Introduction
Periodontitis is a chronic inflammatory disease
caused by certain specific subgingival microorganisms, which
results in the destruction of supporting tissue around the roots
and subsequently leads to a loss of teeth if left untreated. The
periodontal destruction involves a complex interaction, which is
mediated in part by the individual host immune response to
microbial colonization of the periodontal attachment apparatus. The
lipopolysaccharide (LPS)-mediated host responses appear to cause
certain inflammatory reactions, resulting in the expression of
proinflammatory cytokines and amplifying the related host immune
response in periodontal diseases (1).
Toll-like receptors (TLRs) play important roles in
recognizing the invaded micropathogens in chronic inflammation,
such as periodontitis and diabetes mellitus. TLR2 and TLR4 are
receptors for the products of major periodontal pathogens,
including Porphyromonas gingivalis (P. gingivalis)
(2). P. gingivalis LPS
stimulated TLR2 rather than TLR4 (3). TLR2 is involved in T helper 2 (Th2)
orientation of the immune response and P. gingivalis
LPS-induced cellular activation (2,4). The
subsequent pro-inflammatory cytokine products of TLRs, such as
TNF-α, IL-1β and IL-6, modify the occurrence and development of
chronic inflammation, including periodontitis. Human periodontal
ligament cells (hPDLs), which are capable of differentiating into
cells of either cementogenic or osteogenic lineage, play important
roles in maintaining homeostasis during the inflammatory response
to these cytokines (5).
Escin is the major active component of Aesculus
hippocastanum. Escin is a natural mixture of triterpene
saponins, and has shown clinically significant anti-inflammatory
activity post-operatively and in chronic venous insufficiency,
haemorroids and traumatic brain edema. It mainly exerts its
anti-inflammatory and anti-edematous effects through its
anti-histamine and anti-serotoninergic activities, and by reducing
the adhesiveness and migration of neutrophils (6). The aim of the present study was to
investigate the effect of escin on the inflammation induced by LPS
in primary culture of hPDLs.
Materials and methods
Reagents
The escin used in the experiments was sodium
aescinate obtained as a lyophilized powder in a 5 mg vial
manufactured by Wuhu Lvye Pharmaceutical Co. Ltd. (Wuhu, China).
Dulbecco’s modified Eagle’s medium (DMEM), 10% fetal calf serum
(FCS), 0.25% trypsin and 1% penicillin/streptomycin were purchased
from Gibco (Carlsbad, CA, USA). LPS from Escherichia coli
0127:B8 and 3, 4, 5-dimethylthiazol-2-yl-2, 5-diphenyl tetrazolium
bromide (MTT) were purchased from Sigma-Aldrich (St. Louis, MO,
USA). Dimethyl sulfoxide was purchased from Merck Sharp & Dohme
(Darmstadt, Germany). Enzyme-linked immunosorbent assay (ELISA)
kits for TNF-α, IL-1β and IL-6 were from Bender Medsystem (Vienna,
Austria). All chemicals and solvents were of analytical grade.
Anti-TLR-2 antibody was purchased from Santa Cruz Biotechnology
(Santa Cruz, CA, USA). Enhanced chemiluminescence (ECL) detection
reagents and bicinchoninic acid (BCA) protein assay kits were
obtained from Beyotime Institute of Biotechnology (Haimen, China).
This study was approved by the ethics committee of Binzhou Medical
University.
Cell culture
The primary culture of hPDLs was obtained from
healthy 15–22-year-old patients undergoing routine extraction of
their first or second premolars in accordance to the method of
Somerman et al(7), with
minor modifications. All patients were, on an individual basis,
informed in detail of the aim and procedure of this study, and they
signed consent forms. Periodontal ligament tissues were scaled and
isolated from the middle third of the roots. Tissue pieces were
placed in a 6-well plate containing DMEM supplemented with 10% FCS
and 1% penicillin/streptomycin. Plates were cultured in humidified
air containing 5% CO2 at 37°C for approximately 14 days
until cells migrated out of the tissue and reach confluence. The
tissue pieces were then carefully removed and the cells were
collected by trypsinization and cultured in 150 cm2
culturing flasks. Cells of passage 3 to 9 were used in the
subsequent steps.
