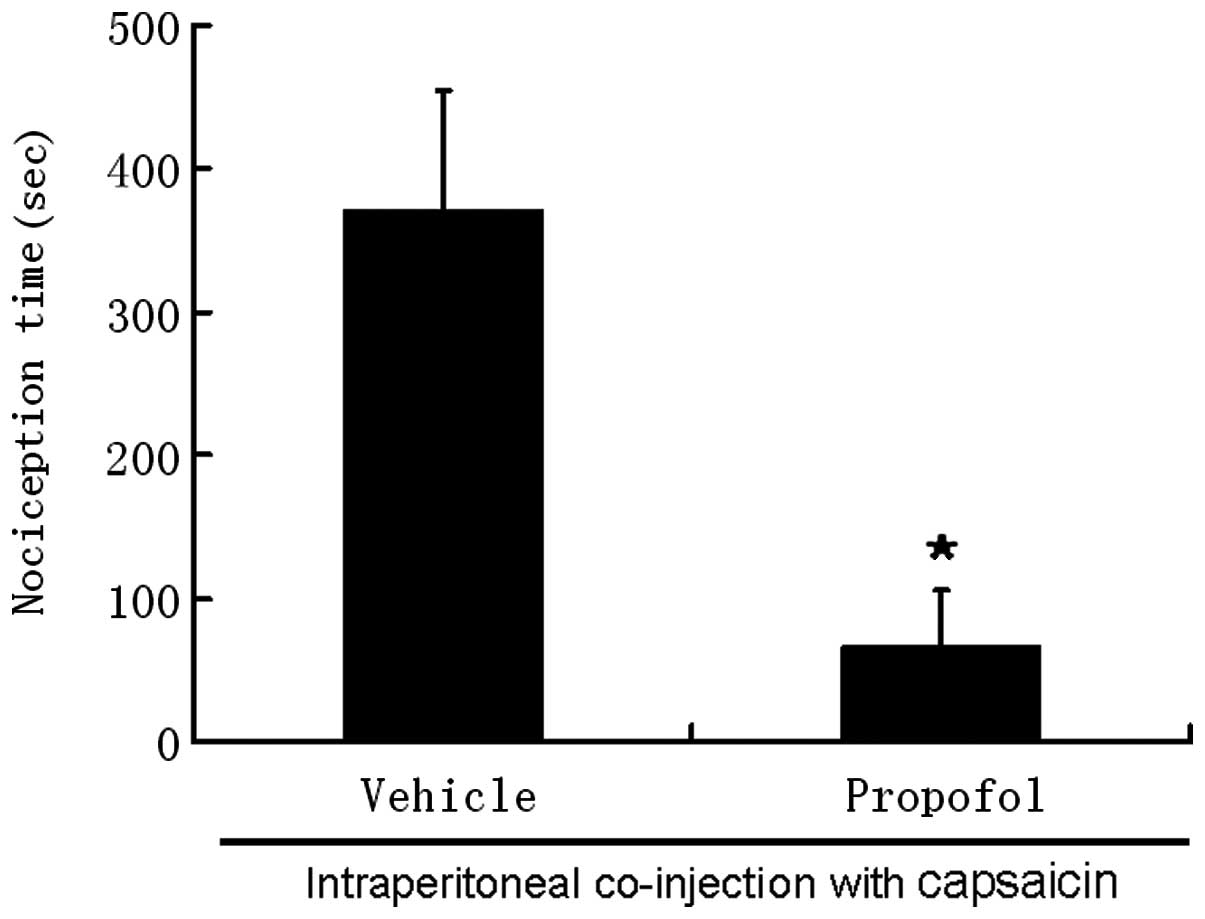
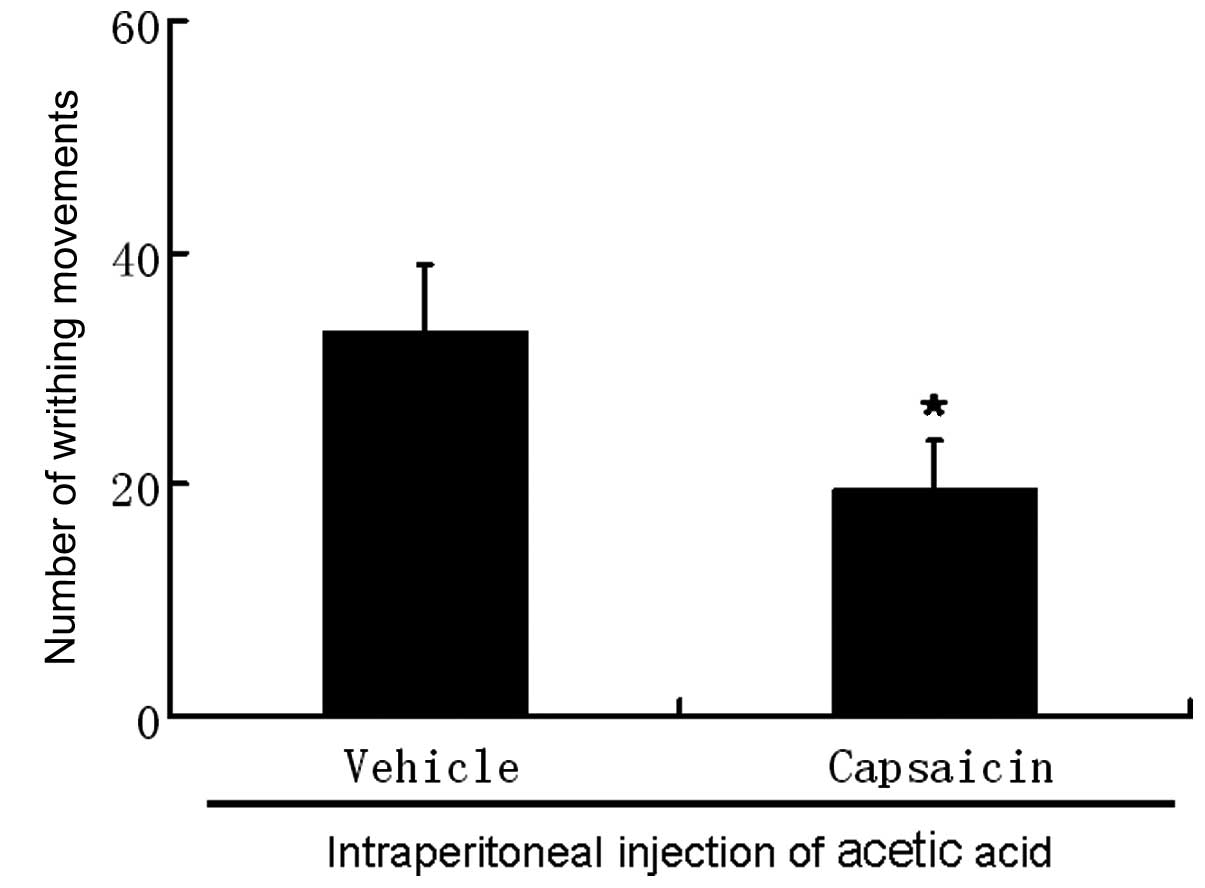
|
1
|
Smith I, White PF, Nathanson M and
Gouldson R: Propofol. An update on its clinical use.
Anesthesiology. 81:1005–1043. 1994. View Article : Google Scholar : PubMed/NCBI
|
|
2
|
Guindon J, LoVerme J, Piomelli D and
Beaulieu P: The antinociceptive effects of local injections of
propofol in rats are mediated in part by cannabinoid CB1 and CB2
receptors. Anesth Analg. 104:1563–1569. 2007. View Article : Google Scholar : PubMed/NCBI
|
|
3
|
Nadeson R and Goodchild CS:
Antinociceptive properties of propofol: involvement of spinal cord
gamma-aminobutyric acid(A) receptors. J Pharmacol Exp Ther.
282:1181–1186. 1997.PubMed/NCBI
|
|
4
|
Xu AJ, Duan SM and Zeng YM: Effects of
intrathecal NMDA and AMPA receptors agonists or antagonists on
antinociception of propofol. Acta Pharmacol Sin. 25:9–14.
2004.PubMed/NCBI
|
|
5
|
Antognini JF, Wang XW, Piercy M and
Carstens E: Propofol directly depresses lumbar dorsal horn neuronal
responses to noxious stimulation in goats. Can J Anaesth.
47:273–279. 2000. View Article : Google Scholar : PubMed/NCBI
|
|
6
|
Sun YY, Li KC and Chen J: Evidence for
peripherally antinociceptive action of propofol in rats: behavioral
and spinal neuronal responses to subcutaneous bee venom. Brain Res.
1043:231–235. 2005. View Article : Google Scholar : PubMed/NCBI
|
|
7
|
Neukom L, Vastani N, Seifert B, Spahn DR
and Maurer K: Propofol decreases the axonal excitability in rat
primary sensory afferents. Life Sci. 90:343–350. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
8
|
Caterina MJ and Julius D: The vanilloid
receptor: a molecular gateway to the pain pathway. Annu Rev
Neurosci. 24:487–517. 2001. View Article : Google Scholar : PubMed/NCBI
|
|
9
|
Caterina MJ, Leffler A, Malmberg AB,
Martin WJ, Trafton J, Petersen-Zeitz KR, Koltzenburg M, Basbaum AI
and Julius D: Impaired nociception and pain sensation in mice
lacking the capsaicin receptor. Science. 288:306–313. 2000.
