Article
Varying levels of 6‑keto‑prostaglandin F1α and thromboxane B2 in serum and endothelialization and hyperplasia in small‑diameter grafts seeded with CD34+ bone marrow cells in canines
- Authors:
- Weishuai Lian
- Huayi Zhang
- Kun Wang
- Junhao Jiang
- Zijie Su
- Zhenhai Yu
-
View Affiliations / Copyright
Affiliations:
Department of Vascular Surgery, Zhongshan Hospital, Fudan University, Shanghai 200032, P.R. China, Department of Vascular Surgery, The Second Affiliated Hospital of Jiaxing Medical College, Jiaxing, Zhejiang 314000, P.R. China, Department of General Surgery, Qianfoshan Hospital, Shandong University, Jinan, Shandong 250014, P.R. China
-
Pages:
1123-1129
|
Published online on:
February 21, 2014
https://doi.org/10.3892/etm.2014.1573
- Expand metrics +
Metrics:
Total
Views: 0
(Spandidos Publications: | PMC Statistics:
)
Metrics:
Total PDF Downloads: 0
(Spandidos Publications: | PMC Statistics:
)
This article is mentioned in:
Abstract
The aim of the present study was to investigate the serum levels of 6‑keto‑prostaglandin (PG)F1α and thromboxane (TX)B2, as well as the endothelialization and hyperplasia of polytetrafluoroethylene (PTFE) and Dacron prostheses seeded with CD34+ cells in medium‑term observation. A total of 24 crossbred dogs were randomly distributed into PTFE or Dacron groups. CD34+ cells were isolated from bone marrow aspirate and collected using an immunomagnetic bead‑based system. The PTFE or Dacron prostheses were implanted into the abdominal aortic artery and inferior vena cava of the dogs. In each group, 8 dogs were implanted with prostheses that had been seeded with CD34+ cells, while 4 dogs were implanted with prostheses that had been seeded with autogenous blood as a control. Serum concentrations of 6‑keto‑PGF1α and TXB2 were determined at days 0, 10, 30 and 60 following surgery. The grafts were removed and examined at days 10, 30, 60 and 100 following surgery. Finally, CD34 factor staining was used to identify endothelial cells, while light and electron microscopy were applied to examine endothelialization and patency. The results revealed that confluent endothelial cells appeared on the neointima of prostheses seeded with CD34+ cells at day 30 following surgery. In the control groups compared with the experimental groups, there were fewer endothelial cells and the neointima was significantly thicker in the arterial (PTFE, 174±1.41 vs. 117±2.83 µm, respectively; P=0.001; Dacron, 187.5±3.5 vs. 100±1.41 µm, respectively; P<0.001) and venous (PTFE, 230.5±6.36 vs. 135±5.66 µm, respectively; P=0.001; Dacron, 249±2.83 vs. 121.5±3.54 µm, respectively; P<0.001) prostheses. In the experimental groups, intimal hyperplasia in the venous prostheses (PTFE, 135±5.66 µm; Dacron, 121.5±3.54 µm) was more severe compared with that in the arterial prostheses (PTFE, 117±2.83 µm; Dacron, 100±1.41 µm) at day 60. Compared with the 6‑keto‑PGF1α concentrations in the experimental groups, those in the control groups were significantly lower on day 10 (PTFE, 135±6.01 vs. 80.5±4.35 pg/l, respectively; P=0.001; Dacron, 145±6.54 vs. 81.2±5.10 pg/l, respectively; P<0.001) and were then maintained at a lower level. By contrast, the TXB2 concentration, following marked increases on day 10 in the experimental and control groups (PTFE, 635±32.8 vs. 1,256±63.5 pg/l, respectively; P<0.001; Dacron, 652±30.9 vs. 1,136±53.2 pg/l, respectively; P=0.001), remained at a high level in the control groups. Therefore, the results of the present study indicate that it is possible to achieve rapid endothelialization in PTFE or Dacron prostheses by implanting CD34+ cells. Endothelialization inhibited the reduction in the concentration of 6‑keto‑PGF1α and the increase in the concentration of TXB2. In addition, endothelialization inhibited excessive intimal hyperplasia and thrombosis. Thus, CD34+ cell seeding provides a theoretical basis for the clinical application of artificial vessel endothelialization.
