Introduction
The prevalence of cerebral aneurysm in the general
population is estimated to be 1–5% (1). Ruptured intracranial aneurysms are the
most important cause of non-traumatic subarachnoid hemorrhage,
which is a medical emergency and can result in severe disability or
mortality (2). Thus, the prompt
diagnosis and treatment of intracranial aneurysm are of
considerable importance for the outcome of the patient.
Conventional digital subtraction angiography (DSA)
has been considered the gold standard for the detection and
characterization of intracranial aneurysms due to its high spatial
resolution and large field of view (3,4);
however, it also has several disadvantages, including the
relatively high cost and the high skill level required to perform
the procedure. Furthermore, DSA is an invasive procedure associated
with a small but definite risk of neurological morbidity (5). There is a requirement, therefore, to
find an accurate, minimally invasive imaging method that is free
from these complications. Computed tomography angiography (CTA), as
a relatively non-invasive imaging method, has been widely used in
the screening of patients with suspected intracranial aneurysms
(6). As a result of the rapid
improvement in multi-detector CTA technology, the diagnostic
performance of multi-detector CTA for the detection of intracranial
aneurysms is now approaching that of DSA (7); however, it exhibits a disadvantage in
the detection of small-sized aneurysms that are located near the
skull base due to the influence of overprojecting bone structures
(8).
As CT technology has evolved and various subtraction
and post-processing techniques have been developed, subtraction
CTA, allowing bone-free visualization of aneurysms, has become
possible for the diagnosis of intracranial aneurysms. There have
been several reports on the potential usefulness of subtraction CTA
in evaluating intracranial aneurysms; however, the results of these
studies have been varied (6–8). The purpose of this meta-analysis was to
calculate the sensitivity and specificity of subtraction CTA for
the detection of cerebral aneurysms, in comparison with the
reference standard of DSA.
Materials and methods
Search strategy
A systematic literature search up to January 1, 2013
was conducted in PubMed to identify any relevant studies. The
search terms included ‘tomography, X-ray computed’ or ‘computed
tomography angiography’, combined with ‘intracranial aneurysm’ or
‘subarachnoid hemorrhage’. Studies that were evidently irrelevant,
based on a scan of the titles and abstracts, were excluded, while
the remaining articles were assessed for relevance to the topic of
interest by reading the full text. Furthermore, a manual search was
conducted by checking the references of retrieved articles to find
any additional published studies. All searches were conducted
independently by 2 authors, prior to the results being compared.
Any questions or discrepancies were resolved through discussion and
consensus.
Study selection
To be eligible for inclusion in the meta-analysis,
the studies had to satisfy 8 inclusion criteria: i) The patients
were clinically suspected of having an intracranial aneurysm; ii)
the diagnostic index test was bone subtraction CTA; iii) the
reference standard was DSA or its combination with neurosurgical
findings; iv) the condition of interest was the presence or absence
of an intracranial aneurysm; v) 2×2 contingency tables were
reconstructed on a per-patient or per-aneurysm basis; vi) the study
had no limitations with regard to specific aneurysm types or
locations; vii) the study included ≥20 patients, due to the
increased likelihood of smaller studies suffering from selection
bias; and viii) the study included ≥5 patients with and 5 patients
without an aneurysm, so that the study provided meaningful numbers
for sensitivity and specificity.
Data extraction
The study data were independently extracted by 2
researchers, and any disagreements were resolved through discussion
and consensus. The following data were collected: Surname of the
first author, year of study publication, country in which the study
was performed, study design, age range of the study participants,
index test and reference standard. The 2×2 count data were
extracted on a per-patient basis and, if reported, on a
per-aneurysm basis.
Assessment of study quality
Study quality was assessed independently by 2
researchers using the Quality Assessment of Diagnostic Accuracy
Studies (QUADAS) tool, which includes 14 quality items (9); disagreement was resolved by consensus.
