Introduction
Psoriasis, which is regarded as a T-cell-mediated
inflammatory skin disease, is characterized by hyperproliferation
and poor differentiation of epidermal keratinocytes, affecting ≤2%
of the population in Northern European countries. It is defined as
an immunological disease, that is coupled with prominently
increased vascularization of the skin, fibroblast activation and
leucocyte infiltration. The underlying pathogenic mechanisms of
this condition have not, however, been entirely clarified.
Recently, numerous studies have confirmed that T helper 17 (Th17)
cells and the inflammatory factors it produces, including
interleukin (IL)-17, IL-22 and IL-23, are detected in psoriatic
skin lesions, serum and are implicated in psoriasis pathogenesis
(1–8).
Currently, it is well established that omega-3
long-chain polyunsaturated fatty acids (PUFAs) have a potential
role in the treatment of numerous diseases, including non-alcoholic
fatty liver disease (9), chronic
experimental colitis (10),
diabetes (11) and pancreatic
ductal adenocarcinoma (12). n-3
PUFAs exert their beneficial effects by inhibiting the actions of
numerous different cytokines in disease progression and are also
essential fatty acids to normal physiological functioning. n-3
PUFAs include alpha-linolenic acid (ALA), eicosapentaenoic acid
(EPA) and docosahexaenoic acid (DHA). Evidence suggests that n-3
PUFAs are promising candidates as a safe adjuvant holistic therapy
for psoriasis, either as an active anti-inflammatory agent by
itself or as a dual action synergistical enhancer for other
anti-psoriatic treatments. The roles of n-3 PUFAs are highly
diverse, including the maturation and differentiation of the
stratum corneum, the inhibition of proinflammatory eicosanoids and
cytokines, and the maintenance of the stratum corneum permeability
barrier (13). Mammals are not
able to endogenously synthesize n-3 PUFAs nor convert n-6 FA to n-3
PUFAs, on account of lacking n-3 desaturase, the enzyme that
catalyzes this reaction. Therefore, the majority of the studies
with n-3 PUFAs in psoriasis utilize oral, intravenous and topical
preparations. In the majoriy of these investigations, n-3 PUFAs are
associated with an observed improvement in patient mean Psoriasis
Area and Severity Index (PASI) score, as well as in clinical
symptoms, including pruritus. Despite the majority of studies
adopting more mature models, the formulation associated with the
fat content isocaloric diet is confounded. Therefore, it is
difficult to control the fat composition between the control and
experimental diets.
In 2004, Kang et al generated transgenic
fat-1 mice based on C57BL6 mice, carrying the fat-1 gene, which
encodes for an n-3 desaturase from Caenorhabditis elegans
(14). Fat-1 transgenic mice have
an n-6/n-3 fatty acid ratio of 1:1 compared with wild-type mice
with a ratio of 20–30:1. In fat-1 transgenic mice, n-3 fatty acids
are endogenously synthesized, which leads to an increase in n-3
PUFAs and a decrease in n-6 fatty acids, and subsequently, a
reduction in the n-6/n-3 fatty acid ratio. As a result, the fat-1
mouse model may avoid the potential confounding factors associated
with other models, including diet, because the same diet is
provided to the wild type (WT) and fat-1 mice. Therefore, the fat-1
mouse represents a significant advance in the development of a more
sophisticated model to investigate the effect of n-3 PUFAs and
n-6/n-3 FA ratios on physiological parameters, including molecular
mechanisms, without the necessity of providing exogenous n-3 fatty
acids. Despite promising accumulating evidence on the potential
benefits of n-3 PUFAs in psoriasis, the underlying mechanisms of
this effect remain elusive. In the present study, we used this
fat-1 transgenic psoriasis mouse model to establish n-3 PUFAs as a
therapeutic agent for psoriasis and to examine the molecular
mechanisms underlying this effect.
Materials and methods
Animals and treatments
Fat-1 transgenic mice and C57BL6 WT control mice
were obtained from Professor Yifan Dai (15) and bred in the Southern Medical
University’s laboratory animal facility (Guangdong, China). Male
fat-1 transgenic mice were mated with wild-type C57BL6 female mice
to obtain female fat-1 positive C57BL6 mice (fat-1) and fat-1
negative C57BL6 mice (WT) identified by genotyping using a
polymerase chain reaction (PCR) kit purchased from (Takara Bio,
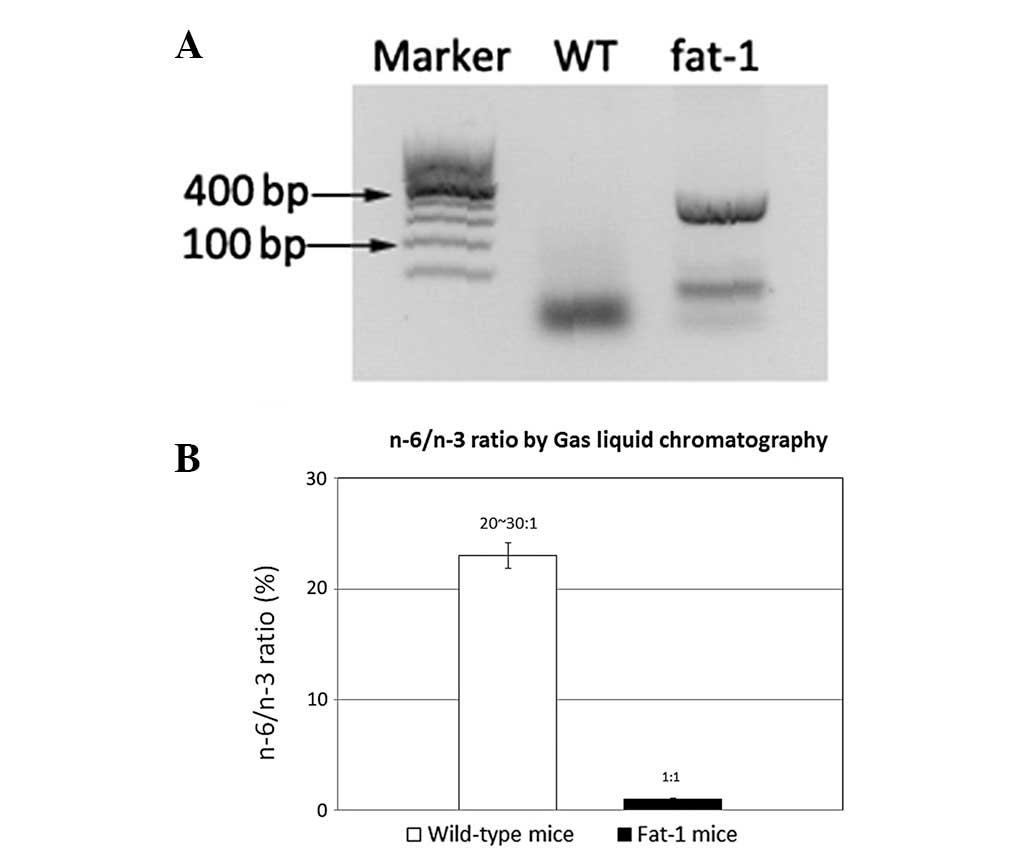
Inc.; Dalian, Liaoning, China; Fig.