Cell treatment
hPDLs were subjected to 1 μg/ml LPS stimulation
(8) for 6, 12, 24, 48 and 72 h,
respectively, for the preliminary investigation of the effect of
LPS on hPDL viability. The hPDLs were then subjected to 1 μg/ml LPS
and escin (2.5, 5.0, 10.0, 20.0 and 40.0 μg/ml) treatment (9) for 24 h at 37°C in a humidified
atmosphere of 95% air and 5% CO2. The cultured hPDLs
undergoing neither LPS nor escin treatment served as controls.
Cell viability assay
hPDL viability was determined using the MTT dye
method. Briefly, hPDLs were seeded in 96-well culture plates at an
initial density of 1×104 cells per well in 100 μl
medium, and allowed to grow to confluence. After treatment as
described above, 10 μl MTT solution (5 mg/ml) was added into each
well and the culture plates were incubated at 37°C for 4 h. The
supernatant was then discarded and 100 μl dimethyl sulfoxide was
added followed by 5 min incubation on a shaker. The absorbance was
measured at 620 nm using an automated ELISA reader (Synergy HT,
Winooski, VT, USA). All samples were tested in duplicate for each
experiment.
Measurement of TNF-α, IL-β and IL-6
levels
hPDLs were subjected to 1 μg/ml LPS and escin at
various concentrations (5.0, 10.0 and 20.0 μg/ml) for 24 h. The
cell culture supernatant was harvested at the end of treatment and
used for the TNF-α, IL-1β and IL-6 assay according to the
manufacturer’s instructions. Absorbance values were measured at 450
nm using an ELISA plate reader (Synergy HT). All samples were
measured in duplicate.
Western blot analysis for TLR-2
hPDLs were detached by 0.02% EDTA and washed three
times with PBS. Whole cell lysates were isolated by lysing in 20
mmol/l Tris-HCl, pH 7.5, 2% SDS (w/v), 2 mmol/l benzamidine and 0.2
mmol/l phenyl-methanesulphonyl fluoride. Protein concentrations
were determined using a BCA protein assay kit. Samples were heated
at 100°C for 5 min prior to gel loading and were then subjected to
SDS-PAGE (8% sodium dodecyl sulfate/polyacrylamide gel
electrophoresis) and transferred to polyvinylidene difluoride
membranes using an electrophoretic transfer system. The membranes
were blocked in Tris-buffered saline mixed with Tween-20 (TBST)
containing 5% skimmed milk for 60 min. Membranes were then
incubated overnight at 4°C with primary antibody against TLR-2
(diluted 1:1000). After washing with TBST, the membranes were
incubated with secondary antibody (diluted 1:2000) for 1 h at 37°C.
After washing with TBST, the binding was detected by ECL detection
reagents. For the quantitative analysis of the density of the
immunoblot bands, densitometry was performed with the Gel-Pro
Analyzer Version 3.0. The ratio of the integrated optical density
of immunoblot bands of the detected protein to that of β-actin was
used for statistical analysis.
Statistical analysis
Data were expressed as the means ± standard
deviation (SD) in this study. One-way ANOVA was used to analyze the
significant differences within the groups. The comparison between
two groups was determined by the post hoc test. A value of
P<0.05 was considered to indicate a statistically significant
difference between groups.
