Introduction
The c-ros oncogene 1 (ROS1)-rearranged lung cancer
is rare and comprises only 1 to 2% of patients with non-small cell
lung cancer (NSCLC), and there were approximately 15,000 new
patients with NSCLC each year considered to be driven by ROS1
rearrangement (1). The
ROS1-rearranged lung cancer was recently reported to have good
response to crizotinib, a tyrosine kinase inhibitor of anaplastic
lymphoma kinase (2,3).
Case report
A 32-year-old woman presented with chest pain and a
prolonged cough. She had a smoking history of 2.5 pack years. Chest
computed tomography (CT) revealed a 57×36-mm tumor in the
mediastinum, with no tumors detected in other regions (Fig. 1A). Positron emission tomography
(PET)-CT showed a strong fluorodeoxyglucose (FDG) accumulation in
the tumor (SUVmax 13.2) (Fig. 1B).
Mediastinal tumor resection was planned by a video-assisted
thoracic surgery approach. No signs of macroscopic tumor invasion
to the right lung or superior vena cava were noted during surgery
(Fig. 1C). The tumor was completely
resected. Pathological examinations revealed the tumor to be 53×37
mm (Fig. 1D), and H&E staining of
the tumor cells showed a solid, alveolar pattern of proliferation
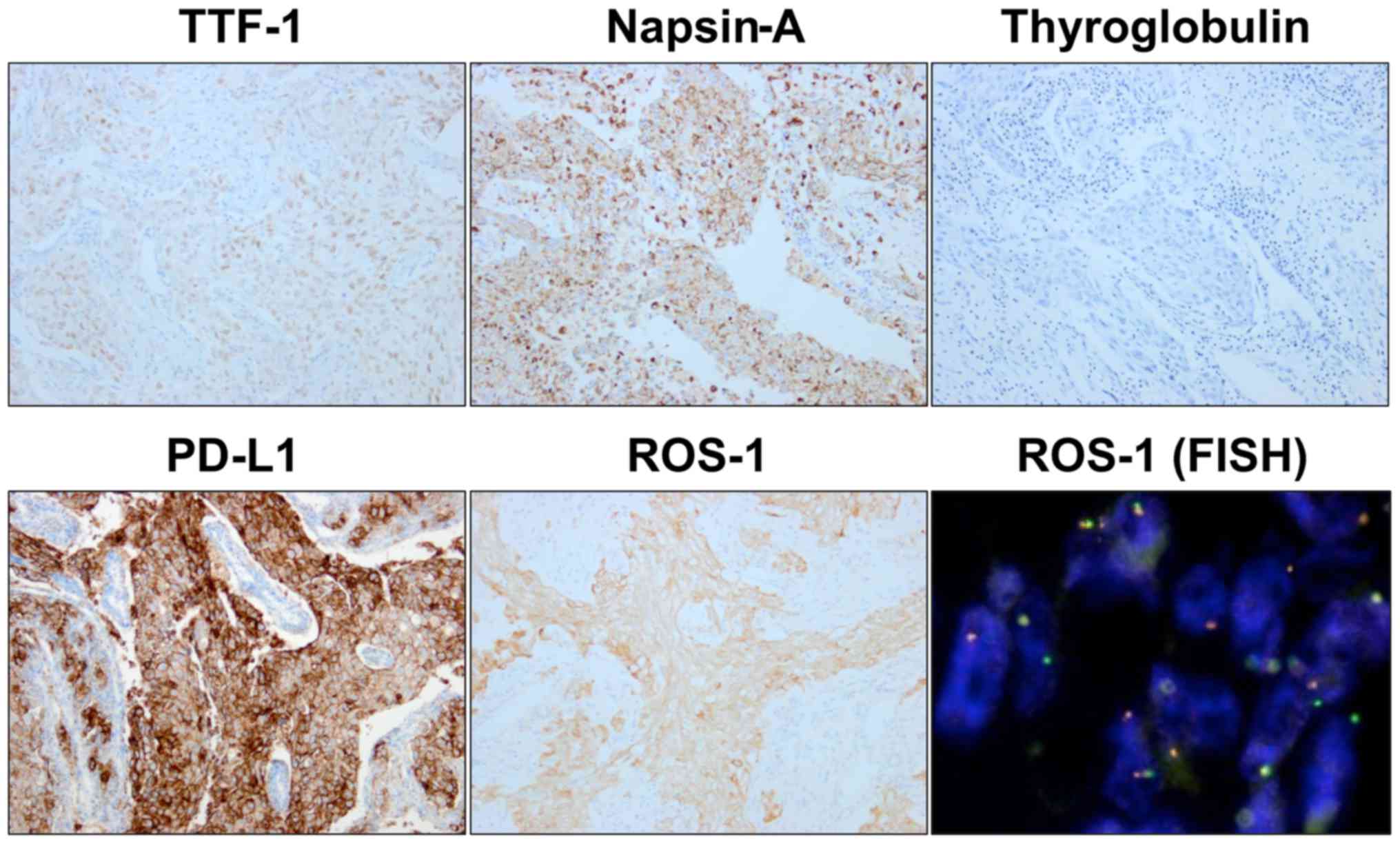
(Fig. 1E). Immunohistochemistry
revealed the tumor cells as positive for TTF-1, Napsin-A, ROS1, and
PD-L1 and negative for Thyroglobulin and EML4-ALK (Fig. 2). The tumor proportion score (TPS) of
PD-L1 was >99% using the clone 22C3 pharmDx kit (Agilent
Technologies, Inc., Santa Clara, CA, USA) (Fig. 2). ROS1 rearrangement was confirmed by
fluorescence in situ hybridization (Fig. 2). The mediastinal tumor was diagnosed
as mediastinal lymph node metastasis of ROS1-rearranged PD-L1
high-expression undifferentiated lung adenocarcinoma (pathological
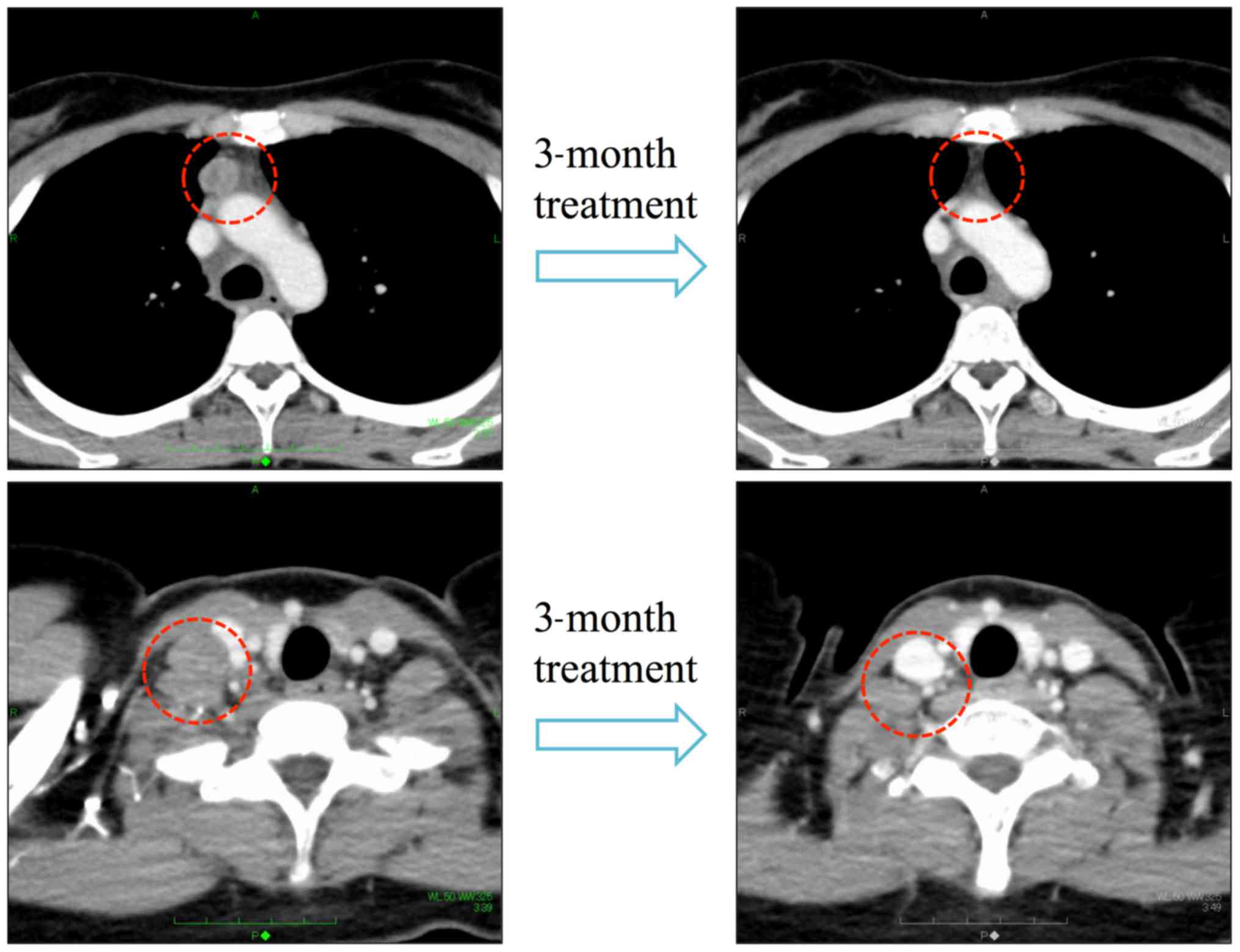
stage 3, TxN2M0). Two months after the operation, CT showed
multiple mediastinum lymph nodes metastases with rapid tumor
growth. The patient achieved a complete response after three cycles
of S-1 plus cisplatin with concurrent radiotherapy 60 Gy/30 Fr
(Fig. 3).
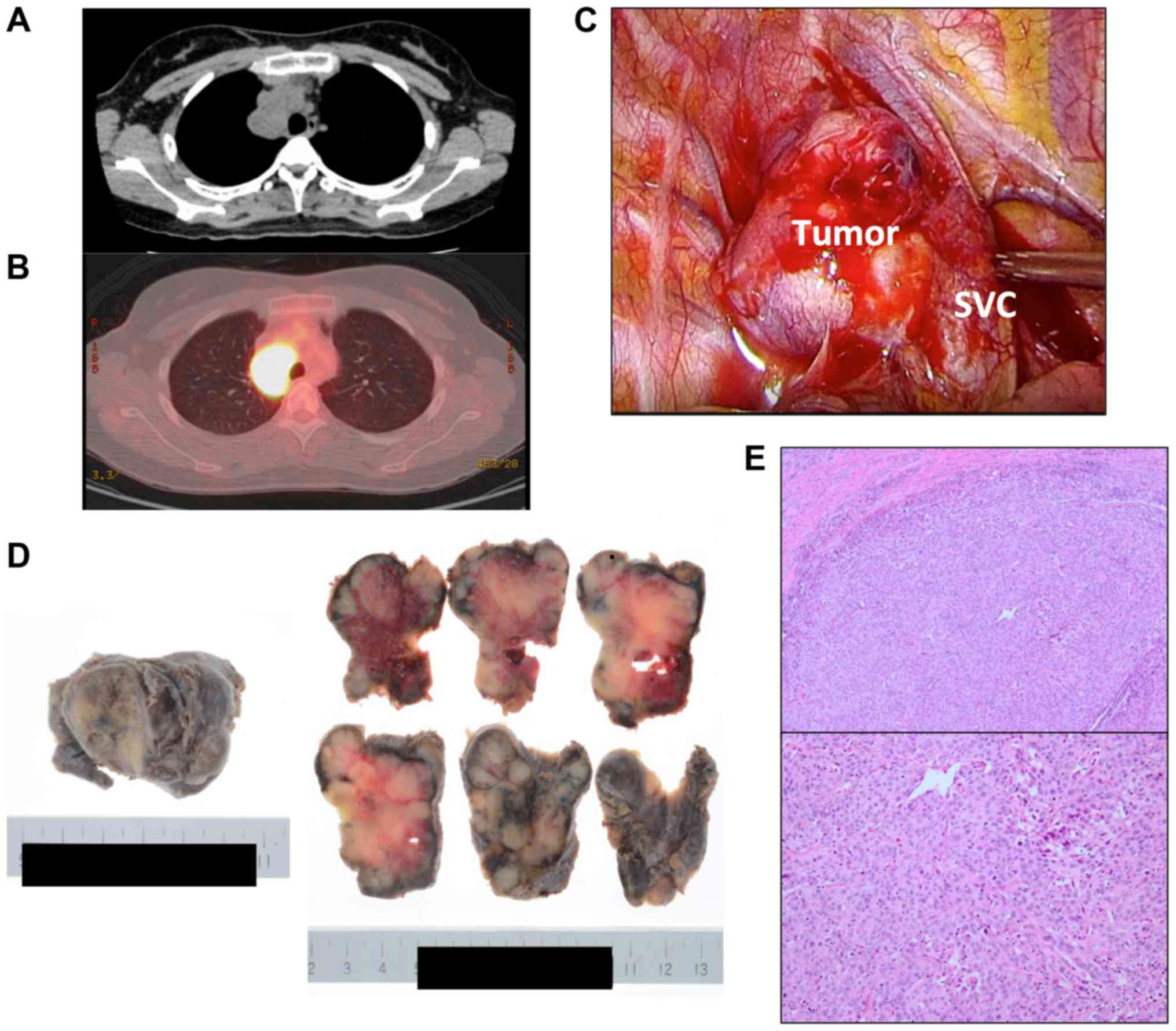 | Figure 1.(A) CT showed a 57×36-mm tumor in the
mediastinum. (B) PET-CT showed a strong accumulation of FDG in the