1). The fatty acid composition of the mouse tails was measured
utilizing gas chromatography (GC; Fig.
1) (16). Weight-matched mice
were housed in a laboratory animal care facility in cages
(n=4/cage), in pathogen-free conditions and subject to a 12 h
light/dark cycle at 24°C and provided with food and water ad
libitum. At the age of 8 weeks, 48 mice (24 WT mice and 24
fat-1 mice) received a daily topical dose of 62.5 mg of
commercially available imiquimod (IMQ) cream (5%; Aldara; 3M
Pharmaceuticals, St. Paul, MN, USA) or control cream (Vaseline
cream; Unilever, Greenwich, CT, USA) on the shaved right ear and
dorsal skin for 14 consecutive days. This dose (translating in a
daily dose of 3.125 mg of active ingredients) was empirically
ascertained to induce optimal and reproducible skin inflammation in
the mice (17). They were divided
into the following four groups (n=12/group): Group A, WT cream;
Group B, WT IMQ; Group C, fat-1 cream and Group D, fat-1 IMQ.
A total of 48 mice were separately fed with a
general diet for two weeks prior to being sacrificed. The present
study was approved by the Animal Research Ethics Committee of
Southern Medical University (Guangdong, China) and the principles
of the National Institutes of Health Guide were strictly followed
in all experimental procedures.
Scoring severity of skin
inflammation
The clinical PASI is a useful tool as an objective
scoring system for the severity of inflammation of the dorsal and
ear skin in mice. The fixed area with IMQ treatment was accounted
for in the overall score, excluding the affected skin area.
Erythema, scaling and thickening were calculated respectively on a
scale from 0–4: 0, none; 1, slight; 2, moderate; 3, marked and 4,
highly marked. The cumulative scores (the amount of erythema,
scaling and thickening) acted as a measure of the severity of
inflammation. Repeated measurements of the thickness of the right
ear with a micrometer (Mahr) were conducted for comparison with the
left ear.
Enzyme-linked immunosorbent assay
Blood serum was obtained on the day of sacrifice by
means of retroorbital bleeding from anesthetized (isoflurane) mice.
Serum was obtained by centrifugation at 300 × g for 15 min at 4°C,
then evaluated for the levels of inflammatory factors. Serum IL-17,
IL-22 and IL-23 were measured using the Quantikine Mouse IL-17A/F
kit (M17AF0; R&D Systems, Collegeville, PA, USA), Quantikine
Mouse IL-22 kit (M2200; R&D Systems) and Quantikine Mouse IL-23
kit (M2300; R&D Systems).
Measurement of body weight and spleen
weight
At the beginning of the treatment, the WT and fat-1
mice were weight matched using a CS 200 balance (Ohaus, Pine Brook,
NJ, USA). The spleens were carefully obtained and weighed at the
time of sacrifice using a Mettler balance (Mettler Toledo,
Columbus, OH, USA).
Flow cytometry
Spleen samples were ground with 200 m meshs and a
syringe piston to obtain single-cell suspensions. Cells were
stimulated with 50 ng/ml of PMA (Sigma, St. Louis, MO, USA) and 1
μg/ml of ionomycin (Sigma) in the presence of Monensin (BD
Golgistop™ protein transport inhibitor), in complete RPMI-1640. The
cells were then diluted to 10 million cells/ml, and centrifuged at
300 × g for 5 min and the buffer was removed. The cell pellet was
carefully suspended in the residual volume of staining buffer and
then 200 μl of freshly prepared cold 1X BD Pharmingen™ Mouse Foxp3
fixation buffer was added. To fix the cells, they were then
incubated for 30 min at 4°C in the dark. Then, the sample was
centrifuged at 300 × g for 5 min and the fixative was removed. To
permeabilize the cells, careful suspension of the cell pellet in
another 200 μl of freshly prepared pre-warmed (37°C) 1X BD
Pharmingen™ Mouse Foxp3 permeabilization buffer was repeated and
the cells were then incubated for 30 min at 37°C in the dark. Next,
the cells were centrifuged at 300 × g for 5 min and the buffer was
removed. To wash the cells, 200 μl of BD Pharmingen™ stain buffer
(FBS) was added to each tube, centrifuged at 300 × g for 5 min and
then the buffer was removed. A total of 20 μl/test of the mouse
Th17/Treg phenotyping cocktail or the appropriate negative staining
control was added and the cells were incubated at room temperature
(RT) for 30 minutes in the dark. Cells were protected from light
throughout the staining and storage, and the washing of the cells
was repeated twice. The cell pellet was suspended in 200 μl stain
buffer and was proceeded by flow cytometry (BD) and analysis with
CellQuest software (BD). The viability of the cells was examined by
staining with propidium iodide.
Histology and immunohistochemistry
The dorsal and ear tissues were formalin-fixed and
paraffin-embedded, and stained with H&E. Epidermal thickness
was accurately measured by ImagePro Plus software (Leeds Precision
Instruments, Minneapolis, MN, USA). The total epidermal area was
calculated using a series of rectangles and the data was divided by
the total length of the epidermis.
For immunohistochemistry, sections from the ear and
dorsal tissues were deparaffinized with xylene and rehydrated, and
then hydrated with a graded alcohol series. The ear and dorsal
sections were incubated in 10 ml citric acid (pH 6.0) at 95°C for
30 min to unmask antigens and the endogenous peroxidase activity
was quenched by treating sections with 3% hydrogen peroxide at RT
for 5 min. The sections were blocked at RT for 60 min followed by
incubation with primary antibodies (Abs): rabbit anti-mouse IL-17A
polyclonal Ab (H-132; Santa Cruz Biotechnology, Inc., Santa Cruz,
CA, USA), rabbit anti-mouse IL-22 polyclonal Ab (ab18564; Abcam,
Cambridge, MA, USA) and rabbit anti-mouse IL-23 polyclonal Ab
(H-113; Santa Cruz Biotechnology, Inc.). This was followed by
treatment with horseradish peroxidase-linked secondary anti-rabbit
GT Vision™ II polymer (Dako, Carpinteria, CA, USA) and DAB
substrate kit for peroxidase (Dako).
Statistical analysis
Results are expressed as the mean ± SEM and data
analysis was performed with the SPSS 13.0 software (SPSS, Inc.,
Chicago, IL, USA) and ANOVA. P<0.05 was considered to indicate a
statistically significant difference.