This evidence-based tool was developed to assess the quality of
primary studies of diagnostic accuracy. The QUADAS item 4 was
scored positive if the delay between the index and reference tests
was ≤3 days in all patients. For each study, a quality score was
accumulated by assigning 1 point for each QUADAS item if fulfilled,
0.5 if unclear and 0 if not fulfilled. A score between 11 and 14
points was considered high quality and a score <11 points as low
quality. A more detailed description of each item, together with a
guide on how to score each item, is provided in the study by
Whiting et al (9).
Statistical analysis
For all studies included in the meta-analysis, the
individual sensitivities and specificities were recalculated from
the 2×2 count data on a per-patient or per-aneurysm basis. Pooled
summary estimates were obtained from a bivariate, mixed-effects,
binary regression modeling framework. Model specification,
estimation and prediction were performed with Stata software,
version 11.0 (Stata Corp, College Station, TX, USA). A forest plot
was generated that contained the individual study sensitivities and
specificities with 95% confidence intervals (CIs) and the pooled
sensitivity and specificity estimates. The areas under the receiver
operating characteristic (ROC) curves (AUCs) were used to analyze
the diagnostic precision.
The I2 statistic was used to
examine whether the results of studies were homogeneous (10). This statistic uses the conventional Q
statistic to calculate the percentage of tool variation
heterogeneity on a scale ranging from 0% (no heterogeneity) to 100%
(all variance due to heterogeneity). In contrast to the Q
statistic, the I2 is less dependent on the number
of studies included in a meta-analysis. I2
>50% suggested notable heterogeneity. When statistical
heterogeneity was detected, sensitivity analyses were also
performed.
In studies assessing test accuracy, one of the
primary causes of heterogeneity is the threshold effect, which
arises due to the use of different cut-offs or thresholds in the
analyzed studies to define a positive (or negative) test result.
The Spearman correlation coefficient between the logit of
sensitivity and the logit of 1-specificity was calculated using
Meta-Disc version 1.4, in order to determine the threshold effect
(11). A threshold effect was
indicated by the presence of a strong, positive correlation
(P<0.05) (11).
The presence of publication bias was visually
assessed through the production of a Deeks' funnel plot and an
asymmetry test (12). In the Stata
software, linear regression of log odds ratios on the inverse root
of effective sample sizes was performed as a test for funnel plot
asymmetry. A P-value of <0.10 was considered to be
representative of statistically significant publication bias,
suggesting that only the small studies that reported a high
accuracy for subtraction CTA had been published, whereas the small
studies that reported a lower accuracy had not been published. Data
were analyzed with Meta-Disc version 1.4 and Stata version 11.0
software.
Results
Literature search
The initial search strategy retrieved a total of
5,224 citations. Following the screening of the titles and
abstracts, 5,209 sources were excluded. The full texts of the
remaining 15 sources were evaluated. Of these, 7 sources were
excluded for reasons given in Fig. 1
(13–19). The remaining 8 studies were included
(20–27).
The study characteristics are shown in Table I. The quality assessment scores
ranged from 10.5 to 13.5, with a median study quality score of
12.5. Four studies were prospective, 2 studies were retrospective,
and in 2 studies this was unclear. The 8 studies included 982
patients. Optional count data on a per-aneurysm basis in addition
to count data on a per-patient basis were provided by all 8 studies
(Table I).
 | Table I.Studies included in the
meta-analysis. |
Table I.
Studies included in the
meta-analysis.
