Introduction
Prostate cancer is the second leading cause of
cancer-related mortality in males worldwide (1–3). In
the Western world, prostate cancer is the second leading cause of
cancer-related mortality in males due to its high prevalence and
metastatic rate (4–5). In Taiwan, prostate cancer is the
seventh leading cause of cancer-related mortality (6). Prostate cancer has a poor prognosis
due in part to tumor invasion and migration. The propensity of
prostate cancer cells to metastasize to the bone greatly reduces
the effectiveness of available treatment options (7–9).
It is well documented that cancer patients succumb
to the disease primarily as a result of metastasis of the cancer to
distant organs (10,11). Metastasis is a multistep process,
including cancer cell adhesion, migration, intravasation,
extravasation and colonization to a secondary site (12–15).
Thus, disruption of the metastatic process is key to reducing
cancer mortality in patients. Mitogen-activated protein kinase
(MAPK) signaling contributes to human cancer cell migration,
invasion and metastasis (16–18).
The molecular mechanics of cancer cell migration has been reported
to involve cell cytoskeletal remodeling and focal adhesion dynamics
(19,20). Matrix metalloproteinases (MMPs) are
important in cell growth, angiogenesis, invasion and metastasis of
cancer cells (16,21–23).
Therapeutic agents that are capable of inhibiting MMPs may be
efficacious. However, studies of MMP inhibitors, including
batimastat (BB-94) for rat mammary carcinoma (24,25)
and FYK-1388 for human fibrosarcoma have thus far been
disappointing due to serious adverse effects (26). Therefore, much attention has been
focused on developing new MMP inhibitors.
Crude extract of Euphorbia formosana (CEEF)
has been used for the treatment of several diseases in the Chinese
population (27). However, the
effects of CEEF on the migration and invasion of prostate cancer
cells has not been examined. In the present study, we investigated
the effects of CEEF on the inhibition of cell migration and
invasion in DU145 human prostate cancer cells. We demonstrated that
CEEF inhibited cell migration and invasion in DU145 cells by
suppressing the MAPK signaling pathway resulting in the inhibition
of MMP-2/9.
Materials and methods
Cell culture and reagents
The DU145 human prostate cancer cell line was
purchased from the Food Industry Research and Development Institute
(Hsinchu, Taiwan). Minimum Essential Medium (MEM), fetal bovine
serum (FBS), L-glutamine, penicillin-streptomycin and trypsin-EDTA
were obtained from Gibco BRL (Carlsbad, CA, USA). Dimethyl
sulfoxide (DMSO), propidium iodide (PI), trypan blue and Tris-HCl
were purchased from Sigma (St. Louis, MO, USA). The crude extract
of Euphorbia formosana was kindly provided by Dr Kuo
(Department of Chinese Pharmaceutical Sciences and Chinese Medicine
Resources, China Medical University, Taichung, Taiwan) (16).
Cell viability
DU145 cells (2×105 cells/well) were
seeded in a 12-well plate at 37°C with 5% CO2 in MEM
(Gibco BRL), supplemented with 10% FBS, 100 mg/ml streptomycin and
100 U/ml penicillin for 24 h. The cells were incubated with 100
μg/ml CEEF or vehicle control (0.5% DMSO) for 0, 6, 12, 24, 48 and
72 h. The cells were then detached with trypsin, harvested, washed
with PBS and stained with trypan blue. The cell number for each
treatment was manually counted using a hemocytometer and presented
as a percentage of viable cells per ml (28).
Wound healing assay
DU145 cells at a density of 1×106
cells/well were cultured in 10 cm petri dishes until cells reached
90–95% confluency. The surface of the plate was then scratched with
a pipette tip and washed three times. Cells were incubated in the
absence and presence of CEEF for 0, 6, 12 and 24 h and then imaged
using a Nikon Eclipse TS100 microscope (29).
Cell migration and invasion assays
The migration of DU145 cells was determined using
Matrigel-uncoated transwell cell culture chambers (8 μm pore size)
as described previously (30).
DU145 cells were incubated with 0, 25, 50 and 75 μg/ml of CEEF for
24 and 48 h. The upper surface of the membrane containing the
non-invaded cells were removed and the invaded cells on the lower
surface of the membrane were fixed and stained with hematoxylin and
eosin (H&E). Migrating cells in the chamber were counted. For
the determination of cell invasion, the same migration assay was
used although the membrane was coated with Matrigel (29).