Results
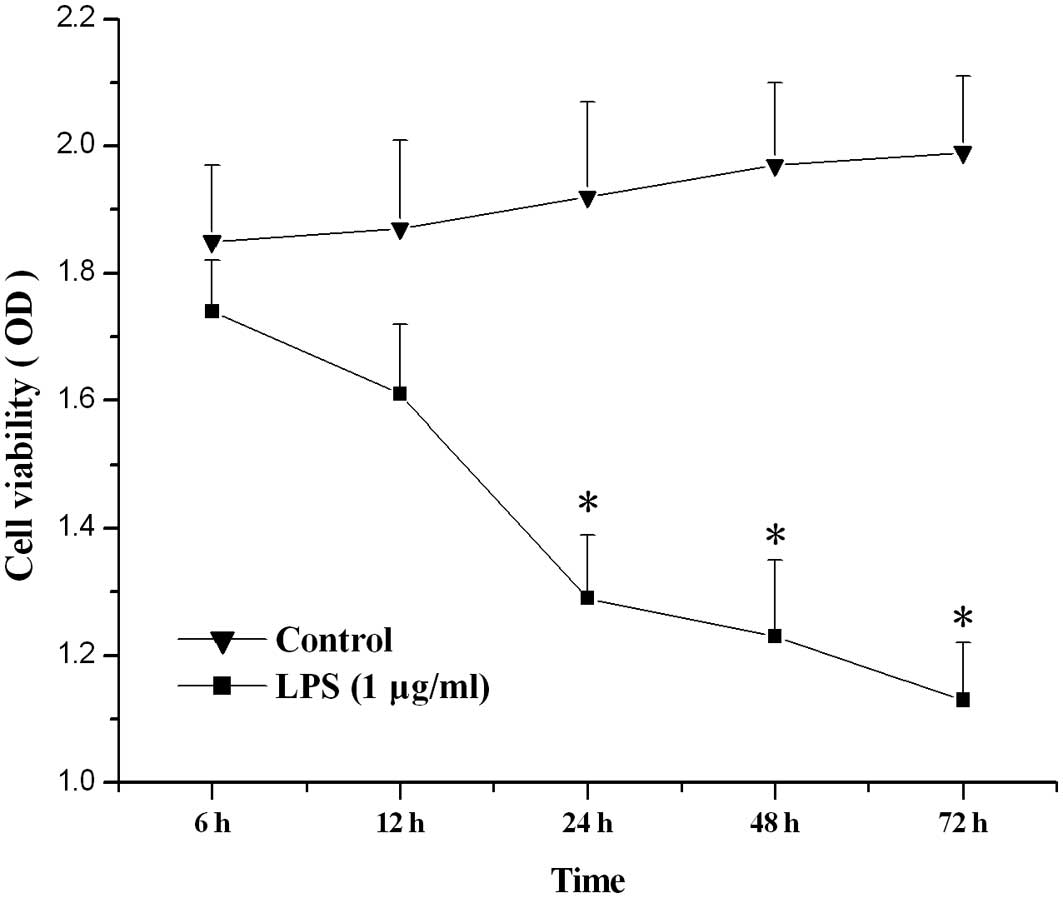
The effect of LPS on the viability of
hPDLs
Compared with control, LPS (1 μg/ml) treatment for
24, 48 or 72 h significantly lowered the viability of hPDLs
(P<0.05; Fig. 1).
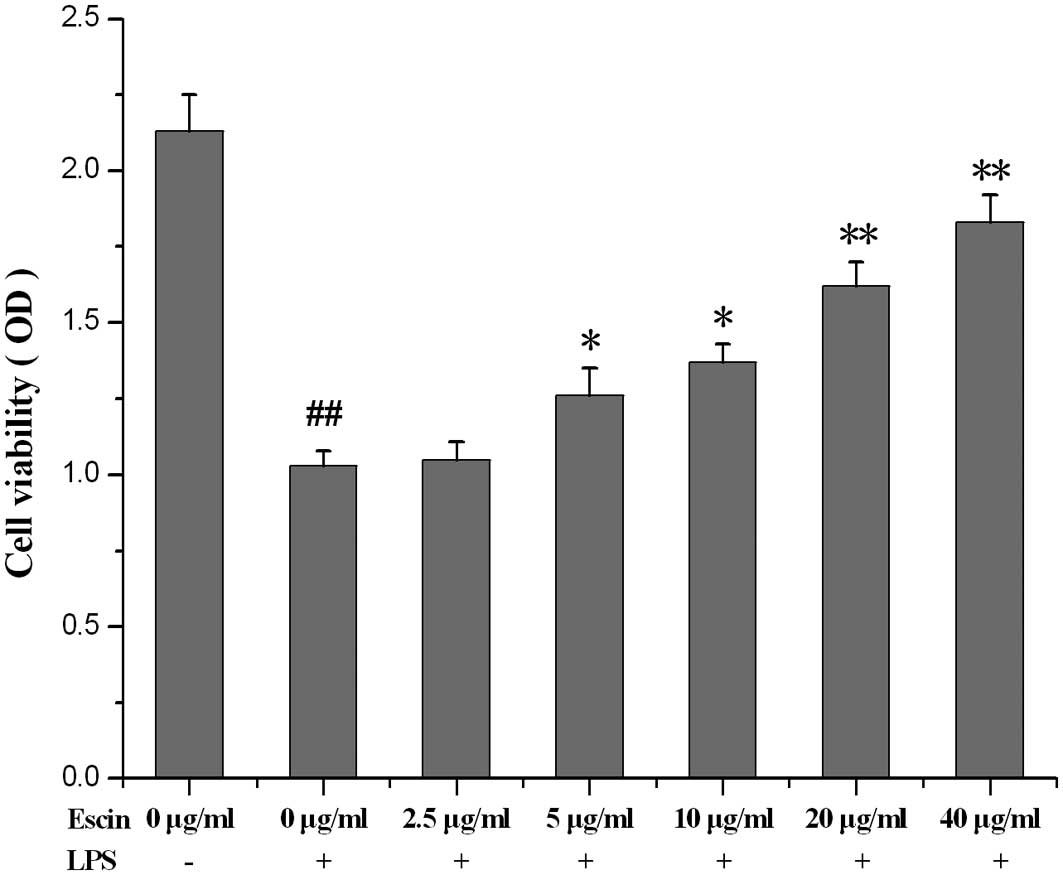
The effect of escin on the viability of
LPS-stimulated hPDLs
Escin (5, 10, 20 and 40 μg/ml) attenuated the