tumor (SUVmax 12.7). The accumulation of FDG in the right 7th
costal bone was also detected. (C) Intraoperative findings. There
were no signs of tumor invasion to the lung, bronchus, SVC, or
azygos vein. (D) Macroscopic pictures of the resected tumor. (E)
H&E staining of the tumor (magnification upper panel, ×40,
lower panel, ×100). CT, computed tomography; PET, positron emission
tomography; FDG, fluorodeoxyglucose; H&E, hematoxylin and
eosin; SVC, superior vena cava. |
Discussion
The mediastinal tumor in the present case was
diagnosed as lymph node metastasis of lung cancer, despite no tumor
being evident in the lung fields on CT or PET-CT. This type of lung
cancer is known as occult lung cancer and is rare (4). Immunohistochemistry was helpful for
diagnosing the origin of this tumor. The tumor was positive for
TTF-1 and Napsin-A, and negative for thyroglobulin which was useful
for distinguishing a lung origin from a thyroid origin. Based on
these findings, the tumor was diagnosed as lung adenocarcinoma.
ROS1-rearranged lung cancer is rare and comprises
only 1% of lung adenocarcinoma, which was recently reported to have
good response to crizotinib, a tyrosine kinase inhibitor of
anaplastic lymphoma kinase (2,3). A high
expression of PD-L1 predicts a good response to immune checkpoint
inhibitor monotherapy with pembrolizumab (5,6). Some
24.9–30.2% of advanced non-small cell lung cancer (NSCLC) had a TPS
of PD-L1 of ≥50% (7). Driver oncogene
mutations with approved therapies seldom coexist with a high
expression of PD-L1, with a reported frequency of only 6% (7). The combination of ROS1 rearrangement and
a high expression of PD-L1 is therefore considered very rare.
We chose the S-1 plus cisplatin with concurrent
radiotherapy regimen for the recurrent lesion as the patient
refused others due to hair loss. This regimen was reported to be
effective for locally advanced non-small cell lung cancer with mild
toxicities (8,9). Although the patient achieved a CR after
this treatment, careful follow-up is needed. If the tumors recur,
crizotinib or pembrolizumab are definitive treatment options
(2).
Acknowledgements
Not applicable.
Funding
No funding was received.
Availability of data and materials
All data generated or analyzed during this study are
included in this published article.