Results
Effect of endogenous n-3 desaturase on
n-6/n-3 ratios in mice
From the PCR analysis (Fig. 1A), it was possible to screen the
fat-1 gene positive C57BL6 mice and fat-1 gene negative C57BL6
mice. Gas liquid chromatography measured the quantity of n-3 PUFAs
and n-6 FAs of the mice tails. The n-3 PUFAs and n-6 PUFA ratios in
WT mice was ~20–30:1, whereas in the fat-1 mouse this ratio was 1:1
(Fig. 1B). The amount of n-3 PUFAs
was enhanced, in contrast with the levels of n-6 FA, which were
decreased, which is a result that may be attributed to the presence
of n-3 desaturase in the transgenic mice model. A total of 48 mice
were grouped utilizing this method in order to obtain accurate
results.
Effect of endogenous n-3 PUFAs on the
structural features of IMQ-induced skin inflammation in mice
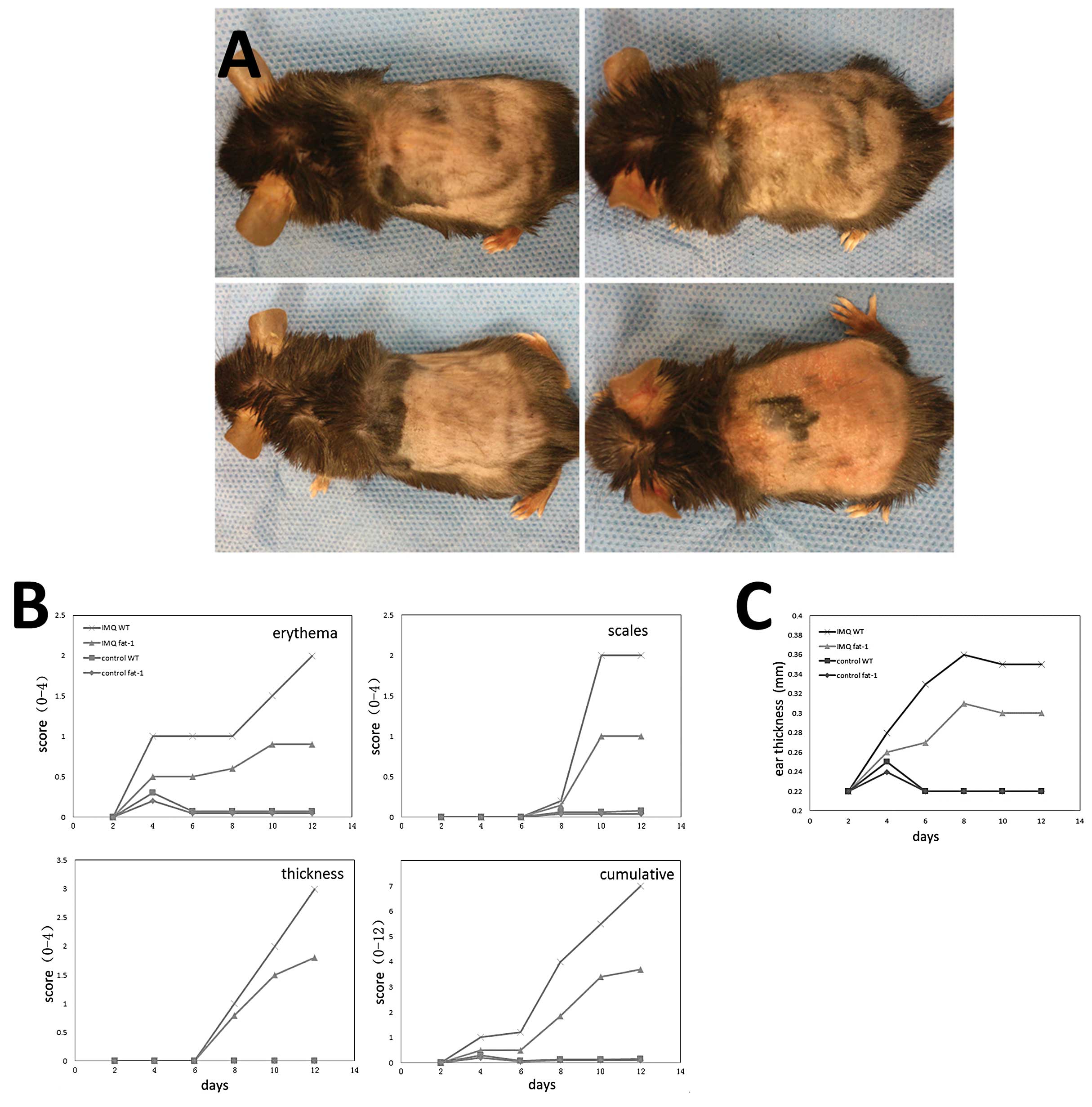
In the present study, IMQ cream and Vaseline cream
were applied on the shaved right ear and dorsal skin of WT mice and
fat-1 mice for 14 consecutive days. Three days following the
application of IMQ onto the ears and dorsal skin of the mice, these
areas began to exhibit symptoms of erythema, scales and thickening.
These signs of inflammation, as observed in groups B and D mice
phenotypically resemble psoriasis. Mice in group D were notably
milder than in group B, who resembled the mice from groups A and C,
where their ears and dorsal skin remained smooth (Fig. 2A). From the images revealed in
Fig. 2B, it is possible to detect
and quantify the severity scores of the mice. Signs of inflammation
in groups B and D continually increased in severity until the end
of the study. Mice in group B had higher scores than group D, while
the mice in the control groups (A and C) treated daily with
Vaseline cream did not present with any signs of inflammation.
Additionally, compared with group A and C, the thickness of the
right ear of mice who received daily IMQ-treatment increased from
days 5–6 onward and were recorded, which in group B was more
notable (Fig. 2C).
As van der Fits et al (17) described in 2009, IMQ-induced
cutitis in mice resembled psoriatic lesions in human patients, not
only in respect of the phenotypic symptoms, but also the
histological characteristics. Furthermore, the development of the
lesion was closely associated with the levels of IL-23 and IL-17
(18). In the present study,
utilizing this model produced consistent results, because the
inflammation of mice in group D was milder than in group B. The
only difference between the two types of mice was the n-6/n-3 PUFA
ratios, which resulted in differential responses to the daily
IMQ-treatment. The data revealed endogenous n-3 PUFAs may protect
against psoriasis-like lesions by means of its anti-inflammatory
action.
