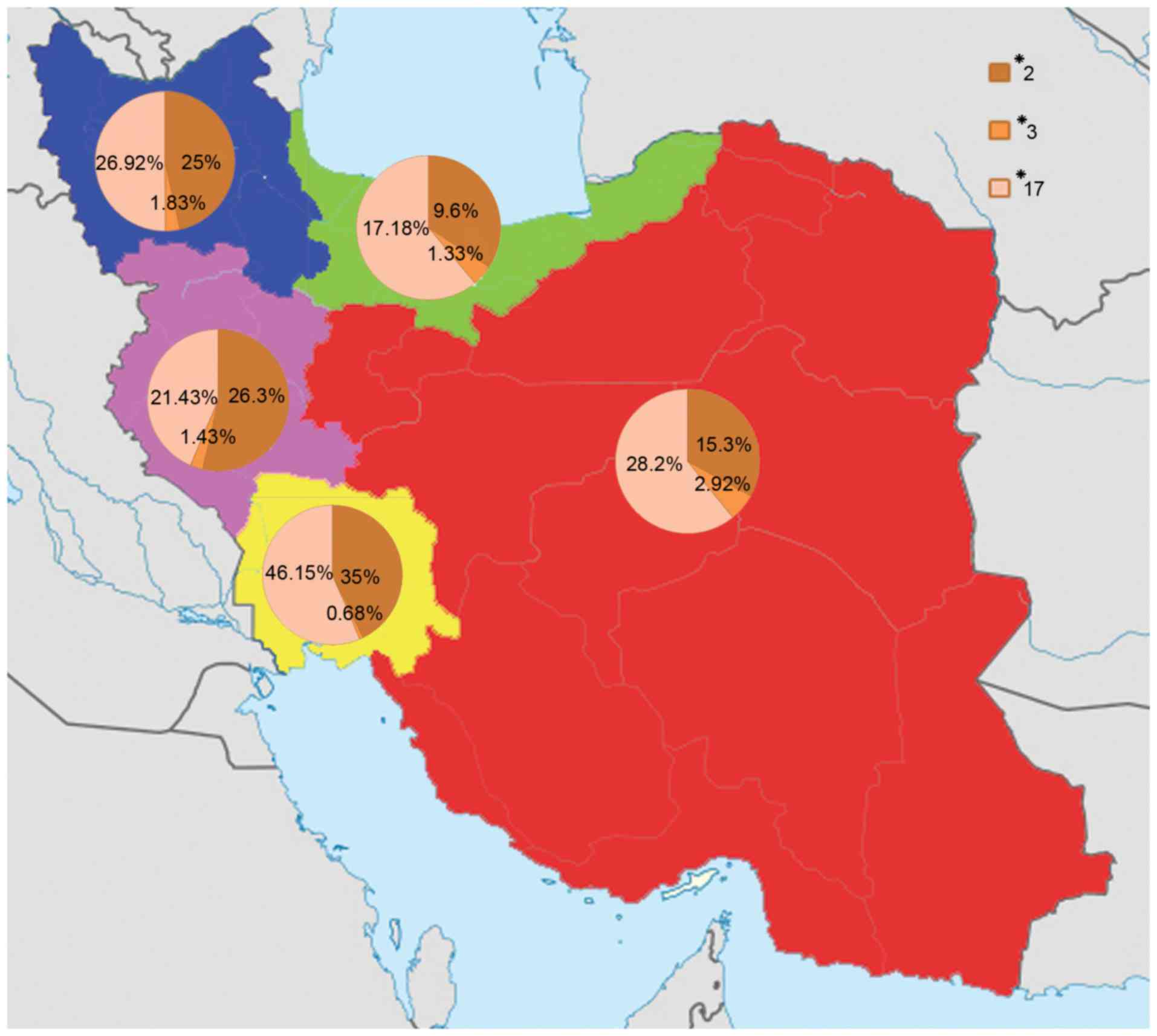
|
1
|
Hamdy SI, Hiratsuka M, Narahara K,
El-Enany M, Moursi N, Ahmed MS and Mizugaki M: Allele and genotype
frequencies of polymorphic cytochromes P450 (CYP2C9, CYP2C19,
CYP2E1) and dihydropyrimidine dehydrogenase (DPYD) in the Egyptian
population. Br J Clin Pharmacol. 53:596–603. 2002. View Article : Google Scholar : PubMed/NCBI
|
|
2
|
Wang H, An N, Wang H, Gao Y, Liu D, Bian
T, Zhu J and Chen C: Evaluation of the effects of 20 nonsynonymous
single nucleotide polymorphisms of CYP2C19 on S-mephenytoin
4′-hydroxylation and omeprazole 5′-hydroxylation. Drug Metab
Dispos. 39:830–837. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
3
|
Chaudhry A, Kochhar R and Kohli K: Genetic
polymorphism of CYP2C19 & therapeutic response to proton pump
inhibitors. Indian J Med Res. 127:521–530. 2008.PubMed/NCBI
|
|
4
|
Yin T and Miyata T: Pharmacogenomics of
clopidogrel: Evidence and perspectives. Thromb Res. 128:307–316.
