1. Introduction
The primary function of telomeres is to prevent the
destruction of the genome from the DNA damage response (DDR), thus
ensuring genomic stability during DNA replication (1). Structurally, the ends of telomeres are
single-stranded, whereas telomeres are double-stranded at all their
length (2). The numerous repetitive
sequences of the hexanucleotide 5'-TTAGGG-3' are organized in a
complex, three-dimensional lariat-like structure known as the
telomere-loop (t-loop) (3). When
the 3' end of a single DNA strand of telomeres is inserted into a
double-stranded duplex of telomeres, the formation of a D-loop
occurs (4). In the t-loop
structure, 3' single-stranded G-rich overhangs, known as
G-overhangs, protrude from the double-stranded telomeric region
(5). The organization of telomeres
in forming such looped structures is crucial and protects them from
degradation. This t-loop conformation requires the presence of
specific telomeric interacting proteins, such as telomeric repeat
binding factor 1 (TRF-1), telomeric repeat binding factor 2
(TRF-2), TRF-1 interacting nuclear protein 2 (TIN-2), telomeric
overhang binding protein 1 (POT-1), TIN-2 and POT-1 interacting
protein 1 and repressor-activator protein 1, that stabilize the
t-loop (2). As mentioned earlier,
telomere-binding proteins comprise the ‘shelterin’ protein complex
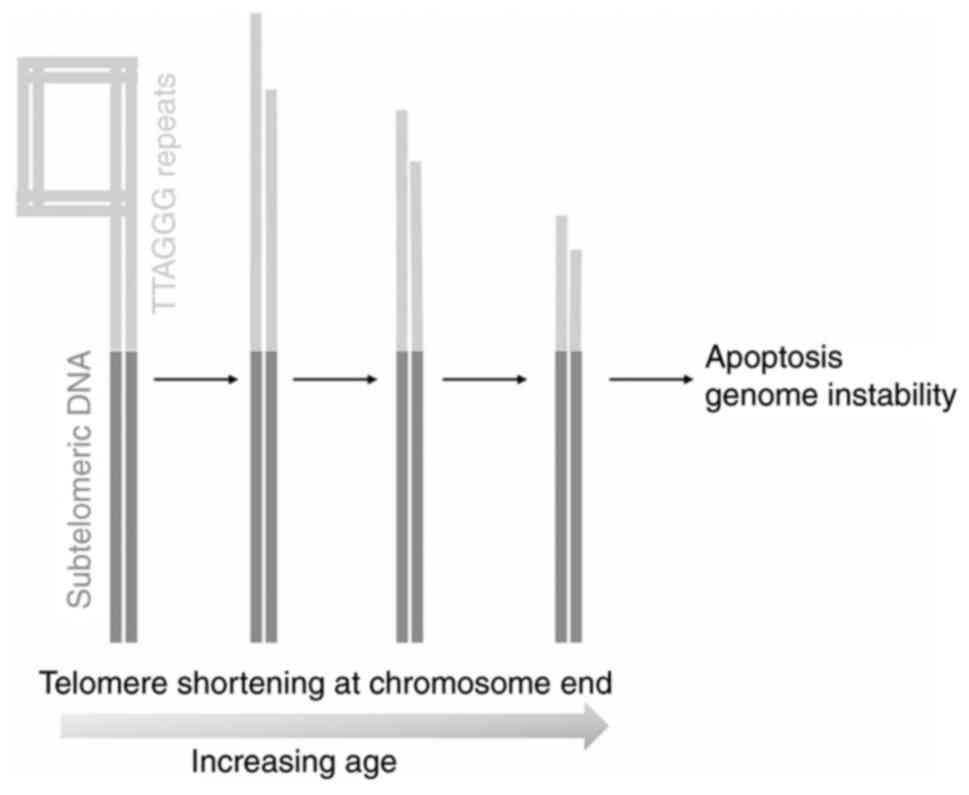
(6). Telomere shortening arises
from incomplete lagging strand DNA synthesis, resulting in
single-stranded overhangs. During the aging process, telomeres are
progressively shortened below a certain threshold due to each cell
division, known as the ‘end-replication problem’ (6). For that reason, it has been
demonstrated that telomere shortening is associated with advanced
aging (7) (Fig. 1). Accordingly, the genetic rescue of
telomerase can compensate for premature aging in
telomerase-deficient mice (8).
Similarly, nutraceutical supplements can sustain the telomere
length (TL) at a greater extent in females than males (9). Additionally, nutraceutical
formulations can attenuate aging by increasing the physical action
of aged animals (10).
Aging arises from multiple mechanisms, such as
cellular senescence, genome instability, metabolic dysfunction,
mitochondrial deterioration, microbial dysbiosis and sustained low
inflammation, epigenetic alterations including DNA methylation,
stem cell exhaustion, disturbed cellular communication, proteasomal
degradation or dysregulated autophagy and telomere shortening
(11). According to the American
Federation for Aging Research (AFAR) (12), the hallmarks of aging have been
defined based on the following criteria: i) The prediction of life
expectancy than natural chronologic age; ii) be subject to
experiments to elucidate characteristics that accelerate aging;
iii) be subject to investigations to shed light on mechanisms
underlying the prevention of aging; iv) be minimally invasive
without harming individuals (13).
The compliance of TL to the criteria noted by the AFAR defines
telomere shortening as a reliable biomarker of aging (14,15).
Notably, telomere shortening represents an essential
hallmark of the aging process, and it is accelerated in age-related
disorders (7). For example, short
telomeres have been observed in leukocytes of osteoporotic patients
compared to long ones of controls, whereas long telomeres have been
reported in females with low osteoporosis risk (16). In agreement with the aforementioned
findings, it has been demonstrated that aged osteocytes that were
senescent through a high p16 expression were associated with bone
loss (17).
Osteoporosis is an aging-related disease of bone
metabolism, is prevalent among the elderly, and at least 200
million cases worldwide have been reported (18). The characteristic features of
osteoporosis are an increased vulnerability to bone fractures and
high fragility due to low bone mass and deterioration of bone
microstructure (19). Osteoporosis
imposes a significant economic burden in the aging society,
necessitating the identification of markers that stratify
individuals to osteoporosis (20).
Post-menopausal women are mainly exposed to bone fractures, such as
vertebral and hip fractures (21).
In Europe and the USA, >30% of women and 20% of men >50 years
of age have osteoporosis (22). At
the same time, it is also estimated that >40% of post-menopausal
women and 30% of men can experience a fracture related to
osteoporosis during their lifetimes (23,24).
A complete understanding of the mechanisms
underlying osteoporosis is crucial in order to develop effective
therapeutic strategies to attenuate its progression. Among the
factors determining bone loss are low estrogen levels in
post-menopausal women and low testosterone levels in men, resulting
in osteoporosis (25).
Estrogen-deprivation-related osteoporosis observed in
post-menopausal women appears to differ from age-related
osteoporosis (26,27). However, estrogen is not the only
factor that contributes to bone loss during aging (28); bone homeostasis can also be affected
by parathyroid hormone, vitamin D (cholecalciferol), calcitonin and
corticosteroids (29) (Fig. 2).