Effect of endogenous n-3 PUFAs on
increased inflammatory cytokines in serum
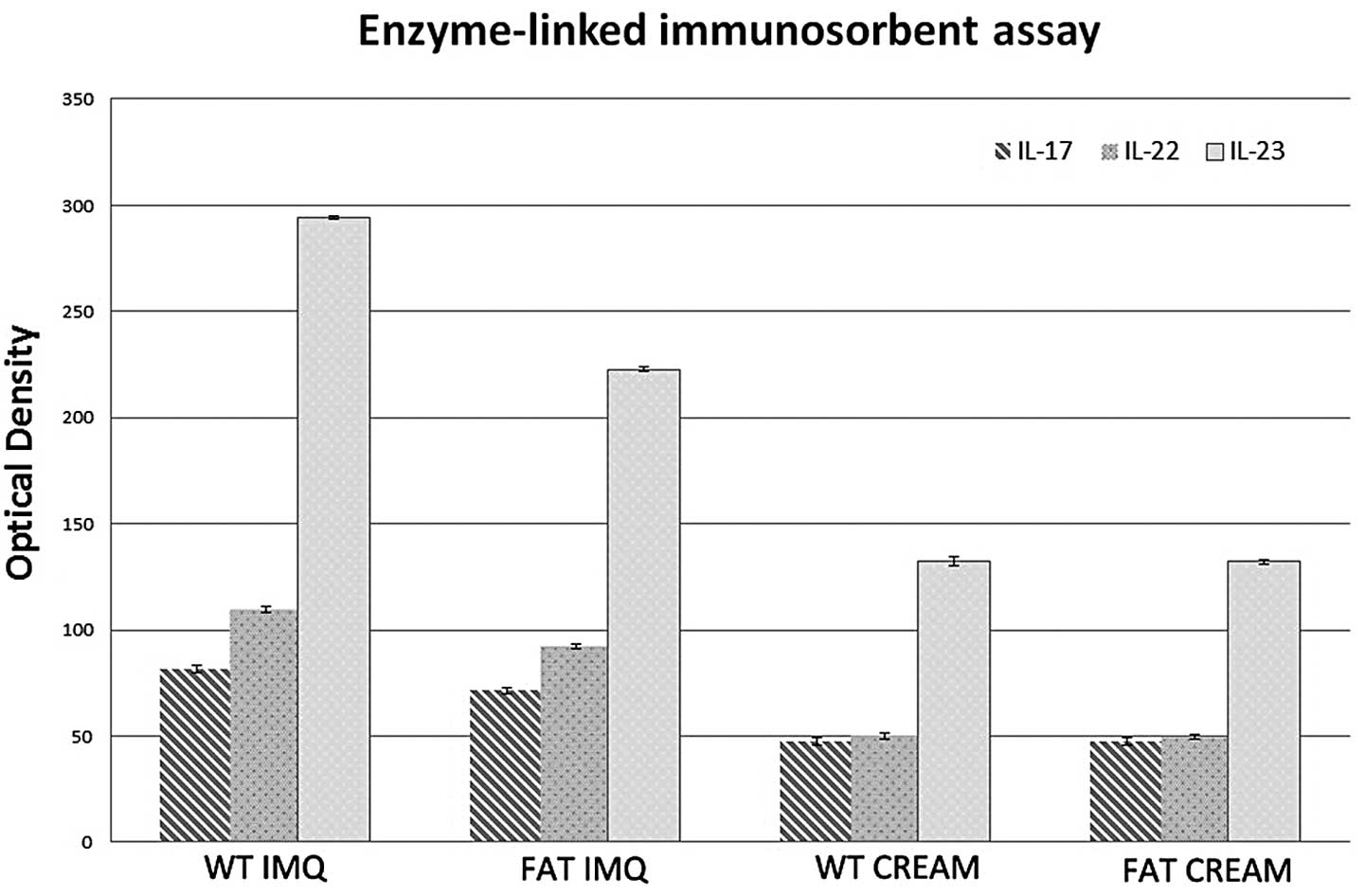
The levels of IL-17, IL-22 and IL-23 in IMQ-treated
groups were significantly higher (P<0.05) in the serum than in
the control groups (Fig. 3). The
levels of the inflammatory factor associated with Th17 cell in the
serum of fat-1 IMQ-treated mice were significantly lower
(P<0.05) compared with that in WT IMQ-treated mice. There was no
significant difference (P<0.05) between WT and fat-1 control
mice in the serum inflammatory factor levels. IL-17, IL-22 and
IL-23 secreted by Th17 cells in the serum of mice in group A and C
remained at low levels prior to their mortality. It is possible
that endogenous n-3 PUFAs prevent Th17 cells from producing
inflammatory factors, such as IL-17. By contrast, endogenous n-3
PUFAs may perform a particular role in decreasing the
differentiation of CD4+ cells into Th17 cells.
Effect of endogenous n-3 PUFAs on spleen
weight in an IMQ-induced psoriasis model
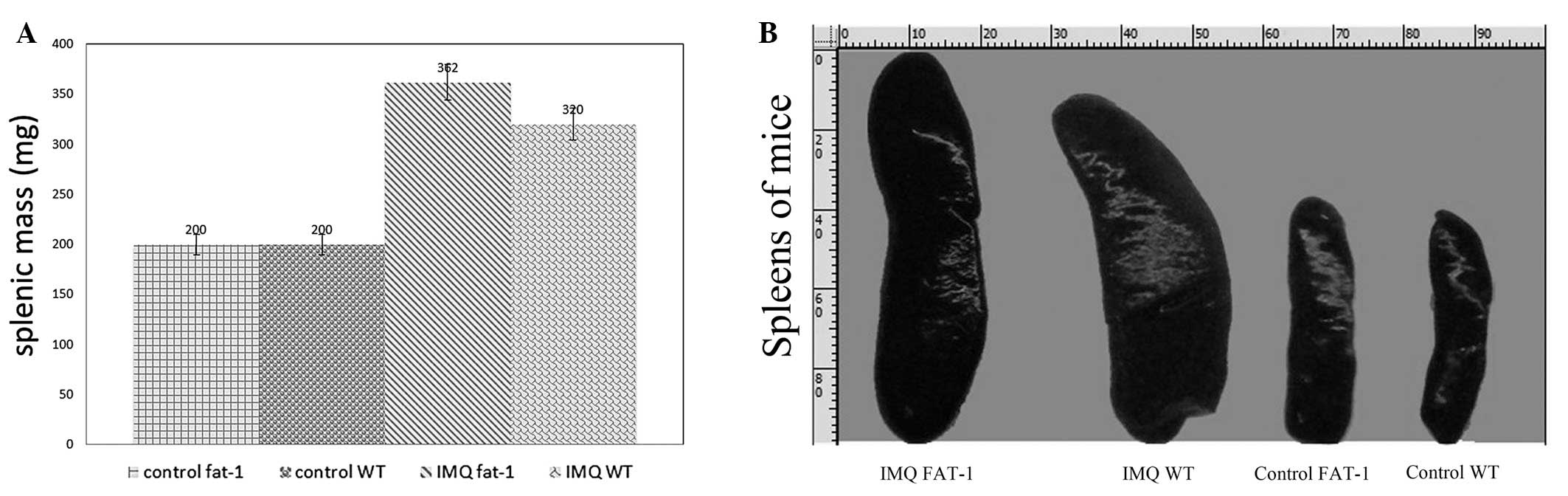
The mice in the four groups were subject to topical
IMQ treatment and any significant changes either in the size or the
weight of spleens were observed. At first, from direct-viewing of
the images, it is possible to note that the lengths of spleens of
mice in group B and D were all ≥22 mm, while the spleens in group A
and C measured ≤16 mm, which was approximately two thirds of the
IMQ-treatment mice (Fig. 4A).