Western blotting assay for the detection
of migration and invasion associated proteins in DU145 cells
DU145 cells at a density of 5×106 cells
were maintained in a 6-well plate overnight and then treated with
100 μg/ml CEEF for 0, 6, 12, 24, 48 and 72 h. Cells were harvested
and lysed with lysis buffer containing 40 mM Tris-HCl (pH 7.4), 10
mM EDTA, 120 mM NaCl, 1 mM dithiothreitol and 0.1% Nonide P-40. The
protein concentration of the lysate was determined using the Bio
Red kit. Proteins from each sample were separated using sodium
dodecyl sulfate-PAGE and transferred to nitrocellulose membranes
(Amersham Pharmacia Biotech, Piscataway, NJ, USA) by
electroblotting. The membranes were probed with primary antibodies
for 24 h and then washed and stained with secondary antibody for
enhanced chemiluminescence (NEN Life Science Products, Inc.,
Boston, MA, USA) as previously described (29).
Real-time PCR of mRNA expression levels
of MMP-2/9 in DU145 cells
Cells (1×106 cells/well) were placed in
6-well plates and incubated with CEEF (50 μg/ml) for 24 h. The
cells were then collected and total RNA was extracted, as
previously described (31).
Collected RNA samples were reverse transcribed using the High
Capacity cDNA Reverse Transcription kit at 42°C for 30 min
according to the manufacturer’s instructions (Applied Biosystems,
Foster City, CA, USA). The primers used were: MMP-2 forward:
CCCCAGACAGGTGATCTTGAC and reverse: GCTTGCGAGGGAAGAAGTTG; MMP-9
forward: CGCTGGGCTTAGATCATTCC and reverse:
AGGTTGGATACATCACTGCATTAGG; GAPDH forward: ACACCCACTCCTCCACCTTT and
reverse: TAGCCAAATTCGTTGTCATACC. An Applied Biosystems 7300
Real-Time PCR system was used for each assay in triplicate and
expression fold changes were derived using the comparative CT
(threshold cycle) method (31).
Statistical analysis
Experiments were repeated at least three times.
Results are shown as the mean ± SD. The Student’s t-test was
performed to determine the statistical difference between the
control- and CEEF-treated groups. P<0.05 was considered to
indicate a statistically significant result.
Results
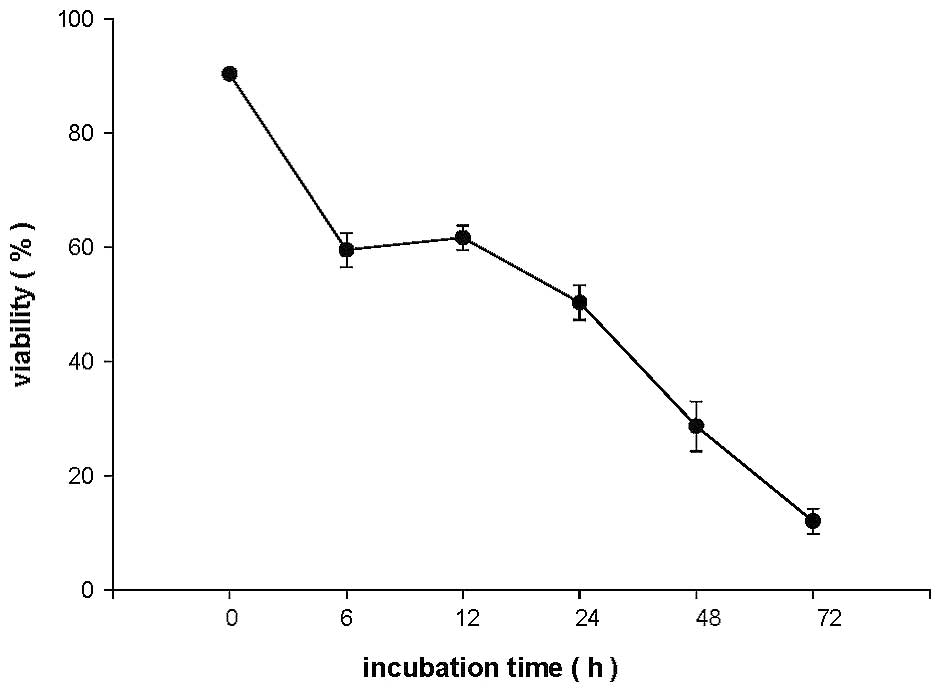
CEEF affects the percentage of viable
DU145 cells
DU145 cells were treated with 100 μg/ml of CEEF for
0, 6, 12, 24, 48 and 72 h, and the percentage of viable cells was
determined by trypan blue exclusion assay. The results are shown in
Fig. 1. Fewer viable cells were
present with increasing time in CEEF-treated cells when compared
with controls.
