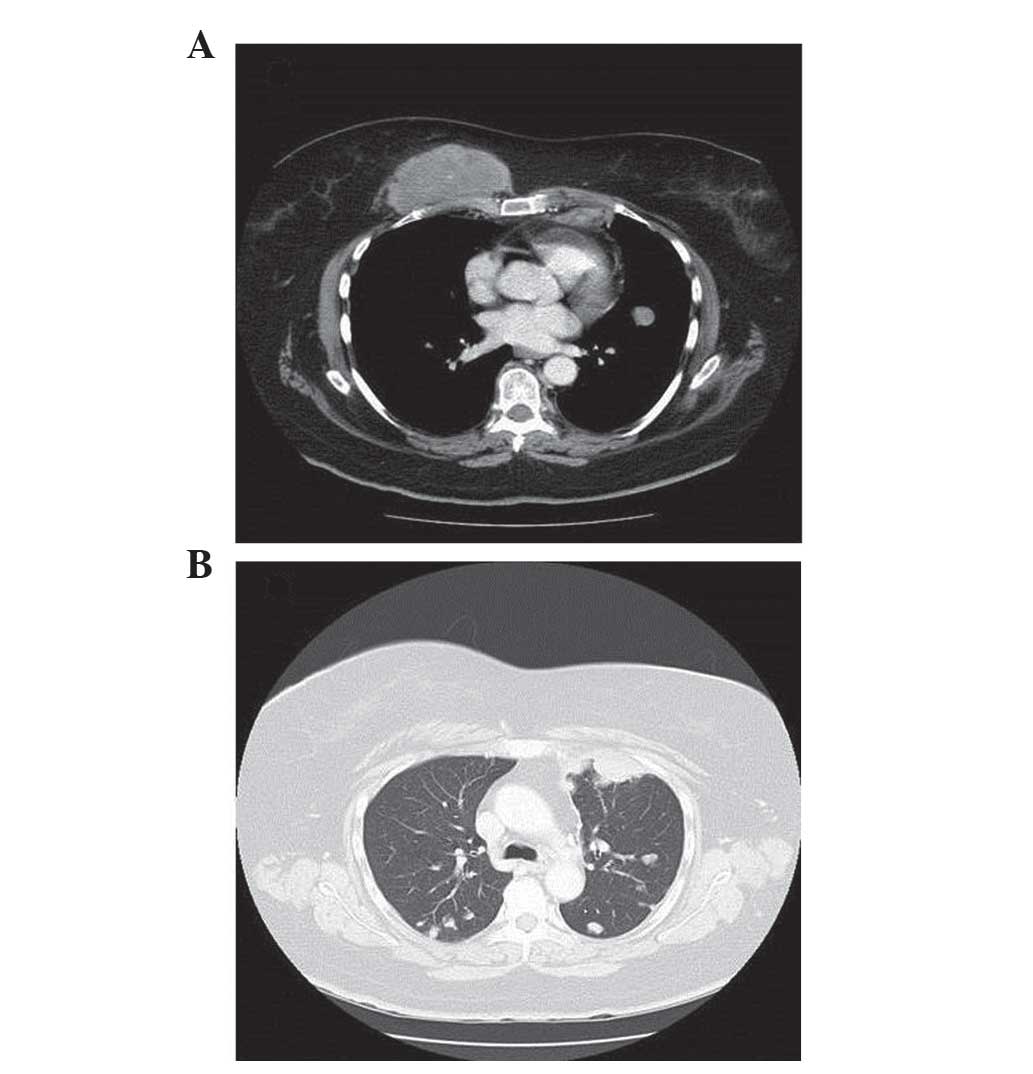
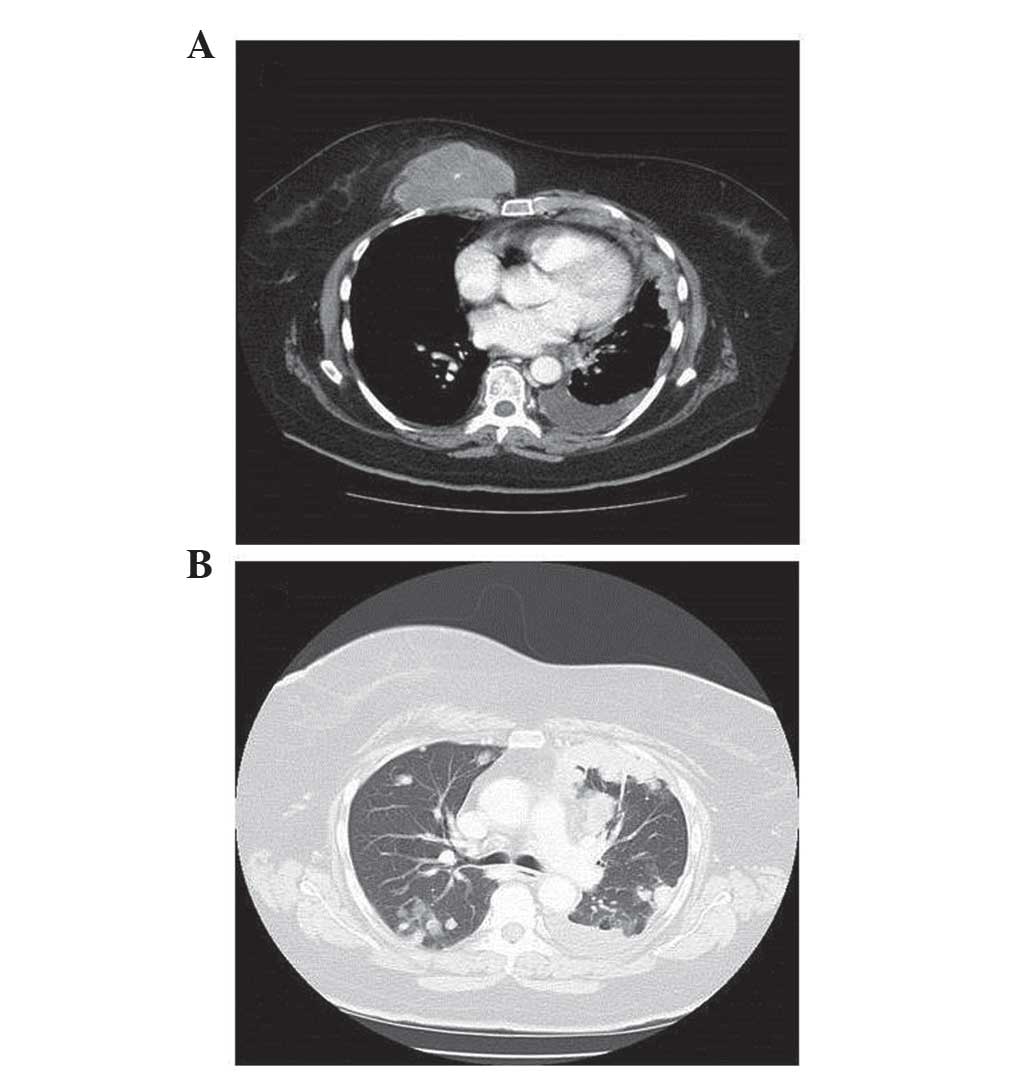
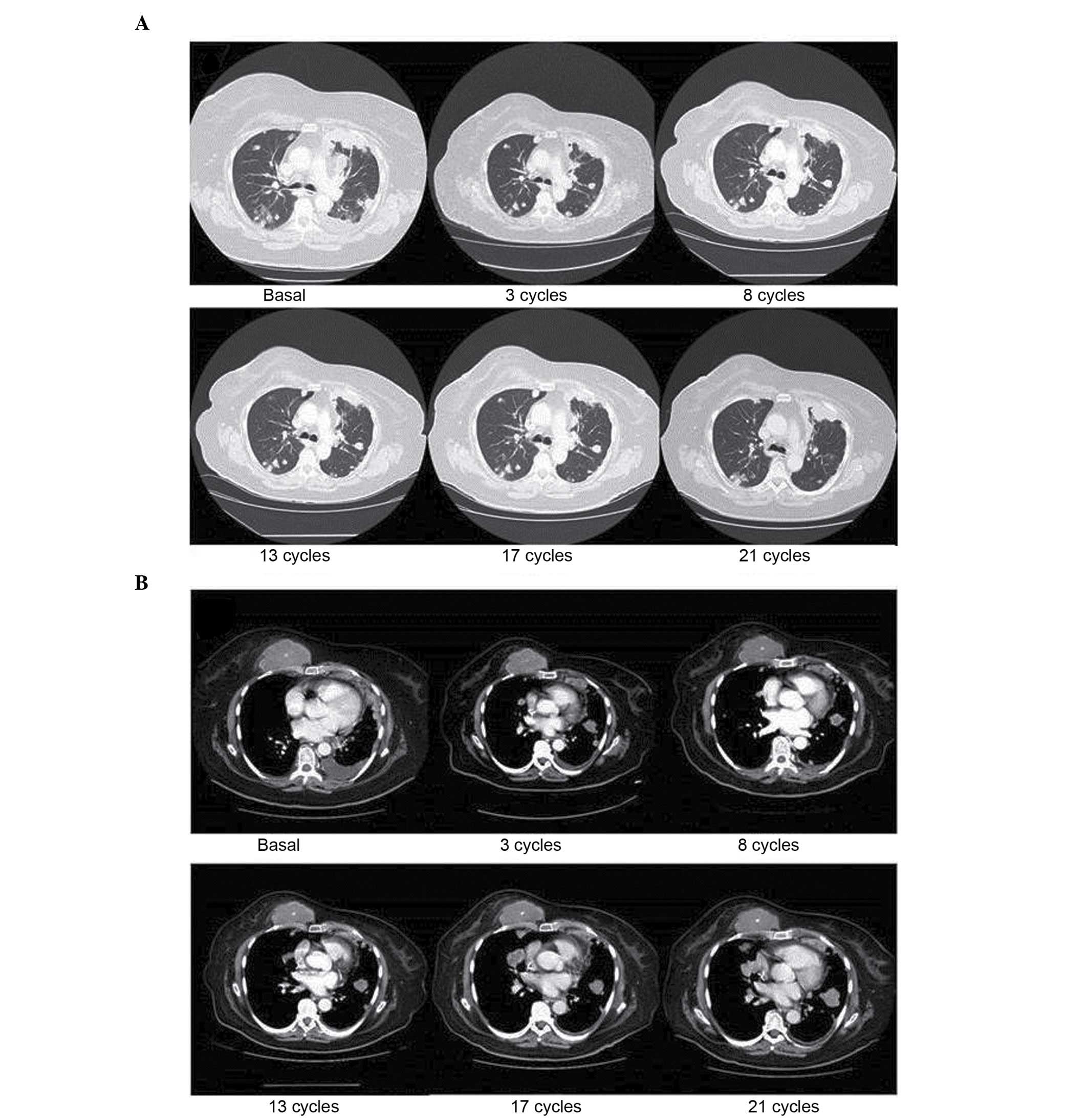
|
1
|
Delattre O, Zucman J, Melot T, Garau XS,
Zucker JM, Lenoir GM, Ambros PF, Sheer D, TurcCarel C, Triche TJ,
et al: The Ewing family of tumors-a subgroup of small-round-cell
tumors defined by specific chimeric transcripts. N Engl J Med.
331:294–299. 1994. View Article : Google Scholar : PubMed/NCBI
|
|
2
|
Ng TL, O'Sullivan MJ, Pallen CJ, Hayes M,
