Introduction
Broca’s aphasia is a condition resulting from damage
to speech areas in the left hemisphere (1). It is easily distinguished by
experienced clinicians from other types of aphasia, such as
Wernicke’s, global and conduction aphasia; however, the prognosis
of patients with Broca’s aphasia remains poor (2). Cerebral venous sinus thrombosis (CVST)
is a rare condition, accounting for 0.5–1% of all strokes and can
cause Broca’s aphasia (2). It
mostly presents as a seizure, intracranial hypertension syndrome,
isolated headache and focal lobar syndrome (3). Diagnosis of CVST is usually based on
the presence of thrombi in the cerebral sinuses and/or veins on
veno computed tomography or magnetic resonance venography (4). Cisplatin is an antineoplastic agent
and has been associated with cerebrovascular incidents (5). Although the exact mechanism of
cisplatin-induced thromboembolic events is not currently well
understood, direct vascular toxicity, apoptosis, endothelial
dysfunction, hypomagnesemia and tumour embolisation are possible
risk factors (6). The current study
presents the case of a young male patient with advanced-stage small
cell lung cancer who developed Broca’s aphasia following
cisplatin-based chemotherapy, and also reviews the literature
surrounding similar cases. Written informed consent was obtained
from the patient.
Case report
A 27-year old male presented to the Department of
Medical Oncology, Gata Haydarpasa Training Hospital (Istanbul,
Turkey) with Broca’s aphasia and general seizures. The patient had
been diagnosed with advanced-stage small cell lung cancer two
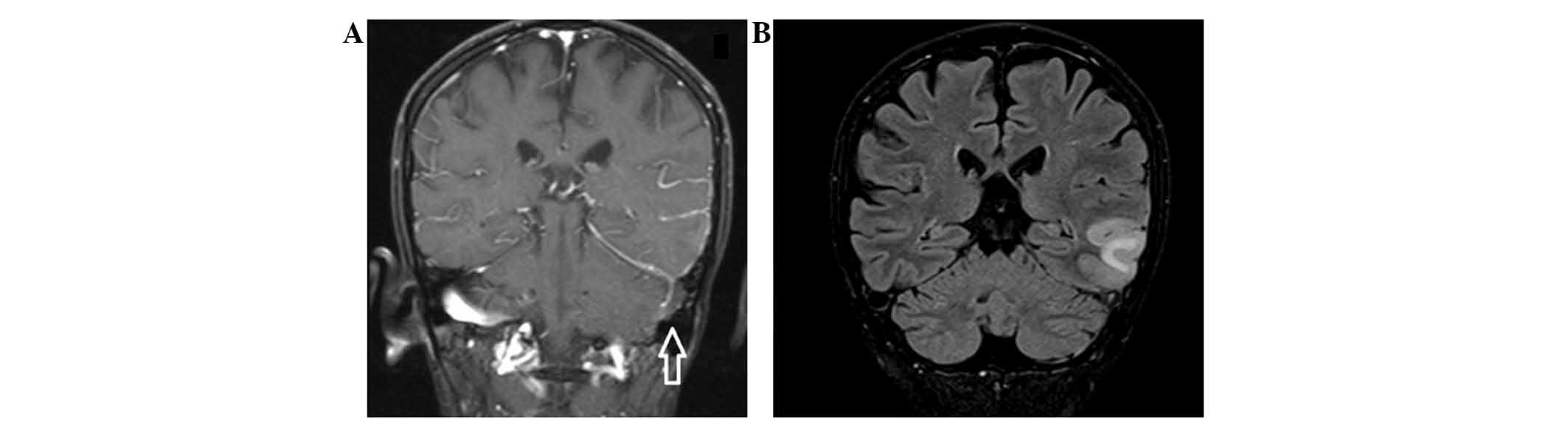
months previously. Baseline magnetic resonance imaging (MRI) of the
patient was normal (Fig. 1). The
patient received 135 mg cisplatin on day one and 180 mg etoposide
on days one to three. On day six of the second cycle, the patient
was admitted to the emergency department 4 h following the onset of
a stroke. The MRI scan revealed a CVST in the left transverse
sinus, as well as in the left sigmoid sinus with venous infarct
(Fig. 2). Laboratory analysis
showed a normal coagulation profile (activated partial
thromboplastin time, 34.5 sec; normal range, 25–38 sec;
international normalized ratio, 1.048; normal range, 0.8–1.2), and
serum magnesium levels (2 mg/dl; normal range, 1.2–2.5 mg/dl),
platelet count (204,000/mm3; normal range,
150,000–450,000/mm3) and lipid profiles (triglycerides:
116 mg/dl; normal range, 40–160 mg/dl) were also normal.