CEEF inhibits migration of DU145
cells
Monolayers of DU145 cells were scratched and treated
with 50 and 75 μg/ml of CEEF for 0, 6, 12 and 24 h, and allowed to
recover to determine the rate of migration using the wound healing
assay (Fig. 2). CEEF suppressed
the migration of DU145 cells. The time required for wound closure
of DU145 cells treated with CEEF for 24 h was significantly longer
than that for the control cells.
CEEF inhibits the migration and invasion
of DU145 cells
The effects of CEEF on the migration and invasion of
DU145 cells are shown in Fig. 3.
CEEF at concentrations between 25, 50 and 75 μg/ml significantly
suppressed the migration of DU145 cells. The percentage of
migration inhibition was 40–84% and 60–92% for cells incubated with
CEEF for 24 and 48 h, respectively, when compared with the control
cells. CEEF inhibited the invasion of DU145 cells (Fig. 3B). Percent inhibition at CEEF
concentrations of 25, 50 and 75 μg/ml were 60–90% and 66–94% for 24
and 48 h, respectively, when compared with the controls.
CEEF alters the levels of proteins
associated with migration and invasion in DU145 cells
Data shown in Fig.
4A demonstrate that CEEF decreased the levels of ERK1/2, p38,
JNK, SOS1, PKC and PI3K protein expression. Protein levels of
MMP-2/9 were also reduced by CEEF treatment in DU145 cells
(Fig. 4B). Based on these results,
CEEF inhibits the ERK and PI3K/AKT signaling pathways which leads
to the suppression of MMP-2/9 expression in DU145 cells.
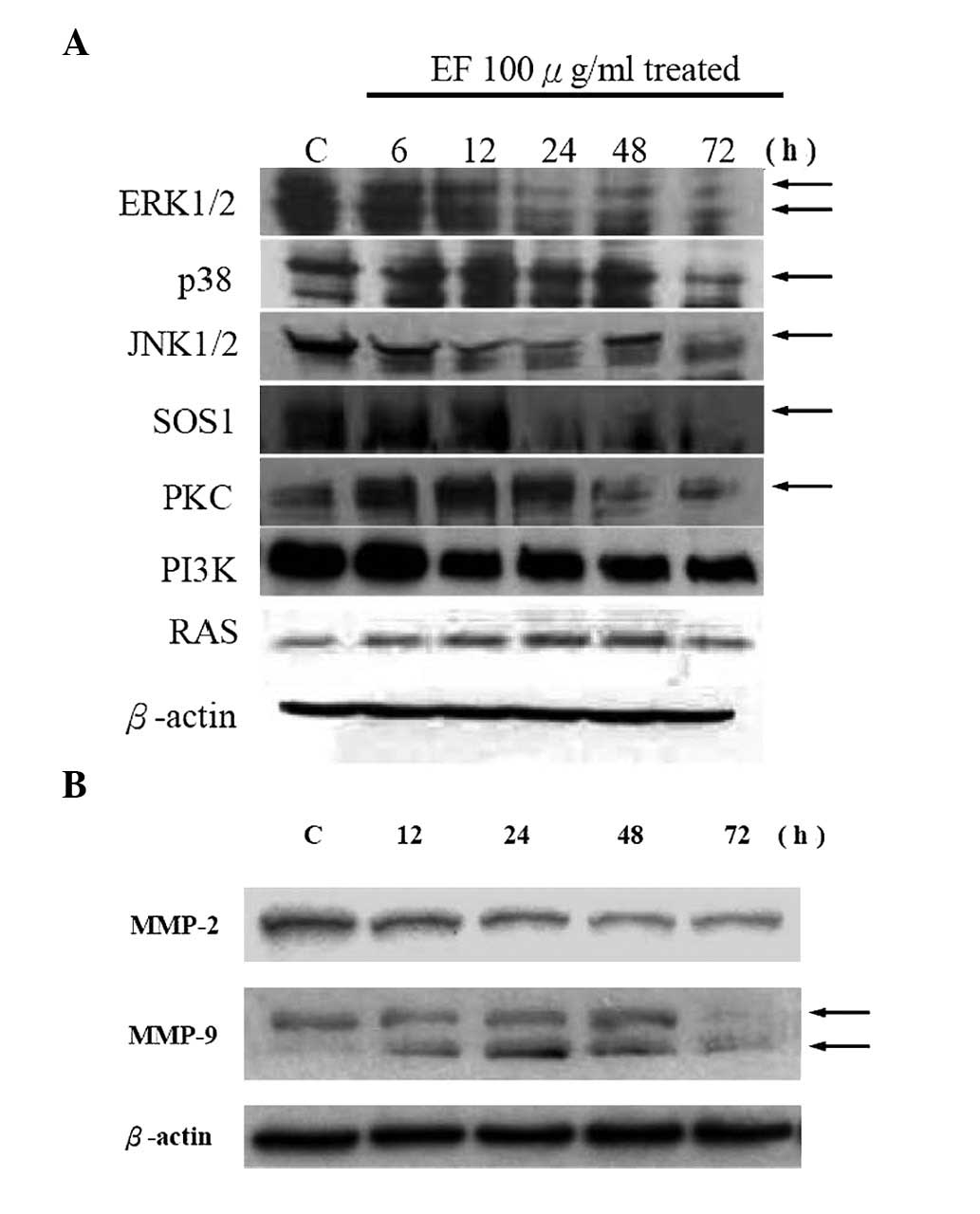 | Figure 4CEEF affects the levels of proteins