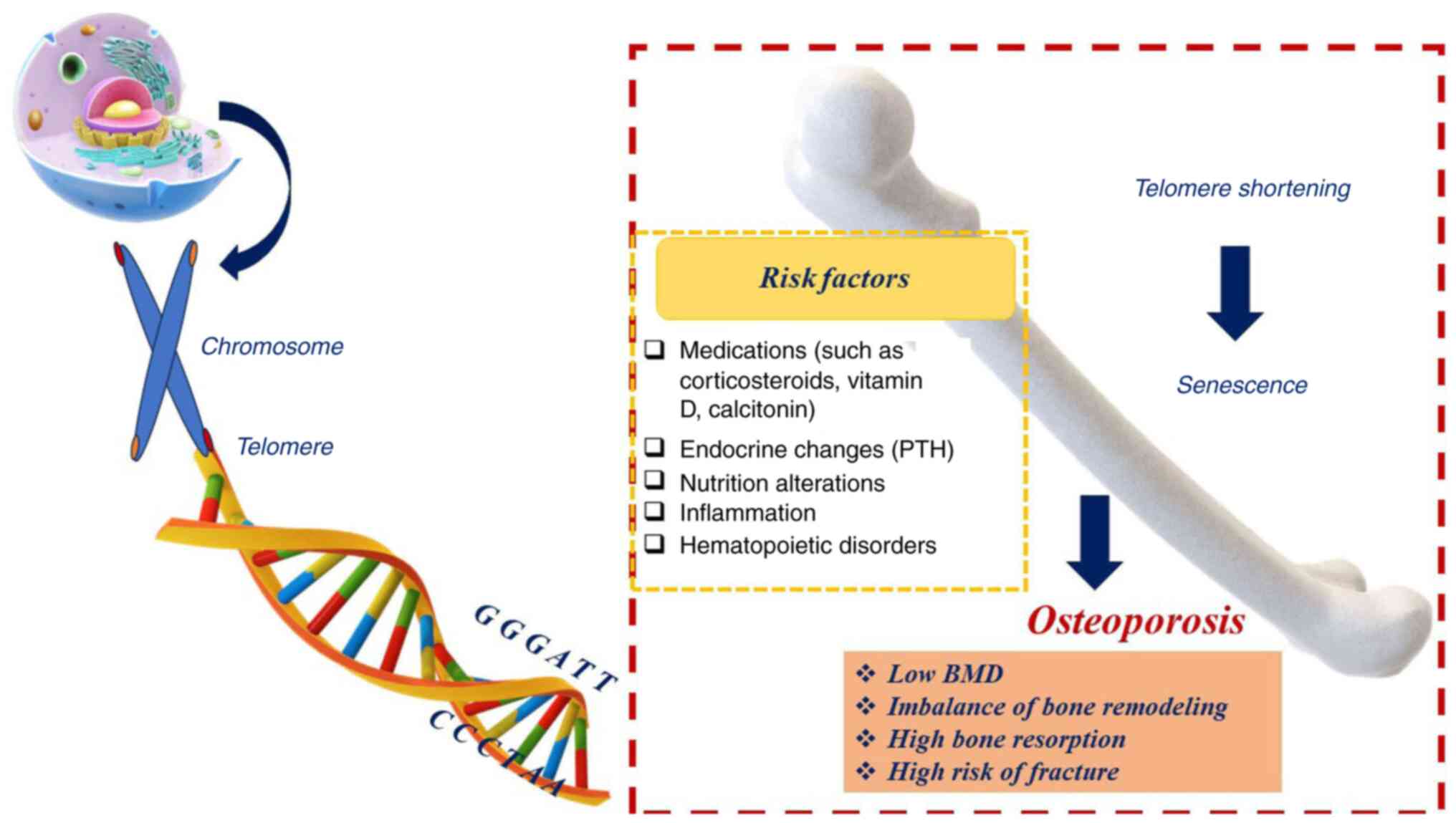 | Figure 2Since osteoporosis is a highly
multifactorial entity, it may be related to multiple risk factors.
Multiple factors, such as PTH, vitamin D (cholecalciferol),
calcitonin, corticosteroids, estrogen, inflammation, lifestyle and
hematopoietic alterations promote telomere shortening, which
increases the risk of developing osteoporosis. BMD, bone mineral
density; PTH, parathyroid hormone. |
Bone remodeling has been reported as a key parameter
for determining the onset and progression of osteoporosis. In bone
remodeling, osteoclasts and osteoblasts are implicated in
orchestrating bone architecture through the degradation of old bone
(bone resorption) and the formation of new bone (bone formation),
respectively (30). Previous
research has analyzed the signaling pathways that regulate the
balance between osteoclastic bone resorption and osteoblastic bone
formation. Such signaling pathways are the following: Receptor
activator of nuclear factor-κB (RANK)/RANK ligand
(RANKL)/osteoprotegerin (OPG) and canonical Wnt (31). For example, OPG prevents the binding
of RANKL to the RANK receptor, thus preventing osteoclast function
(27) (Fig. 3). Currently, several pharmaceutical
agents, including bisphosphonates, estrogens, the selective
estrogen receptor modulator, raloxifene, the human monoclonal
antibody, denosumab, and the recombinant human parathormone, 1-34
teriparatide, are commonly used to treat osteoporosis effectively
(18) (Fig. 3). Recently, romosozumab (ROMO), an
approved monoclonal antibody directed against sclerostin in several
countries for the treatment of osteoporosis in post-menopausal
women who are at a high risk of fractures, has attracted
significant attention owing to its ability to reinforce bone
formation. At the same time, it can attenuate bone resorption after
its short-term use (32,33). The underlying molecular mechanism of
ROMO relies on the induction of the Wnt signaling pathway since the
action of ROMO is directed to prevent sclerostin-a glycoprotein,
which exerts inhibitory effects on osteoblasts and further induces
bone resorption (32,33). Following the review of three-phase
clinical trials, researchers have previously evaluated the superior
effect of ROMO compared with teriparatide in post-menopausal women
with osteoporosis who were at a high risk of fractures and they
were previously treated with bisphosphonate therapy, thereby
causing bone remodeling and increasing bone mineral density (BMD)
(34). Despite therapeutic options,
their clinical efficacy is hindered due to the emerging
side-effects after their long-term use, making it urgent to
identify novel and effective therapeutic interventions against
osteoporosis (35). In this
direction, further investigations are required to elucidate the
possible cardiovascular events after using ROMO in postmenopausal
women with a risk of high fractures (33).