View Figures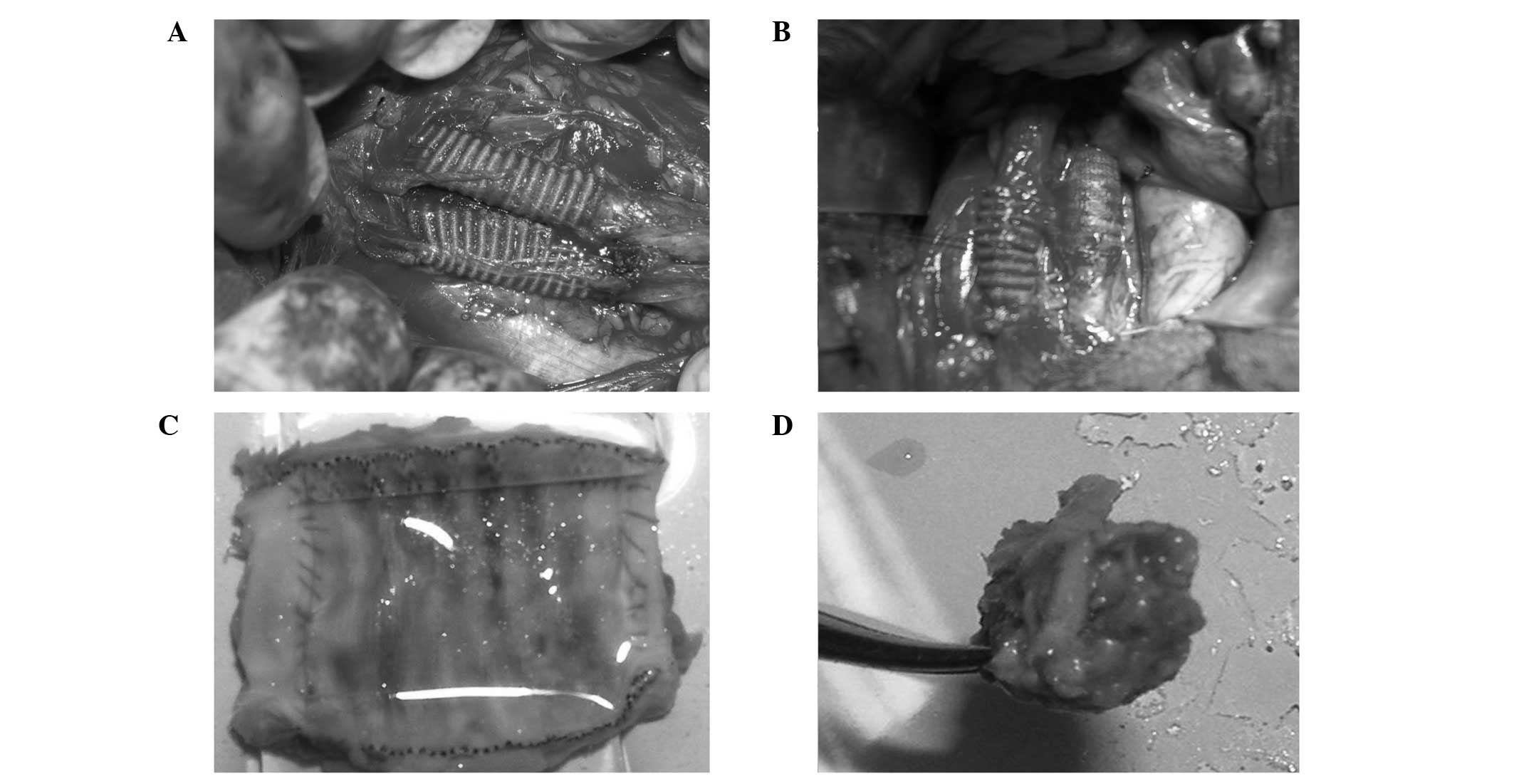 |
Figure 1
|
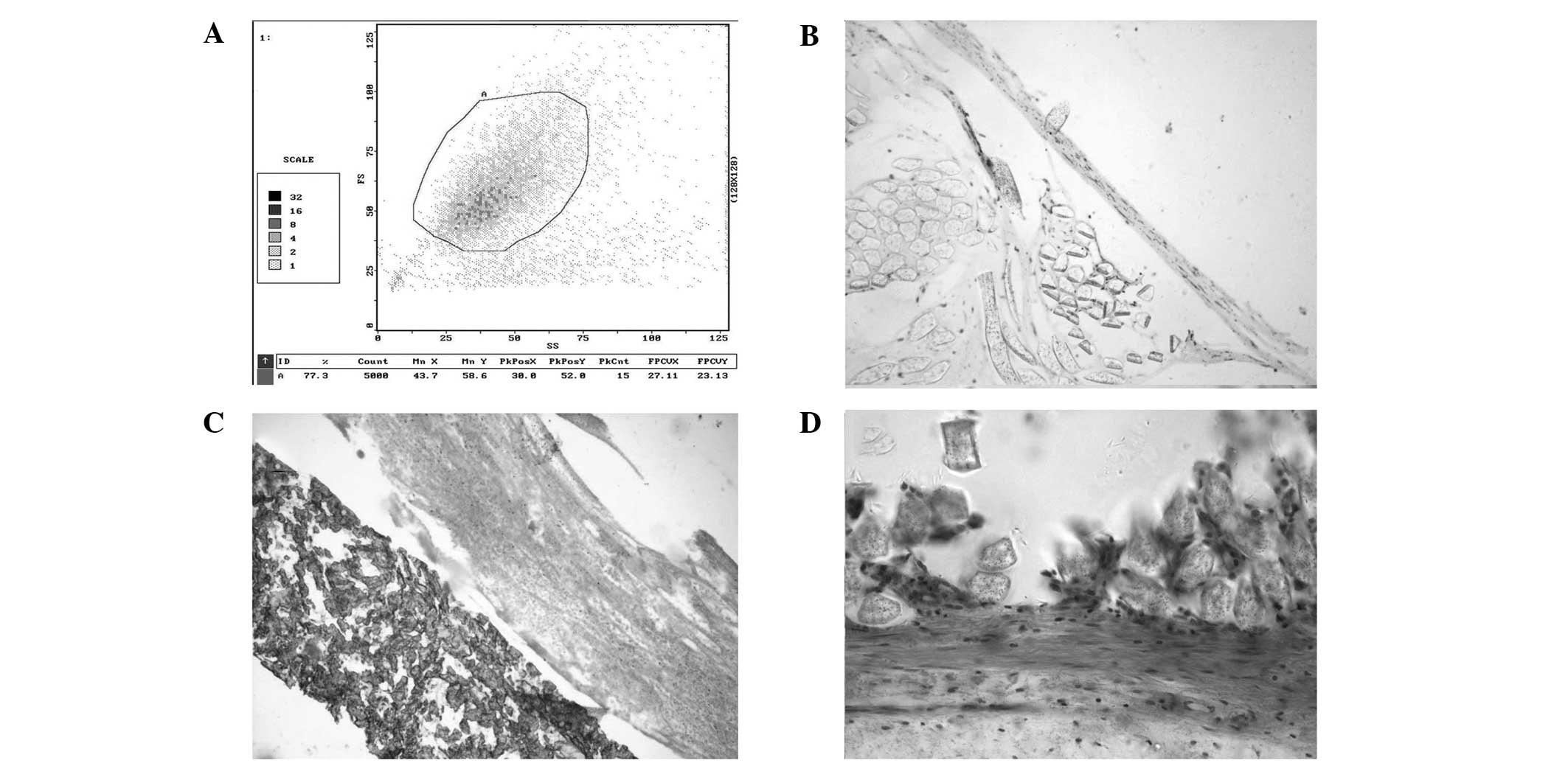 |
Figure 2
|
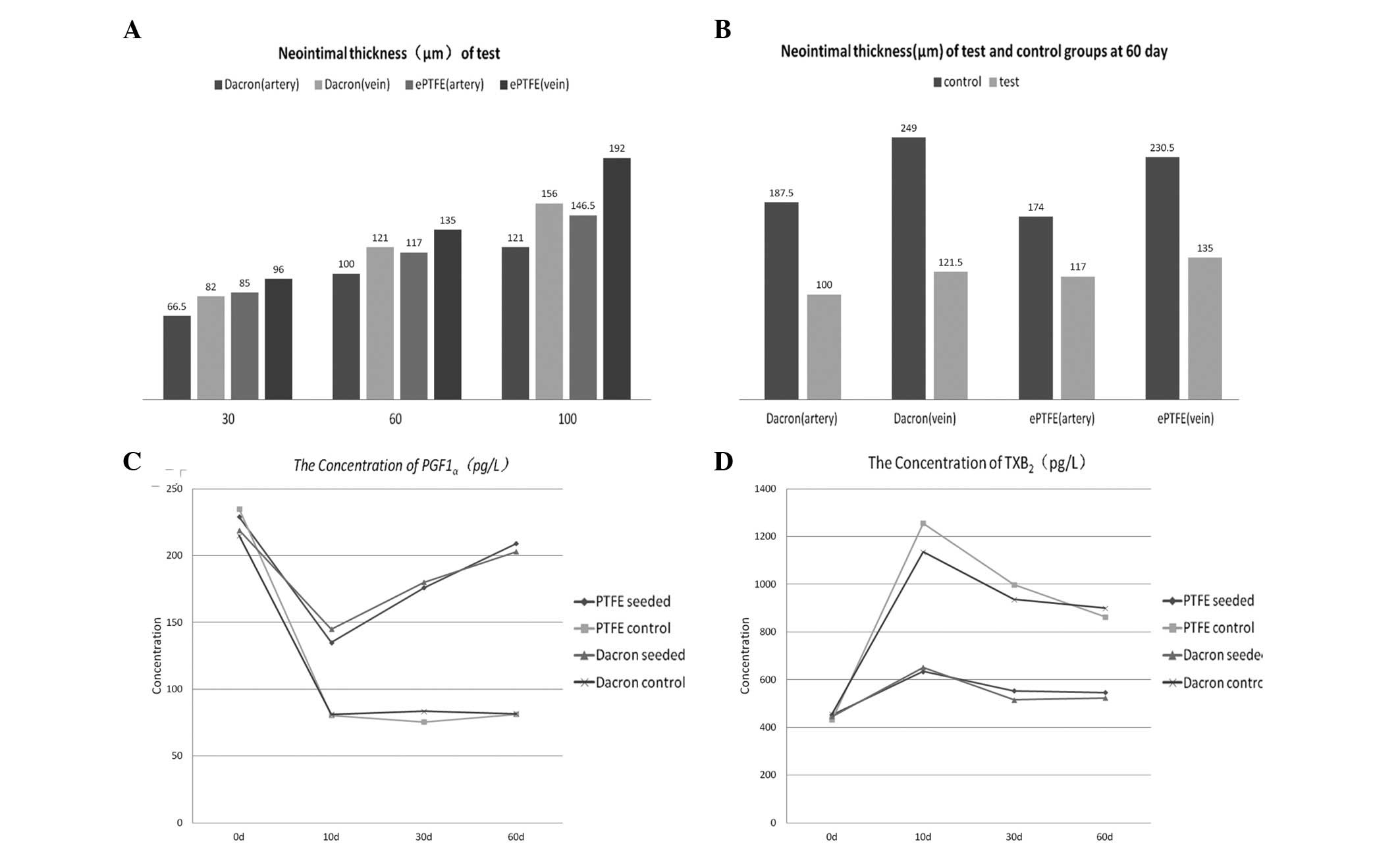 |
Figure 3
|
 |
Figure 4
|
 |
Figure 5
|
 |
Figure 6
|
View References
|
1
|
Kidd K, Patula VB and Williams SK:
Accelerated endothelialization of interpositional 1-mm vascular
grafts. J Surg Res. 113:234–242. 2003. View Article : Google Scholar : PubMed/NCBI
|
|
2
|
Bhattacharya V, McSweeney PA, Shi Q, et
al: Enhanced endothelialization and microvessel formation in
polyester grafts seeded with CD34(+) bone marrow cells. Blood.