associated with the migration and invasion of DU145 cells. Cells
were treated with CEEF at 0 and 100 μg/ml for 0, 6, 12, 24, 48 and
72 h and then collected. The total protein was obtained as
described in Materials and methods. The levels of (A) ERK, JNK,
p38, SOS1, PKC and PI3K, and (B) MMP-2/9 expression were estimated
by Western blotting as described in Materials and methods. CEEF,
crude extract of Euphorbia formosana; MMP, matrix
metalloproteinases. |
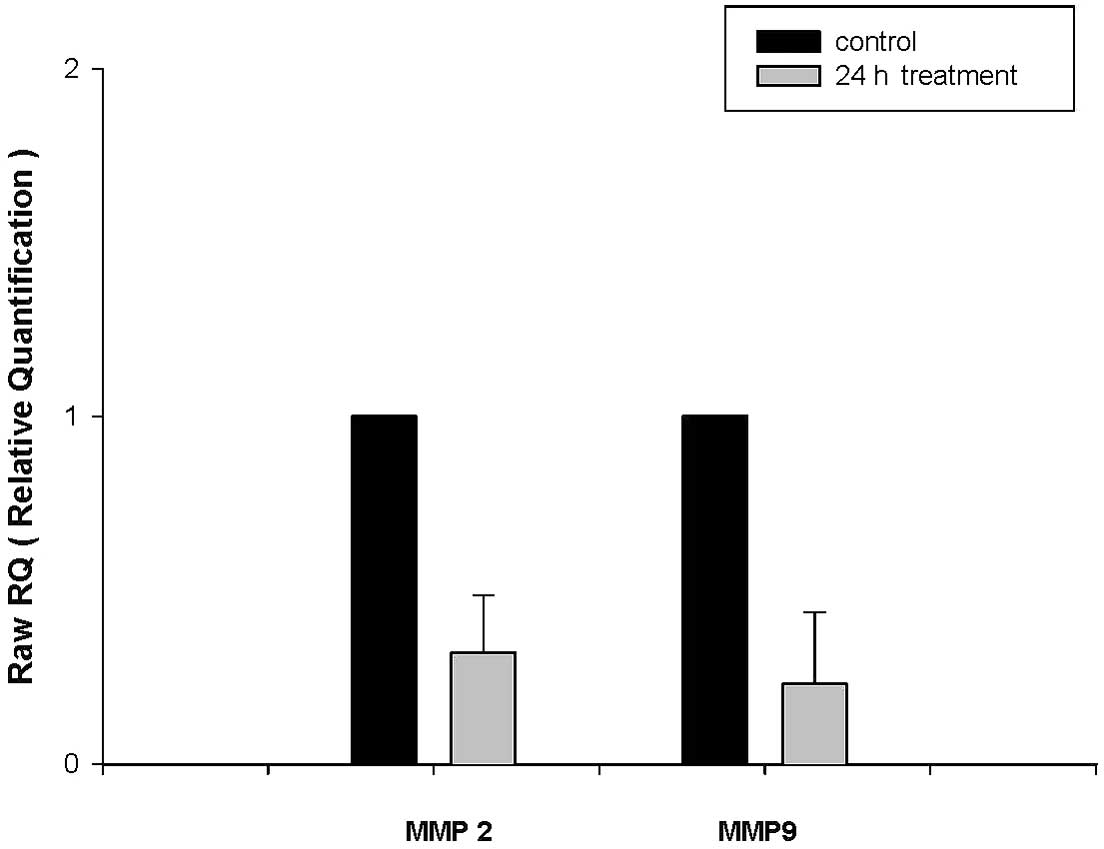
CEEF alters mRNA expression levels of
MMP-2/9 in DU145 cells
Cells incubated with 100 μg/ml CEEF for 24 h and
mRNA expression levels of MMP-2/9 were determined using real-time
PCR. Fig. 5 demonstrates that CEEF
significantly inhibits gene expression levels of MMP-2/9.
Discussion
Metastasis is one of the major causes of
cancer-related mortality (31–35).
At present, there is an intense effort to identify potential
therapeutic agents that inhibit metastasis (35–38).
We previously demonstrated that CEEF treatment significantly
inhibits cell proliferation and induces cell cycle arrest and
apoptosis in DU145 prostate cancer cells (data not shown). In the
present study, we investigated the mechanisms of the anti-migratory
and anti-invasive effects of CEEF in DU145 prostate cancer cells.
At the CEEF concentrations used, no effects on the growth rate of
DU145 cells were observed (Fig.
1A). However, anti-migratory and anti-invasive effects were
observed at CEEF concentrations which did not significantly inhibit
cell growth (between 12.5 and 200 μg/ml). We conclude that the
effects of CEEF on migration and invasion were not due to
