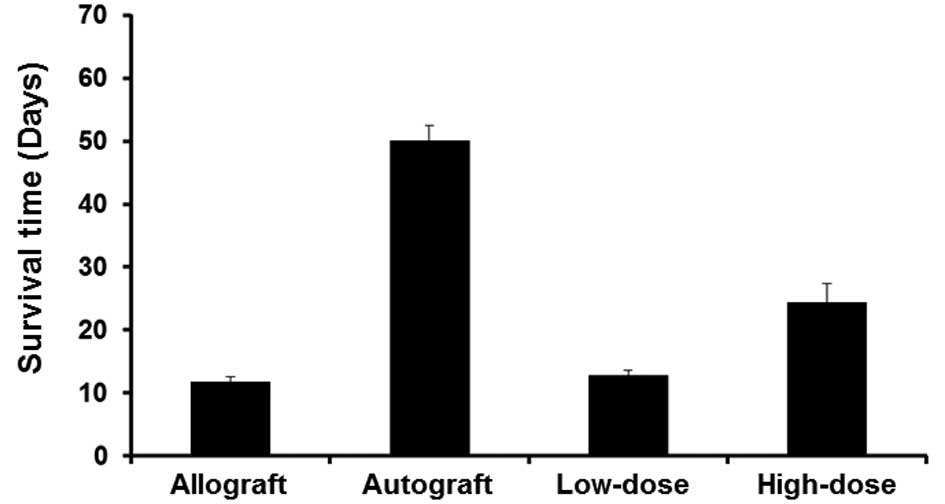
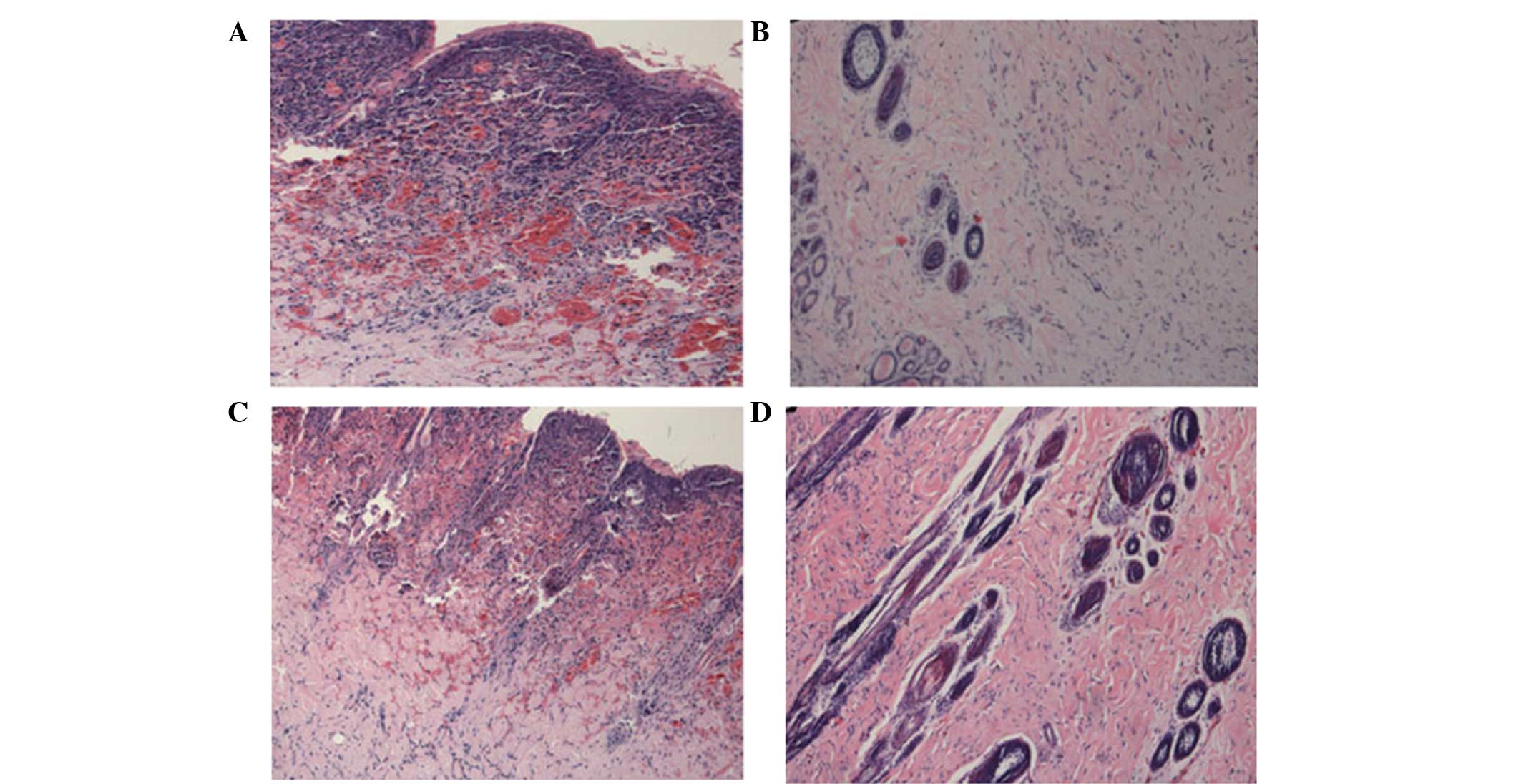
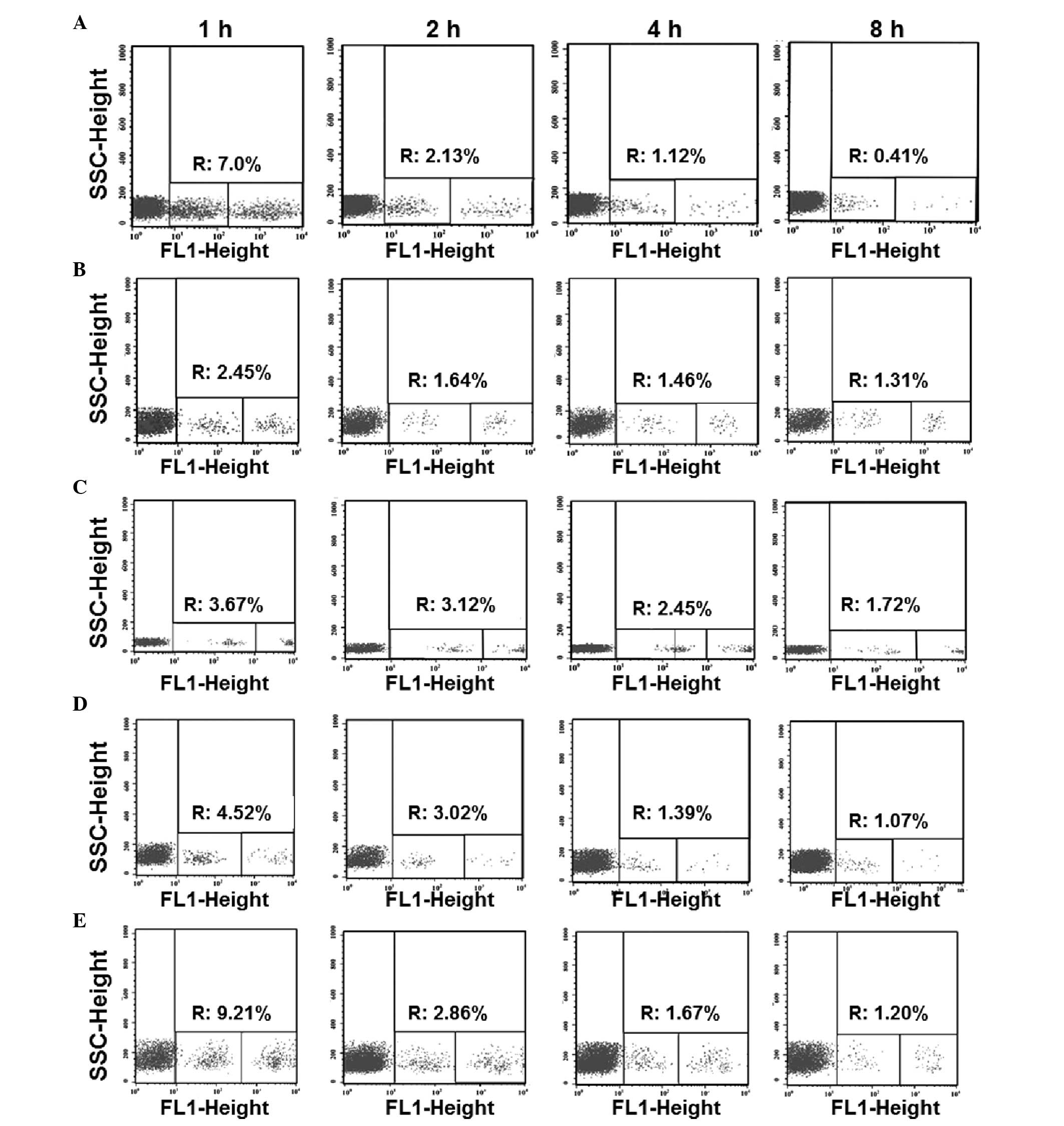
|
1
|
Segoloni GP: New immunodepressant drugs
for the prevention and control of kidney transplant rejection. G
Ital Nefrol. 22:3–15. 2005.(In Italian). PubMed/NCBI
|
|
2
|
Cobbold SP, Adams E, Graca L, Daley S,
Yates S, Paterson A, et al: Immune privilege induced by regulatory
T cells in transplantation tolerance. Immunol Rev. 213:239–255.