| First author
(ref.) | Year | Country | Design | No. of patients | Age, years | QUADAS score | No. of detector
rows | RS | No. of aneurysms |
|---|
| Jayaraman (20) | 2004 | US | P | 35 | 54 | 13.5 | Multi-detector | DSA | 26 |
| Yoon (21) | 2007 | Korea | P | 85 | 49.6 | 13.5 | 16 | DSA | 93 |
| Romijn (22) | 2008 | Netherlands | NR | 108 | 56 | 12.0 | 4 | DSA | 117 |
| Li (23) | 2009 | China | P | 76 | 48.0 | 12.5 | 64 | DSA | 76 |
| Zhang (24) | 2010 | China | P | 61 | 52.0 | 11.5 | Dual-source | DSA | 47 |
| Ramasundara (25) | 2010 | Australia | R | 36 | NA | 10.5 | 16/64 | DSA | 34 |
| Luo (26) | 2012 | China | NR | 56 |
48.0 | 12.5 | 320 | DSA | 51 |
| Lu (27) | 2012 | China | R | 525 |
52.0 | 13.5 | Dual-source | 3D DSA | 459 |
Assessment of publication bias
On a per-patient and per-aneurysm basis, the funnel
plot and regression test showed no significant publication bias
(P=0.30 and P=0.53) (Fig. 2). This
suggested that there were no smaller studies with lower diagnostic
accuracies that had not been published.
Analysis of heterogeneity and pooled
sensitivity and specificity on a per-patient basis
The sensitivities ranged from 86 to 100% (Table II). Concerning sensitivity, the
selected studies showed moderate heterogeneity
(I2=81.2%). For specificity, low heterogeneity was
observed (I2=36.0%); the specificity ranged from 89 to
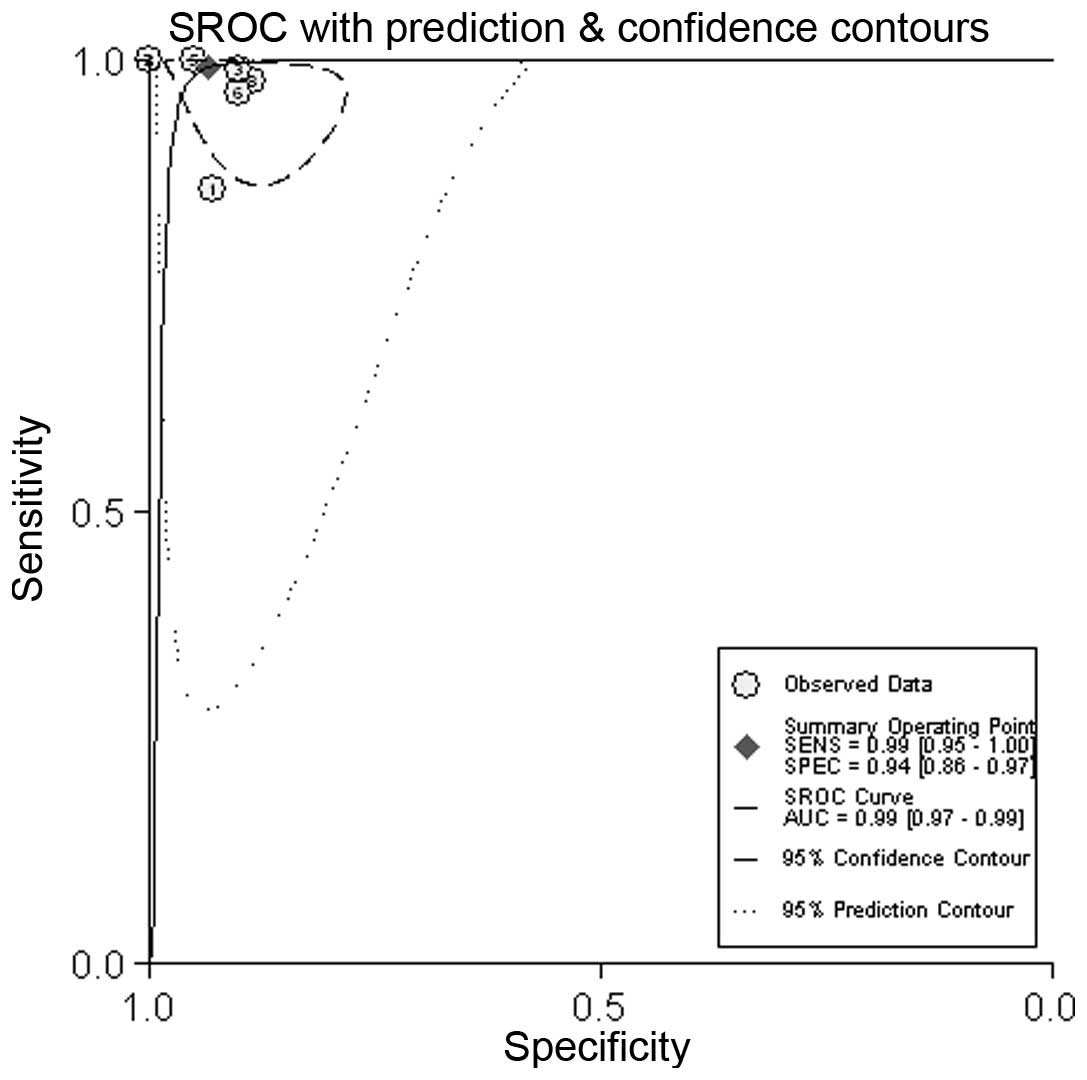
100%. The overall pooled sensitivity was 99% (95% CI, 95–100%), and
the pooled specificity was 94% (95% CI, 86–97%) (Fig. 3). The AUC was 0.99 (95% CI,
0.97–0.99) (Fig. 4).
 | Table II.Count data, sensitivities and
specificities of the included studies. |