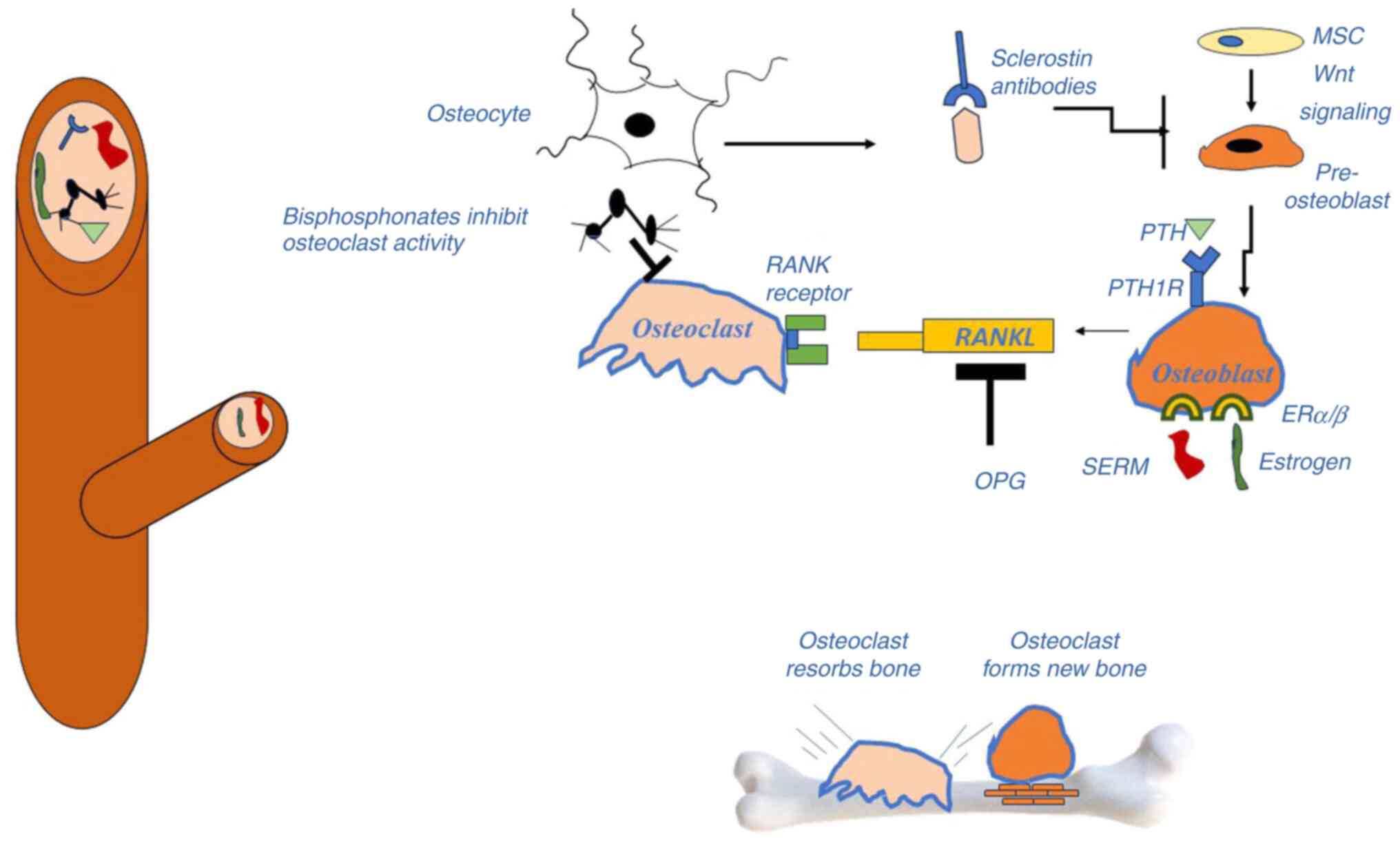 | Figure 3Bone is a tissue that consists of
diverse cell types (osteoblasts, osteoclasts) that interact in a
complex manner. In particular, bone marrow-derived stem cells
differentiate into osteoblasts, which in turn are converted into
osteocytes following their incorporation into the mineralized
matrix. As cells age, they undergo apoptosis or senescence. An
imbalance between osteoclasts and osteoblasts may result in the
development of fragile porous bone. RANKL is required for
osteoclastogenesis, whereas OPG hinders the osteoclast route.
Sclerostin, the Wnt pathway, SERM and estrogen are key factors
involved in the differentiation of MSCs into osteoblasts. When
sclerostin antibodies are present owing to their production by
osteocytes, the Wnt signaling pathway renders osteoblastogenesis
inactive. ER, estrogen receptor; MSC, mesenchymal stem cell; OPG,
osteoprotegerin; PTH, parathyroid hormone; RANKL, receptor
activator of nuclear factor-κB ligand; SERM, selective estrogen
receptor modulator. |
Since telomere shortening is considered a molecular
hallmark of aging (11), a summary
of the state of the field provides clues that telomere shortening
can be a driver for the onset and progression of osteoporosis. In
the present review, a discussion was performed of the current
research data on the possible association of TL with osteoporosis
in humans.
2. Clinical studies regarding telomere
length and osteoporosis
Initially, it was generally questionable whether TL
can contribute independently to aging as a biomarker (36-38).
To answer that question, a number of studies have demonstrated that
TL can fulfill the main prerequisites defined by the AFAR.
Epidemiological and clinical studies have provided evidence that
telomere shortening is the most widely and reliable biomarker that
characterizes the aging process (15,39).
Numerous studies have illustrated a strong association between
different biomarkers and the onset of aging; current aging markers
are not without drawbacks. Indeed, other features have been
employed to evaluate various aspects of the aging process since the
mechanism underlying the aging process in humans is incredibly
complicated.
Apart from the crucial involvement of telomere
shortening in aging, biomarkers of aging are usually used for
evaluating the progression of age-related disorders, such as
osteoporosis (40). Identifying
preventive strategies against osteoporosis requires precise and
reliable biomarkers for assessing the rate of osteoporosis. Over
the past decades, age-related telomere shortening has attracted
increasing attention in research as one of the most promising
fields.
The literature search performed herein resulted in
the isolation of relevant published articles; there were three
non-randomized-controlled studies and eight longitudinal cohort
studies. The main findings of the review are summarized in Table I.
 | Table IThe main findings of the published
articles. |
Table I
The main findings of the published
articles.
| Authors, year of
publication, country | Participants | Outcome
measures | Main findings | (Refs.) |
|---|
| Kveiborg et
al, 1999, Denmark | Young (aged 20-26
years), elderly (aged 48-85 years, and patients with osteoporosis
(aged 52-81 years) | TL in peripheral
blood leukocytes | No significant
changes were observed between patients with osteoporosis and the
age-matched controls | (60) |
| Bekaert et
al, 2005, Belgium | 352 Healthy males,
71-86 years of age | TL in the
leucocytes of peripheral blood | Age-corrected mean
telomere restriction fragment length was associated with
longitudinal bone loss at the total forearm, particularly at the
mid-region of the forearm and at the ultra-distal forearm | (41) |
| Valdes et
al, 2007, UK | 2,150 Healthy and
osteoporotic women, 18-79 years of age | Telomere leukocyte
length, bone mineral density, and osteoporosis | TL positively
related to bone mineral density of both the forearm and the spinal
column | (16) |
| Sanders et
al, 2009, USA | 2,750 Healthy and
osteoporotic adults, 71-79 years of age | Telomere leukocyte
length, bone mineral density, and osteoporosis | TL was not related
to bone mineral density, osteoporosis, and risk of fractures | (65) |
| Tang et al,
2010, China | 1,867 Elderly
adults (mean age, 72 years) | Telomere leukocyte
length and hip bone mineral density | No association was
found between TL and baseline bone mineral density or bone loss
over a 4-year period | (61) |
| Tamayo et
al, 2010, Spain | 35 Adults aged
>40 years with osteoporosis vs. 130 healthy individuals | Telomere leukocyte
length and osteoporosis | Telomere leukocyte
length was statistically significantly shorter in patients with
osteoporosis | (45) |
| Nielsen et
al, 2015, Denmark | 420 Women (mean
age, 63.9 years; range, 25-93 years) | Telomere leukocyte
length and bone mineral density | No statistically
significant associations were found between leukocyte TL and bone
mineral density | (46) |
| Kalyan et
al, 2018, Canada | 73 Women with HIV
(mean age, 43 years) | Telomere leukocyte
length and bone mineral density | Reduction of
leukocyte TL was statistically significantly related to lower bone
mineral density | (49) |
| Tao et al,
2019, China | 1,017 Elderly
Chinese adults (mean age, 66.4 years) from whom 433 were males and
584 cases were females at the post-menopausal stage and probably
osteoporotic | Telomere leukocyte
length and bone mineral density | Leukocyte TL was
found to be associated with a lower bone mineral density and
osteoporosis in elderly women, but not in the male population | (51) |
| Kirk et al,
2022, Australia | 20,400 Elderly
adults with osteosarcopenia (n=205) compared to the matched
controls in the UK Biobank (mean age 67.8 years, 53% male) | Telomere leukocyte
length and osteosarcopenia | No association was
found between leukocyte TL and osteosarcopenia or femoral neck bone
mineral density | (62) |
| Curtis et
al, 2022, UK | 111,395 Adults in
the UK (mean age, 56.7 years) | Telomere leukocyte
length and risk of fractures | Weak association in
females, even weaker in males | (53) |
Initially, Bekaert et al (41) performed an early observational study
comprising 352 elderly healthy male subjects (aged 71-86 years).
