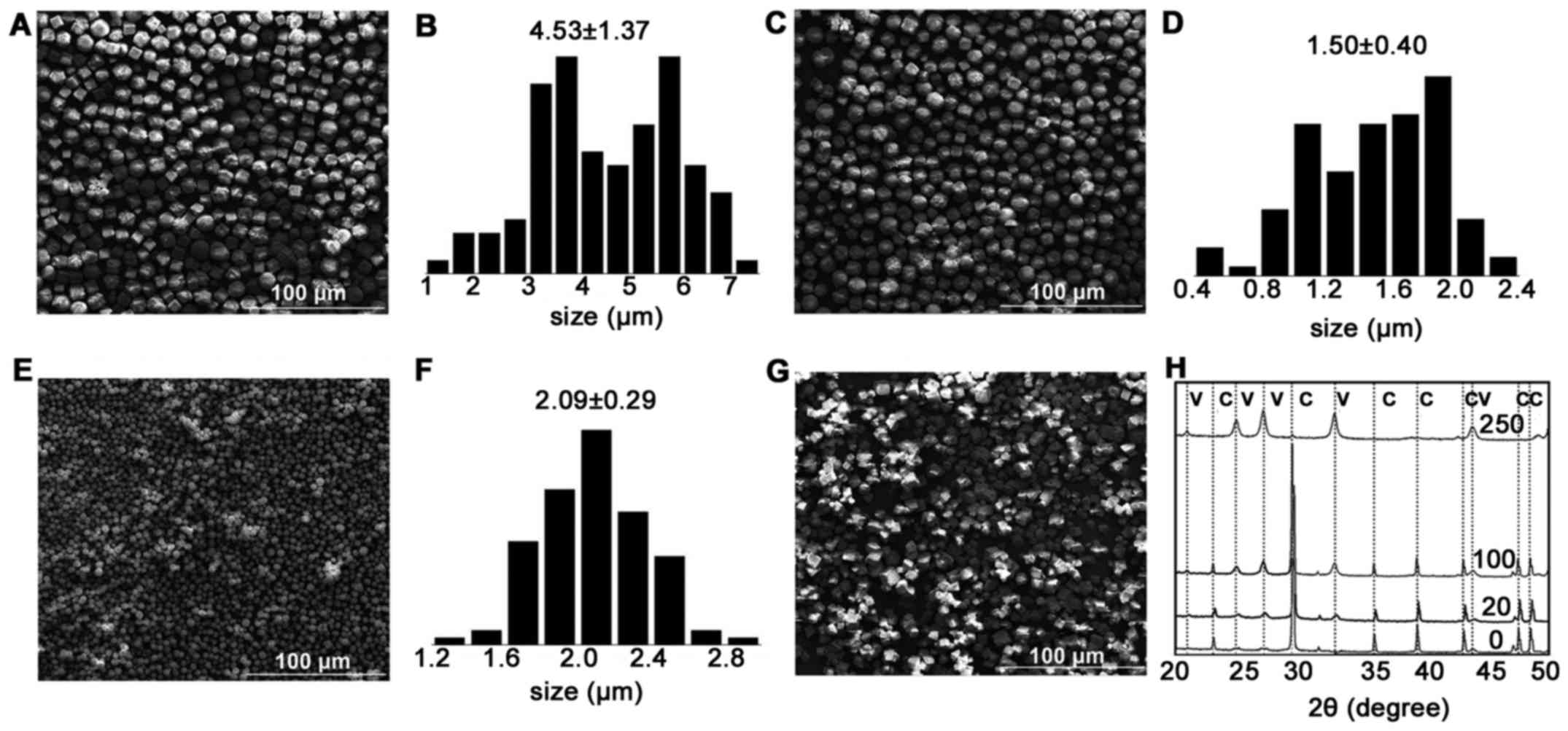
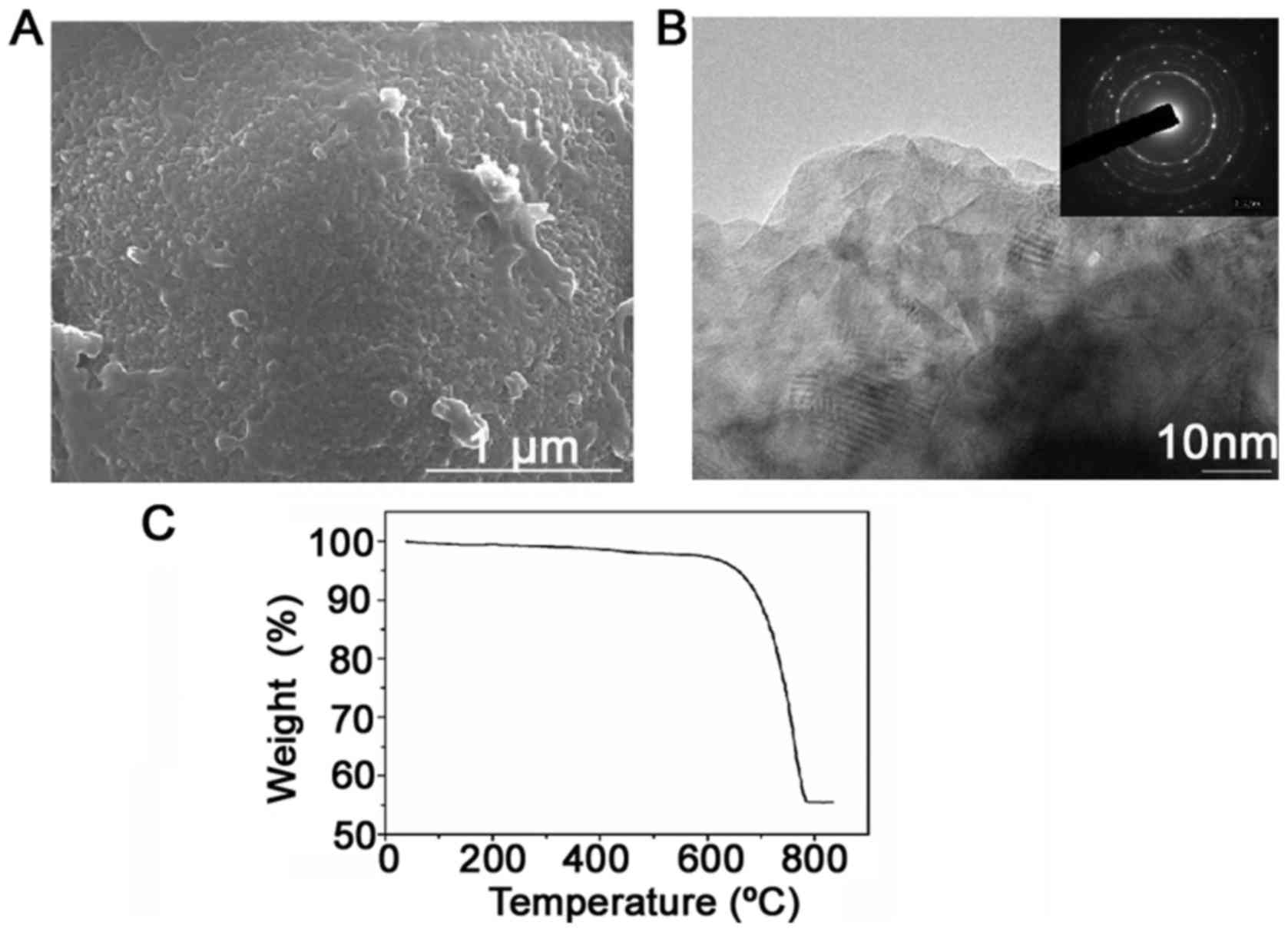
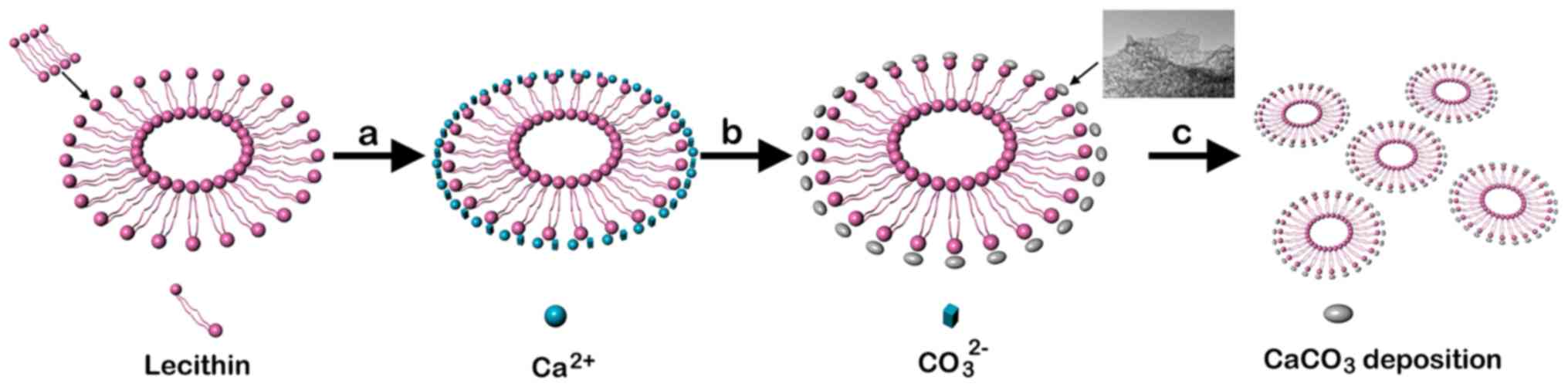
|
1
|
Chi Y and Lin S: Research advances in the
extraction and application of egg-yolk lecithin. Food Ferment Ind.
