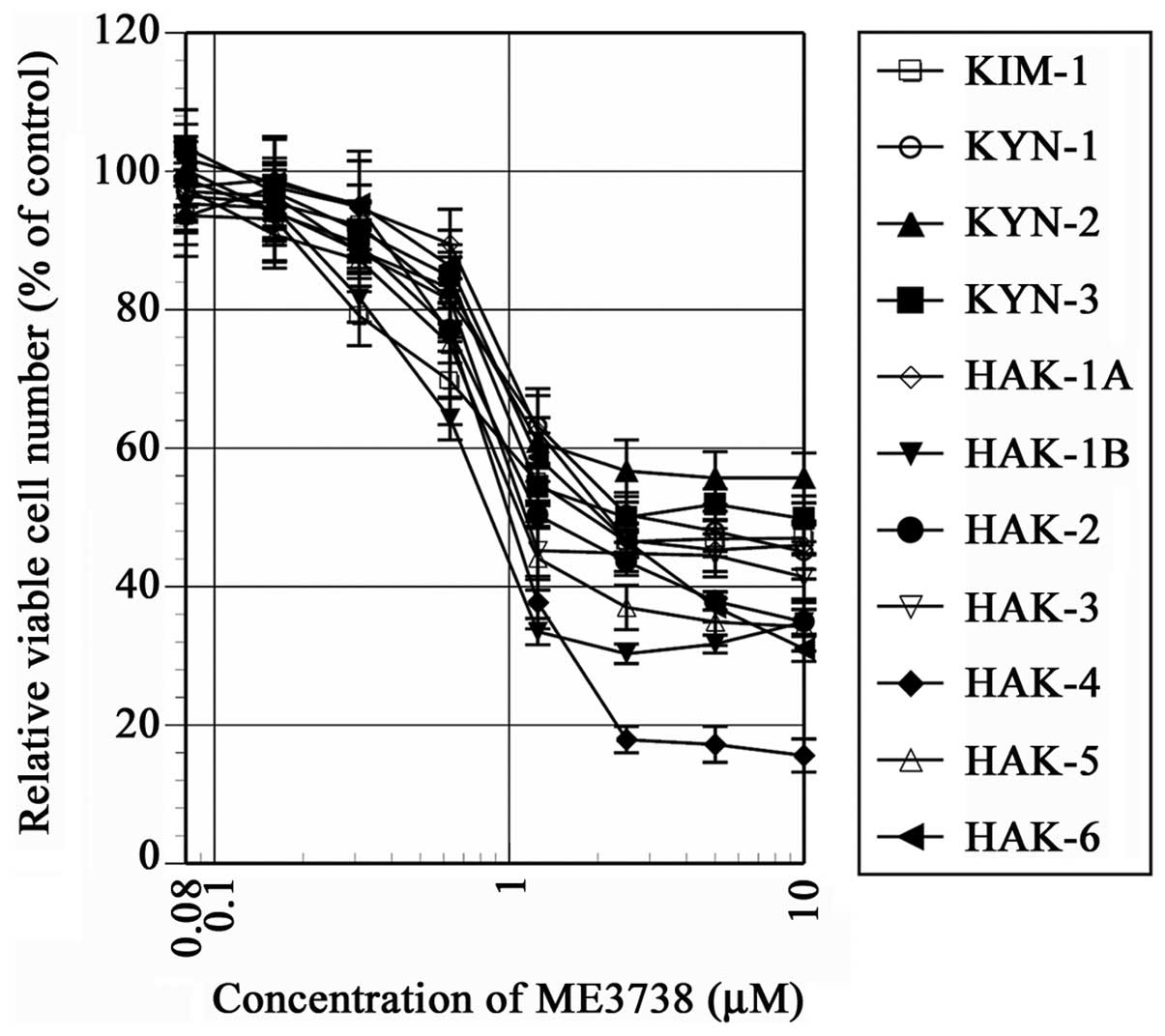
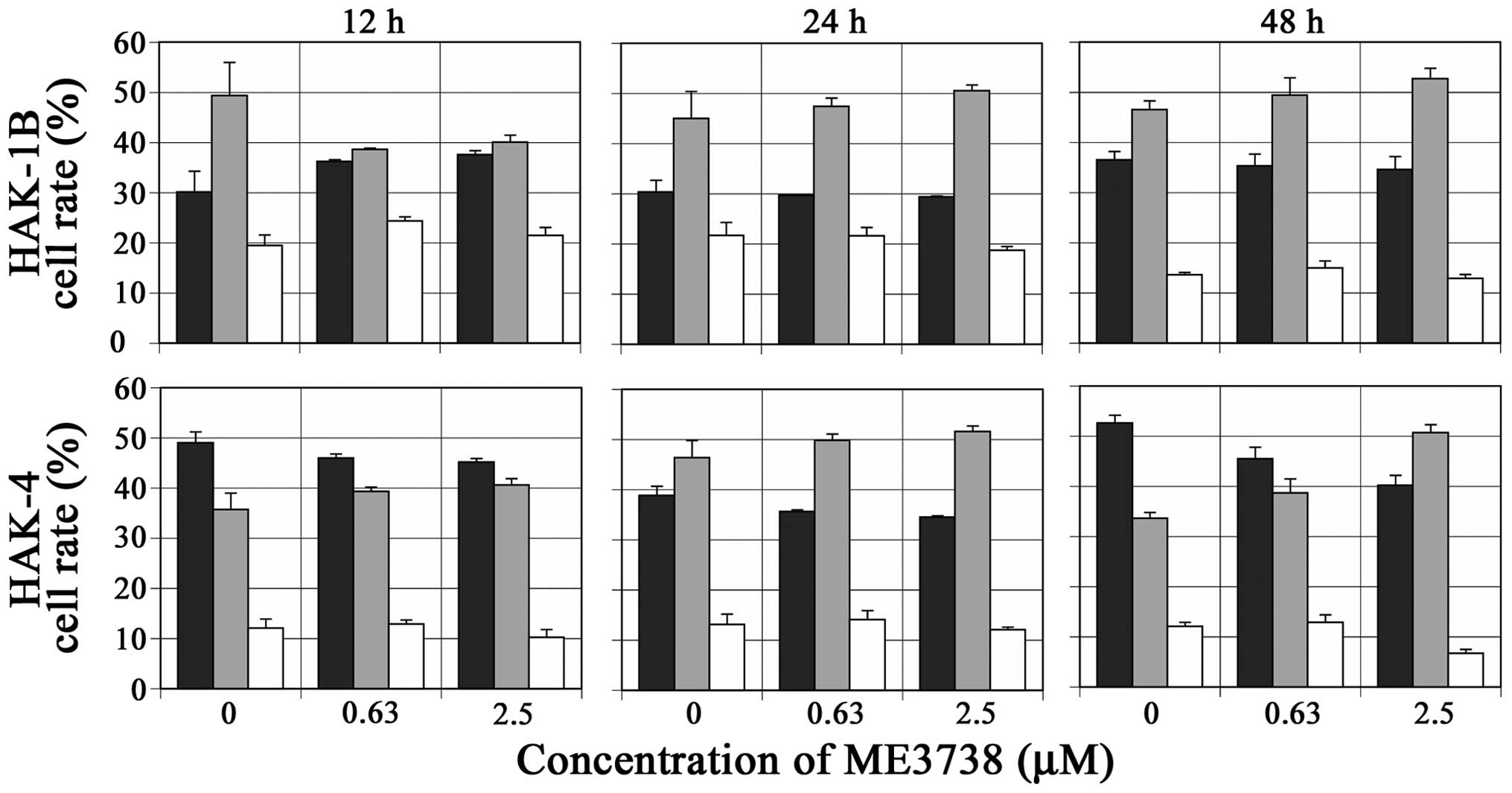
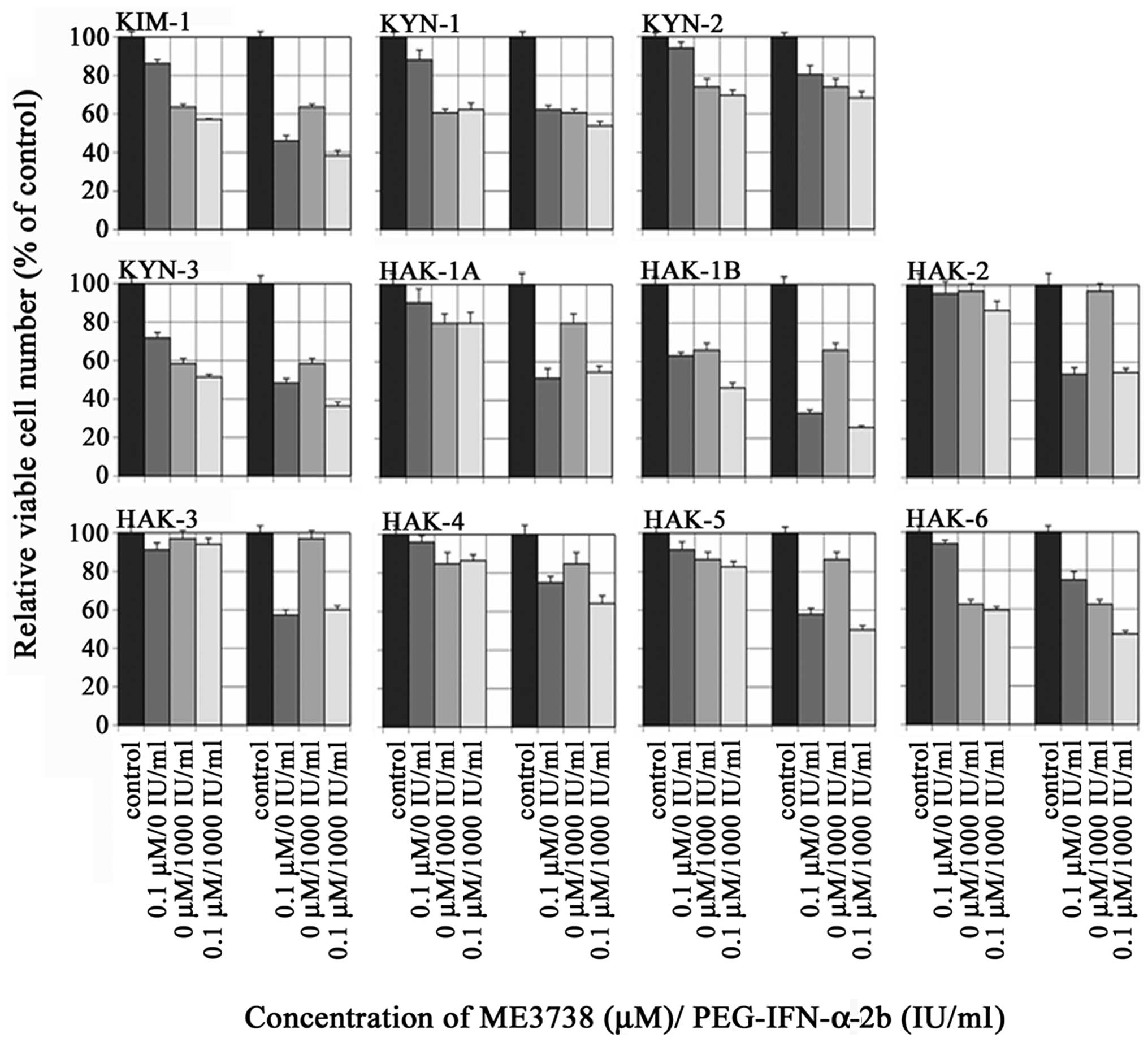
|
1
|
Rao AV and Gurfinkel DM: The bioactivity
of saponins: Triterpenoid and steroidal glycosides. Drug Metabol
Drug Interact. 17:211–235. 2000. View Article : Google Scholar : PubMed/NCBI
|
|
2
|
Hayashi K, Hayashi H, Hiraoka N and
Ikeshiro Y: Inhibitory activity of soyasaponin II on virus
replication in vitro. Planta Med. 63:102–105. 1997. View Article : Google Scholar : PubMed/NCBI
|
|
3
|
Simões CM, Amoros M and Girre L: Mechanism
of antiviral activity of triterpenoid saponins. Phytother Res.