They found that the mean leukocyte TL (LTL) was reduced with an
advanced age. The age-corrected TL was positively associated with
bone loss, as confirmed by biochemical analysis of bone turnover
and BMD at different distal forearm sites, including the mid-region
of the forearm, ultra-distal forearm and the total forearm
(41). Furthermore, they elucidated
the use of mean LTL as a predictive marker for bone loss, according
to the sex steroid status of elderly healthy male individuals
(41). For that purpose, telomere
restriction fragment (TRF) length analysis was performed along with
various examinations of testosterone, estradiol and sex
hormone-binding globulin in the blood of males (41). The results proved that the short
length of telomere restriction fragment determined bone loss at
various sites of their radius and ulna (P<0.05), which was
inversely associated with the age of the participants (P=0.049),
irrespective of the hormonal status of healthy older adults
(41). Furthermore, aged
individuals with bone loss had a mean TRF length 423 base pairs
(bps), shorter than that of age-matched controls without bone loss,
given that the telomere erosion rate in leukocytes appears to be
shortened by 23 bps per year (42).
Based on experiments revealing a tight association
between telomere shortening and the senescence of osteoblasts in
vitro (43,44), a large population-based study was
performed in a series of 2,150 female twins (aged 18-80 years),
where the association between LTL and BMD was evaluated. A positive
association between TL with BMD of the spine and distal forearm
(but not with the femoral neck) was revealed, and longer telomeres
were associated with a reduced risk of developing osteoporosis at
two or more sites in women >50 years of age (16). In a clinical setting, women with
osteoporosis had a shorter TL by 117 bps than their matched
controls, implying accelerating skeletal aging due to TL. As a
result, it was proposed that the LTL could be used as a marker for
skeletal aging (16).
In the same year, Tamayo et al (45), in a non-randomized controlled study
of 35 elderly patients (aged 59-95 years) with osteoporosis and 130
healthy individuals, found that LTL was statistically significantly
shorter (P=0.001) in the osteoporosis group than that in the
control group. Nielsen et al (46), in a longitudinal cohort study
involving 420 women (mean age, 64 years; range, 25-93 years), did
not find a statistically significant association between LTL and
BMD; nevertheless, a statistically significant association was
indeed observed between LTL and age as well as between body mass
index-adjusted age and BMD (46).
It was also increasingly apparent that accelerating aging was
related to human immunodeficiency virus (HIV) infection, which in
turn accounted for telomere damage and mitochondrial DNA damage
(47,48). In this context, in a
non-randomized-controlled study involving 73 women (mean age, 43
years) living with HIV aged >50 years, it was highlighted that
the observed shortening of LTL was statistically significantly
related to a lower BMD at the lumbar spine [mean difference, -0.39;
95% confidence interval (CI), -0.61, -0.17] and total hip (mean
difference, -0.29; 95% CI, -0.52, -0.07), suggesting that LTL was
negatively associated with a low bone mass in women with HIV, and
that this connection may be related to the pathophysiology of
premature aging in HIV-infected women (49). The aforementioned results were
important, given that aged individuals living with HIV had a higher
prevalence for osteoporosis, as shown by a pooled odds ratio (OR)
at 3.7 compared to their age-matched controls, following a
meta-analysis of 20 studies (50).
In agreement with the aforementioned findings, Tao
et al (51) conducted a
cohort study of 1,017 elderly Chinese adults (mean age, 66.4
years), from whom 584 were older women at the post-menopausal stage
and 433 were older men, providing substantial evidence that there
was a positive association between LTL and BMD through analysis of
the results with multiple linear and ordinal logistic regressions,
thereby increasing risk of developing osteoporosis in women at
post-menopausal stage (51).
Importantly, that positive association of LTL and BMD reduced as
women aged. On the contrary, no association was observed between
the two parameters mentioned above in older males. As a result, it
was concluded that the predictive value of LTL in osteoporosis was
sex-dependent (51).
A significant association between short TLs and a
low telomerase activity has also been shown to be associated with
skeletal pathologies, such as osteoporosis and osteoarthritis, in
which the dysregulated restitution of the subchondral bone occurs
in the elderly (52).
Recently, Curtis et al (53) analyzed the positive association
between LTL and the risk of fractures in a longitudinal cohort
study. The authors of that study examined a population of 51,900
males and 59,500 females from the UK Biobank (mean age, 56.7
years). They showed that there was a weak association between a
longer LTL and a reduced risk of any fracture in women [hazard
ratio (HR)/standard deviation (SD), 0.96], with less evidence found
in males (HR/SD, 0.98) (53).
According to that study, this was the most extensive relevant
study, showing only a weak association between LTL and the risk of
fractures (53).
The majority of studies have reported a positive
association between telomere shortening and the pathological
characteristics of osteoporosis. In particular, telomere shortening
is strongly associated with a low BMD (13,52).
The mechanism by which telomere shortening causes osteoporosis has
come to light through experiments using mice or individuals
harboring germline mutations in genes implicated in telomere
maintenance (15,40,44,45,48,50,52-59).
However, only a limited number of studies contradicted the positive
association between telomere shortening and the aggravation of
skeletal pathology (60-63).
Initially, Kveiborg et al (60) investigated the LTL derived from 49
healthy women aged 20-26 and 48-85 years, and compared the value of
LTL to that of osteoporotic women of 52-81 years of age. They did
not observe any marked differences among groups (60). Moreover, that study had no
statistical power due to its small study group (<30
individuals), given that the analysis of the results is affected by
sample size (64). Similar results
were obtained from a longitudinal cohort study in which 2,750
elderly adults (aged 71-79 years) were enrolled (65). The results revealed no association
between LTL and BMD at the total hip or femoral neck, changes in
BMD over time, the presence of osteoporosis and the risk of
fractures in this population of elderly males and females (65). Apart from the negative association
between TL and osteoporosis, it was reported that the TL was
independent of the following factors: weight, fasting insulin, and
fasting glucose in elderly males and females (65). Since systemic inflammation is
regarded as the primary mechanism underlying the association
between telomere shortening and osteoporosis, and women with
osteoporosis are characterized by higher serum levels of C-reactive
protein (CRP) than the controls (16,65,66),
Sanders et al substantiated the inverse correlation of TL
with serum levels of CRP and IL-6(65). Compared to the results from the
study by Valdes et al (16),
the association of TL with BMD was sex- and age-dependent (16,65).
In another longitudinal cohort study of 1,867 elderly Chinese
adults (mean age, 72 years), Tang et al (61) found no statistically significant
association between TL and baseline BMD at the total hip and
femoral neck in both males and females (61).
Recently, Kirk et al (62) provided insight into the association
between LTL and the incidence of osteosarcopenia in a population of
20,400 elderly adults (mean age, 68 years; 47% females) from the UK
Biobank (62). Osteosarcopenia,
according to the World Health Organization (WHO) criteria, is
defined as follows: i) Bone density of the femoral neck T-score
≤-1; along with ii) a low appendicular lean mass as calculated from
the relation mass/height2 or low grip strength; and iii)
a slow walking pace (63). One of
the results of that study was that LTL was not associated either
with osteosarcopenia or the low bone density of the femoral neck.
The authors suggest it may be worth studying the same outcome
measures in an older population (>74 years) (62).