Table II.
Count data, sensitivities and
specificities of the included studies.
|
| Per-patient | Per-aneurysm |
|---|
|
|
|
|
|---|
| First author
(ref.) | Year | TP, n | FP, n | FN, n | TNa, n | Sensitivity,
%a | Specificity,
%a | TP, n | FP, n | FN, n | TNb, n | Sensitivity, % | Specificity, % |
|---|
| Jayaraman (20) | 2004 | 18 | 1 | 3 | 13 | 85.7
(63.7–97.0) | 92.9
(66.1–99.8) | 20 | 1 | 6 | 13 | 76.9
(56.4–91.0) | 92.9
(66.1–99.8) |
| Yoon (21) | 2007 | 71 | 0 | 0 | 14 | 100.0
(94.9–100.0) | 100.0
(76.8–100.0) | 86 | 1 | 7 | 14 | 92.5
(85.1–96.9) | 93.3
(68.1–99.8) |
| Romijn (22) | 2008 | 87 | 2 | 1 | 18 | 98.9
(93.8–100.0) | 90.0
(68.3–98.8) | 106 | 3 | 11 | 18 | 90.6
(83.8–95.2) | 85.7
(63.7–97.0) |
| Li (23) | 2009 | 64 | 0 | 0 | 12 | 100.0
(94.4–100.0) | 100.0
(73.5–100.0) | 75 | 0 | 0 | 12 | 100.0
(95.2–100.0) | 100.0
(73.5–100.0) |
| Zhang (24) | 2010 | 41 | 1 | 0 | 19 | 100.0
(91.4–100.0) | 95.0
(75.1–99.9) | 45 | 1 | 2 | 19 | 95.7
(85.5–99.5) | 95.0
(75.1–99.9) |
| Ramasundara
(25) | 2010 | 26 | 1 | 1 | 9 | 96.3
(81.0–99.9) | 90.0
(55.5–99.7) | 33 | 1 | 1 | 9 | 97.1
(84.7–99.9) | 90.0
(55.5–99.7) |
| Luo (26) | 2012 | 42 | 0 | 0 | 14 | 100.0
(91.6–100.0) | 100.0
(76.8–100.0) | 51 | 0 | 0 | 14 | 100.0
(93.0–100.0) | 100.0
(76.8–100.0) |
| Lu (27) | 2012 | 398 | 12 | 9 | 94 | 97.8
(95.8–99.0) | 88.7
(81.1–94.0) | 443 | 13 | 16 | 94 | 96.5
(94.4–98.0) | 87.9
(80.1–93.4) |
A Spearman rank correlation was performed as a
further test for the threshold effect and was determined to be
0.558 (P=0.151), which indicated that there was an absence of a
notable threshold effect in the accuracy estimates among the
individual studies.