There was a significant difference in the value for spleen weight
between the fat-1 and WT IMQ-treatment mice (P<0.05) following
14 days of treatment, however, there is no significant difference
in the spleen weight between fat-1 and WT cream-treatment mice
(P>0.05). IMQ-treatment groups induced an increase in the spleen
weight compared with the control groups (Fig. 4B).
The spleen is the largest immune organ in the human
body, owing to various immunocompetent cytokines, which have an
important role in anti-infection and anti-tumor activities. The
increased weight and size observed in the IMQ-treated mice,
suggests that the amount of cells in spleen had increased to a high
level, which may be a sign of enhancing the immunoreaction.
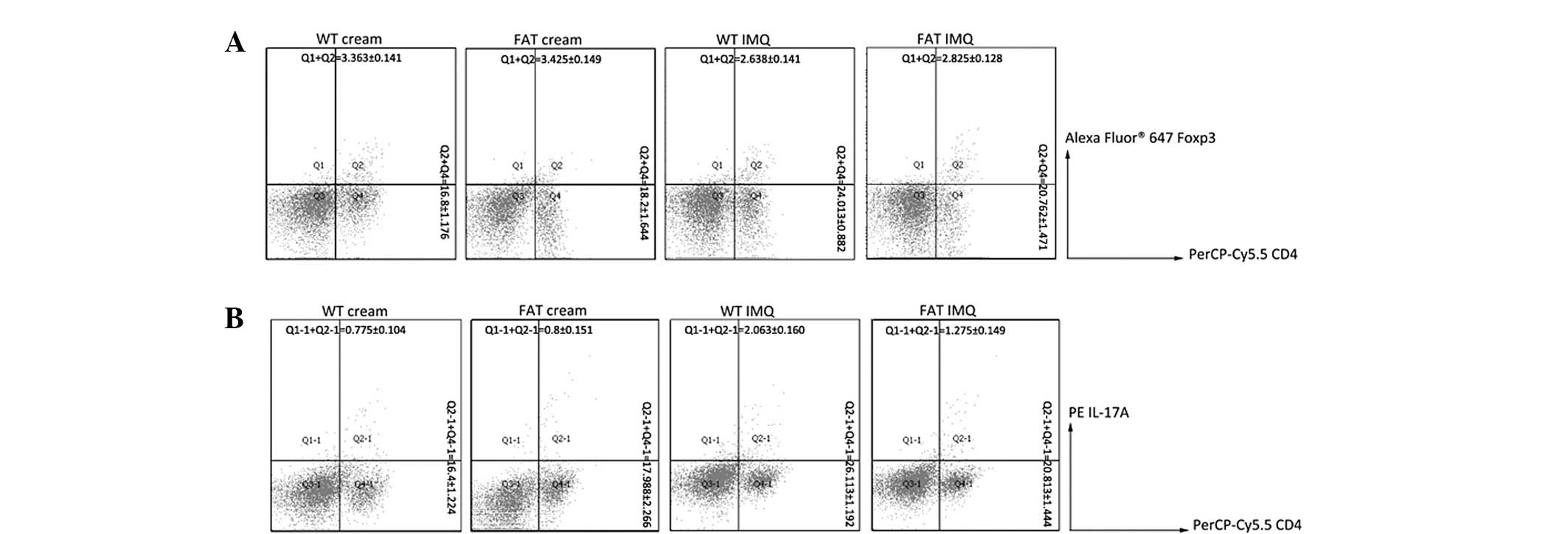
Effect of endogenous n-3 PUFAs on
IMQ-induced splenomegaly with increased numbers of Th17 cells
Following 14 days of IMQ and Vaseline treatment, we
identified a consistently significant spleen enlargement in WT mice
and fat-1 mice (Fig. 4A). To
determine the percentages of CD4+T, Treg and Th17
cytokine positive cells in the spleen, splenic cells were activated
ex vivo by phorbol myristate acetate (PMA; protein kinase C
activator) plus ionomycin (Ca2+ ionophore), stained
intracellularly for ‘Mouse Th17/Treg Phenotyping Cocktail’
containing ‘Mouse CD4 PerCP-Cy5.5’, ‘Mouse IL-17A PE’ and ‘Foxp3
Alexa Fluor® 647’ and analyzed using flow cytometry.
The percentage of CD4+T cells, IL-17A and
Foxp3 of the spleens of the four groups of mice was detected by
flow cytometry. IL-17A and Foxp3 were secreted by Th17 cells and
Treg cells, respectively, and therefore represent the quantity of
Th17 cells and Treg cells. An increase in the percentage of IL-17A
in IMQ-treated groups was observed through the amount of
IL-17A+ following IMQ treatment (Fig. 5), whereas the percentage of Foxp3
was decreased. Additionally, the percentage of Th17 cells in fat-1
IMQ group were significantly lower (P<0.05) in spleens than in
the WT IMQ group and the percentage of Treg cells in the spleens of
fat-1 IMQ mice was significantly higher (P<0.05) compared with
that in WT IMQ mice. However, there was no significant difference
(P>0.05) between WT and fat-1 control mice in the cellular
composition of the spleen. These results imply that endogenous n-3
PUFAs could upregulate the Foxp3 levels and reduce IL-17A to
inhibit the inflammatory response.
