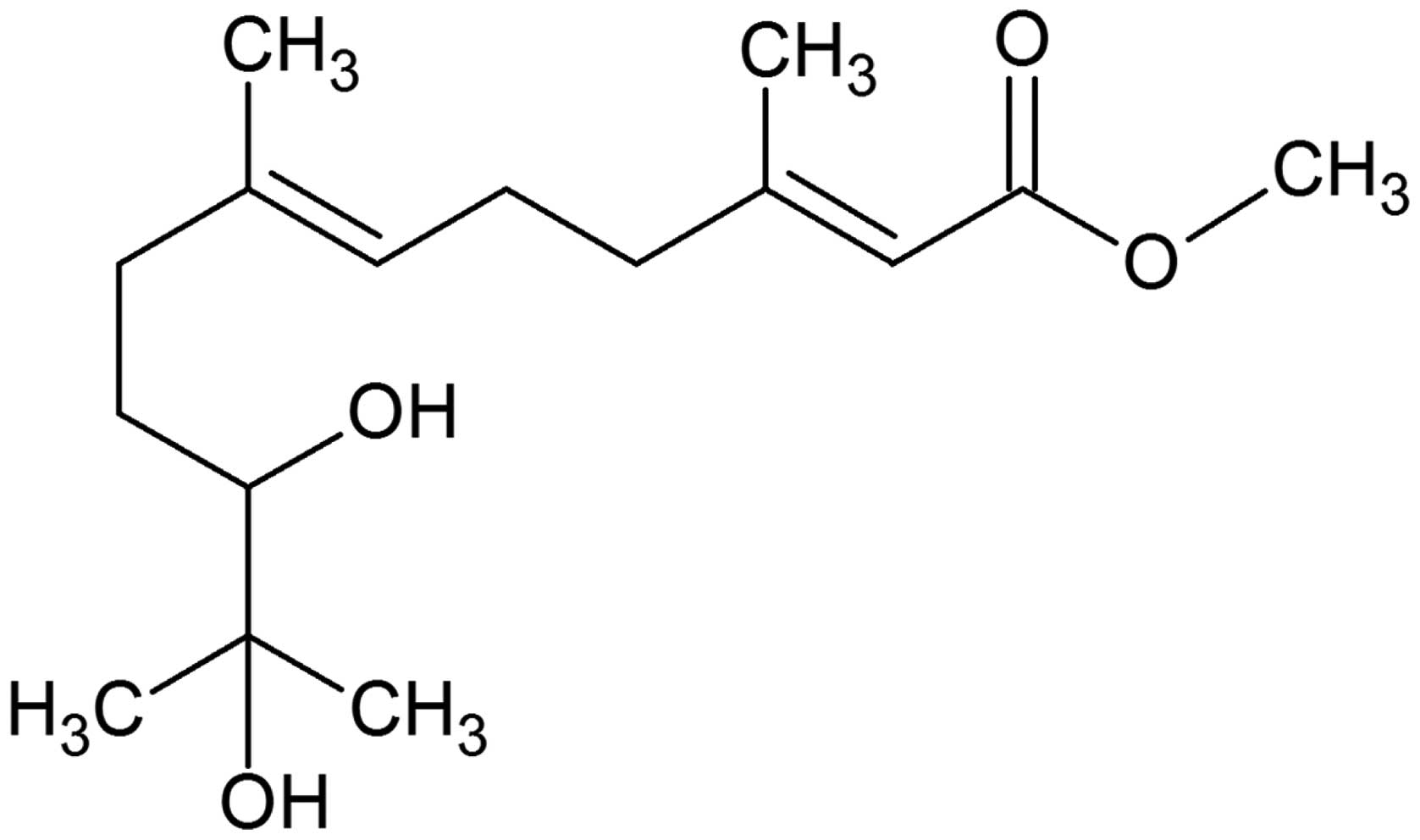
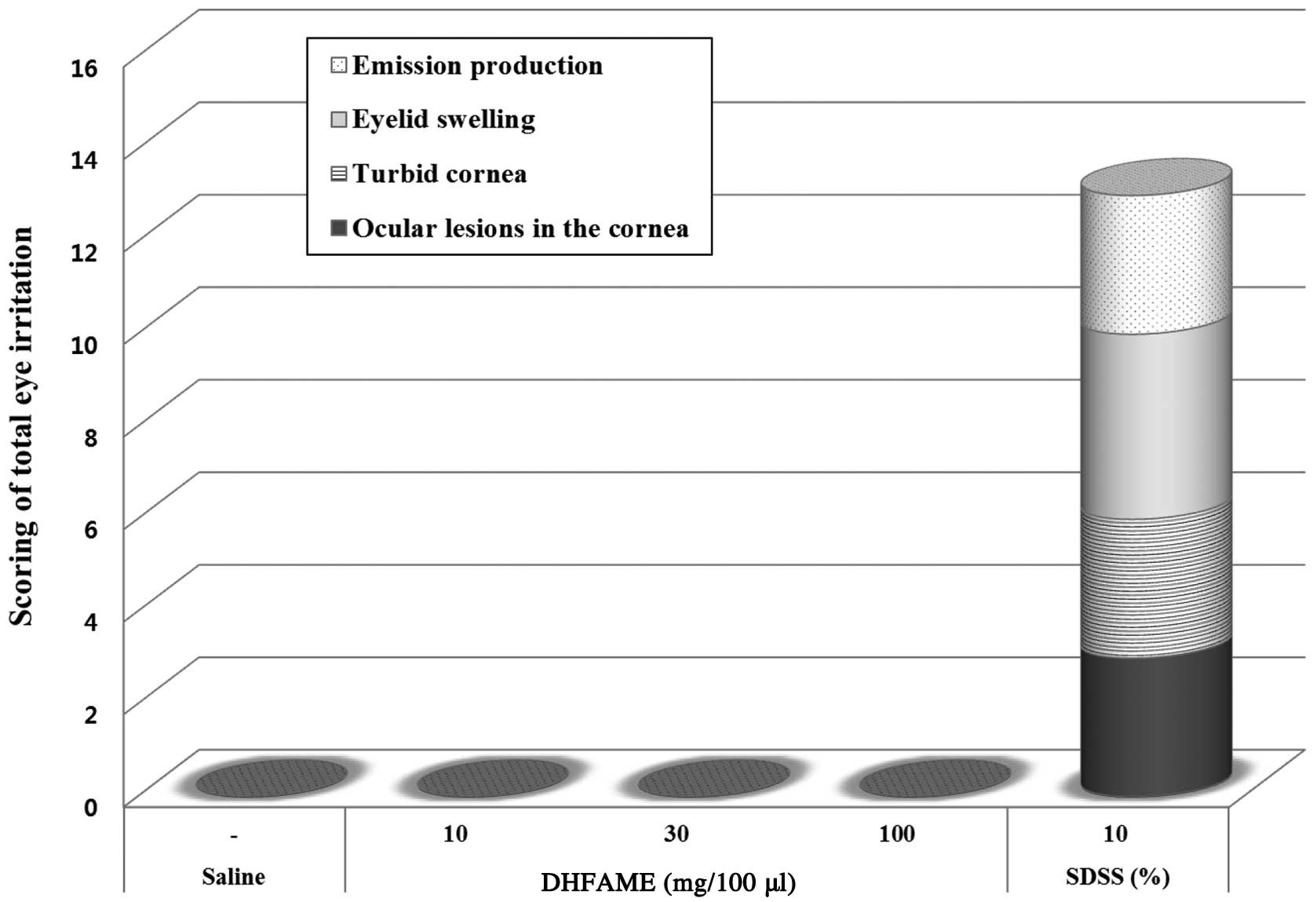
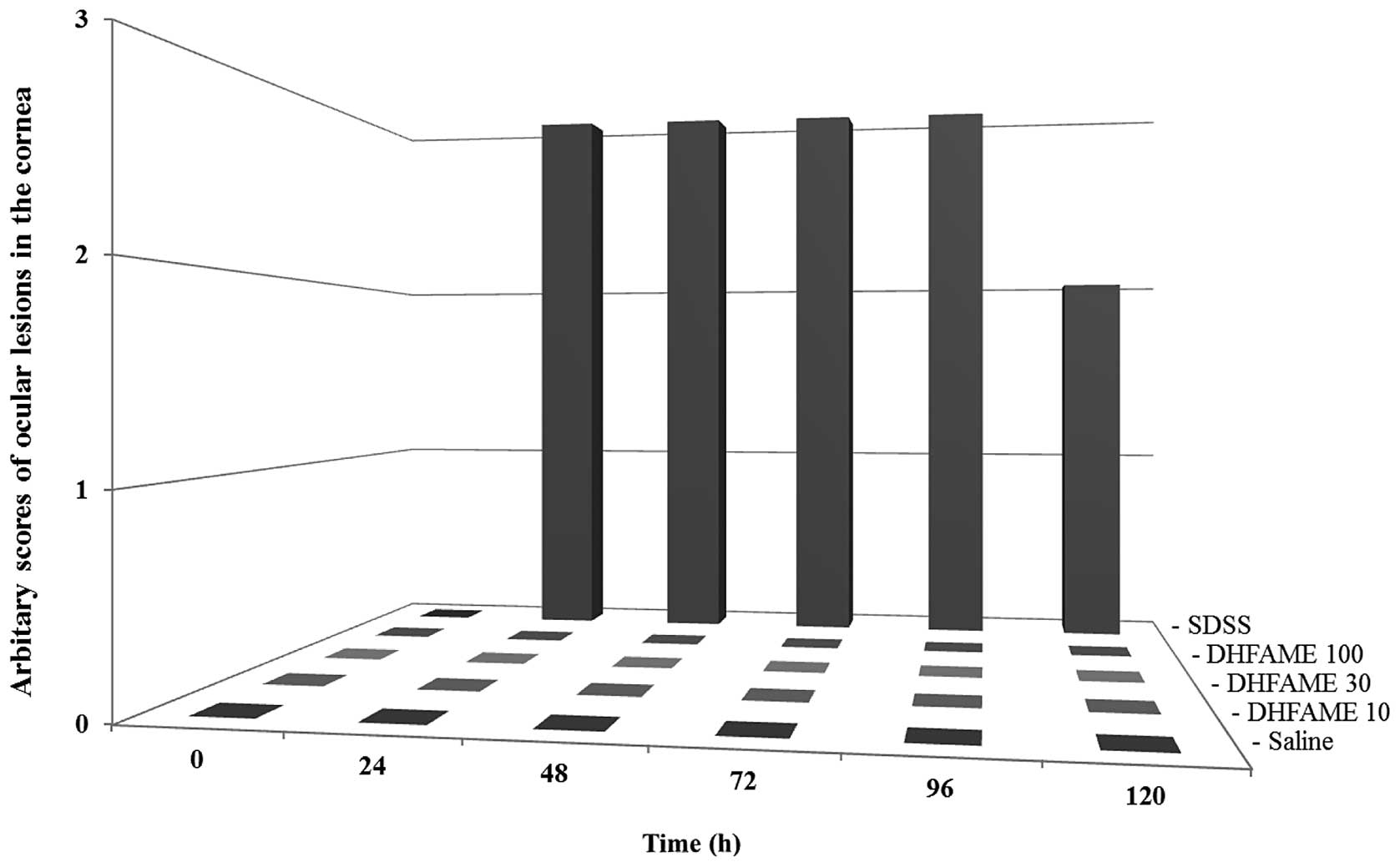
|
1.
|
Figueira L, Pinheiro D, Moreira R, Pinto
E, Simões J, Camisa E, Torrão L, Palmares J and Falcão-Reis F:
Beauveria bassiana keratitis in bullous keratopathy:
antifungal sensitivity testing and management. Eur J Ophthalmol.
22:814–818. 2012.
|
|
2.
|
Pedrini N, Crespo R and Juárez MP:
Biochemistry of insect epicuticle degradation by entomopathogenic
fungi. Comp Biochem Physiol C Toxicol Pharmacol. 146:124–137. 2007.