decreased viability induced by LPS in hPDLs in a dose-dependent
manner (Fig. 2).
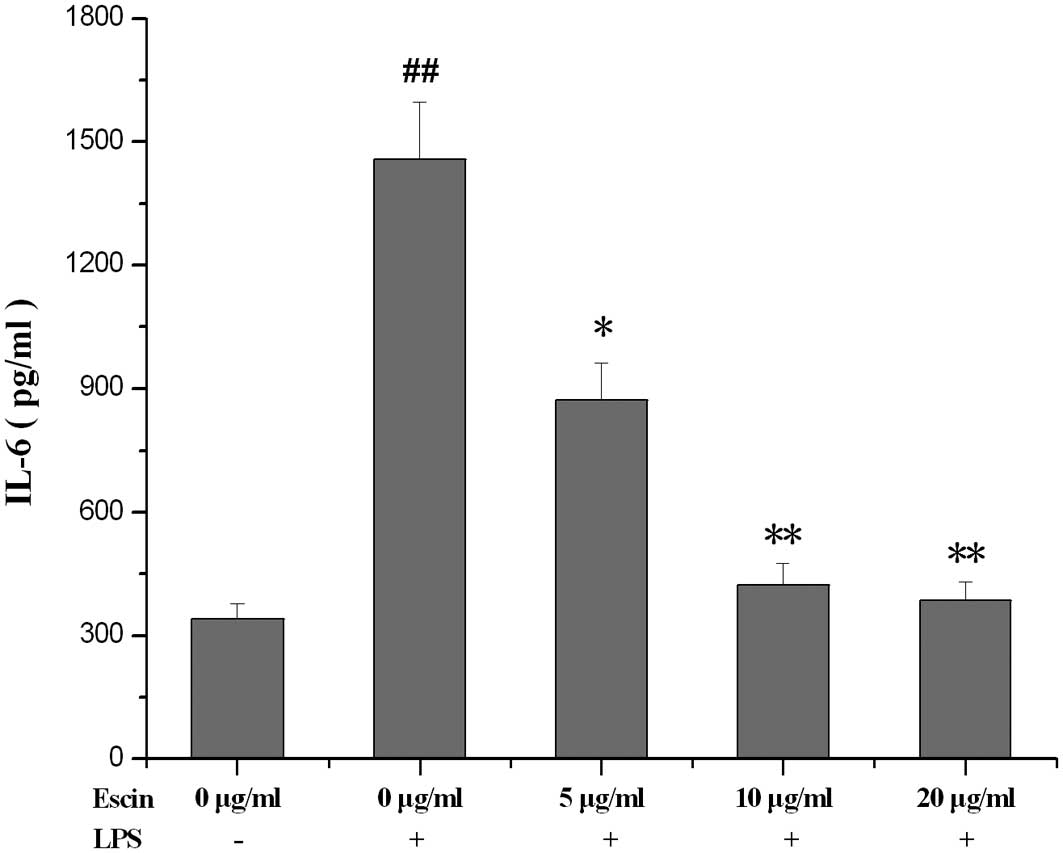
The effect of escin on the IL-6 level in
supernatant
Compared with the control, the IL-6 level in the
supernatant of LPS-treated hPDL culture was significantly
increased. Treatment with escin for 24 h reduced the IL-6 level in
a dose-dependent manner (Fig.