3. Challenges of studying telomere length in
osteoporosis
The inconsistency among studies presented herein may
be related to variability in the population of studies, the
methodology of studies and the size of the groups, establishing
different aspects of statistical analysis. Furthermore, a
discrepancy can arise through measuring TL in leukocytes and not in
bone cells like osteoblasts.
After adjusting for age, two epidemiological
research studies have reported a statistically significant
association between LTL and BMD, though measuring LTL via Southern
blot analysis (16,41). On the contrary, when LTL was
evaluated through quantitative polymerase chain reaction (qPCR), no
link was found between LTL and BMD (61,65).
It is generally accepted that the qPCR is more accurate and
reliable than Southern blot analysis, due to its high-throughput
nature and sensitivity (67);
however, this comparison appears to be underestimated as no report
comparing the two methodologies has been performed to date, at
least to the best of our knowledge (64).
Indeed, the high inconsistency of results may be
attributed to different methods used to measure TL. In this
direction, several reliable methods exist to evaluate TL with
various forms of sensitivity, either at the population or at the
single-cell level. Initially, studies used Southern blot analysis
to evaluate the length distribution of the terminal restriction
fragments in cells. Then, other studies performed qPCR to measure
the copy number of telomere repeats in homogenized cells, obtaining
a general image of the enrichment of telomere ends in cells
(14). Significant advances in the
telomere field have been noted, using telomere shortest length
assay (TeSLA) . In the former technique, the length of all
telomeres can be evaluated. In the TeSLA technique, PCR amplifies
telomeres at the single-chromosome level, and gel electrophoresis
then enables the evaluation of their length (14,68).
Recently, the most precise method to evaluate TL at a single-cell
level and at a single-chromosome level is quantitative fluorescence
in situ hybridization (qFISH) with the use of telomere
peptide nucleic acid (PNA) probe (14). Epidemiologists isolating peripheral
blood (PB) usually follow the latter. Among all these techniques,
only qFISH allows the measurement of individual TLs at single cell
level (14). Despite the
superiority of qFISH compared to other techniques, the qFISH can
recognize the telomeric repeats that the PNA probe defines.
However, DNA methylation-based methods have been
used to evaluate the TL (69). The
sensitivity and the reproducibility of the aforementioned
innovative methods regarding TL have been enhanced at a single-cell
level; however, they are not considered complete. The inconsistency
of DNA methylation-based methods arises from differences among
techniques (70). It should also be
taken into consideration that the reproducibility of TL is hampered
by other parameters involving the processing and storage of samples
(71). Accordingly, the differences
among studies can arise from the different statistical methods used
and intrinsic variations among the study groups of each study. The
mechanisms underlying age-related telomere shortening remain
incompletely understood, since the phenomenon is complex and
multifaceted. In addition to the above, the inter-individual
variability of samples themselves can complicate the TL
measurements to be precise (72),
rendering it very difficult to use telomere shortening as a single
routine biomarker in clinical practice (73). Those limitations need to be
addressed before establishing telomere shortening as a biomarker of
osteoporosis in clinical settings.
4. Role of telomere shortening in premature
aging disorders associated with osteoporosis
Clues of aging mechanisms arise from segmental
progeroid disorders in humans. Apart from the direct significance
of telomere shortening in osteoporosis, dyskeratosis congenita and
Werner syndrome (WS) constitute two genetic diseases that are
characterized by common signs of telomere shortening and premature
osteoporosis (74-76).
Notably, the standard features of the two diseases are premature
osteoporosis and telomere shortening owing to the loss of function
mutations in maintaining TL homeostasis (77). In the aforementioned genetic
disorders, osteoporosis is accomplished to a greater extent than
that achieved with natural aging, and the distribution of included
osteoporosis in WS appears at the limbs more than the axial
skeleton (78,79). In both diseases, increased systemic
inflammation and impaired immune system are implicated in their
pathophysiology, as represented by telomere shortening in their
lymphocytes (80).
Since Wrn helicase plays a crucial role in
sustaining TL (81), it is apparent
that dysfunctional telomeres are accelerated in double mutant
Wrn-/- telomerase RNA component (Terc)-/-
mice. Double-deficient mice have been shown to exhibit an abnormal
proliferation of their bone-forming cells, and they are
characterized by bone loss and age-related osteoporosis owing to
the disrupted differentiation of osteoblasts, accompanied by
advanced senescence observed in mesenchymal stem cells and the
normal differentiation of osteoclasts (82). As a result, the osteoporosis
observed in double-deficient mice appears to arise from damage in
the distribution and the functionality of osteoblasts (57), suggesting the significance of a
short TL in the risk of fractures. Consistent with the
aforementioned findings, the transfection of telomerase reverse
transcriptase (TERT) into osteoblasts that are implanted into
Wrn-/- Terc-/- mice has been shown to restore
bone homeostasis (57).
5. Conclusions and future perspectives
The present review discussed different results
regarding the usefulness of TL for identifying individuals with a
higher susceptibility for developing osteoporosis and also
highlighted the significance of TL in patients with osteoporosis.
Indeed, the present review proposes that TL shortening may be used
as a prognostic or predictive marker to evaluate the onset and
progression of osteoporosis. However, further large and
well-characterized cohorts with large sample sizes and proper
design are urgently required to gain new insight into the
mechanisms through which telomere shortening leads to the
development of osteoporosis and to encourage the use of telomere
shortening as a predictive tool for the onset and progression of
osteoporosis.
Thus, it remains questionable whether telomere
shortening can be a reliable biomarker of osteoporosis or
comprising a part of a composite panel of biomarkers for
osteoporosis. Moreover, a combination of panel markers can exert
greater predictive value in evaluating the progression of
osteoporosis than single measures. In parallel, telomere shortening
can serve as a predictive tool for assessing the risk of developing
osteoporosis.
Acknowledgements
Not applicable.
Funding
Funding: No funding was received.
Availability of data and materials
Not applicable.
Authors' contributions
All authors (FK, SP, EVakonaki, AB, MNT, PF, DAS,
SB, AT, PI, EH, DN and EVasileiadis) contributed to the conception
and design of the study. FK, SB, EV, AB, PF and MNT searched the
literature for articles to be included in the present review, which
were then examined and reviewed by SP, EH, SB, PI, DAS, DN, AT and
EVasileiadis. FK, EVasileiadis, SB, EV and AB drafted and wrote the
manuscript. MNT, DN, EH, SB, PI, DAS, DN, AT and SP revised the
manuscript. AB, SB and DN designed the figures. FK and EVakonaki
designed the table and SB revised the table. All authors have read
and approved the final version of the manuscript. Data
authentication is not applicable.
Ethics approval and consent to
participate
Not applicable.
Patient consent for publication
Not applicable.
Competing interests
DAS is the editor-in-chief for the journal, but had
no personal involvement in the reviewing process, or any influence
in terms of adjudicating on the final decision for this article.
The other authors declare that they have no competing
interests.
References
|
1
|
Shay JW: Telomeres and aging. Curr Opin
Cell Biol. 52:1–7. 2018.PubMed/NCBI View Article : Google Scholar
|
|
2
|
Gruber HJ, Semeraro MD, Renner W and
Herrmann M: Telomeres and Age-Related Diseases. Biomedicines.
9(1335)2021.PubMed/NCBI View Article : Google Scholar
|
|
3
|
Griffith JD, Comeau L, Rosenfield S,
Stansel RM, Bianchi A, Moss H and de Lange T: Mammalian Telomeres
End in a Large Duplex Loop. Cell. 97:503–514. 1999.PubMed/NCBI View Article : Google Scholar
|
|
4
|
Greider CW: Telomeres Do D-Loop-T-Loop.