Echocardiography for embolic sources and electrocardiography for
arrhythmia were normal. The patient was subjected to
anticoagulation therapy with low molecular weight heparin and
speech therapy was commenced. The symptoms resolved completely
within one month and cisplatin-etoposide chemotherapy was continued
in addition to the anticoagulation therapy.
Discussion
Chemotherapy leads to an increased risk of
thromboembolic complications in germ cell, urethral, head and neck
and breast cancer, lymphoma and leukemia (7,8).
Venous thromboembolic events are observed more frequently than
arterial thromboembolic events. Weijl et al (7) reported that germ cell cancer patients
who receive cisplatin-based chemotherapy, particulary those who
receive high doses of corticosteroids or have liver metastases, are
at considerable risk of developing thromboembolic events.
Prophylactic administration of heparin may be considered in this
group. Li et al (8) reviewed
10,963 patients and reported that the risk of ischemic stroke
stroke after chemotherapy is predicted by the use of
cisplatin-based chemotherapy, not cancer histological type. The
worldwide incidence of venous thromboembolic events during
cisplatin-based chemotherapy for a various advanced solid tumors is
1.92% (10). We reviewed the
literature with respect to CVST occurring in cisplatin-based
chemotherapy. To date, very few cases of patients with CVST
following cisplatin-based chemotherapy have been reported (Table I) (11–13)
and to the best of our knowledge, this is the first report of
Broca’s aphasia and CVST associated with a cisplatin-based
chemotherapy regimen.
 | Table IPreviously reported cases of cerebral
venous sinus thrombosis following cisplatin-based chemotherapy. |
Table I
Previously reported cases of cerebral
venous sinus thrombosis following cisplatin-based chemotherapy.
| First author (year)
[ref] | Age,
years/gender | Tumor | Chemotherapy | Symptoms | Imaging | Site | Treatment |
|---|
| Unal (2008) [10] | 16/F | Ewing sarcoma | Cis+Ifo+Adr+Vin | Headache diplopia
ptosis | MR MRA | SSSTS | LMWH |
| Karam (2008)
[11] | 33/M | Germ-cell carcinoma
of the testis | Cis+Eto | Headache, partial
seizures | MR, CA | SSS
RLS | Anticoagulant sodium
valproate |
| Karam (2008)
[9] | 60/F | PDP carcinoma | Cis+5
Fluorouracil | Headache, partial
seizures | MR, CA | SSS
RLS | Heparin, clopidogrel
sodium valproate |
| Papet (2011)
[12] | 47/M | Embryonal
carcinoma | Cis+Eto+Ble | Headache, weakness in
left limbs | MR | SSS
SS
TS | Anticoagulant |
| Papet (2011)
[12] | 29/M | Embryonal
carcinoma | Cis+Eto+Ble | Headache, general
seizure | CT | SSS | LMWH |
The patient in the current study presented with
Broca’s aphasia due to a CVST in the left transverse sinus,
following cisplatin-based chemotherapy. Increased von Willebrand
factor, hypomagnesemia and damage to the vascular endothelium are
known risk factors for the cisplatin-associated vascular toxicity.
Chemotherapy-induced venous thrombosis usually develops within 10
days following the most recent chemotherapy cycle and 63% of cases
occurred following the first cycle (9). CVST is rare condition responsible for
approximately 1–2% of all cerebral strokes (14). The clinical presentation of CVST is
highly variable and non-specific; symptoms including headaches,
nausea and vomiting, visual disturbances, aphasia, coma and seizure
may be observed in CVST. The treatment of this condition ranges
from anticoagulation with intravenous heparin or subcutaneous
low-molecular-weight heparin, to endovascular thrombectomy or
thrombolysis. To date, there are no evidence-based guidelines on
prophylaxis of thromboembolic events in cancer patients that are
treated with cisplatin-based chemoterapy. Prophylactic heparin may
be used in patients with curable germ cell tumors and early stage
of lung cancer. The aim of this study was to increase the awareness
of the possibility of CVST in patients with lung cancer treated
with cisplatin-based chemotherapy.