2006. View Article : Google Scholar : PubMed/NCBI
|
|
3
|
Pretagostini R, Cinti P, Lai Q, Poli L and
Berloco PB: Minimization of immunosuppressive therapy and
immunological monitoring of kidney transplant recipients with
long-term allograft survival. Transpl Immunol. 20:3–5. 2008.
View Article : Google Scholar : PubMed/NCBI
|
|
4
|
Quatra F, Lowenberg DW, Buncke HJ, Romeo
OM, Brooks D, Buntic RF and Baxter-Lowe LA: Induction of tolerance
to composite tissue allograft in a rat model. Microsurgery.
26:573–578. 2006. View Article : Google Scholar : PubMed/NCBI
|
|
5
|
Tryphonopoulos P, Ruiz P, Weppler D,
Nishida S, Levi DM, Moon J, Tekin A, Velez M, Neuman DR, Island E,
Selvaggi and Tzakis AG: Long-term follow-up of 23 operational
tolerant liver transplant recipients. Transplantation.
90:1556–1561. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
6
|
Ashokkumar C, Talukdar A, Sun Q, Higgs BW,
Janosky J, Wilson P, Mazariegos G, Jaffe R, Demetris A, Dobberstein
J, Soltys K, Bond G, Thomson AW, Zeevi A and Sindhi R: Allospecific
CD154+ T cells associate with rejection risk after
pediatric liver transplantation. Am J Transplant. 9:179–191. 2009.
View Article : Google Scholar : PubMed/NCBI
|
|
7
|
Weimar W, Rischen-Vos J, de Kuiper P,
Gregoor PJ, IJzermans N, van Besouw NM, et al: Tapering
immunosuppression in recipients of living donor kidney transplants.
Nephrol Dial Transplant. 19 (Suppl 4):iv61–iv63. 2004. View Article : Google Scholar : PubMed/NCBI
|
|
8
|
Amirzargar A, Lessanpezeshki M, Fathi A,
Amirzargar M, Khosravi F, Ansaripour B and Nikbin B: TH1/TH2
cytokine analysis in Iranian renal transplant recipients.
Transplant Proc. 37:2985–2987. 2005. View Article : Google Scholar : PubMed/NCBI
|
|
9
|
Sengul S, Keven K, Gormez U, Kutlay S,
Erturk S and Erbay B: Identification of patients at risk of acute
rejection by pretransplantation and posttransplantation monitoring
of soluble CD30 levels in kidney transplantation. Transplantation.
81:1216–1219. 2006. View Article : Google Scholar : PubMed/NCBI
|
|
10
|
Cinti P, Pretagostini R, Arpino A,
Tamburro ML, Mengasini S, Lattanzi R, et al: Evaluation of
pretransplant immunologic status in kidney-transplant recipients by
panel reactive antibody and soluble CD30 determinations.
Transplantation. 79:1154–1156. 2005. View Article : Google Scholar : PubMed/NCBI
|
|
11
|
Süsal C, Döhler B, Sadeghi M, Salmela KT,
Weimer R, Zeier M and Opelz G: Posttransplant sCD30 as a predictor
of kidney graft outcome. Transplantation. 91:1364–1369. 2011.
View Article : Google Scholar : PubMed/NCBI
|
|
12
|
Gebel HM, Bray RA and Nickerson P:
Pre-transplant assessment of donor-reactive, HLA-specific
antibodies in renal transplantation: Contraindication vs. risk. Am
J Transplant. 3:1488–1500. 2003. View Article : Google Scholar : PubMed/NCBI
|
|
13
|
Worthington JE, Martin S, Al-Husseini DM,
Dyer PA and Johnson RW: Posttransplantation production of donor
HLA-specific antibodies as a predictor of renal transplant outcome.
Transplantation. 75:1034–1040. 2003. View Article : Google Scholar : PubMed/NCBI
|
|
14
|
Israeli M, Klein T, Sredni B, Avitzur Y,
Mor E, Bar-Nathen N, et al: A new parameter in immune monitoring of
pediatric liver transplantation recipients. Liver Transpl.
14:893–898. 2008. View
Article : Google Scholar : PubMed/NCBI
|
|
15
|
Kowalski RJ, Post DR, Mannon RB, Sebastian
A, Wright HI, Sigle G, Burdick J, Elmagd KA, Zeevi A, Lopez-Cepero
M, Daller JA, Gritsch HA, Reed EF, Jonsson J, Hawkins D and Britz
JA: Assessing relative risks of infection and rejection: A
meta-analysis using an immune function assay. Transplantation.