13:323–328. 1999. View Article : Google Scholar : PubMed/NCBI
|
|
4
|
Gurfinkel DM and Rao AV: Soyasaponins: The
relationship between chemical structure and colon anticarcinogenic
activity. Nutr Cancer. 47:24–33. 2003. View Article : Google Scholar : PubMed/NCBI
|
|
5
|
Kim HY, Yu R, Kim JS, Kim YK and Sung MK:
Antiproliferative crude soy saponin extract modulates the
expression of IkappaBalpha, protein kinase C, and cyclooxygenase-2
in human colon cancer cells. Cancer Lett. 210:1–6. 2004. View Article : Google Scholar : PubMed/NCBI
|
|
6
|
Sung MK, Kendall CW, Koo MM and Rao AV:
Effect of soybean saponins and gypsophilla saponin on growth and
viability of colon carcinoma cells in culture. Nutr Cancer.
23:259–270. 1995. View Article : Google Scholar : PubMed/NCBI
|
|
7
|
Spector D, Anthony M, Alexander D and Arab
L: Soy consumption and colorectal cancer. Nutr Cancer. 47:1–12.
2003. View Article : Google Scholar : PubMed/NCBI
|
|
8
|
Hiwatashi K, Shirakawa H, Hori K, Yoshiki
Y, Suzuki N, Hokari M, Komai M and Takahashi S: Reduction of blood
pressure by soybean saponins, renin inhibitors from soybean, in
spontaneously hypertensive rats. Biosci Biotechnol Biochem.
74:2310–2312. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
9
|
Klein C, Wüstefeld T, Heinrich PC, Streetz
KL, Manns MP and Trautwein C: ME3738 protects from concanavalin
A-induced liver failure via an IL-6-dependent mechanism. Eur J
Immunol. 33:2251–2261. 2003. View Article : Google Scholar : PubMed/NCBI
|
|
10
|
Kuzuhara H, Nakano Y, Yamashita N, Imai M,
Kawamura Y, Kurosawa T and Nishiyama S: Protective effects of
alpha1-acid glycoprotein and serum amyloid A on concanavalin
A-induced liver failure via interleukin-6 induction by ME3738. Eur
J Pharmacol. 541:205–210. 2006. View Article : Google Scholar : PubMed/NCBI
|
|
11
|
Fukumura A, Tsutsumi M, Tsuchishima M,
Hayashi N, Fukura M, Yano H, Ozaki K and Takase S: Effect of the
inducer of interleukin-6 (ME3738) on rat liver treated with
ethanol. Alcohol Clin Exp Res (Suppl 1). 31:S49–S53. 2007.
View Article : Google Scholar
|
|
12
|
Nomoto M, Miyata M, Shimada M, Yoshinari
K, Gonzalez FJ, Shibasaki S, Kurosawa T, Shindo Y and Yamazoe Y:
ME3738 protects against lithocholic acid-induced hepatotoxicity,
which is associated with enhancement of biliary bile acid and
cholesterol output. Eur J Pharmacol. 574:192–200. 2007. View Article : Google Scholar : PubMed/NCBI
|
|
13
|
Maeda K, Koda M, Matono T, Sugihara T,
Yamamoto S, Ueki M, Murawaki Y, Yamashita N and Nishiyama S:
Preventive effects of ME3738 on hepatic fibrosis induced by bile
duct ligation in rats. Hepatol Res. 38:727–735. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
14
|
Abe H, Imamura M, Hiraga N, Tsuge M,
Mitsui F, Kawaoka T, Takahashi S, Ochi H, Maekawa T, Hayes CN, et
al: ME3738 enhances the effect of interferon and inhibits hepatitis
C virus replication both in vitro and in vivo. J Hepatol. 55:11–18.