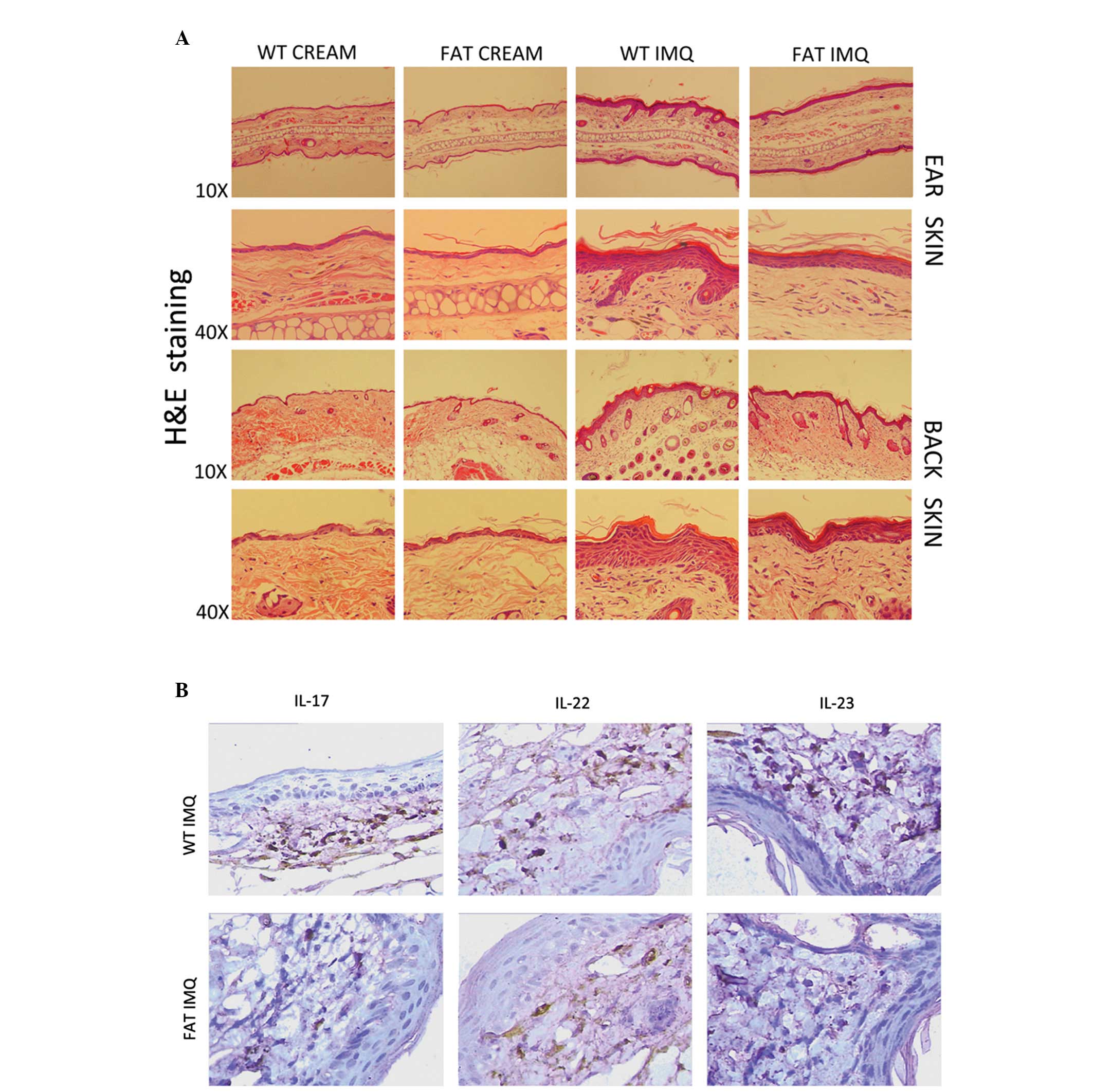
Effect of endogenous n-3 PUFAs on
IMQ-induced increased proliferation and altered differentiation of
keratinocytes
It has been previously revealed that IMQ treatment
induces increased epidermal thickening, hyperproliferous
keratinocytes, parakeratosis and altered differentiated epidermis
symptomatic of psoriatic skin lesions (19). In the present study, through the
analysis of H&E-stained sections from the IMQ-treated dorsal
tissue and ear skin, we observed increasing epidermal thickeness,
stratum corneum, prickle cell layer in the IMQ-treated group
compared with the control group (P<0.05) for the WT and fat-1
mice. However, the indication of inflammation of fat-1 mice (group
D) was evidently weaker than in the WT mice (group B) in each
respect (Fig. 6A).
Stained-inflammatory factors secreted by Th17 cells from
histological analyses confirmed multiplicity of IL-17, IL-22 and
IL-23 accumulation in the WT IMQ-treated group vs. fat-1
IMQ-treated group (P<0.05; Fig.
6B). There was no evident change in groups A or C.
Discussion
The IMQ-induced mouse is utilized as a model of
human psoriatic lesions, as it exhibits similar characteristics,
including erythema, epidermal thickening, scaling, neoangiogenesis,
and the inflammatory infiltrate of T cells, neutrophils and
dendritic cells (DCs). A previous study revealed that IMQ-induced
skin inflammation markedly and consistently reflected the
characteristics of psoriasis, including activated T cells,
epidermal alterations by keratinocyte hyperproliferation and
differentiation, existence of inflammatory cells consisting of T
cells, neutrophils, DCs and vascular proliferation (17).
A study by van der Fits et al provided new
insights into how Th1-Th17 challenge and IL-17 receptor signaling
are critical to the development of psoriasis, since genetic
knockout of these molecules leads to nearly a complete blockade of
disease. Another study revealed that blockade of
phosphatidylinositol 3-kinase (PI3K)δ or PI3Kγ ameliorated
IMQ-induced psoriasis-like dermatitis, correlating with reduced
IL-17 levels in the spleen serum and lesions (20). To further investigate the effect of
IL-17 signaling in psoriasis, El Malki et al generated IL-17
receptorA deficient IL-17RA (del) mice and treated these mice with
IMQ (4). The authors identified
that psoriatic skin was partly reduced and delayed when compared
with the controls. Of note, in the naive state, the skin of IL-17RA
(del) mice contained markedly elevated numbers of Th17- and
IL-17-producing γδ T cells. It is assumed that IL-17RA signaling
regulates the population size of Th17 and γδ T cells. Therefore,
the IL-23/IL-17 axis is critical in psoriasis-like lesions, which
are triggered by the interaction between immune mediators from
innate mechanisms and adaptive immunity. Therefore, in current
studies, this classical IMQ-induced psoriasis model of mice is
selected to examine the beneficial effects of n-3 PUFAs in
psoriasis.
It has been reported that populations which consume
a diet high in fish and other marine-based products, have a lower
risk of heart disease. As a result, n-3 fatty acids as nutritional
supplements have attracted notable attention and numerous studies
have since focused on deciphering the beneficial effects of n-3
fatty acids in a number of different disease conditions (18,21).
The relative ratio between n-6 and n-3 FAs is important in the
overall health benefits of consuming n-3 FAs. The n-6 FAs,
particularly arachidonic acid (AA), is a precursor of leukotrienes,
prostaglandins (PGs) and other related compounds, affecting the
synthesis of eicosanoids, which may enhance inflammation (19). In our previous study, it was
identified that elongation and desaturation were inhibited by the
presence of n-3 FA because the reduced levels of AA in cell
membranes in fat-1 mice were partially replaced by EPA and DHA due
to consuming increased amounts of n-3 PUFAs (19,22).