82:663–668. 2006. View Article : Google Scholar : PubMed/NCBI
|
|
16
|
Li Y, Koshiba T, Yoshizawa A, Yonekawa Y,
Masuda K, Ito A, Ueda M, Mori T, Kawamoto H, Tanaka Y, Sakaguchi S,
Minato N, Wood KJ and Tanaka K: Analyses of peripheral blood
mononuclear cells in operational tolerance after pediatric living
donor liver transplantation. Am J Transplant. 4:2118–2125. 2004.
View Article : Google Scholar : PubMed/NCBI
|
|
17
|
Cortesini R, Renna-Molajoni E, Cinti P,
Pretagostini R, Ho E, Rossi P and Suciu-Foca Cortesini N: Tailoring
of immunosuppression in renal and liver allograft recipients
displaying donor specific T-suppressor cells. Hum Immunol.
63:1010–1018. 2002. View Article : Google Scholar : PubMed/NCBI
|
|
18
|
Wolf T, Oumeraci T, Gottlieb J, Pich A,
Brors B, Eils R, Haverich A, Schlegelberger B, Welte T, Zapatka M
and von Neuhoff N: Proteomic bronchiolitis obliterans syndrome risk
monitoring in lung transplant recipients. Transplantation.
92:477–485. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
19
|
Cookson S, Doherty DG, Todryk S, Gibbs P,
Portmann B, O'Grady J, et al: Hepatic expression of IL-15 mRNA is
associated with liver graft acceptance. Transpl Immunol. 11:39–48.
2003. View Article : Google Scholar : PubMed/NCBI
|
|
20
|
Schaub S, Rush D, Wilkins J, Gibson IW,
Weiler T, Sangster K, et al: Proteomic-based detection of urine
proteins associated with acute renal allograft rejection. J Am Soc
Nephrol. 15:219–227. 2004. View Article : Google Scholar : PubMed/NCBI
|
|
21
|
El Essawy B, Otu HH, Choy B, Zheng XX,
Libermann TA and Strom TB: Proteomic analysis of the allograft
response. Transplantation. 82:267–274. 2006. View Article : Google Scholar : PubMed/NCBI
|
|
22
|
Sigdel TK and Sarwal MM: The proteogenomic
path towards biomarker discovery. Pediatr Transplant. 12:737–747.
2008. View Article : Google Scholar : PubMed/NCBI
|
|
23
|
Truong DQ, Bourdeaux C, Wieërs G, Saussoy
P, Latinne D and Reding R: The immunological monitoring of kidney
and liver transplants in adult and pediatric recipients. Transpl
Immunol. 22:18–27. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
24
|
Rodrigo E, López-Hoyos M, Corral M,
Fábrega E, Fernández-Fresnedo G, San Segundo D, et al: ImmuKnow as
a diagnostic tool for predicting infection and acute rejection in
adult liver transplant recipients: A systematic review and
meta-analysis. Liver Transpl. 18:1245–1253. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
25
|
Li B, Tian L, Diao Y, Li X, Zhao L and
Wang X: Exogenous IL-10 induces corneal transplantation immune
tolerance by a mechanism associated with the altered Th1/Th2
cytokine ratio and the increased expression of TGF-β. Mol Med Rep.
9:2245–2250. 2014.PubMed/NCBI
|
|
26
|
Hermans IF, Silk JD, Yang J, Palmowski MJ,
Gileadi U, McCarthy C, et al: The VITAL assay: A versatile
fluorometric technique for assessing CTL- and NKT-mediated
cytotoxicity against multiple targets in vitro and in vivo. J
Immunol Methods. 285:25–40. 2004. View Article : Google Scholar : PubMed/NCBI
|
|
27
|
Ritchie DS, Hermans IF, Lumsden JM, Scanga
CB, Roberts JM, Yang J, et al: Dendritic cell elimination as an
assay of cytotoxic T lymphocyte activity in vivo. J Immunol
Methods. 246:109–117. 2000. View Article : Google Scholar : PubMed/NCBI
|
|
28
|
Weist BM, Hernandez JB and Walsh CM: Loss
of DRAK2 signaling enhances allogeneic transplant survival by
limiting effector and memory T cell responses. Am J Transplant.
12:2220–2227. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
29
|
Barchet W, Oehen S, Klenerman P, Wodarz D,
Bocharov G, Lloyd AL, et al: Direct quantitation of rapid
elimination of viral antigen-positive lymphocytes by antiviral
CD8(+) T cells in vivo. Eur J Immunol. 30:1356–1363. 2000.
View Article : Google Scholar : PubMed/NCBI
|
|
30
|
Jiang Z, Gao Y, Pan M and Zhong L: Reagent
for monitoring immune state of rabit after skin grafting, and
preparation method thereof. Chinese Patent CN201210013937. Filed.
January 17–2012 Issued. July 18–2012
|