The results of meta-regression indicated that the
year the study was published, study design, quality score and
blinding method were strongly associated with sensitivity, but not
with specificity (Fig. 5).
Analysis of heterogeneity and pooled
sensitivity and specificity on a per-aneurysm basis
The sensitivities ranged from 77 to 100% (Table II). Concerning sensitivity, the
selected studies showed moderate heterogeneity
(I2=84.3%). For specificity, low heterogeneity was
observed (I2=6.9%); the specificity ranged from 86 to
100%. The overall pooled sensitivity was 96% (95% CI, 90–99%), and
the pooled specificity was 91% (95% CI, 85–95%). The AUC was 0.96
(95% CI, 0.94–0.97).
A Spearman rank correlation was performed as a
further test for the threshold effect and was determined to be
0.548 (P=0.160), which indicated that there was an absence of a
notable threshold effect in the accuracy estimates among the
individual studies.
False-negative CTA results
Forty-three intracranial aneurysms were missed at
subtraction CTA. The location of the false-negative aneurysm was
specified for 37 aneurysms (Table
III). The size of the false-negative aneurysms was also
provided in 37 cases: 19 were <3 mm, 15 were <5 mm and 3 were
5–10 mm in diameter. At least 22 of the missed aneurysms could be
detected retrospectively.
 | Table III.Location of false-negative
intracranial aneurysms. |
Table III.
Location of false-negative
intracranial aneurysms.
| Location | No. of
aneurysms |
|---|
| Anterior
circulation |
|
|
Pericallosal artery/ophthalmic
artery | 2 |
|
Anterior communicating
artery/anterior cerebral artery | 5 |
|
Internal carotid artery | 11 |
|
Posterior communicating
artery | 5 |
| Middle
cerebral artery | 9 |
| Posterior
circulation |
|
|
Posterior cerebral artery | 1 |
|
Posterior inferior cerebellar
artery | 2 |
|
Anterior superior cerebellar
artery | 1 |
|
Vertebral and basilar
artery | 1 |
Discussion
To the best of our knowledge, this study is the
first meta-analysis of the diagnostic performance of subtraction
CTA to detect cerebral aneurysms. The results show that subtraction
CTA has a high diagnostic value for the detection of intracranial
aneurysms. According to this meta-analysis of 8 studies,
subtraction CTA has an overall sensitivity of ~99% (95% CI,
95–100%) and a high specificity of ~94% (95% CI, 86–97%) for
diagnosing cerebral aneurysms on a per-patient basis. On a
per-aneurysm basis, the diagnostic accuracy was only slightly
lower.
Among the studies included in this meta-analysis, a
total of 22 false-negative aneurysms at CTA could be identified
retrospectively (20,21,23,24).
These false-negative interpretations can therefore be categorized
as perceptual in nature and could have been substantially bypassed
by double reading.
The I2 statistic for sensitivity
indicated the presence of notable heterogeneity, a finding that is
consistent with previous meta-analyses investigating CTA and
cerebral aneurysms (7,28). The Spearman correlation coefficient
on a per-patient basis was 0.558 (P=0.151), which suggested the
absence of a significant threshold effect. To determine whether
other sources of heterogeneity existed, in addition to the
threshold effect, a subgroup analysis was conducted to identify
factors affecting heterogeneity. The results of the meta-regression
showed that the year the study was published, study design, quality
score and blinding method had a strong association with
sensitivity.
Non-subtraction multi-detector CTA has a relatively
high sensitivity and specificity for the detection of cerebral
aneurysms (6–8); however, the detection of aneurysms
adjacent to bone remains a challenging issue due to overlying bone
structures. To potentially circumvent this limitation, a number of
bone removal techniques, including subtraction and manual or
automated bone editing, have been developed; however, these methods
are associated with several disadvantages, such as the complexity
of use, dependence on the user and the high dose of radiation.