View Article : Google Scholar : PubMed/NCBI
|
|
10
|
Davis JB, Gray J, Gunthorpe MJ, Hatcher
JP, Davey PT, Overend P, Harries MH, Latcham J, Clapham C, et al:
Vanilloid receptor-1 is essential for inflammatory thermal
hyperalgesia. Nature. 405:183–187. 2000. View Article : Google Scholar : PubMed/NCBI
|
|
11
|
Urban L, Campbell EA, Panesar M, Patel S,
Chaudhry N, Kane S, Buchheit K, Sandells B and James IF: In vivo
pharmacology of SDZ 249–665, a novel, non-pungent capsaicin
analogue. Pain. 89:65–74. 2000.
|
|
12
|
Rigoni M, Trevisani M, Gazzieri D,
Nadaletto R, Tognetto M, Creminon C, Davis JB, Campi B, et al:
Neurogenic responses mediated by vanilloid receptor-1 (TRPV1) are
blocked by the high affinity antagonist, iodo-resiniferatoxin. Br J
Pharmacol. 138:977–985. 2003. View Article : Google Scholar : PubMed/NCBI
|
|
13
|
Ikeda Y, Ueno A, Naraba H and Oh-ishi S:
Involvement of vanilloid receptor VR1 and prostanoids in the
acid-induced writhing responses of mice. Life Sci. 69:2911–2919.
2001. View Article : Google Scholar : PubMed/NCBI
|
|
14
|
Yamamoto H, Kawamata T, Ninomiya T, Omote
K and Namiki A: Endothelin-1 enhances capsaicin-evoked
intracellular Ca2+ response via activation of endothelin
a receptor in a protein kinase Cepsilon-dependent manner in dorsal
root ganglion neurons. Neuroscience. 137:949–960. 2006.PubMed/NCBI
|
|
15
|
Moriyama T, Higashi T, Togashi K, Iida T,
Segi E, Sugimoto Y, Tominaga T, Narumiya S and Tominaga M:
Sensitization of TRPV1 by EP1 and IP reveals peripheral nociceptive
mechanism of prostaglandins. Mol Pain. 1:32005. View Article : Google Scholar : PubMed/NCBI
|
|
16
|
Chuang HH, Prescott ED, Kong H, Shields S,
Jordt SE, Basbaum AI, Chao MV and Julius D: Bradykinin and nerve
growth factor release the capsaicin receptor from PtdIns(4,5)
P2-mediated inhibition. Nature. 411:957–962. 2001. View Article : Google Scholar : PubMed/NCBI
|
|
17
|
Wickley PJ, Yuge R, Russell MS, Zhang H,
Sulak MA and Damron DS: Propofol modulates agonist-induced
transient receptor potential vanilloid subtype-1 receptor
desensitization via a protein kinase Cepsilon-dependent pathway in
mouse dorsal root ganglion sensory neurons. Anesthesiology.
113:833–844. 2010. View Article : Google Scholar
|
|
18
|
Zhang H, Wickley PJ, Sinha S, Bratz IN and
Damron DS: Propofol restores transient receptor potential vanilloid
receptor subtype-1 sensitivity via activation of transient receptor
potential ankyrin receptor subtype-1 in sensory neurons.
Anesthesiology. 114:1169–1179. 2011. View Article : Google Scholar
|
|
19
|
Wang X, Miyares RL and Ahern GP:
Oleoylethanolamide excites vagal sensory neurones, induces visceral
pain and reduces short-term food intake in mice via capsaicin
receptor TRPV1. J Physiol. 564:541–547. 2005. View Article : Google Scholar
|
|
20
|
Rashid MH, Inoue M, Kondo S, Kawashima T,
Bakoshi S and Ueda H: Novel expression of vanilloid receptor 1 on
capsaicin-insensitive fibers accounts for the analgesic effect of
capsaicin cream in neuropathic pain. J Pharmacol Exp Ther.
304:940–948. 2003. View Article : Google Scholar : PubMed/NCBI
|
|
21
|
Szallasi A, Cortright DN, Blum CA and Eid
SR: The vanilloid receptor TRPV1: 10 years from channel cloning to
antagonist proof-of-concept. Nat Rev Drug Discov. 6:357–372.