View Article : Google Scholar : PubMed/NCBI
|
|
3.
|
Zhou X, Gong Z, Su Y, Lin J and Tang K:
Cordyceps fungi: natural products, pharmacological functions and
developmental products. J Pharm Pharmacol. 61:279–291. 2009.
View Article : Google Scholar : PubMed/NCBI
|
|
4.
|
Tomoda H and Doi T: Discovery and
combinatorial synthesis of fungal metabolites beauveriolides, novel
antiatherosclerotic agents. Acc Chem Res. 41:32–39. 2008.PubMed/NCBI
|
|
5.
|
Liu L, Liu Y, Li J, Du G and Chen J:
Microbial production of hyaluronic acid: current state, challenges,
and perspectives. Microb Cell Fact. 10:992011. View Article : Google Scholar : PubMed/NCBI
|
|
6.
|
Draize JH: Appraisal of the Safety of
Chemicals in Foods, drugs and cosmetics. Association of Food and
Drug Officials of the United States; CA: pp. 46–59. 1959
|
|
7.
|
Nigam PK: Adverse reactions to cosmetics
and methods of testing. Indian J Dermatol Venereol Leprol.
75:10–18. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
8.
|
Tavaszi J, Budai P, Pálovics A and
Kismányoki A: An alternative test battery in detecting ocular
irritancy of agrochemicals. Commun Agric Appl Biol Sci. 73:891–895.
2008.PubMed/NCBI
|
|
9.
|
Scott L, Eskes C, Hoffmann S, Adriaens E,
et al: A proposed eye irritation testing strategy to reduce and
replace in vivo studies using bottom-up and top-down approaches.
Toxicol In vitro. 24:1–9. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
10.
|
Osborne R, Perkins MA and Roberts DA:
Development and intralaboratory evaluation of an in vitro human
cell-based test to aid ocular irritancy assessments. Fundam Appl
Toxicol. 28:139–153. 1995. View Article : Google Scholar : PubMed/NCBI
|
|
11.
|
Zimmermann M: Ethical guidelines for
investigations of experimental pain in conscious animals. Pain.
16:109–110. 1983. View Article : Google Scholar : PubMed/NCBI
|
|
12.
|
Nam SH, Yoon CS, Jeon JY, Lee SH, Lee KG,
Yeo JH and Hwang JS: Composition exhibiting melanin-inhibiting
activity. KR Patent 10-1239631. Filed March 28, 2011; issued
February 27, 2013.
|
|
13.
|
Draize JH, Woodard G and Calvery HO:
Methods for the study of irritation and toxicity of substances
applied topically to the skin and mucous membranes. J Pharmacol Exp
Ther. 82:377–390. 1944.
|
|
14.
|
Aoshima H, Saitoh Y, Ito S, Yamana S and
Miwa N: Safety evaluation of highly purified fullernenes (HPFs):
based on screening of eye and skin damage. J Toxicol Sci.
34:555–562. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
15.
|
Son HU, Yoon EK, Cha YS, Kim MA, Shin YK,
Kim JM, Choi YH and Lee SH: Comparison of the toxicity of aqueous
and ethanol fraction of Angelica keiskei leaf using the eye
irritancy test. Exp Ther Med. 4:820–824. 2012.PubMed/NCBI
|
|
16.
|
Korting HC, Herzinger T, Hartinger A,
Kerscher M, Angerpointner T and Maibach HI: Discrimination of the
irritancy potential of surfactants in vitro by two cytotoxicity
assays using normal human keratinocytes, HaCaT cells and 3T3 mouse
fibro-blasts: correlation with in vivo data from a soap chamber
assay. J Dermatol Sci. 7:119–129. 1994. View Article : Google Scholar
|
|
17.
|
Tardiff RG, Hubner RP and Graves CG:
Harmonization of thresholds for primary skin irritation from
results of human repeated insult patch tests and laboratory animal
skin irritation tests. J Appl Toxicol. 23:279–281. 2003. View Article : Google Scholar : PubMed/NCBI
|
|
18.
|
Basketter DA and Kimber I: Skin
irritation, false positives and the local lymph node assay: a
guideline issue? Regul Toxicol Pharmacol. 61:137–140. 2011.
View Article : Google Scholar : PubMed/NCBI
|