In conclusion, the possibility of Broca’s aphasia
and CVST in patients with lung cancer, receiving platinum-based
chemotherapy, must be considered when determining the differential
diagnosis. Broca’s aphasia is a fatal and rare complication, but
can be treated successfully with low-molecular-weight heparin. An
accurate diagnosis of CVST may be determined using MRI. To date,
there are no evidence-based guidelines for the prophylaxis of
thromboemboli in cancer; for patients with a history of
thromboembolism, the concomitant use of low-molecular-weight
heparin and acetylsalicylic acid during each cycle of
cisplatin-based chemotherapy must be considered (15).
References
|
1
|
Burns MS and Fahy J: Broca’s area:
rethinking classical concepts from a neuroscience perspective. Top
Stroke Rehabil. 17:401–410. 2010. View Article : Google Scholar
|
|
2
|
Bousser MG and Ferro JM: Cerebral venous
thrombosis: an update. Lancet Neurol. 6:162–170. 2007. View Article : Google Scholar : PubMed/NCBI
|
|
3
|
Fridriksson J, Fillmore P, Guo D and
Rorden C: Chronic Broca’s aphasia is caused by damage to Broca’s
and Wernicke’s areas. Cereb Cortex. July 11–2014.(Epub ahead of
print). View Article : Google Scholar
|
|
4
|
Ferro JM and Canhão P: Cerebral venous
sinus thrombosis: update on diagnosis and manangement. Curr Cardiol
Rep. 16:5232014. View Article : Google Scholar
|
|
5
|
Karagoz B, Bilgi O, Akyol I, Ozgun A,
Turken O and Kandemir EG: Cerebrovasculer accident after
chemotherapy for testicular cancer. Military medicine. 174:320–321.
2009. View Article : Google Scholar
|
|
6
|
Fernández-Domínguez J, García Rodríguez R,
Valle Pereda M and Mateos Marcos V: Cerebral infarction after
cisplatin-gemcitabine chemotherapy: probable cause-effect.
Neurologia. 27:245–247. 2012. View Article : Google Scholar
|
|
7
|
Weijl NI, Rutten MF, Zwinderman AH, Keizer
HJ, Nooy MA, Rosendaal FR, Cleton FJ and Osanto S: Thromboembolic
events during chemotherapy for germ cell cancer: a cohort study and
review of the literature. J Clin Oncol. 18:2169–2178.
2000.PubMed/NCBI
|
|
8
|
Li SH, Chen WH, Tang Y, Rau KM, Chen YY,
Huang TL, Lis JS and Huang CH: Incidence of ischemic stroke
post-chemotherapy: a retrospective review of 10,963 patients. Clin
Neurol Neurosurg. 108:150–156. 2006. View Article : Google Scholar : PubMed/NCBI
|
|
9
|
Moore RA, Adel N, Riedel E, Bhutani M,
Feldman DR, Tabarra NE, et al: High incidence or thromboembolic
events in patients treated with cisplatin-based chemotherapy: a
large retrospective analysis. J Clin Oncol. 29:3466–3473. 2011.
View Article : Google Scholar : PubMed/NCBI
|
|
10
|
Seng S, Liu Z, Chiu SK, et al: Risk venous
thromboembolism in patients with cancer treated with Cisplatin: a
systematic review and meta-analysis. J Clin Oncol. 30:4416–4426.
2012. View Article : Google Scholar : PubMed/NCBI
|
|
11
|
Unal E, Yazar A, Koksal Y, Caliskan U,
Paksoy Y and Kalkan E: Cerebral venous sinus thrombosis in an
adolescent with Ewing sarcoma. Childs Nerv Syst. 24:983–986. 2008.
View Article : Google Scholar : PubMed/NCBI
|
|
12
|
Karam C and Koussa S: Cerebral dural sinus
thrombosis following cisplatin chemotherapy. J Clin Neurosci.
15:1274–1275. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
13
|
Papet C, Gutzeit A and Pless M: Two cases
of cerebral sinus venous thrombosis following chemotherapy for
non-seminomatous germ cell tumor. Case Rep Oncol. 4:555–559. 2011.
View Article : Google Scholar
|
|
14
|
Coutinho JM and Stam J: How to treat
cerebral venous and sinus thrombosis. J Thromb Haemost. 8:877–883.
2010.PubMed/NCBI
|
|
15
|
Etgen T, Weidenhöfer G and Kubin T:
Cisplatin-associated occlusion of the internal carotid artery.
Onkologie. 32:754–757. 2009. View Article : Google Scholar : PubMed/NCBI
|