2007.PubMed/NCBI
|
|
22
|
Nakane M and Iwama H: A potential
mechanism of propofol-induced pain on injection based on studies
using nafamostat mesilate. Br J Anaesth. 83:397–404. 1999.
View Article : Google Scholar : PubMed/NCBI
|
|
23
|
Ando R and Watanabe C: Characteristics of
propofol-evoked vascular pain in anaesthetized rats. Br J Anaesth.
95:384–392. 2005. View Article : Google Scholar : PubMed/NCBI
|
|
24
|
Cayla C, Labuz D, Machelska H, Bader M,
Schäfer M and Stein C: Impaired nociception and peripheral opioid
antinociception in mice lacking both kinin B1 and B2 receptors.
Anesthesiology. 116:448–457. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
25
|
Wang QY, Cao JL, Zeng YM and Dai TJ: GABAA
receptor partially mediated propofol-induced hyperalgesia at
superspinal level and analgesia at spinal cord level in rats. Acta
Pharmacol Sin. 25:1619–1625. 2004.PubMed/NCBI
|
|
26
|
Petersen-Felix S, Arendt-Nielsen L, Bak P,
Fischer M and Zbinden AM: Psychophysical and electrophysiological
responses to experimental pain may be influenced by sedation:
comparison of the effects of a hypnotic (propofol) and an analgesic
(alfentanil). Br J Anaesth. 77:165–171. 1996. View Article : Google Scholar
|
|
27
|
Goto T, Marota JJA and Crosby G:
Pentobarbitone, but not propofol, produce pre-emptive analgesia in
the rat formalin model. Br J Anaesth. 72:662–667. 1994. View Article : Google Scholar : PubMed/NCBI
|
|
28
|
Matta JA, Cornett PM, Miyares RL, Abe K,
Sahibzada N and Ahern GP: General anesthetics activate a
nociceptive ion channel to enhance pain and inflammation. Proc Natl
Acad Sci USA. 105:8784–8789. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
29
|
Patwardhan A, Edelmayer R, Annabi E, Price
T, Malan P and Dussor G: Brief report: receptor specificity defines
algogenic properties of propofol and fospropofol. Anesth Analg.
115:837–840. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
30
|
Hirota K, Smart D and Lambert DG: The
effects of local and intravenous anesthetics on recombinant rat VR1
vanilloid receptors. Anesth Analg. 96:1656–1660. 2003. View Article : Google Scholar : PubMed/NCBI
|
|
31
|
Tsutsumi S, Tomioka A, Sudo M, Nakamura A,
Shirakura K, Takagishi K and Kohama K: Propofol activates vanilloid
receptor channels expressed in human embryonic kidney 293 cells.
Neurosci Lett. 312:45–49. 2001. View Article : Google Scholar : PubMed/NCBI
|
|
32
|
Fischer MJ, Leffler A, Niedermirtl F,
Kistner K, Eberhardt M, Reeh PW and Nau C: The general anesthetic
propofol excites nociceptors by activating TRPV1 and TRPA1 rather
than GABAA receptors. J Biol Chem. 285:34781–3492. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
33
|
Tominaga M, Caterina MJ, Malmberg AB,
Rosen TA, Gilbert H, Skinner K, Raumann BE, Basbaum AI and Julius
D: The cloned capsaicin receptor integrates multiple pain-producing
stimuli. Neuron. 21:531–543. 1998. View Article : Google Scholar : PubMed/NCBI
|
|
34
|
Jordt SE, Tominaga M and Julius D: Acid
potentiation of the capsaicin receptor determined by a key
extracellular site. Proc Natl Acad Sci USA. 97:8134–8139. 2000.
View Article : Google Scholar : PubMed/NCBI
|
|
35
|
Jordt SE and Julius D: Molecular basis for
species-specific sensitivity to ‘hot’ chili peppers. Cell.
108:421–430. 2002.
|
|
36
|
Ahern GP, Brooks IM, Miyares RL and Wang
XB: Extracellular cations sensitize and gate capsaicin receptor
TRPV1 modulating pain signaling. J Neurosci. 25:5109–5116. 2005.
View Article : Google Scholar : PubMed/NCBI
|
|
37
|
Orser BA, Bertlik M, Wang LY and MacDonald
JF: Inhibition by propofol (2,6 di-isopropylphenol) of the
N-methyl-D-aspartate subtype of glutamate receptor in cultured
hippocampal neurones. Br J Pharmacol. 116:1761–1768. 1995.
View Article : Google Scholar : PubMed/NCBI
|
|
38
|
Franks NP and Lieb WR: Molecular and
cellular mechanisms of general anaesthesia. Nature. 367:607–614.
1994. View
Article : Google Scholar : PubMed/NCBI
|