Manual bone editing in CTA is a time-consuming and user-dependent
process that relies on a knowledge of vascular anatomy (13,14).
Matched mask bone elimination (MMBE) is a relatively new technique
that enables bone removal in an automatic and user-independent way
(18,22). In CTA-MMBE, a second, non-enhanced
scan is necessary for the identification of bony structures that
could be masked in the CTA scan. The two consecutive volumetric
scans expose the patient to more radiation, although low-dose CT
techniques are used in the non-enhanced CT. Dual-energy CTA is an
immediate automatic bone removal CTA technique that offers the
advantage that images from a single CT acquisition can be used to
remove bone structures. This technique enables simultaneous
dual-energy image acquisition in the same phase of contrast
enhancement, which reduces the radiation dose. The limitation of
dual-energy CT is that it is not widely available and it requires
more expensive hardware (24).
A number of factors should be taken into
consideration in the interpretation of the present results. First,
homogeneity tests revealed moderate heterogeneity in the
sensitivity of the selected studies. The potential sources of
variability among the studies were variations in the quality
scores, the year that the study was published, the study design and
the blinding method used. Secondly, 3 studies were excluded due to
the data not enabling the reconstruction of the required 2×2 count
tables. It is also possible that the search of the literature did
not identify all the eligible studies, but the random omission of
studies is less likely to have caused a systematic bias. Although
no significant publication bias was suggested by the funnel plot
and regression test, unnoticed publication bias may still have been
present. Thirdly, the included studies were limited to populations
with a high disease prevalence. The extrapolation of the results of
the meta-analysis to screening populations with a disease
prevalence that is inherently lower could introduce bias. Finally,
the number of studies included in this meta-analysis was relatively
small; however, in a previous systematic review (29) investigating the characteristics of
meta-analyses and their included studies in the Cochrane Database,
it was revealed that, irrespective of the medical field, relatively
few studies are typically eligible for meta-analysis for all
outcomes and interventions covered by the Cochrane reviews.
Furthermore, the methodological quality of the studies included in
a meta-analysis has a greater impact on the estimated effects than
the numbers of included studies (30,31).
QUADAS assessment revealed a high overall quality of the studies
included in the present meta-analysis. In conclusion, the results
of this study show that subtraction CTA is a highly sensitive,
specific and non-invasive imaging method for the diagnosis and
evaluation of intracranial aneurysms.
References
|
1
|
Wiebers DO, Whisnant JP, Huston J III,
Meissner I, Brown RD Jr, Piepgras DG, Forbes GS, Thielen K, Nichols
D, O'Fallon WM, et al: International Study of Unruptured
Intracranial Aneurysms Investigators: Unruptured intracranial
aneurysms: Natural history, clinical outcome and risks of surgical
and endovascular treatment. Lancet. 362:103–110. 2003. View Article : Google Scholar : PubMed/NCBI
|
|
2
|
Linn FH, Rinkel GJ, Algra A and van Gijn
J: Incidence of subarachnoid hemorrhage: Role of region, year and
rate of computed tomography: A meta-analysis. Stroke. 27:625–629.
1996. View Article : Google Scholar : PubMed/NCBI
|
|
3
|
Han X, Zhan Y and Chen J: Comparative
study of multi-slice CT angiography with digital subtraction
angiography in the blood supply of meningiomas. Exp Ther Med.
3:31–36. 2012.PubMed/NCBI
|
|
4
|
Brisman JL, Song JK and Newell DW:
Cerebral aneurysms. N Engl J Med. 355:928–939. 2006. View Article : Google Scholar : PubMed/NCBI
|
|
5
|
Kaufmann TJ, Huston J III, Mandrekar JN,
Schleck CD, Thielen KR and Kallmes DF: Complications of diagnostic
cerebral angiography: Evaluation of 19,826 consecutive patients.