2011. View Article : Google Scholar : PubMed/NCBI
|
|
15
|
Saibara T, Enomoto N, Kaneko S, Chayama K,
Sata M, Imawari M, Onishi S and Okita K: Clinical efficacy of
combination therapy with ME3738 and pegylated interferon-alpha-2a
in patients with hepatitis C virus genotype 1. Hepatol Res.
44:410–419. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
16
|
Murakami T: Establishment and
characterization of human hepatoma cell line (KIM-1). Act Hepatol
Jpn. 25:532–539. 1984. View Article : Google Scholar
|
|
17
|
Yano H, Kojiro M and Nakashima T: A new
human hepatocellular carcinoma cell line (KYN-1) with a
transformation to adenocarcinoma. In Vitro Cell Dev Biol.
22:637–646. 1986. View Article : Google Scholar : PubMed/NCBI
|
|
18
|
Yano H, Maruiwa M, Murakami T, Fukuda K,
Ito Y, Sugihara S and Kojiro M: A new human pleomorphic
hepatocellular carcinoma cell line, KYN-2. Acta Pathol Jpn.
38:953–966. 1988.PubMed/NCBI
|
|
19
|
Murakami T, Maruiwa M, Fukuda K, Kojiro M,
Tanaka M and Tanikawa K: Characterization of a new human hepatoma
cell line (KYN-3) derived from the ascites of the hepatoma patient.
Proceedings of the Japanese Cancer Association. Jpn J Cancer Res.
Tokyo. pp. 2921988;
|
|
20
|
Yano H, Iemura A, Fukuda K, Mizoguchi A,
Haramaki M and Kojiro M: Establishment of two distinct human
hepatocellular carcinoma cell lines from a single nodule showing
clonal dedifferentiation of cancer cells. Hepatology. 18:320–327.
1993. View Article : Google Scholar : PubMed/NCBI
|
|
21
|
Haramaki M, Yano H, Iemura A, Momosaki S,
Ogasawara S, Inoue M, Yamaguchi R, Kusaba A, Utsunomiya I and
Kojiro M: A new human hepatocellular carcinoma cell line (HAK-2)
forms various structures in collagen gel matrices. Hum Cell.
10:183–192. 1997.PubMed/NCBI
|
|
22
|
Utsunomiya I, Iemura A, Yano H, Akiba J
and Kojiro M: Establishment and characterization of a new human
hepatocellular carcinoma cell line, HAK-3, and its response to
growth factors. Int J Oncol. 15:669–675. 1999.PubMed/NCBI
|
|
23
|
Yano H, Iemura A, Haramaki M, Ogasawara S,
Takayama A, Akiba J and Kojiro M: Interferon alfa receptor
expression and growth inhibition by interferon alfa in human liver
cancer cell lines. Hepatology. 29:1708–1717. 1999. View Article : Google Scholar : PubMed/NCBI
|
|
24
|
Ogasawara S, Yano H, Momosaki S, Akiba J,
Nishida N, Kojiro S, Moriya F, Ishizaki H, Kuratomi K and Kojiro M:
Growth inhibitory effects of IFN-beta on human liver cancer cells
in vitro and in vivo. J Interferon Cytokine Res. 27:507–516. 2007.
View Article : Google Scholar : PubMed/NCBI
|
|
25
|
Hiasa Y, Kuzuhara H, Tokumoto Y, Konishi
I, Yamashita N, Matsuura B, Michitaka K, Chung RT and Onji M:
Hepatitis C virus replication is inhibited by
22beta-methoxyolean-12-ene-3beta, 24(4beta)-diol (ME3738) through
enhancing interferon-beta. Hepatology. 48:59–69. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
26
|
Kovalovich K, Li W, DeAngelis R, Greenbaum
LE, Ciliberto G and Taub R: Interleukin-6 protects against
Fas-mediated death by establishing a critical level of
anti-apoptotic hepatic proteins FLIP, Bcl-2, and Bcl-xL. J Biol
Chem. 276:26605–26613. 2001. View Article : Google Scholar : PubMed/NCBI
|
|
27
|
Haga S, Ogawa W, Inoue H, Terui K, Ogino
T, Igarashi R, Takeda K, Akira S, Enosawa S, Furukawa H, et al:
Compensatory recovery of liver mass by Akt-mediated hepatocellular
hypertrophy in liver-specific STAT3-deficient mice. J Hepatol.