Cell. 97:419–422. 1999.PubMed/NCBI View Article : Google Scholar
|
|
5
|
Turner KJ, Vasu V and Griffin DK: Telomere
biology and human phenotype. Cells. 8(73)2019.PubMed/NCBI View Article : Google Scholar
|
|
6
|
de Lange T: Shelterin: The protein complex
that shapes and safeguards human telomeres. Genes Dev.
19:2100–2110. 2005.PubMed/NCBI View Article : Google Scholar
|
|
7
|
Blasco MA: Telomere length, stem cells and
aging. Nat Chem Biol. 3:640–649. 2007.PubMed/NCBI View Article : Google Scholar
|
|
8
|
Jaskelioff M, Muller FL, Paik JH, Thomas
E, Jiang S, Adams AC, Sahin E, Kost-Alimova M, Protopopov A,
Cadiñanos J, et al: Telomerase reactivation reverses tissue
degeneration in aged telomerase-deficient mice. Nature.
469:102–106. 2011.PubMed/NCBI View Article : Google Scholar
|
|
9
|
Tsoukalas D, Fragkiadaki P, Docea AO,
Alegakis AK, Sarandi E, Vakonaki E, Salataj E, Kouvidi E, Nikitovic
D, Kovatsi L, et al: Association of nutraceutical supplements with
longer telomere length. Int J Mol Med. 44:218–226. 2019.PubMed/NCBI View Article : Google Scholar
|
|
10
|
Tsoukalas D, Buga AM, Docea AO, Sarandi E,
Mitrut R, Renieri E, Spandidos DA, Rogoveanu I, Cercelaru L,
Niculescu M, et al: Reversal of brain aging by targeting
telomerase: A nutraceutical approach. Int J Mol Med.
48(199)2021.PubMed/NCBI View Article : Google Scholar
|
|
11
|
López-Otín C, Blasco MA, Partridge L,
Serrano M and Kroemer G: The Hallmarks of Aging. Cell.
153:1194–1217. 2013.PubMed/NCBI View Article : Google Scholar
|
|
12
|
Lederman S: American Federation for Aging
Research. In: Encyclopedia of Gerontology and Population Aging. Gu
D and Dupre ME (eds). Springer, Cham, pp1–5, 2020.
|
|
13
|
Vaiserman A and Krasnienkov D: Telomere
length as a marker of biological age: State-of-the-Art, open
issues, and future perspectives. Front Genet.
11(630186)2021.PubMed/NCBI View Article : Google Scholar
|
|
14
|
Rossiello F, Jurk D, Passos JF and d'Adda
di Fagagna F: Telomere dysfunction in ageing and age-related
diseases. Nat Cell Biol. 24:135–147. 2022.PubMed/NCBI View Article : Google Scholar
|
|
15
|
Sanders JL and Newman AB: Telomere length
in epidemiology: A biomarker of aging, age-related disease, both,
or neither? Epidemiol Rev. 35:112–131. 2013.PubMed/NCBI View Article : Google Scholar
|
|
16
|
Valdes AM, Richards JB, Gardner JP,
Swaminathan R, Kimura M, Xiaobin L, Aviv A and Spector TD: Telomere
length in leukocytes correlates with bone mineral density and is
shorter in women with osteoporosis. Osteoporos Int. 18:1203–1210.
2007.PubMed/NCBI View Article : Google Scholar
|
|
17
|
Farr JN and Khosla S: Cellular senescence
in bone. Bone. 121:121–133. 2019.PubMed/NCBI View Article : Google Scholar
|
|
18
|
Sozen T, Ozisik L and Basaran NC: An
overview and management of osteoporosis. Eur J Rheumatol. 4:46–56.
2017.PubMed/NCBI View Article : Google Scholar
|
|
19
|
Chen X, Hu Y, Geng Z and Su J: The ‘Three
in One’ Bone repair strategy for osteoporotic fractures. Front
Endocrinol (Lausanne). 13(910602)2022.PubMed/NCBI View Article : Google Scholar
|
|
20
|
Dimai HP, Redlich K, Peretz M, Borgström
F, Siebert U and Mahlich J: Economic burden of osteoporotic
fractures in Austria. Health Econ Rev. 2(12)2012.PubMed/NCBI View Article : Google Scholar
|
|
21
|
Johnston CB and Dagar M: Osteoporosis in
older adults. Med Clin North Am. 104:873–884. 2020.PubMed/NCBI View Article : Google Scholar
|
|
22
|
Pignolo RJ, Law SF and Chandra A: Bone
aging, cellular senescence, and osteoporosis. JBMR Plus.
5(e10488)2021.PubMed/NCBI View Article : Google Scholar
|
|
23
|
Wright NC, Looker AC, Saag KG, Curtis JR,
Delzell ES, Randall S and Dawson-Hughes B: The recent prevalence of
osteoporosis and low bone mass in the united states based on bone
mineral density at the femoral neck or lumbar Spine. J Bone Miner
Res. 29:2520–2526. 2014.PubMed/NCBI View Article : Google Scholar
|
|
24
|
Watts NB, Bilezikian JP, Camacho PM,
Greenspan SL, Harris ST, Hodgson SF, Kleerekoper M, Luckey MM,
McClung MR, Pollack RP, et al: American association of clinical
endocrinologists medical guidelines for clinical practice for the
diagnosis and treatment of postmenopausal osteoporosis. Endocr
Pract. 16 Suppl 3(Suppl 3):S1–S37. 2010.PubMed/NCBI View Article : Google Scholar
|
|
25
|
Khosla S: Pathogenesis of Age-Related Bone
Loss in Humans. J Gerontol A Biol Sci Med Sci. 68:1226–1235.
2013.PubMed/NCBI View Article : Google Scholar
|
|
26
|
Chandra A, Lagnado AB, Farr JN, Monroe DG,
Park S, Hachfeld C, Tchkonia T, Kirkland JL, Khosla S, Passos JF
and Pignolo RJ: Targeted reduction of senescent cell burden
alleviates focal radiotherapy-related bone loss. J Bone Miner Res.
35:1119–1131. 2020.PubMed/NCBI View Article : Google Scholar
|
|
27
|
Chandra A and Rajawat J: Skeletal aging
and osteoporosis: Mechanisms and Therapeutics. Int J Mol Sci.
22(3553)2021.PubMed/NCBI View Article : Google Scholar
|
|
28
|
Farr JN, Rowsey JL, Eckhardt BA, Thicke
BS, Fraser DG, Tchkonia T, Kirkland JL, Monroe DG and Khosla S:
Independent roles of estrogen deficiency and cellular senescence in
the pathogenesis of osteoporosis: Evidence in young adult mice and
older humans. J Bone Miner Res. 34:1407–1418. 2019.PubMed/NCBI View Article : Google Scholar
|
|
29
|
Hachemi Y, Rapp AE, Picke AK, Weidinger G,
Ignatius A and Tuckermann J: Molecular mechanisms of
glucocorticoids on skeleton and bone regeneration after fracture. J
Mol Endocrinol. 61:R75–R90. 2018.PubMed/NCBI View Article : Google Scholar
|
|
30
|
Siddiqui JA and Partridge NC:
Physiological bone remodeling: Systemic regulation and growth
factor involvement. Physiology (Bethesda). 31:233–245.