28:50–53. 2002.
|
|
2
|
Li Y, Liu JB and Lin SY: Study on
extraction of high pure egg yolk lecithin. Food Sci. 27:851–853.
2006.
|
|
3
|
Palacios LE and Wang T: Extraction of
egg-yolk lecithin. J Am Oil Chem Soc. 82:565–569. 2005. View Article : Google Scholar
|
|
4
|
Chang H, Wang EL, Gong XT and Liu JB: Over
view on study of yolk lecithin. Sci Tech Food Ind. 5:414–420.
2010.
|
|
5
|
Ali AH, Zou X, Lu J, Abed SM, Yao Y, Tao
G, Jin Q and Wang X: Identification of phospholipids classes and
molecular species in different types of egg yolk by using
UPLC-Q-TOF-MS. Food Chem. 221:58–66. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
6
|
Rossi J and Leroux JC: Principles in the
development of intravenous lipid emulsionsWasan KM and Wiley: John
Wiley & Sons, Inc.; New York: pp. 88–123. 2006, PubMed/NCBI
|
|
7
|
Foran E, Weiner S and Fine M: Biogenic
fish-gut calcium carbonate is a stable amorphous phase in the
gilt-head seabream, sparus aurata. Sci Rep. 3:17002013. View Article : Google Scholar : PubMed/NCBI
|
|
8
|
Vlieg E: Materials science. Complexity
from simplicity. Science. 340:822–823. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
9
|
Kim S and Park CB: Bio-inspired synthesis
of minerals for energy, environment, and medicinal applications.
Adv Funct Mater. 23:10–25. 2013. View Article : Google Scholar
|
|
10
|
Zhao Y, Luo Z, Li M, Qu Q, Ma X, Yu SH and
Zhao Y: A preloaded amorphous calcium carbonate/doxorubicin@silica
nanoreactor for pH-responsive delivery of an anticancer drug. Angew
Chem Int Ed Engl. 54:919–922. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
11
|
Wang C, Chen SQ, Yu Q, Hu FQ and Yuan H:
Taking advantage of the disadvantage: Employing the high aqueous
instability of amorphous calcium carbonate to realize burst drug
release within cancer cells. J Mater Chem B. 5:2068–2073. 2017.
View Article : Google Scholar
|
|
12
|
Zan G and Wu Q: Biomimetic and bioinspired
synthesis of nanomaterials/nanostructures. Adv Mater. 28:2099–2147.
2016. View Article : Google Scholar : PubMed/NCBI
|
|
13
|
Han MR, Kwon MC, Lee HY, Kim JC, Kim JD,
Yoo SK, SIN IS and Kim SM: pH-dependent release property of
alginate beads containing calcium carbonate particles. J
Microencapsul. 24:787–796. 2007. View Article : Google Scholar : PubMed/NCBI
|
|
14
|
Wei W, Ma GH, Hu G, Yu D, Mcleish T, Su ZG
and Shen ZY: Preparation of hierarchical hollow CaCO3
Particles and the application as anticancer drug carrier. J Am Chem
Soc. 130:15808–15810. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
15
|
Xiaoguang C, Hongyan L, Xiaohong L, Zhaodi
F, Yan L, Lihua T and Rui H: Cancer chemopreventive and therapeutic
activities of red ginseng. J Ethnopharmacol. 60:71–78. 1998.
View Article : Google Scholar : PubMed/NCBI
|
|
16
|
Gao L, Chen L, Fei XH, Qiu HY, Zhou H and
Wang JM: STI571 combined with vincristine greatly suppressed the
tumor formation of multidrug-resistant K562 cells in a human-nude
mice xenograft model. Chin Med J (Engl). 119:911–918.
2006.PubMed/NCBI
|
|
17
|
Jin Y, Li J, Rong LF, Li YH, Guo L and Xu
SY: Anti-hepatocarcinoma effects of 5-fluorouracil encapsulated by
galactosylceramide liposomes in vivo and in vitro. World J
Gastroenterol. 11:2643–2646. 2005. View Article : Google Scholar : PubMed/NCBI
|
|
18
|
Chen H, Takahashi S, Imamura M, Okutani E,
Zhang ZG, Chayama K and Chen BA: Earthworm fibrinolytic enzyme:
Anti-tumor activity on human hepatoma cells in vitro and in vivo.