cytotoxicity.
It is well known that the uncontrolled degradation
of the extracellular matrix and basement is associated with tumor
cell invasion and migration, and that MMPs are important in cancer
cell migration and invasion (23,27).
In the present study, the secretion levels of MMP-2/9 (Figs. 4B and 5) were downregulated by CEEF treatment
and these effects were concentration- and time-dependent. Western
blotting also demonstrated that MMP-2/9 protein levels were reduced
by CEEF (Fig. 5B). Overexpression
of MMP-2/9 has been observed in different human cancer types and
those proteins are associated with a high potential for metastasis
(31,39,40).
We hypothesize that a significant downregulation of MMP-2/9
secretion levels in CEEF-treated DU145 cells may be involved in
reducing protein levels of MMP-2/9. In addition, CEEF reduced the
protein levels of ERK1/2, JNK1/2, p38, SOS1 and PI3K (Fig. 4A). MAPK pathways involving ERK,
JNK, p38 and ERK signaling have been reported to upregulate the
expression of MMPs (31,41).
A model of the potential action of CEEF on the
migration and invasion of DU145 cells is shown in Fig. 6. CEEF may inhibit the migration and
invasion of DU145 cells via the MAPK (ERK1/2, JNK1/2 and p38)
signaling pathway resulting in the subsequent downregulation of
MMP-2/9 expression levels. Future studies are needed to address
whether CEEF inhibits tumor migration and invasion in animal
models.
Acknowledgements
This study was supported in part by research grants
CMU99-COL-06-1 and CMU99-COL-06-2 from China Medical
University.