Clarkson PW, Winstanley M, Sorensen PH, Nielsen TO and Horsman DE:
Ewing sarcoma with novel translocation t(2;16) producing an
in-frame fusion of FUS and FEV. J Mol Diagn. 9:459–463. 2007.
View Article : Google Scholar : PubMed/NCBI
|
|
3
|
Urano F, Umezawa A, Yabe H, Hong W,
Yoshida K, Fujinaga K and Hata J: Molecular analysis of Ewing's
sarcoma: Another fusion gene, EWS-E1AF, available for diagnosis.
Jpn J Cancer Res. 89:703–711. 1998. View Article : Google Scholar : PubMed/NCBI
|
|
4
|
Grier HE, Krailo MD, Tarbell NJ, Link MP,
Fryer CJ, Pritchard DJ, Gebhardt MC, Dickman PS, Perlman EJ, Meyers
PA, et al: Addition of ifosfamide and etoposide to standard
chemotherapy for Ewing's sarcoma and primitive neuroectodermal
tumor of bone. N Engl J Med. 348:694–701. 2003. View Article : Google Scholar : PubMed/NCBI
|
|
5
|
Mora J, Cruz CO, Parareda A and de Torres
C: Treatment of relapsed/refractory pediatric sarcomas with
gemcitabine and docetaxel. J Pediatr Hematol Oncol. 31:723–729.