3).
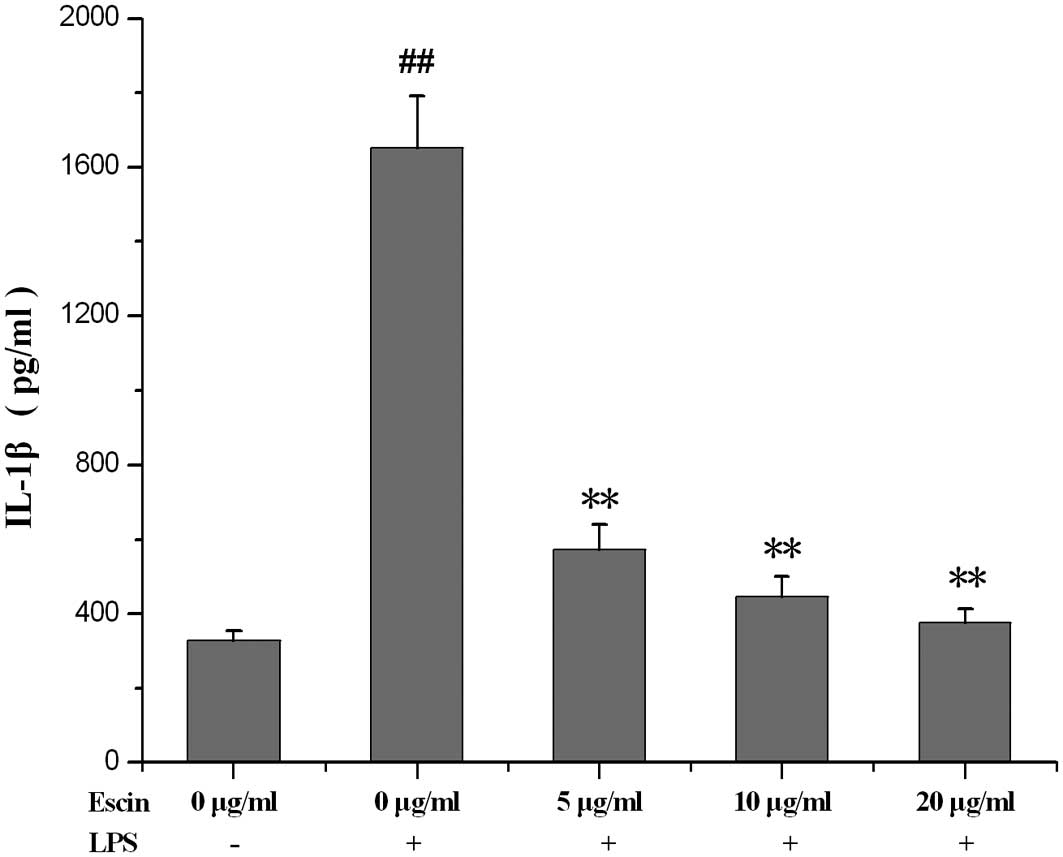
The effect of escin on the IL-1β level in
supernatant
The IL-1β level was increased in the supernatant of
LPS-treated hPDL culture. Following treatment with escin for 24 h,
the IL-1β level in the supernatant significantly decreased
(Fig. 4).
The effect of escin on the TNF-α level in
supernatant
The TNF-α level in the supernatant of LPS-treated
hPDL culture was increased compared to that of the control. Escin
treatment for 24 h significantly lowered the TNF-α level (Fig. 5).