References
|
1
|
Baumunk D, Blana A, Ganzer R, Henkel T,
Köllermann J, Roosen A, Machtens S, Salomon G, Sentker L, Witzsch
U, Köhrmann KU and Schostak M: Focal prostate cancer therapy:
Capabilities, limitations and prospects. Urologe A. Oct
18–2012.(Epub ahead of print) (In German).
|
|
2
|
Obertova Z, Brown C, Holmes M and
Lawrenson R: Prostate cancer incidence and mortality in rural men -
a systematic review of the literature. Rural Remote Health.
12:20392012.PubMed/NCBI
|
|
3
|
van der Meer S, Löwik SA, Hirdes WH,
Nijman RM, Van der Meer K, Hoekstra-Weebers JE and Blanker MH:
Prostate specific antigen testing policy worldwide varies greatly
and seems not to be in accordance with guidelines: a systematic
review. BMC Fam Pract. 13:1002012.
|
|
4
|
Cheng L, Montironi R, Bostwick DG,
Lopez-Beltran A and Berney DM: Staging of prostate cancer.
Histopathology. 60:87–117. 2012. View Article : Google Scholar
|
|
5
|
Provenzano M: New biomarkers in prostate
cancer. Praxis (Bern 1994). 101:115–121. 2012.(In German).
|
|
6
|
Ou YC, Chen JT, Cheng CL, Ho HC and Yang
CR: Radical prostatectomy for prostate cancer patients with
prostate-specific antigen >20 ng/ml. Jpn J Clin Oncol.
33:574–579. 2003. View Article : Google Scholar
|
|
7
|
Mener DJ: Prostate specific antigen
reduction following statin therapy: Mechanism of action and review
of the literature. IUBMB Life. 62:584–590. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
8
|
Roy M, Kung HJ and Ghosh PM: Statins and
prostate cancer: role of cholesterol inhibition vs. prevention of
small GTP-binding proteins. Am J Cancer Res. 1:542–561.
2011.PubMed/NCBI
|
|
9
|
Zhang Y, Ma B and Fan Q: Mechanisms of
breast cancer bone metastasis. Cancer Lett. 292:1–7. 2010.
View Article : Google Scholar : PubMed/NCBI
|
|
10
|
Karakiewicz PI and Hutterer GC: Predictive
models and prostate cancer. Nat Clin Pract Urol. 5:82–92. 2008.
View Article : Google Scholar : PubMed/NCBI
|
|
11
|
Karlou M, Tzelepi V and Efstathiou E:
Therapeutic targeting of the prostate cancer microenvironment. Nat
Rev Urol. 7:494–509. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
12
|
Khamis ZI, Iczkowski KA and Sang QX:
Metastasis suppressors in human benign prostate, intraepithelial
neoplasia, and invasive cancer: their prospects as therapeutic
agents. Med Res Rev. 32:1026–1077. 2012. View Article : Google Scholar
|
|
13
|
Minato N, Takada T, Koga M and Sugao H:
Prostate cancer with disseminated carcinomatosis of bone marrow
initially presenting with disseminated intravascular coagulation
syndrome: a case report. Hinyokika Kiyo. 58:249–253. 2012.(In
Japanese).
|
|
14
|
Payne H, Khan A, Chowdhury S and Davda R:
Hormone therapy for radiorecurrent prostate cancer. World J Urol.
Sep 21–2012.(Epub ahead of print).
|
|
15
|
Smith BN and Odero-Marah VA: The role of
Snail in prostate cancer. Cell Adh Migr. 6:433–441. 2012.
View Article : Google Scholar : PubMed/NCBI
|
|
16
|
Hour MJ, Tsai SC, Wu HC, Lin MW, Chung JG,
Wu JB, Chiang JH, Tsuzuki M and Yang JS: Antitumor effects of the
novel quinazolinone MJ-33: Inhibition of metastasis through the
MAPK, AKT, NF-κB and AP-1 signaling pathways in DU145 human
prostate cancer cells. Int J Oncol. Jul 18–2012.(Epub ahead of
print).
|
|
17
|
Lee KH, Choi EY, Hyun MS and Kim JR:
Involvement of MAPK pathway in hypoxia-induced up-regulation of
urokinase plasminogen activator receptor in a human prostatic
cancer cell line, PC3MLN4. Exp Mol Med. 36:57–64. 2004. View Article : Google Scholar : PubMed/NCBI
|
|
18
|
Mulholland DJ, Kobayashi N, Ruscetti M,
Zhi A, Tran LM, Huang J, Gleave M and Wu H: Pten loss and RAS/MAPK
activation cooperate to promote EMT and metastasis initiated from
prostate cancer stem/progenitor cells. Cancer Res. 72:1878–1889.