2009. View Article : Google Scholar : PubMed/NCBI
|
|
6
|
Rapkin L, Qayed M, Brill P, Martin M,
Clark D, George BA, Olson TA, WasilewskiMasker K, Alazraki A and
Katzenstein HM: Gemcitabine and docetaxel (GEMDOX) for the
treatment of relapsed and refractory pediatric sarcomas. Pediatr
Blood Cancer. 59:854–858. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
7
|
Fox E, Patel S, Wathen JK, Schuetze S,
Chawla S, Harmon D, Reinke D, Chugh R, Benjamin RS and Helman LJ:
Phase II study of sequential gemcitabine followed by docetaxel for
recurrent Ewing sarcoma, osteosarcoma, or unresectable or locally
recurrent chondrosarcoma: Results of Sarcoma Alliance for Research
Through Collaboration Study 003. Oncologist. 17:3212012. View Article : Google Scholar : PubMed/NCBI
|
|
8
|
García-del-Muro X, López-Pousa A, Maurel
J, Martín J, Martínez-Trufero J, Casado A, Gómez-España A, Fra J,
Cruz J, Poveda A, et al: Randomized phase II study comparing
gemcitabine plus dacarbazine versus dacarbazine alone in patients
with previously treated soft tissue sarcoma: A Spanish Group for
Research on Sarcomas study. J Clin Oncol. 29:2528–2533. 2011.
View Article : Google Scholar : PubMed/NCBI
|
|
9
|
D'Incalci M and Galmarini CM: A review of
trabectedin (ET-743): A unique mechanism of action. Mol Cancer
Ther. 9:2157–2163. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
10
|
GarciaCarbonero R, Supko JG, Manola J,
Seiden MV, Harmon D, Ryan DP, Quigley MT, Merriam P, Canniff J,
Goss G, et al: Phase II and pharmacokinetic study of ecteinascidin
743 in patients with progressive sarcomas of soft tissues
refractory to chemotherapy. J Clin Oncol. 22:1480–1490. 2004.
View Article : Google Scholar : PubMed/NCBI
|
|
11
|
LeCesne A, Blay JY, Judson I, van Oosterom
A, Verweij J, Radford J, Lorigan P, Rodenhuis S, Ray-Coquard I,
Bonvalot S, et al: Phase II study of ET-743 in advanced soft tissue
sarcomas: A European Organisation for the Research and Treatment of
Cancer (EORTC) soft tissue and bone sarcoma group trial. J Clin
Oncol. 23:576–584. 2005. View Article : Google Scholar : PubMed/NCBI
|
|
12
|
Yovine A, Riofrio M, Blay JY, Brain E,
Alexandre J, Kahatt C, Taamma A, Jimeno J, Martin C, Salhi Y, et
al: Phase II study of ecteinascidin-743 in advanced pretreated soft
tissue sarcoma patients. J Clin Oncol. 22:890–899. 2004. View Article : Google Scholar : PubMed/NCBI
|
|
13
|
Demetri GD, Chawla SP, von Mehren M, Ritch
P, Baker LH, Blay JY, Hande KR, Keohan ML, Samuels BL, Schuetze S,
et al: Efficacy and safety of trabectedin in patients with advanced
or metastatic liposarcoma or leiomyosarcoma after failure of prior
anthracyclines and ifosfamide: Results of a randomized phase II
study of two different schedules. J Clin Oncol. 27:4188–4196. 2009.
View Article : Google Scholar : PubMed/NCBI
|
|
14
|
LeCesne A, Cresta S, Maki RG, Blay JY,
Verweij J, Poveda A, Casali PG, Balaña C, Schöffski P, Grosso F, et
al: A retrospective analysis of antitumor activity with trabectedin
in translocation-related sarcomas. Eur J Cancer. 48:3036–3044.
2012. View Article : Google Scholar : PubMed/NCBI
|
|
15
|
Pink D, BertzLepel J, Busemann C, Bitz U
and Reichardt P: Efficacy of trabectedin in patients with advanced
or metastatic alveolar soft-part sarcoma. Onkologie. 35:249–252.