2011. View Article : Google Scholar : PubMed/NCBI
|
|
5
|
Blaisdell J, Mohrenweiser H, Jackson J,
Ferguson S, Coulter S, Chanas B, Xi T, Ghanayem B and Goldstein JA:
Identification and functional characterization of new potentially
defective alleles of human CYP2C19. Pharmacogenetics. 12:703–711.
2002. View Article : Google Scholar : PubMed/NCBI
|
|
6
|
Buzoianu AD, Trifa AP, Popp RA, Militaru
MS, Militaru CF, Bocşan CI, Farcaş MF and Pop IV: Screening for
CYP2C19*2, *3 and *4 gene variants in a Romanian population study
group. Farmacia. 58:806–818. 2010.
|
|
7
|
Beitelshees AL, Horenstein RB, Vesely MR,
Mehra MR and Shuldiner AR: Pharmacogenetics and clopidogrel
response in patients undergoing percutaneous coronary
interventions. Clin Pharmacol Ther. 89:455–459. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
8
|
Scott SA, Sangkuhl K, Gardner EE, Stein
CM, Hulot JS, Johnson JA, Roden DM, Klein TE and Shuldiner AR;
Clinical Pharmacogenetics Implementation Consortium, : Clinical
Pharmacogenetics Implementation Consortium guidelines for
cytochrome P450-2C19 (CYP2C19) genotype and clopidogrel therapy.
Clin Pharmacol Ther. 90:328–332. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
9
|
Gawrońska-Szklarz B, Adamiak-Giera U,
Wyska E, Kurzawski M, Gornik W, Kaldonska M and Drozdzik M: CYP2C19
polymorphism affects single-dose pharmacokinetics of oral
pantoprazole in healthy volunteers. Eur J Clin Pharmacol.
68:1267–1274. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
10
|
Sim SC, Risinger C, Dahl ML, Aklillu E,
Christensen M, Bertilsson L and Ingelman-Sundberg M: A common novel
CYP2C19 gene variant causes ultrarapid drug metabolism relevant for
the drug response to proton pump inhibitors and antidepressants.
Clin Pharmacol Ther. 79:103–113. 2006. View Article : Google Scholar : PubMed/NCBI
|
|
11
|
Ikebuchi J, Yamada M, Ogura Y, Yamamoto Y,
Nishimura A, Nishi K, Yamada K and Irizawa Y: Individual difference
in drug metabolism and disposition: Toxicological significance of
genotypes and phenotypes of S-mephenytoin 4′-hydroxylase (CYP2C19).
Int Congress Series. 1239:589–592. 2003. View Article : Google Scholar
|
|
12
|
Shastry BS: Pharmacogenetics and the
concept of individualized medicine. Pharmacogenomics J. 6:16–21.