2012. View Article : Google Scholar : PubMed/NCBI
|
|
19
|
Guo LW, Gao L, Rothschild J, Su B and
Gelman IH: Control of protein kinase C activity, phorbol
ester-induced cytoskeletal remodeling, and cell survival signals by
the scaffolding protein SSeCKS/GRAVIN/AKAP12. J Biol Chem.
286:38356–38366. 2011. View Article : Google Scholar
|
|
20
|
Marques RB, Dits NF, Erkens-Schulze S, van
Ijcken WF, van Weerden WM and Jenster G: Modulation of androgen
receptor signaling in hormonal therapy-resistant prostate cancer
cell lines. PLoS One. 6:e231442011. View Article : Google Scholar : PubMed/NCBI
|
|
21
|
Kanoh Y, Ohtani H, Egawa S, Baba S and
Akahoshi T: Changes of proteases and proteinase inhibitors in
androgen-dependent advanced prostate cancer patients with
alpha2-macroglobulin deficiency. Clin Lab. 58:217–225. 2012.
|
|
22
|
Ramsay AK, McCracken SR, Soofi M, Fleming
J, Yu AX, Ahmad I, Morland R, Machesky L, Nixon C, Edwards DR,
Nuttall RK, Seywright M, Marquez R, Keller E and Leung HY: ERK5
signalling in prostate cancer promotes an invasive phenotype. Br J
Cancer. 104:664–672. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
23
|
Xiao LJ, Lin P, Lin F, Liu X, Qin W, Zou
HF, Guo L, Liu W, Wang SJ and Yu XG: ADAM17 targets MMP-2 and MMP-9
via EGFR-MEK-ERK pathway activation to promote prostate cancer cell
invasion. Int J Oncol. 40:1714–1724. 2012.PubMed/NCBI
|
|
24
|
Low JA, Johnson MD, Bone EA and Dickson
RB: The matrix metalloproteinase inhibitor batimastat (BB-94)
retards human breast cancer solid tumor growth but not ascites
formation in nude mice. Clin Cancer Res. 2:1207–1214. 1996.
|
|
25
|
Wojtowicz-Praga SM, Dickson RB and Hawkins
MJ: Matrix metalloproteinase inhibitors. Invest New Drugs.
15:61–75. 1997. View Article : Google Scholar
|
|
26
|
Shinoda K, Shibuya M, Hibino S, Ono Y,
Matsuda K, Takemura A, Zou D, Kokubo Y, Takechi A and Kudoh S: A
novel matrix metalloproteinase inhibitor, FYK-1388 suppresses tumor
growth, metastasis and angiogenesis by human fibrosarcoma cell
line. Int J Oncol. 22:281–288. 2003.PubMed/NCBI
|
|
27
|
Yu CC, Hsieh CR, Hsiao G, Chen PY, Chang
ML, Yin HW, Lee TH and Lee CK: Regulated expressions of MMP-2, -9
by diterpenoids from Euphorbia formosana Hayata. Molecules.
17:2082–2090. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
28
|
Lee MH, Yang HI, Lu SN, Jen CL, You SL,
Wang LY, Wang CH, Chen WJ and Chen CJ: Chronic hepatitis C virus
infection increases mortality from hepatic and extrahepatic
diseases: a community-based long-term prospective study.
R.E.V.E.A.L-HCV Study Group. J Infect Dis. 206:469–477. 2012.
View Article : Google Scholar : PubMed/NCBI
|
|
29
|
Lu CC, Yang JS, Chiang JH, Hour MJ,
Amagaya S, Lu KW, Lin JP, Tang NY, Lee TH and Chung JG: Inhibition
of invasion and migration by newly synthesized quinazolinone MJ-29
in human oral cancer CAL 27 cells through suppression of MMP-2/9
expression and combined down-regulation of MAPK and AKT signaling.