2016.PubMed/NCBI View Article : Google Scholar
|
|
31
|
Kenkre JS and Bassett J: The bone
remodelling cycle. Ann Clin Biochem. 55:308–327. 2018.PubMed/NCBI View Article : Google Scholar
|
|
32
|
Rauner M, Taipaleenmäki H, Tsourdi E and
Winter EM: Osteoporosis treatment with anti-sclerostin
antibodies-mechanisms of action and clinical application. J Clin
Med. 10(787)2021.PubMed/NCBI View Article : Google Scholar
|
|
33
|
Fabre S, Funck-Brentano T and Cohen-Solal
M: Anti-Sclerostin antibodies in osteoporosis and other bone
diseases. J Clin Med. 9(3439)2020.PubMed/NCBI View Article : Google Scholar
|
|
34
|
Shakeri A and Adanty C: Romosozumab
(sclerostin monoclonal antibody) for the treatment of osteoporosis
in postmenopausal women: A review. J Popul Ther Clin Pharmacol.
27:e25–e31. 2020.PubMed/NCBI View Article : Google Scholar
|
|
35
|
Ensrud KE and Crandall CJ: Osteoporosis.
Ann Intern Med. 167:ITC17–ITC32. 2017.PubMed/NCBI View Article : Google Scholar
|
|
36
|
Mather KA, Jorm AF, Parslow RA and
Christensen H: Is telomere length a biomarker of aging? A review. J
Gerontol A Biol Sci Med Sci. 66A:202–213. 2011.PubMed/NCBI View Article : Google Scholar
|
|
37
|
Der G, Batty GD, Benzeval M, Deary IJ,
Green MJ, McGlynn L, McIntyre A, Robertson T and Shiels PG: Is
telomere length a biomarker for aging: Cross-Sectional evidence
from the west of scotland? PLoS One. 7(e45166)2012.PubMed/NCBI View Article : Google Scholar
|
|
38
|
Simons MJ: Questioning causal involvement
of telomeres in aging. Ageing Res Rev. 24(Pt B):191–196.
2015.PubMed/NCBI View Article : Google Scholar
|
|
39
|
Gorenjak V, Akbar S, Stathopoulou MG and
Visvikis-Siest S: The future of telomere length in personalized
medicine. Front Biosci (Landmark Ed). 23:1628–1654. 2018.PubMed/NCBI View Article : Google Scholar
|
|
40
|
Fasching CL: Telomere length measurement
as a clinical biomarker of aging and disease. Crit Rev Clin Lab
Sci. 55:443–465. 2018.PubMed/NCBI View Article : Google Scholar
|
|
41
|
Bekaert S, Van Pottelbergh I, De Meyer T,
Zmierczak H, Kaufman JM, Van Oostveldt P and Goemaere S: Telomere
length versus hormonal and bone mineral status in healthy elderly
men. Mech Ageing Dev. 126:1115–1122. 2005.PubMed/NCBI View Article : Google Scholar
|
|
42
|
Cawthon RM, Smith KR, O'Brien E,
Sivatchenko A and Kerber RA: Association between telomere length in
blood and mortality in people aged 60 years or older. Lancet.
361:393–395. 2003.PubMed/NCBI View Article : Google Scholar
|
|
43
|
Kassem M and Marie PJ:
Senescence-associated intrinsic mechanisms of osteoblast
dysfunctions: Age-related mechanisms of osteoblast dysfunctions.
Aging Cell. 10:191–197. 2011.PubMed/NCBI View Article : Google Scholar
|
|
44
|
Wang D and Wang H: Cellular Senescence in
Bone. In: Physiology. Heshmati H (ed). IntechOpen, vol. 15 M,
2022.
|
|
45
|
Tamayo M, Mosquera A, Rego JI,
Fernández-Sueiro JL, Blanco FJ and Fernández JL: Differing patterns
of peripheral blood leukocyte telomere length in rheumatologic
diseases. Mutat Res. 683:68–73. 2010.PubMed/NCBI View Article : Google Scholar
|
|
46
|
Nielsen BR, Linneberg A, Bendix L, Harboe
M, Christensen K and Schwarz P: Association between leukocyte
telomere length and bone mineral density in women 25-93 years of
age. Exp Gerontol. 66:25–31. 2015.PubMed/NCBI View Article : Google Scholar
|
|
47
|
Smith RL, de Boer R, Brul S, Budovskaya Y
and van Spek H: Premature and accelerated aging: HIV or HAART?
Front Genet. 3(328)2013.PubMed/NCBI View Article : Google Scholar
|
|
48
|
Deeks SG: HIV infection, inflammation,
immunosenescence, and aging. Annu Rev Med. 62:141–155.
2011.PubMed/NCBI View Article : Google Scholar
|
|
49
|
Kalyan S, Pick N, Mai A, Murray MCM,
Kidson K, Chu J, Albert AYK, Côté HCF, Maan EJ, Goshtasebi A, et
al: Premature spinal bone loss in women living with HIV is
associated with shorter leukocyte telomere length. Int J Environ
Res Public Health. 15(1018)2018.PubMed/NCBI View Article : Google Scholar
|
|
50
|
Brown TT and Qaqish RB: Antiretroviral
therapy and the prevalence of osteopenia and osteoporosis: A
meta-analytic review. AIDS. 20:2165–2174. 2006.PubMed/NCBI View Article : Google Scholar
|
|
51
|
Tao L, Huang Q, Yang R, Dai Y, Zeng Y, Li
C, Li X, Zeng J and Wang Q: The age modification to leukocyte
telomere length effect on bone mineral density and osteoporosis
among Chinese elderly women. J Bone Miner Metab. 37:1004–1012.
2019.PubMed/NCBI View Article : Google Scholar
|
|
52
|
Fragkiadaki P, Nikitovic D, Kalliantasi K,
Sarandi E, Thanasoula M, Stivaktakis PD, Nepka C, Spandidos DA,
Tosounidis T and Tsatsakis A: Telomere length and telomerase
activity in osteoporosis and osteoarthritis. Exp Ther Med.
19:1626–1632. 2020.PubMed/NCBI View Article : Google Scholar
|
|
53
|
Curtis EM, Codd V, Nelson C, D'Angelo S,
Wang Q, Allara E, Kaptoge S, Matthews PM, Tobias JH, Danesh J, et
al: Telomere length and risk of incident fracture and arthroplasty:
Findings from UK Biobank. J Bone Miner Res. 37:1997–2004.
2022.PubMed/NCBI View Article : Google Scholar
|
|
54
|
Farr JN, Fraser DG, Wang H, Jaehn K,
Ogrodnik MB, Weivoda MM, Drake MT, Tchkonia T, LeBrasseur NK,
Kirkland JL, et al: Identification of senescent cells in the bone
microenvironment. J Bone Miner Res. 31:1920–1929. 2016.PubMed/NCBI View Article : Google Scholar
|
|
55
|
Haapanen MJ, Perälä MM, Salonen MK,
Guzzardi MA, Iozzo P, Kajantie E, Rantanen T, Simonen M,
Pohjolainen P, Eriksson JG and von Bonsdorff MB: Telomere length
and frailty: The Helsinki birth cohort study. J Am Med Dir Assoc.
19:658–662. 2018.PubMed/NCBI View Article : Google Scholar
|
|
56
|
Hong Z, Lin X, Zhou Y, Zheng G, Liao X,
Wei Q, Zhang Z and Liang J: Lean body mass but not body fat mass is
related with leukocyte telomere length in children. Int J Obes
(Lond). 47:67–74. 2023.PubMed/NCBI View Article : Google Scholar
|
|
57
|
Pignolo RJ, Suda RK, McMillan EA, Shen J,
Lee SH, Choi Y, Wright AC and Johnson FB: Defects in telomere
maintenance molecules impair osteoblast differentiation and promote
osteoporosis. Aging Cell. 7:23–31. 2008.PubMed/NCBI View Article : Google Scholar
|
|
58
|
Brennan TA, Egan KP, Lindborg CM, Chen Q,
Sweetwyne MT, Hankenson KD, Xie SX, Johnson FB and Pignolo RJ:
Mouse models of telomere dysfunction phenocopy skeletal changes
found in human age-related osteoporosis. Dis Model Mech. 7:583–592.