Chin Med J (Engl). 120:898–904. 2007.PubMed/NCBI
|
|
19
|
Naka K, Huang SC and Chujo Y: Formation of
stable vaterite with poly(acrylic acid) by the delayed addition
method. Langmuir. 22:7760–7767. 2006. View Article : Google Scholar : PubMed/NCBI
|
|
20
|
Naka K, Tanaka Y and Chujo Y: Effect of
anionic starburst dendrimers on the crystallization of
CaCO3 in aqueous solution: Size control of spherical
vaterite particles. Langmuir. 18:3655–3658. 2002. View Article : Google Scholar
|
|
21
|
Helmlinger G, Yuan F, Dellian M, Dellian M
and Jain RK: Interstitial pH and pO2 gradients in solid tumors in
vivo: High-resolution measurements reveal a lack of correlation.
Nat Med. 3:177–182. 1997. View Article : Google Scholar : PubMed/NCBI
|
|
22
|
Hirschhaeuser F, Menne H, Dittfeld C, West
J, Mueller-Klieser W and Kunz-Schughart LA: Multicellular tumor
spheroids: An underestimated tool is catching up again. J
Biotechnol. 148:3–15. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
23
|
Chauhan VP and Jain RK: Strategies for
advancing cancer nanomedicine. Nat Mater. 12:958–962. 2013.
View Article : Google Scholar : PubMed/NCBI
|
|
24
|
Parakhonskiy BV, Haase A and Antolini R:
Sub-micrometer vaterite containers: Synthesis, substance loading,
and release. Angew Chem Int Ed Engl. 51:1195–1197. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
25
|
Schmidt S and Volodkin D: Microparticulate
biomolecules by mild CaCO3 templating. J Mater Chem B.
1:1210–1218. 2013. View Article : Google Scholar
|
|
26
|
Qi C, Zhu YJ and Chen F: Microwave
hydrothermal transformation of amorphous calcium carbonate
nanospheres and application in protein adsorption. ACS Appl Mater
Interfaces. 6:4310–4320. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
27
|
Svenskaya Y, Parakhonskiy B, Haase A,
Atkin V, Lukyanets E, Gorin D and Antolini R: Anticancer drug
delivery system based on calcium carbonate particles loaded with a
photosensitizer. Biophys Chem. 182:11–15. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
28
|
Yang YH, Liu CH, Liang YH, Lin FH and Wu
KCW: Hollow mesoporous hydroxyapatite nanoparticles (hmHANPs) with
enhanced drug loading and PH-responsive release properties for
intracellular drug delivery. J Mater Chem B. 1:2447–2450. 2013.
View Article : Google Scholar
|
|
29
|
Qi C, Zhu YJ and Chen F: Microwave
hydrothermal transformation of amorphous calcium carbonate
nanospheres and application in protein adsorption. ACS Appl Mater
Interfaces. 6:4310–4320. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
30
|
Wang J, Chen JS, Zong JY, Zhao D, Li F,
Zhou RX and Cheng SX: Calcium carbonate/carboxymethyl chitosan
hybrid microspheres and nanospheres for drug delivery. J Phys Chem
C. 114:18940–18945. 2010. View Article : Google Scholar
|
|
31
|
Ying X, Shan C, Jiang K, Chen Z and Du Y:
Intracellular pH-sensitive delivery CaCO3 nanoparticles
templated by hydrophobic modified starch micelles. RSC Adv.
4:10841–10844. 2014. View Article : Google Scholar
|
|
32
|
Kurapati R and Raichur AM: Composite
cyclodextrin-calcium carbonate porous microparticles and modified
multilayer capsules: Novel carriers for encapsulation of
hydrophobic drugs. J Mater Chem B. 1:3175–3184. 2013. View Article : Google Scholar
|
|
33
|
Peng C, Zhao Q and Gao C: Sustained
delivery of doxorubicin by porous CaCO3 and
chitosan/alginate multilayers-coated CaCO3
microparticles. Colloids Sur A: Physicochem Eng Aspects.
353:132–139. 2010. View Article : Google Scholar
|
|
34
|
Parakhonskiy BV, Foss C, Carletti E, Fedel
M, Haase A, Motta A, Migliaresi C and Antolini R: Tailored
intracellular delivery via a crystal phase transition in 400 nm
vaterite particles. Biomater Sci. 1:1273–1281. 2013. View Article : Google Scholar
|