This results in decreased production of pro-inflammatory mediators
by AA, including PGE2. Therefore, n-3 PUFAs may restrain the
hyperkeratosis and parakeratosis in psoriasis via reducing the
levels of circulating inflammatory mediators. We also previously
demonstrated that dietary n-3 PUFAs fed mice exhibit less
IMQ-induced Th17 cell changes accompanied by psoriasis-like lesions
(20), however, the affect of
endogenous n-3 PUFAs on the psoriatic lesions remains poorly
understood. To improve our understanding of the effect of
endogenous n-3 PUFAs, a transgenic fat-1 gene overexpression mice
model was used, which exhibit a characteristic reduction in the n-6
to n-3 ratio of 1:1, as compared with WT mice littermates with a
ratio of 20–30:1, which may be optimal for health. Therefore, for
the first time, to the best of our knowledge, we introduce this
model to investigate the pathogenesis underlying psoriasis, with
its advantages of steady endogenous n-3 PUFA ratio.
In the present study, the effect of endogenous n-3
PUFAs on psoriasis-like lesions was investigated in a fat-1
transgenic IMQ-induced model. The accuracy of grouping was
guaranteed with PCR genotyping of the mice. The right ear and
dorsal skin of the four groups of mice were separately treated with
equal IMQ and cream for 14 consecutive days. As it may have been
expected, the skin coated with IMQ exhibited thickening, erythema
and scales, in the fat-1 transgenic mice, however, these
inflammatory effects were more mild than those in WT mice. We noted
that IMQ treatment resulted in hyperproliferative keratinocytes,
parakeratosis, incrassate stratum corneum and stratum spinosum, all
of which correspond with the characteristic histological features
of psoriasis. The symptoms of the condition in fat-1 mice was
notably weaker compared with the WT mice. Following this, we
determined the anti-inflammatory effect of endogenous n-3 PUFAs and
lower n-6/n-3 FA level for psoriasis-like lesions in fat-1
mice.
As the largest immune organ in body, the spleen
participates in the process of systemic immune adjustment. In
measuring the spleens, it was identified that those in the
IMQ-treated mice were approximately twice the size and weight of
those in the control groups, which provided evidence that
inflammation increases the size, weight and cell number of the
spleen. The evident changes observed in the spleens of fat-1
IMQ-treated mice may be due to endogenous n-3 PUFAs stimulating a
more aggressive and active immune response. With a balanced role in
the majority of the inflammatory reaction, Treg cells are usually
presented by anti-inflammatory factor Foxp3. Under the condition of
IMQ-treatment, the amount of Treg cells reduced, but the Th17 cells
exhibited an opposing response. Fat-1 and wild-type mice had
similar T cell and cytokine levels in vivo, as well as the
presence of keratinocytes in the skin. We observed that the mildly
decreased levels of Foxp3 secreted by regulatory T cells in the
spleen of fat-1 IMQ-treated mice was accompanied by a lower level
IL-17A from Th17 cells than that of the WT IMQ-treated group. It is
believed that higher levels of n-3 PUFAs and lower n-6/n-3 ratios
could maintain regulatory T cells and reduce the increasing Th17
cells in the spread of inflammation when compared with that of WT
mice. Th17 cells are an important source of inflammatory factors,
including IL-17, IL-22 and IL-23, that may all have an effect on
psoriasis, which leads to hyperkeratosis and parakeratosis. Th17
cells not only specifically secrete IL-17, IL-22 and IL-23 but also
are involved in promoting the differentiation from CD4+T
cells. With regard to the affect of fatty acid on psoriasis, the
levels of inflammatory factors in serum should always be
considered. In the blood serum, IL-17, IL-22 and IL-23 levels were
enhanced in the IMQ-treated mice, as compared with the control
groups. In the fat-1 IMQ-induced group, the factors remained at
lower levels compared with those of the WT IMQ-induced mice. It may
be due to the impact of endogenous n-3 PUFAs on regulatory T cells
and Th17 cells. In the present study, a significantly lower
expression of IL-17, IL-22 and IL-23 was observed in the dorsal and
ear skin of IMQ-treated fat-1 mice when compared with the WT mice,
which confirmed the inhibitory impact of endogenous n-3 PUFAs on
epidermis inflammation theoretically.
The results from the present study have revealed
that endogenous lowering of the n-6/n-3 ratio and higher n-3 PUFA
levels not only suppresses Th17 cells and maintains the level of
anti-inflammatory cytokines Foxp3 from Treg cells, but also
inhibits the expression of pro-inflammatory or inflammatory
cytokines, including IL-17, IL-22 and IL-23 in the serum,
preventing their accumulation in the lesion, and subsequently
reducing thickening, erythema and scales. These data indicate the
potential beneficial effects of endogenous n-3 PUFAs on IMQ-induced
psoriasis. Currently the anti-psoriasis drugs, such as infliximab,
have been widely used in prevention and treatment of TNF-α targeted
prevention and treatment of psoriasis. One study identified that
dietary n-3 PUFAs induced moderate clinical improvement and
inhibited the inflammation in psoriasis (23). In the present study, the fat-1
transgenic mouse was selected to expound the molecular mechanisms
underlying n-3 PUFA effects in psoriasis, due to its advantages
over other models, in eliminating confounding factors with regard
to exogenous diets. This study on fat-1 transgenic mice provided
compelling evidence that the IL-17/IL-23 axis is a critical
therapeutic target of inflammation in psoriasis and endogenous n-3
PUFAs are potential candidates for the prevention of hyperkeratosis
and parakeratosis.
Recently, fish oils rich in n-3 FAs have been
approved by the FDA as a prescription drug to treat cardiovascular
diseases and high triglyceride levels owing to its cardioprotective
effect (24), anti-carcinogenic
effect (25), triglyceride
lowering effect (26) and
protective effect against inflammatory diseases (16,27),
as a supplementation. Future studies should investigate the effect
of endogenous n-3 PUFAs on a genetic level.
Acknowledgements
The authors are grateful to Mr. Sheng-Fa Li and Mr.
Yuan-Jian Huang for their technical assistance and to Professor
Yi-Fan Dai for providing the fat-1 transgenic mice. This study was
supported by Guangdong No. 2 Provincial People’s Hospital.
Abbreviations:
|
ALA
|
alpha-linolenic acid
|
|
DCs
|
dendritic cells
|
|
DHA
|
docosahexaenoic acid
|
|
EPA
|
eicosapentaenoic acid
|
|
FDA
|
Food and Drug Administration
|
|
GC
|
gas chromatography
|
|
IMQ
|
imiquimod
|
|
IL
|
interleukin
|
|
n-3 FAs
|
n-3 fatty acids
|
|
PASI
|
Psoriasis Area and Severity Index
|
|
WT
|
wild-type
|
References
|
1
|
Yoo IS, Lee JH, Song ST, Kim JH, Lee HJ
and Kang SW: T-helper 17 cells: the driving force of psoriasis and
psoriatic arthritis. Int J Rheum Dis. 15:531–537. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
2
|
Tokura Y: Th17 cells and skin diseases.