2005. View Article : Google Scholar
|
|
13
|
Nakamoto K, Kidd JR, Jenison RD, Klaassen
CD, Wan YJ, Kidd KK and Zhong XB: Genotyping and haplotyping of
CYP2C19 functional alleles on thin-film biosensor chips.
Pharmacogenet Genomics. 17:103–114. 2007. View Article : Google Scholar : PubMed/NCBI
|
|
14
|
Sugimoto K, Uno T, Yamazaki H and Tateishi
T: Limited frequency of the CYP2C19*17 allele and its minor role in
a Japanese population. Br J Clin Pharmacol. 65:437–439. 2008.
View Article : Google Scholar : PubMed/NCBI
|
|
15
|
Yamada H, Dahl ML, Lannfelt L, Viitanen M,
Winblad B and Sjöqvist F: CYP2D6 and CYP2C19 genotypes in an
elderly Swedish population. Eur J Clin Pharmacol. 54:479–481. 1998.
View Article : Google Scholar : PubMed/NCBI
|
|
16
|
Brockmöller JJ, Rost K, Gross D, Schenkel
A and Roots I: Phenotyping of CYP2C19 with enantiospecific
HPLC-quantification of R- and S-mephenytoin and comparison with the
intron4/exon5 G->A-splice site mutation. Pharmacogenetics.
5:80–88. 1995.PubMed/NCBI
|
|
17
|
Persson I, Aklillu E, Rodrigues F,
Bertilsson L and Ingelman-Sundberg M: S-mephenytoin hydroxylation
phenotype and CYP2C19 genotype among Ethiopians. Pharmacogenetics.
6:521–526. 1996. View Article : Google Scholar : PubMed/NCBI
|
|
18
|
Dandara C, Masimirembwa CM, Magimba A,
Sayi J, Kaaya S, Sommers DK, Snyman JR and Hasler JA: Genetic
polymorphism of CYP2D6 and CYP2C19 in east-and southern African
populations including psychiatric patients. Eur J Clin Pharmacol.
57:11–17. 2001. View Article : Google Scholar : PubMed/NCBI
|
|
19
|
Goldstein JA, Ishizaki T, Chiba K, de
Morais SM, Bell D, Krahn PM and Evans DA: Frequencies of the
defective CYP2C19 alleles responsible for the mephenytoin poor
metabolizer phenotype in various Oriental, Caucasian, Saudi Arabian
and American black populations. Pharmacogenetics. 7:59–64. 1997.
View Article : Google Scholar : PubMed/NCBI
|
|
20
|
Sistonen J, Fuselli S, Palo JU, Chauhan N,
Padh H and Sajantila A: Pharmacogenetic variation at CYP2C9,
CYP2C19, and CYP2D6 at global and microgeographic scales.
Pharmacogenet Genomics. 19:170–179. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
21
|
Jurima-Romet M, Goldstein JA, LeBelle M,
Aubin RA, Foster BC, Walop W and Rode A: CYP2C19 genotyping and
associated mephenytoin hydroxylation polymorphism in a Canadian
Inuit population. Pharmacogenetics. 6:329–339. 1996. View Article : Google Scholar : PubMed/NCBI
|
|
22
|
Bathum L, Andersen-Ranberg K, Boldsen J,
Brøsen K and Jeune B: Genotypes for the cytochrome P450 enzymes
CYP2D6 and CYP2C19 in human longevitY. Role of CYP2D6 and CYP2C19
in longevity. Eur J Clin Pharmacol. 54:427–430. 1998. View Article : Google Scholar : PubMed/NCBI
|
|
23
|
Herrlin K, Massele AY, Jande M, Alm C,
Tybring G, Abdi YA, Wennerholm A, Johansson I, Dahl ML, Bertilsson
L and Gustafsson LL: Bantu Tanzanians have a decreased capacity to
metabolize omeprazole and mephenytoin in relation to their CYP2C19
genotype. Clin Pharmacol Ther. 64:391–401. 1998. View Article : Google Scholar : PubMed/NCBI
|
|
24
|
Hsu HL, Woad KJ, Woodfield DG and Helsby
NA: A high incidence of polymorphic CYP2C19 variants in archival
blood samples from Papua New Guinea. Hum Genomics. 3:17–23. 2008.