Radiology. 243:812–819. 2007. View Article : Google Scholar : PubMed/NCBI
|
|
6
|
Chappell ET, Moure FC and Good MC:
Comparison of computed tomographic angiography with digital
subtraction angiography in the diagnosis of cerebral aneurysms: A
meta-analysis. Neurosurgery. 52:624–631. 2003. View Article : Google Scholar : PubMed/NCBI
|
|
7
|
Westerlaan HE, van Dijk JM, Jansen-van der
Weide MC, de Groot JC, Groen RJ, Mooij JJ and Oudkerk M:
Intracranial aneurysms in patients with subarachnoid hemorrhage: CT
angiography as a primary examination tool for diagnosis-systematic
review and meta-analysis. Radiology. 258:134–145. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
8
|
Donmez H, Serifov E, Kahriman G, Mavili E,
Durak AC and Menkü A: Comparison of 16-row multislice CT
angiography with conventional angiography for detection and
evaluation of intracranial aneurysms. Eur J Radiol. 80:455–461.
2011. View Article : Google Scholar : PubMed/NCBI
|
|
9
|
Whiting P, Rutjes AW, Reitsma JB, Bossuyt
PM and Kleijnen J: The development of QUADAS: A tool for the
quality assessment of studies of diagnostic accuracy included in
systematic reviews. BMC Med Res Methodol. 3:252003. View Article : Google Scholar : PubMed/NCBI
|
|
10
|
Higgins JP, Thompson SG, Deeks JJ and
Altman DG: Measuring inconsistency in meta-analyses. BMJ.
327:557–560. 2003. View Article : Google Scholar : PubMed/NCBI
|
|
11
|
Zamora J, Abraira V, Muriel A, Khan K and
Coomarasamy A: Meta-DiSc: A software for meta-analysis of test
accuracy data. BMC Med Res Methodol. 6:312006. View Article : Google Scholar : PubMed/NCBI
|
|
12
|
Deeks JJ, Macaskill P and Irwig L: The
performance of tests of publication bias and other sample size
effects in systematic reviews of diagnostic test accuracy was
assessed. J Clin Epidemiol. 58:882–893. 2005. View Article : Google Scholar : PubMed/NCBI
|
|
13
|
Schwartz RB, Tice HM, Hooten SM, Hsu L and
Stieg PE: Evaluation of cerebral aneurysms with helical CT:
Correlation with conventional angiography and MR angiography.
Radiology. 192:717–722. 1994. View Article : Google Scholar : PubMed/NCBI
|
|
14
|
Imakita S, Onishi Y, Hashimoto T, Motosugi
S, Kuribayashi S, Takamiya M, Hashimoto N, Yamaguchi T and Sawada
T: Subtraction CT angiography with controlled-orbit helical
scanning for detection of intracranial aneurysms. AJNR Am J
Neuroradiol. 19:291–295. 1998.PubMed/NCBI
|
|
15
|
Tomandl BF, Hammen T, Klotz E, Ditt H,
Stermper B and Lell M: Bone-subtraction CT angiography for the
evaluation of intracranial aneurysms. AJNR Am J Neuroradiol.
27:55–59. 2006.PubMed/NCBI
|
|
16
|
Sakamoto S, Kiura Y, Shibukawa M, Ohba S,
Arita K and Kurisu K: Subtracted 3D CT angiography for evaluation
of internal carotid artery aneurysms: Comparison with conventional
digital subtraction angiography. AJNR Am J Neuroradiol.
27:1332–1337. 2006.PubMed/NCBI
|
|
17
|
Zhang LJ, Wu SY, Niu JB, Zhang ZL, Wang
HZ, Zhao YE, Chai X, Zhou CS and Lu GM: Dual-energy CT angiography
in the evaluation of intracranial aneurysms: Image quality,
radiation dose and comparison with 3D rotational digital
subtraction angiography. AJR Am J Roentgenol. 194:23–30. 2010.
View Article : Google Scholar : PubMed/NCBI
|
|
18
|
Venema HW, Hulsmans FJ and den Heeten GJ:
CT angiography of the circle of Willis and intracranial internal
carotid arteries: Maximum intensity projection with matched mask
bone elimination-feasibility study. Radiology. 218:893–898. 2001.