The effect of escin on TLR2 expression in
LPS-treated hPDLs
LPS led to an increase in TLR2 expression. Escin
treatment partially blocked the increase of TLR2 expression induced
by LPS (Fig. 6).
Discussion
Escin, a natural mixture of triterpenoid asponin
isolated from the seed of the horse chestnut, possesses
anti-cancerous, anti-oedematous and anti-inflammatory effects
(10). In this study, escin
increased the viability of hPDLs pre-treated by LPS in a
dose-dependent manner, and the findings suggest that escin may
protect the hPDLs from injury induced by LPS.
Periodontal bacteria stimulate, via TLR2 and TLR4,
the production of proinflammatory cytokines in the host, such as
IL-1β and TNF, which induce alveolar bone resorption (11). TLR2 is responsible for the
predominantly Th2-oriented immune response observed in progressive
periodontitis (4). TLR2 was
associated with exacerbation of P. gingivalis-induced
periodontal bone loss due to TLR2-induced TNF-α-dependent
osteoclastogenesis (12).
TLR2 is important in P. gingivalis-induced
Th2 oriented immune responses. The reduction of the TLR-2 level in
LPS-induced hPDLs treated by escin indicates that escin may play a
role in the Th2 oriented immune response in periodontitis.
Extensive studies have shown that IL-6, TNF-α and
IL-1β (the products of the TLR2 signaling pathway) are the major
products of inflammatory cells and contribute to the progression of
inflammation (13,14). The present results showed that
escin treatment attenuated the inflammatory cytokine responses of
hPDLs in response to P. gingivalis LPS (Figs. 3–5).
TNF-α and IL-1β are considered to be pivotal
cytokines in periodontal disease, since they are expressed in
higher levels in the gingival crevicular fluid and inflamed
periodontal tissues, and cause alveolar bone resorption (15,16).
It was also proven that the inflammatory response and bone loss in
ligature-induced periodontitis in monkeys were reduced by local
inhibition of the IL-1β pathway in periodontal tissues (17).
IL-6 is a major mediator of the host response to
injury and infections by inducing the differentiation of activated
B lymphocytes into antibody-secreting cells (18). The diseased gingiva of
periodontitis patients contained higher levels of IL-6, which is
capable of stimulating bone resorption (19). However, IL-6 secretion was
upregulated by P. gingivalis LPS, the presence of escin
reduced IL-6 secretion in hPDLs, indicating that escin may prevent
periodontal tissue destruction, including alveolar bone resorption.
The possible mechanism by which escin affects cytokine production
is likely to be due to an altered sensitivity of the isolated hPDLs
induced by the LPS of P. gingivalis(14).
In conclusion, escin has an anti-inflammatory effect
in hPDLs. The molecular mechanism of the anti-inflammatory effect
of escin in periodontitis is related to the downregulation of
TNF-α, IL-1β and IL-6 through the TLR2 signaling pathway in the
Th2-oriented immune response, which requires further study.
Acknowledgements
This research was supported by the ‘Taishan
Scholars’ project of the Government of Shandong Province,
China.
References
|
1
|
Bascones-Martinez A, Matesanz-Perez P,
Escribano-Bermejo M, et al: Periodontal disease and diabetes -
Review of the Literature. Med Oral Patol Oral Cir Bucal.
16:722–729. 2011. View Article : Google Scholar
|
|
2
|
Kikkert R, Laine ML, Aarden LA and van
Winkelhoff AJ: Activation of Toll-like receptors 2 and 4 by
Gram-negative periodontal bacteria. Oral Microbiol Immunol.
22:145–151. 2007. View Article : Google Scholar : PubMed/NCBI
|
|
3
|
Re F and Strominger JL: Toll-like receptor
2 (TLR2) and TLR4 differentially activate human dendritric cells. J
Biol Chem. 276:37692–37699. 2001. View Article : Google Scholar : PubMed/NCBI
|
|
4
|
Rojo-Botello NR, García-Hernández AL and
Moreno-Fierros L: Expression of toll-like receptors 2, 4 and 9 is
increased in gingival tissue from patients with type 2 diabetes and
chronic periodontitis. J Periodontal Res. 47:62–73. 2012.