Anticancer Res. 32:2895–2903. 2012.PubMed/NCBI
|
|
30
|
Chen YY, Chiang SY, Lin JG, Ma YS, Liao
CL, Weng SW, Lai TY and Chung JG: Emodin, aloe-emodin and rhein
inhibit migration and invasion in human tongue cancer SCC-4 cells
through the inhibition of gene expression of matrix
metalloproteinase-9. Int J Oncol. 36:1113–1120. 2010.PubMed/NCBI
|
|
31
|
Ma CY, Ji WT, Chueh FS, Yang JS, Chen PY,
Yu CC and Chung JG: Butein inhibits the migration and invasion of
SK-HEP-1 human hepatocarcinoma cells through suppressing the ERK,
JNK, p38, and uPA signaling multiple pathways. J Agric Food Chem.
59:9032–9038. 2011. View Article : Google Scholar
|
|
32
|
Hu WG, Li JW, Feng B, Beveridge M, Yue F,
Lu AG, Ma JJ, Wang ML, Guo Y, Jin XL and Zheng MH: Vascular
endothelial growth factors C and D represent novel prognostic
markers in colorectal carcinoma using quantitative image analysis.
Eur Surg Res. 39:229–238. 2007. View Article : Google Scholar
|
|
33
|
Ma D, Gerard RD, Li XY, Alizadeh H and
Niederkorn JY: Inhibition of metastasis of intraocular melanomas by
adenovirus-mediated gene transfer of plasminogen activator
inhibitor type 1 (PAI-1) in an athymic mouse model. Blood.
90:2738–2746. 1997.PubMed/NCBI
|
|
34
|
Maarouf A, Adham M, Scoazec JY and
Partensky C: Mixed hepato/cholangiocarcinoma with paraneoplastic
hypercalcemia. J Hepatobiliary Pancreat Surg. 15:224–227. 2008.
View Article : Google Scholar : PubMed/NCBI
|
|
35
|
Yang D, Gu T, Wang T, Tang Q and Ma C:
Effects of osthole on migration and invasion in breast cancer
cells. Biosci Biotechnol Biochem. 74:1430–1434. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
36
|
Lin SS, Lai KC, Hsu SC, Yang JS, Kuo CL,
Lin JP, Ma YS, Wu CC and Chung JG: Curcumin inhibits the migration
and invasion of human A549 lung cancer cells through the inhibition
of matrix metalloproteinase-2 and -9 and Vascular Endothelial
Growth Factor (VEGF). Cancer Lett. 285:127–133. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
37
|
Liu JW, Cai MX, Xin Y, Wu QS, Ma J, Yang
P, Xie HY and Huang DS: Parthenolide induces proliferation
inhibition and apoptosis of pancreatic cancer cells in vitro. J Exp
Clin Cancer Res. 29:1082010. View Article : Google Scholar : PubMed/NCBI
|
|
38
|
Qin JM, Yin PH, Li Q, Sa ZQ, Sheng X, Yang
L, Huang T, Zhang M, Gao KP, Chen QH, Ma JW and Shen HB: Anti-tumor
effects of brucine immuno-nanoparticles on hepatocellular
carcinoma. Int J Nanomedicine. 7:369–379. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
39
|
Weigel MT, Kramer J, Schem C, Wenners A,
Alkatout I, Jonat W, Maass N and Mundhenke C: Differential
expression of MMP-2, MMP-9 and PCNA in endometriosis and
endometrial carcinoma. Eur J Obstet Gynecol Reprod Biol. 160:74–78.
2012. View Article : Google Scholar : PubMed/NCBI
|
|
40
|
Zheng W, Zhang Y, Ma D, Shi Y, Liu C and
Wang P: ()Equol inhibits invasion in prostate cancer DU145 cells
possibly via down-regulation of matrix metalloproteinase-9, matrix
metalloproteinase-2 and urokinase-type plasminogen activator by
antioxidant activity. J Clin Biochem Nutr. 51:61–67. 2012.
View Article : Google Scholar
|
|
41
|
Ho YT, Yang JS, Li TC, Lin JJ, Lin JG, Lai
KC, Ma CY, Wood WG and Chung JG: Berberine suppresses in vitro
migration and invasion of human SCC-4 tongue squamous cancer cells
through the inhibitions of FAK, IKK, NF-kappaB, u-PA and MMP-2 and
-9. Cancer Lett. 279:155–162. 2009. View Article : Google Scholar : PubMed/NCBI
|