2012. View Article : Google Scholar : PubMed/NCBI
|
|
16
|
Grosso F, Dileo P, Sanfilippo R,
Stacchiotti S, Bertulli R, Piovesan C, Jimeno J, D'Incalci M,
Gescher A and Casali PG: Steroid premedication markedly reduces
liver and bone marrow toxicity of trabectedin in advanced sarcoma.
Eur J Cancer. 42:1484–1490. 2006. View Article : Google Scholar : PubMed/NCBI
|
|
17
|
Sanfilippo R, Dileo P, Blay JY,
Constantinidou A, Le Cesne A, Benson C, Vizzini L, Contu M, Daldi
GG, Dei Tos AP and Casali PG: Trabectedin in advanced synovial
sarcomas: A multicenter retrospective study from four European
institutions and the Italian Rare Cancer Network. Anticancer Drugs.
26:678–681. 2015.PubMed/NCBI
|
|
18
|
Takahashi S, Araki N, Sugiura H, Ueda T,
Takahashi M, Morioka H, Yonemoto T, Hiraga H, Hiruma T, Kunisada T,
et al: A randomized phase II study comparing trabectedin (T) and
best supportive care (BSC) in patients (pts) with
translocation-related sarcomas (TRS). J Clin Oncol (ASCO Annual
Meeting abstracts). 32:((Suppl; abstr 10524)). 5s2014.
|
|
19
|
Grohar PJ, Griffin LB, Yeung C, Chen QR,
Pommier Y, Khanna C, Khan J and Helman LJ: Ecteinascidin 743
interferes with the activity of EWS-FLI1 in Ewing sarcoma cells.
Neoplasia. 13:145–153. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
20
|
Lau L, Supko JG, Blaney S, Hershon L,
Seibel N, Krailo M, Qu W, Malkin D, Jimeno J, Bernstein M, et al: A
phase I and pharmacokinetic study of ecteinascidin-743 (Yondelis)
in children with refractory solid tumors. A Children's Oncology
Group study. Clin Cancer Res. 11:672–677. 2005.PubMed/NCBI
|
|
21
|
Chu Q, Mita A, Forouzesh B, Tolcher AW,
Schwartz G, Nieto A, SotoMatos A, Alfaro V, Lebedinsky C and
Rowinsky EK: Phase I and pharmacokinetic study of sequential
paclitaxel and trabectedin every 2 weeks in patients with advanced
solid tumors. Clin Cancer Res. 16:2656–2665. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
22
|
Dileo P, Grosso F, Casanova M, Jimeno J,
Marsoni S, Sanfilippo R, Podda M, Ferrari S, Bertulli R and Casali
P: Trabectedin (T) in metastatic Ewing's family tumors (EFT)
patients (pts) progressing after standard chemotherapy. Journal of
Clinical Oncology, 2007. In: ASCO Annual Meeting Proceedings
(Post-Meeting Edition). 25(18S): (June 20 Supplement). pp.
100402007
|
|
23
|
Baruchel S, Pappo A, Krailo M, Baker KS,
Wu B, Villaluna D, LeeScott M, Adamson PC and Blaney SM: A phase II
trial of trabectedin in children with recurrent rhabdomyosarcoma,
Ewing sarcoma and non-rhabdomyosarcoma soft tissue sarcomas: A
report from the Children's Oncology Group. Eur J Cancer.
48:579–585. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
24
|
Grohar PJ, Segars LE, Yeung C, Pommier Y,
D'Incalci M, Mendoza A and Helman LJ: Dual targeting of EWS-FLI1
activity and the associated DNA damage response with trabectedin
and SN38 synergistically inhibits Ewing sarcoma cell growth. Clin
Cancer Res. 20:1190–1203. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
25
|
Cesne AL, Judson I, Maki R, Grosso F,
Schuetze S, Mehren MV, Chawla SP, Demetri GD, Nieto A, Tanovic A
and Blay JY: Trabectedin is a feasible treatment for soft tissue
sarcoma patients regardless of patient age: A retrospective pooled
analysis of five phase II trials. Br J Cancer. 109:1717–1724. 2013.
View Article : Google Scholar : PubMed/NCBI
|