View Article : Google Scholar : PubMed/NCBI
|
|
19
|
Hwang SB, Kwak HS, Han YM and Chung GH:
Detection of intracranial aneurysms using three-dimensional
multidetector-row CT angiography: Is bone subtraction necessary?
Eur J Radiol. 79:e18–e23. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
20
|
Jayaraman MV, Mayo-Smith WW, Tung GA, Haas
RA, Rogg JM, Mehta NR and Doberstein CE: Detection of intracranial
aneurysms: Multi-detector row CT angiography compared with DSA.
Radiology. 230:510–518. 2004. View Article : Google Scholar : PubMed/NCBI
|
|
21
|
Yoon DY, Lim KJ, Choi CS, Cho BM, Oh SM
and Chang SK: Detection and characterization of intracranial
aneurysms with 16-channel multidetector row CT angiography: A
prospective comparison of volume-rendered images and digital
subtraction angiography. AJNR Am J Neuroradiol. 28:60–67.
2007.PubMed/NCBI
|
|
22
|
Romijn M, van Andel Gratama HA, van
Walderveen MA, Sprengers ME, van Rijn JC, van Rooij WJ, Venema HW,
Grimbergen CA, den Heeten GJ and Majoie CB: Diagnostic accuracy of
CT angiography with matched mask bone elimination for detection of
intracranial aneurysms: Comparison with digital subtraction
angiography and 3D rotational angiography. AJNR Am J Neuroradiol.
29:134–139. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
23
|
Li Q, Lv F, Li Y, Li K, Luo T and Xie P:
Subtraction CT angiography for evaluation of intracranial
aneurysms: Comparison with conventional CT angiography. Eur Radiol.
19:2261–2267. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
24
|
Zhang LJ, Wu SY, Poon CS, Zhao YE, Chai X,
Zhou CS and Lu GM: Automatic bone removal dual-energy CT
angiography for the evaluation of intracranial aneurysms. J Comput
Assist Tomogr. 34:816–824. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
25
|
Ramasundara S, Mitchell PJ and Dowling RJ:
Bone subtraction CT angiography for the detection of intracranial
aneurysms. J Med Imaging Radiat Oncol. 54:526–533. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
26
|
Luo Z, Wang D, Sun X, Zhang T, Liu F, Dong
D, Chan NK and Shen B: Comparison of the accuracy of subtraction CT
angiography performed on 320-detector row volume CT with
conventional CT angiography for diagnosis of intracranial
aneurysms. Eur J Radiol. 81:118–122. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
27
|
Lu L, Zhang LJ, Poon CS, Wu SY, Zhou CS,
Luo S, Wang M and Lu GM: Digital subtraction CT angiography for
detection of intracranial aneurysms: Comparison with
three-dimensional digital subtraction angiography. Radiology.
262:605–612. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
28
|
Menke J, Larsen J and Kallenberg K:
Diagnosing cerebral aneurysms by computed tomographic angiography:
Meta-analysis. Ann Neurol. 69:646–654. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
29
|
Davey J, Turner RM, Clarke MJ and Higgins
JP: Characteristics of meta-analyses and their component studies in
the Cochrane Database of Systematic Reviews: A cross-sectional,
descriptive analysis. BMC Med Res Methodol. 11:1602011. View Article : Google Scholar : PubMed/NCBI
|
|
30
|
Gluud LL, Thorlund K, Gluud C, Woods L,
Harris R and Sterne JA: Correction: Reported methodologic quality
and discrepancies between large and small randomized trials in
meta-analyses. Ann Intern Med. 149:2192008. View Article : Google Scholar : PubMed/NCBI
|
|
31
|
Zheng Z, Li X, Li Z and Ma X: Artificial
and bioartificial liver support systems for acute and
acute-on-chronic hepatic failure: A meta-analysis and
meta-regression. Exp Ther Med. 6:929–936. 2013.PubMed/NCBI
|