View Article : Google Scholar : PubMed/NCBI
|
|
5
|
Quintero JC, Piesco NP and Langkamp HH:
LPS responsiveness in periodontal ligament cells is regulated by
tumor necrosis factor-a. J Dent Res. 74:1802–1811. 1995. View Article : Google Scholar : PubMed/NCBI
|
|
6
|
Wang T, Fu FH, Zhang LM, et al: Effect of
of escin on acute inflammation and the immune system in mice.
Pharmacol Rep. 61:697–704. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
7
|
Somerman MJ, Archer SY, Imm GR and Forster
RA: A comparative study of human periodontal ligament cells and
gingival fibroblasts in vitro. J Dent Res. 67:66–70. 1988.
View Article : Google Scholar : PubMed/NCBI
|
|
8
|
Pi SH, Jeong GS, Oh HW, et al: Heme
oxygenase-1 mediates nicotine- and lipopolysaccharide-induced
expression of cyclooxygenase-2 and inducible nitric oxide synthase
in human periodontal ligament cells. J Periodont Res. 45:177–183.
2010. View Article : Google Scholar : PubMed/NCBI
|
|
9
|
Zhang LM, Fu FH, Zhang XM, et al: Escin
attenuates cognitive deficits and hippocampal injury after
transient global cerebral ischemia in mice via regulating certain
inflammatory genes. Neurochem Int. 57:119–127. 2010. View Article : Google Scholar
|
|
10
|
Yang XW, Zhao J, Cui JR and Guo W: Studies
on the biotransformation of escin Ia by human intestinal bacteria
and the anti-tumor activities of desacylla I. Beijing Da Xue Xue
Bao. 36:31–35. 2004.(In Chinese).
|
|
11
|
Graves DT and Cochran D: The contribution
of interleukin-1 and tumor necrosis factor to periodontal tissue
destruction. J Periodontol. 74:391–401. 2003. View Article : Google Scholar : PubMed/NCBI
|
|
12
|
Ukai T, Yumoto H, Gibson FC and Genco CA:
Macrophage-elicited osteoclastogenesis in response to bacterial
stimulation requires Toll-like receptor 2-dependent tumor necrosis
factor-α production. Infect Immun. 76:812–819. 2008.PubMed/NCBI
|
|
13
|
Cheung KL, Khor TO and Kong AN:
Synergistic effect of combination of phenethyl isothiocyanate and
sulforaphane or curcumin and sulforaphane in the inhibition of
inflammation. Pharm Res. 26:224–231. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
14
|
Antonietta R, Rossella P, Luigi G, Marco
A, Nazario B and Maria AT: Effect of metronidazole and modulation
of cytokine production on human periodontal ligament cells. Int
Immunopharmacol. 10:744–750. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
15
|
Chiang CY, Kyritsis G, Graves DT and Amar
S: Interleukin-1 and tumor necrosis factor activities partially
account for calvarial bone resorption induced by local injection of
lipopolysaccharide. Infect Immun. 67:4231–4236. 1999.PubMed/NCBI
|
|
16
|
Gemmell E, Marshall RI and Seymour GJ:
Cytokines and prostaglandins in immune homeostasis and tissue
destruction in periodontal disease. Periodontol 2000. 14:112–143.
1997. View Article : Google Scholar : PubMed/NCBI
|
|
17
|
Assuma R, Oates T, Cochran D, Amar S and
Graves DT: IL-1 and TNF antagonists inhibit the inflammatory
response and bone loss in experimental periodontitis. J Immunol.
160:403–409. 1998.PubMed/NCBI
|
|
18
|
Muraguchi A, Hirano T, Tang B, et al:
Essential role of B-cell stimulatory factor 2 (BSF-2/IL-6) for the
terminal differentiation of B-cells. J Exp Med. 167:332–344. 1988.
View Article : Google Scholar : PubMed/NCBI
|
|
19
|
Takahashi K, Takashiba S, Nagai A, et al:
Assessment of interleukin-6 in the pathogenesis of periodontal
disease. J Periodontol. 65:147–153. 1994. View Article : Google Scholar : PubMed/NCBI
|