95:581–585. 2000.PubMed/NCBI
|
|
3
|
Solovey A, Lin Y, Browne P, Choong S,
Wayner E and Hebbel RP: Circulating activated endothelial cells in
sickle cell anemia. N Engl J Med. 337:1584–1590. 1997. View Article : Google Scholar : PubMed/NCBI
|
|
4
|
Shi Q, Wu MH, Hayashida N, Wechezak AR,
Clowes AW and Sauvage LR: Proof of fallout endothelialization of
impervious Dacron grafts in the aorta and inferior vena cava of the
dog. J Vasc Surg. 20:546–557. 1994. View Article : Google Scholar : PubMed/NCBI
|
|
5
|
Shi Q, Rafii S, Wu MH, et al: Evidence for
circulating bone marrow-derived endothelial cells. Blood.
92:362–367. 1998.PubMed/NCBI
|
|
6
|
Asahara T, Murohara T, Sullivan A, et al:
Isolation of putative progenitor endothelial cells for
angiogenesis. Science. 275:964–967. 1997. View Article : Google Scholar : PubMed/NCBI
|
|
7
|
Moncada S, Gryglewski R, Bunting S and
Vane JR: An enzyme isolated from arteries transforms prostaglandin
endoperoxides to an unstable substance that inhibits platelet
aggregation. Nature. 263:663–665. 1976. View Article : Google Scholar
|
|
8
|
Wen SJ, Zhao LM, Wang SG, et al: Human
vascular smooth muscle cells and endothelial cells cocultured on
polyglycolic acid (70/30) scaffold in tissue engineered vascular
graft. Chin Med J (Engl). 120:1331–1335. 2007.PubMed/NCBI
|
|
9
|
Nakahata N: Thromboxane A2:
physiology/pathophysiology, cellular signal transduction and
pharmacology. Pharmacol Ther. 118:18–35. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
10
|
Ranjan AK, Kumar U and Hardikar AA, Poddar
P, Nair PD and Hardikar AA: Human blood vessel-derived endothelial
progenitors for endothelialization of small diameter vascular
prosthesis. PLoS One. 4:e77182009. View Article : Google Scholar : PubMed/NCBI
|
|
11
|
Li CM, Wang ZG, Gu YQ, et al: Preliminary
investigation of seeding mesenchymal stem cells on biodegradable
scaffolds for vascular tissue engineering in vitro. ASAIO J.
55:614–619. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
12
|
Civin CI, Strauss LC, Brovall C, et al:
Antigenic analysis of hematopoiesis. III A hematopoietic progenitor
cell surface antigen defined by a monoclonal antibody raised
against KG-la cells. J Immunol. 133:157–165. 1984.
|
|
13
|
Yoder MC: Is endothelium the origin of
endothelial progenitor cells? Arterioscler Thromb Vasc Biol.
30:1094–1103. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
14
|
Case J, Mead LE, Bessler WK, Prater D,
White HA, Saadatzadeh MR, Bhavsar JR, Yoder MC, Haneline LS and
Ingram DA: Human CD34+AC133+VEGFR-2+ cells are not endothelial
progenitor cells but distinct, primitive hematopoietic progenitors.
Exp Hematol. 35:1109–1118. 2007.
|
|
15
|
Timmermans F, Van Hauwermeiren F, De Smedt
M, Raedt R, Plasschaert F, De Buyzere ML, Gillebert TC, Plum J and
Vandekerckhove B: Endothelial outgrowth cells are not derived from
CD133+ cells or CD45+ hematopoietic precursors. Arterioscler Thromb
Vasc Biol. 27:1572–1579. 2007.
|
|
16
|
van der Zijpp YJ, Poot AA and Feijen J:
Endothelialization of small-diameter vascular prostheses. Arch
Physiol Biochem. 111:415–427. 2003.PubMed/NCBI
|




