43:799–807. 2005. View Article : Google Scholar : PubMed/NCBI
|
|
28
|
Ellington AA, Berhow M and Singletary KW:
Induction of macroautophagy in human colon cancer cells by soybean
B-group triterpenoid saponins. Carcinogenesis. 26:159–167. 2005.
View Article : Google Scholar : PubMed/NCBI
|
|
29
|
Kumada H, Suzuki Y, Ikeda K, Toyota J,
Karino Y, Chayama K, Kawakami Y, Ido A, Yamamoto K, Takaguchi K, et
al: Daclatasvir plus asunaprevir for chronic HCV genotype 1b
infection. Hepatology. 59:2083–2091. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
30
|
Asahina Y, Tsuchiya K, Nishimura T,
Muraoka M, Suzuki Y, Tamaki N, Yasui Y, Hosokawa T, Ueda K,
Nakanishi H, et al: α-fetoprotein levels after interferon therapy
and risk of hepatocarcinogenesis in chronic hepatitis C.
Hepatology. 58:1253–1262. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
31
|
Ikeda M, Fujiyama S, Tanaka M, Sata M, Ide
T, Yatsuhashi H and Watanabe H: Risk factors for development of
hepatocellular carcinoma in patients with chronic hepatitis C after
sustained response to interferon. J Gastroenterol. 40:148–156.
2005. View Article : Google Scholar : PubMed/NCBI
|
|
32
|
Oze T, Hiramatsu N, Yakushijin T, Miyazaki
M, Yamada A, Oshita M, Hagiwara H, Mita E, Ito T, Fukui H, et al:
Osaka Liver Forum: Post-treatment levels of α-fetoprotein predict
incidence of hepatocellular carcinoma after interferon therapy.
Clin Gastroenterol Hepatol. 12:1186–1195. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
33
|
Nagaoki Y, Aikata H, Nakano N, Shinohara
F, Nakamura Y, Hatooka M, Morio K, Kan H, Fujino H, Kobayashi T, et
al: Hiroshima Liver Study Group: Development of hepatocellular
carcinoma in patients with hepatitis C virus infection who achieved
sustained virological response following interferon therapy: A
large-scale, long-term cohort study. J Gastroenterol Hepatol.
31:1009–1015. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
34
|
Nagaoki Y, Aikata H, Miyaki D, Murakami E,
Hashimoto Y, Katamura Y, Azakami T, Kawaoka T, Takaki S, Hiramatsu
A, et al: Clinical features and prognosis in patients with
hepatocellular carcinoma that developed after hepatitis C virus
eradication with interferon therapy. J Gastroenterol. 46:799–808.
2011. View Article : Google Scholar : PubMed/NCBI
|
|
35
|
Tokita H, Fukui H, Tanaka A, Kamitsukasa
H, Yagura M, Harada H and Okamoto H: Risk factors for the
development of hepatocellular carcinoma among patients with chronic
hepatitis C who achieved a sustained virological response to
interferon therapy. J Gastroenterol Hepatol. 20:752–758. 2005.
View Article : Google Scholar : PubMed/NCBI
|
|
36
|
Makiyama A, Itoh Y, Kasahara A, Imai Y,
Kawata S, Yoshioka K, Tsubouchi H, Kiyosawa K, Kakumu S, Okita K,
et al: Characteristics of patients with chronic hepatitis C who
develop hepatocellular carcinoma after a sustained response to
interferon therapy. Cancer. 101:1616–1622. 2004. View Article : Google Scholar : PubMed/NCBI
|