View Article : Google Scholar : PubMed/NCBI
|
|
25
|
Blendon RJ, Schoen C, DesRoches C, Osborn
R and Zapert K: Common concerns amid diverse systems: Health care
experiences in five countries. Health Aff (Millwood). 22:106–121.
2003. View Article : Google Scholar : PubMed/NCBI
|
|
26
|
Lindley CM, Tully MP, Paramsothy V and
Tallis RC: Inappropriate medication is a major cause of adverse
drug reactions in elderly patients. Age and Ageing. 21:294–300.
1992. View Article : Google Scholar : PubMed/NCBI
|
|
27
|
Pedersen RS, Brasch-Andersen C, Sim SC,
Bergmann TK, Halling J, Petersen MS, Weihe P, Edvardsen H,
Kristensen VN, Brøsen K and Ingelman-Sundberg M: Linkage
disequilibrium between the CYP2C19*17 allele and wildtype CYP2C8
and CYP2C9 alleles: Identification of CYP2C haplotypes in healthy
Nordic populations. Eur J Clin Pharmacol. 66:1199–1205. 2010.
View Article : Google Scholar : PubMed/NCBI
|
|
28
|
Rudberg I, Mohebi B, Hermann M, Refsum H
and Molden E: Impact of the ultrarapid CYP2C19*17 allele on serum
concentration of escitalopram in psychiatric patients. Clin
Pharmacol Ther. 83:322–327. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
29
|
Kurzawski M, Gawrońska-Szklarz B,
Wrześniewska J, Siuda A, Starzyńska T and Droździk M: Effect of
CYP2C19*17 gene variant on Helicobacter pylori eradication in
peptic ulcer patients. Eur J Clin Pharmacol. 62:877–880. 2006.
View Article : Google Scholar : PubMed/NCBI
|
|
30
|
Ruas JL and Lechner MC: Allele frequency
of CYP2C19 in a Portuguese population. Pharmacogenetics. 7:333–335.
1997. View Article : Google Scholar : PubMed/NCBI
|
|
31
|
Hoskins JM, Shenfield GM and Gross AS:
Relationship between proguanil metabolic ratio and CYP2C19 genotype
in a Caucasian population. Br J Clin Pharmacol. 46:499–504. 1998.
View Article : Google Scholar : PubMed/NCBI
|
|
32
|
Lamba JK, Dhiman RK and Kohli KK: CYP2C19
genetic mutations in North Indians. Clin Pharmacol Ther.
68:328–335. 2000. View Article : Google Scholar : PubMed/NCBI
|
|
33
|
Ramsjö M, Aklillu E, Bohman L,
Ingelman-Sundberg M, Roh HK and Bertilsson L: CYP2C19 activity
comparison between Swedes and Koreans: Effect of genotype, sex,
oral contraceptive use, and smoking. Eur J Clin Pharmacol.
66:871–877. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
34
|
Kim KA, Song WK, Kim KR and Park JY:
Assessment of CYP2C19 genetic polymorphisms in a Korean population
using a simultaneous multiplex pyrosequencing method to
simultaneously detect the CYP2C19*2, CYP2C19*3, and CYP2C19*17
alleles. J Clin Pharm Ther. 35:697–703. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
35
|
Chen L, Qin S, Xie J, Tang J, Yang L, Shen
W, Zhao X, Du J, He G and Feng G: Genetic polymorphism analysis of
CYP2C19 in Chinese Han populations from different geographic areas
of mainland China. Pharmacogenomics. 9:691–702. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
36
|
Kearns GL, Leeder JS and Gaedigk A: Impact
of the CYP2C19*17 allele on the pharmacokinetics of omeprazole and
pantoprazole in children: Evidence for a differential effect. Drug
Metab Dispos. 38:894–897. 2010. View Article : Google Scholar : PubMed/NCBI
|