Authors' contributions
HO and YT collaborated in the conception of the
present study. KU, NS, TD and DH collected the data and prepared
the pictures presented in the figures. MT, YT and YM critically
revised the manuscript and were involved in data interpretation.
All authors contributed to writing the manuscript and approved the
final version.
Ethics approval and consent to
participate
Written informed consent was obtained from the
patients.
Patient consent for publication
Written informed consent was obtained from the
patients for the publication of any accompanying images.
Competing interests
The authors declare that they have no competing
interests.
References
|
1
|
Wu YL, Yang JC, Kim DW, Lu S, Zhou J, Seto
T, Yang JJ, Yamamoto N, Ahn MJ, Takahashi T, et al: Phase II study
of Crizotinib in east asian patients with ROS1-positive advanced
non-small-cell lung cancer. J Clin Oncol. 36:1405–1411. 2018.
View Article : Google Scholar : PubMed/NCBI
|
|
2
|
Shaw AT, Ou SH, Bang YJ, Camidge DR,
Solomon BJ, Salgia R, Riely GJ, Varella-Garcia M, Shapiro GI, Costa
DB, et al: Crizotinib in ROS1-rearranged non-small-cell lung
cancer. N Engl J Med. 371:1963–1971. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
3
|
Mazières J, Zalcman G, Crino L, Biondani
P, Barlesi F, Filleron T, Dingemans AM, Léna H, Monnet I,
Rothschild SI, et al: Crizotinib therapy for advanced lung
adenocarcinoma and a ROS1 rearrangement: Results from the EUROS1
cohort. J Clin Oncol. 33:992–999. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
4
|
Isohata N, Naritaka Y, Shimakawa T, Asaka
S, Katsube T, Konno S, Murayama M, Shiozawa S, Yoshimatsu K, Aiba
M, et al: Occult lung cancer incidentally found during surgery for
esophageal and gastric cancer: A case report. Anticancer Res.
28:1841–1847. 2008.PubMed/NCBI
|
|
5
|
Reck M, Rodríguez-Abreu D, Robinson AG,
Hui R, Csőszi T, Fülöp A, Gottfried M, Peled N, Tafreshi A, Cuffe
S, et al: Pembrolizumab versus chemotherapy for PD-L1-positive
non-small-cell lung cancer. N Engl J Med. 375:1823–1833. 2016.
View Article : Google Scholar : PubMed/NCBI
|
|
6
|
Herbst RS, Baas P, Kim DW, Felip E,
Pérez-Gracia JL, Han JY, Molina J, Kim JH, Arvis CD, Ahn MJ, et al:
Pembrolizumab versus docetaxel for previously treated,
PD-L1-positive, advanced non-small-cell lung cancer (KEYNOTE-010):
A randomised controlled trial. Lancet. 387:1540–1550. 2016.
View Article : Google Scholar : PubMed/NCBI
|
|
7
|
Rangachari D, VanderLaan PA, Shea M, Le X,
Huberman MS, Kobayashi SS and Costa DB: Correlation between classic
driver oncogene mutations in EGFR, ALK, or ROS1 and 22C3-PD-L1 ≥50%
expression in lung adenocarcinoma. J Thorac Oncol. 12:878–883.
2017. View Article : Google Scholar : PubMed/NCBI
|
|
8
|
Nogami N, Takigawa N, Hotta K, Segawa Y,
Kato Y, Kozuki T, Oze I, Kishino D, Aoe K, Ueoka H, et al: A phase
II study of cisplatin plus S-1 with concurrent thoracic
radiotherapy for locally advanced non-small-cell lung cancer: The
Okayama Lung Cancer Study Group Trial 0501. Lung Cancer.
87:141–997. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
9
|
Ichinose Y, Seto T, Sasaki T, Yamanaka T,
Okamoto I, Takeda K, Tanaka M, Katakami N, Sawa T, Kudoh S, et al:
S-1 plus cisplatin with concurrent radiotherapy for locally
advanced non-small cell lung cancer: A multi-institutional phase II
trial (West Japan Thoracic Oncology Group 3706). J Thorac Oncol.
6:2069–2075. 2011. View Article : Google Scholar : PubMed/NCBI
|