2014.PubMed/NCBI View Article : Google Scholar
|
|
59
|
Singh L, Brennan TA, Kim JH, Egan KP,
McMillan EA, Chen Q, Hankenson KD, Zhang Y, Emerson SG, Johnson FB
and Pignolo RJ: Long-Term functional engraftment of mesenchymal
progenitor cells in a mouse model of accelerated aging. Stem Cells.
31:607–611. 2013.PubMed/NCBI View Article : Google Scholar
|
|
60
|
Kveiborg M, Kassem M, Langdahl B, Eriksen
EF, Clark BF and Rattan SI: Telomere shortening during aging of
human osteoblasts in vitro and leukocytes in vivo: Lack of
excessive telomere loss in osteoporotic patients. Mech Ageing Dev.
106:261–271. 1999.PubMed/NCBI View Article : Google Scholar
|
|
61
|
Tang NL, Woo J, Suen EW, Liao CD, Leung JC
and Leung PC: The effect of telomere length, a marker of biological
aging, on bone mineral density in elderly population. Osteoporos
Int. 21:89–97. 2010.PubMed/NCBI View Article : Google Scholar
|
|
62
|
Kirk B, Kuo CL, Xiang M and Duque G:
Associations between leukocyte telomere length and osteosarcopenia
in 20,400 adults aged 60 years and over: Data from the UK Biobank.
Bone. 161(116425)2022.PubMed/NCBI View Article : Google Scholar
|
|
63
|
Sepúlveda-Loyola W, Phu S, Bani Hassan E,
Brennan-Olsen SL, Zanker J, Vogrin S, Conzade R, Kirk B, Al Saedi
A, Probst V and Duque G: The joint occurrence of osteoporosis and
sarcopenia (Osteosarcopenia): Definitions and Characteristics. J Am
Med Dir Assoc. 21:220–225. 2020.PubMed/NCBI View Article : Google Scholar
|
|
64
|
Aviv A, Valdes AM and Spector TD: Human
telomere biology: Pitfalls of moving from the laboratory to
epidemiology. Int J Epidemiol. 35:1424–1429. 2006.PubMed/NCBI View Article : Google Scholar
|
|
65
|
Sanders JL, Cauley JA, Boudreau RM, Zmuda
JM, Strotmeyer ES, Opresko PL, Hsueh WC, Cawthon RM, Li R, Harris
TB, et al: Leukocyte Telomere length is not associated with BMD,
osteoporosis, or fracture in older adults: Results from the health,
aging and body composition study. J Bone Miner Res. 24:1531–1536.
2009.PubMed/NCBI View Article : Google Scholar
|
|
66
|
Saeed H, Abdallah BM, Ditzel N,
Catala-Lehnen P, Qiu W, Amling M and Kassem M: Telomerase-deficient
mice exhibit bone loss owing to defects in osteoblasts and
increased osteoclastogenesis by inflammatory microenvironment. J
Bone Miner Res. 26:1494–1505. 2011.PubMed/NCBI View Article : Google Scholar
|
|
67
|
Baird D: New developments in telomere
length analysis. Exp Gerontol. 40:363–368. 2005.PubMed/NCBI View Article : Google Scholar
|
|
68
|
Lai TP, Zhang N, Noh J, Mender I, Tedone
E, Huang E, Wright WE, Danuser G and Shay JW: A method for
measuring the distribution of the shortest telomeres in cells and
tissues. Nat Commun. 8(1356)2017.PubMed/NCBI View Article : Google Scholar
|
|
69
|
Lu AT, Seeboth A, Tsai PC, Sun D, Quach A,
Reiner AP, Kooperberg C, Ferrucci L, Hou L, Baccarelli AA, et al:
DNA methylation-based estimator of telomere length. Aging (Albany
NY). 11:5895–5923. 2019.PubMed/NCBI View Article : Google Scholar
|
|
70
|
Dagnall CL, Hicks B, Teshome K, Hutchinson
AA, Gadalla SM, Khincha PP, Yeager M and Savage SA: Effect of
pre-analytic variables on the reproducibility of qPCR relative
telomere length measurement. PLoS One. 12(e0184098)2017.PubMed/NCBI View Article : Google Scholar
|
|
71
|
Lin J, Smith DL, Esteves K and Drury S:
Telomere length measurement by qPCR-Summary of critical factors and
recommendations for assay design. Psychoneuroendocrinology.
99:271–278. 2019.PubMed/NCBI View Article : Google Scholar
|
|
72
|
Bodelon C, Savage SA and Gadalla SM:
Telomeres in Molecular Epidemiology Studies. Prog Mol Biol Transl
Sci. 125:113–131. 2014.PubMed/NCBI View Article : Google Scholar
|
|
73
|
Semeraro MD, Smith C, Kaiser M, Levinger
I, Duque G, Gruber HJ and Herrmann M: Physical activity, a
modulator of aging through effects on telomere biology. Aging
(Albany NY). 12:13803–13823. 2020.PubMed/NCBI View Article : Google Scholar
|
|
74
|
Kirwan M and Dokal I: Dyskeratosis
congenita, stem cells and telomeres. Biochim Biophys Acta.
1792:371–379. 2009.PubMed/NCBI View Article : Google Scholar
|
|
75
|
McGrath JA: Dyskeratosis congenita: New
clinical and molecular insights into ribosome function. Lancet.
353:1204–1205. 1999.PubMed/NCBI View Article : Google Scholar
|
|
76
|
Tsuge K and Shimamoto A: Research on
werner syndrome: Trends from past to present and future prospects.
Genes (Basel). 13(1802)2022.PubMed/NCBI View Article : Google Scholar
|
|
77
|
Du X, Shen J, Kugan N, Furth EE, Lombard
DB, Cheung C, Pak S, Luo G, Pignolo RJ, DePinho RA, et al: Telomere
shortening exposes functions for the mouse werner and bloom
syndrome genes. Mol Cell Biol. 24:8437–8446. 2004.PubMed/NCBI View Article : Google Scholar
|
|
78
|
Hofer AC, Tran RT, Aziz OZ, Wright W,
Novelli G, Shay J and Lewis M: Shared phenotypes among segmental
progeroid syndromes suggest underlying pathways of aging. J
Gerontol A Biol Sci Med Sci. 60:10–20. 2005.PubMed/NCBI View Article : Google Scholar
|
|
79
|
Mason PJ, Wilson DB and Bessler M:
Dyskeratosis Congenita-A disease of dysfunctional telomere
maintenance. Curr Mol Med. 5:159–170. 2005.PubMed/NCBI View Article : Google Scholar
|
|
80
|
Salminen A, Suuronen T, Huuskonen J and
Kaarniranta K: NEMO shuttle: A link between DNA damage and
NF-kappaB activation in progeroid syndromes? Biochem Biophys Res
Commun. 367:715–718. 2008.PubMed/NCBI View Article : Google Scholar
|
|
81
|
Crabbe L, Verdun RE, Haggblom CI and
Karlseder J: Defective telomere lagging strand synthesis in cells
lacking WRN Helicase activity. Science. 306:1951–1953.
2004.PubMed/NCBI View Article : Google Scholar
|
|
82
|
Majors AK, Boehm CA, Nitto H, Midura RJ
and Muschler GF: Characterization of human bone marrow stromal
cells with respect to osteoblastic differentiation. J Orthop Res.
15:546–557. 1997.PubMed/NCBI View Article : Google Scholar
|