Nihon Rinsho Meneki Gakkai Kaishi. 35:388–392. 2012.(In
Japanese).
|
|
3
|
Mudigonda P, Mudigonda T, Feneran AN,
Alamdari HS, Sandoval L and Feldman SR: Interleukin-23 and
interleukin-17: importance in pathogenesis and therapy of
psoriasis. Dermatol Online J. 18:12012.PubMed/NCBI
|
|
4
|
El Malki K, Karbach SH, Huppert J, et al:
An alternative pathway of imiquimod-induced psoriasis-like skin
inflammation in the absence of interleukin-17 receptor a signaling.
J Invest Dermatol. 133:441–451. 2013.PubMed/NCBI
|
|
5
|
Wang WJ, Yin XY, Zuo XB, et al: Gene-gene
interactions in IL23/Th17 pathway contribute to psoriasis
susceptibility in Chinese Han population. J Eur Acad Dermatol
Venereol. 27:1156–1162. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
6
|
Zhang L, Yang XQ, Cheng J, Hui RS and Gao
TW: Increased Th17 cells are accompanied by FoxP3(+) Treg cell
accumulation and correlated with psoriasis disease severity. Clin
Immunol. 135:108–117. 2010.
|
|
7
|
Res PC, Piskin G, de Boer OJ, et al:
Overrepresentation of IL-17A and IL-22 producing CD8 T cells in
lesional skin suggests their involvement in the pathogenesis of
psoriasis. PLoS One. 5:e141082010. View Article : Google Scholar : PubMed/NCBI
|
|
8
|
Kagami S, Rizzo HL, Lee JJ, Koguchi Y and
Blauvelt A: Circulating Th17, Th22, and Th1 cells are increased in
psoriasis. J Invest Dermatol. 130:1373–1383. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
9
|
Kim EH, Bae JS, Hahm KB and Cha JY:
Endogenously synthesized n-3 polyunsaturated fatty acids in fat-1
mice ameliorate high-fat diet-induced non-alcoholic fatty liver
disease. Biochem Pharmacol. 84:1359–1365. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
10
|
Monk JM, Jia Q, Callaway E, et al: Th17
cell accumulation is decreased during chronic experimental colitis
by (n-3) PUFA in Fat-1 mice. J Nutr. 142:117–124. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
11
|
Bellenger J, Bellenger S, Bataille A, et
al: High pancreatic n-3 fatty acids prevent STZ-induced diabetes in
fat-1 mice: inflammatory pathway inhibition. Diabetes.
60:1090–1099. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
12
|
Mohammed A, Janakiram NB, Brewer M, et al:
Endogenous n-3 polyunsaturated fatty acids delay progression of
pancreatic ductal adenocarcinoma in
Fat-1-p48(Cre/+)-LSL-Kras(G12D/+) mice. Neoplasia. 14:1249–1259.
2012.PubMed/NCBI
|
|
13
|
McCusker MM and Grant-Kels JM: Healing
fats of the skin: the structural and immunologic roles of the
omega-6 and omega-3 fatty acids. Clin Dermatol. 28:440–451. 2010.
View Article : Google Scholar : PubMed/NCBI
|
|
14
|
Kang JX, Wang J, Wu L and Kang ZB:
Transgenic mice: fat-1 mice convert n-6 to n-3 fatty acids. Nature.
427:5042004. View
Article : Google Scholar : PubMed/NCBI
|
|
15
|
Wei D, Li J, Shen M, et al: Cellular
production of n-3 PUFAs and reduction of n-6-to-n-3 ratios in the
pancreatic beta-cells and islets enhance insulin secretion and
confer protection against cytokine-induced cell death. Diabetes.
59:471–478. 2010. View Article : Google Scholar
|
|
16
|
Bhattacharya A, Chandrasekar B, Rahman MM,
Banu J, Kang JX and Fernandes G: Inhibition of inflammatory
response in transgenic fat-1 mice on a calorie-restricted diet.
Biochem Biophys Res Commun. 349:925–930. 2006. View Article : Google Scholar : PubMed/NCBI
|
|
17
|
van der Fits L, Mourits S, Voerman JS, et
al: Imiquimod-induced psoriasis-like skin inflammation in mice is
mediated via the IL-23/IL-17 axis. J Immunol. 182:5836–5845.
2009.PubMed/NCBI
|
|
18
|
Ma DW, Ngo V, Huot PS and Kang JX: N-3
polyunsaturated fatty acids endogenously synthesized in fat-1 mice
are enriched in the mammary gland. Lipids. 41:35–39. 2006.
View Article : Google Scholar : PubMed/NCBI
|
|
19
|
Hudert CA, Weylandt KH, Lu Y, et al:
Transgenic mice rich in endogenous omega-3 fatty acids are
protected from colitis. Proc Natl Acad Sci USA. 103:11276–11281.
2006. View Article : Google Scholar : PubMed/NCBI
|
|
20
|
Roller A, Perino A, Dapavo P, et al:
Blockade of phosphatidylinositol 3-kinase PI3Kδ or PI3Kγ reduces
IL-17 and ameliorates imiquimod-induced psoriasis-like dermatitis.
J Immunol. 189:4612–4620. 2012.
|
|
21
|
Gago-Dominguez M, Yuan JM, Sun CL, Lee HP
and Yu MC: Opposing effects of dietary n-3 and n-6 fatty acids on
mammary carcinogenesis: The Singapore Chinese Health Study. Br J
Cancer. 89:1686–1692. 2003. View Article : Google Scholar : PubMed/NCBI
|
|
22
|
Kang JX: Fat-1 transgenic mice: a new
model for omega-3 research. Prostaglandins Leukot Essent Fatty
Acids. 77:263–267. 2007. View Article : Google Scholar : PubMed/NCBI
|
|
23
|
Blok WL, Katan MB and van der Meer JW:
Modulation of inflammation and cytokine production by dietary (n-3)
fatty acids. J Nutr. 126:1515–1533. 1996.PubMed/NCBI
|
|
24
|
Wang C, Harris WS, Chung M, et al: n-3
Fatty acids from fish or fish-oil supplements, but not
alpha-linolenic acid, benefit cardiovascular disease outcomes in
primary- and secondary-prevention studies: a systematic review. Am
J Clin Nutr. 84:5–17. 2006.PubMed/NCBI
|
|
25
|
Jia Q, Lupton JR, Smith R, et al: Reduced
colitis-associated colon cancer in Fat-1 (n-3 fatty acid
desaturase) transgenic mice. Cancer Res. 68:3985–3991. 2008.
View Article : Google Scholar : PubMed/NCBI
|
|
26
|
Qi K, Fan C, Jiang J, et al: Omega-3 fatty
acid containing diets decrease plasma triglyceride concentrations
in mice by reducing endogenous triglyceride synthesis and enhancing
the blood clearance of triglyceride-rich particles. Clin Nutr.
27:424–430. 2008. View Article : Google Scholar
|
|
27
|
Calder PC: Immunomodulation by omega-3
fatty acids. Prostaglandins Leukot Essent Fatty Acids. 77:327–335.
2007. View Article : Google Scholar : PubMed/NCBI
|