Introduction
Arthritis is a common medical problem associated
with chronic pain, which may further cause a pain syndrome
characterized by spontaneous pain, allodynia and hyperalgesia.
Joint inflammation inducing peripheral and central sensitization is
thought to be the main cause of arthritic pain (1,2), and
thus, targeting joint inflammation may be a promising therapeutic
strategy for the treatment of arthritis.
Dexmedetomidine (DEX) is a highly selective agonist
of α 2-adrenoceptors (α2AR) and has been found to bind α2AR more
avidly than clonidine (3), which has
demonstrated significant analgesic effects on acute inflammatory
pain (2), post-operative pain
(4) and neuropathic pain
unresponsive to opioid analgesics (5,6). In
addition, various α2AR agonists have been demonstrated to have
anti-nociceptive properties, and the roles of α2AR in pain
regulation have been widely investigated in the spinal cord
(7–9).
Toll-like receptor 4 (TLR4), a member of the TLR
family, is an important transmembrane protein involved in signal
transduction. Previous studies have demonstrated that TLR4
participates in the defense against Gram-negative bacteria, and
lipopolysaccharide (LPS) is its most common ligand (10). The downstream signaling molecules of
TLR4 mainly include nuclear factor (NF)-κB, an important
transcriptional factor (11). The
activated NF-κB protein enters into the nucleus, initiating the
transcription of numerous genes, including the pro-inflammatory
tumor necrosis factor (TNF)-α, interleukin (IL)-1β and IL-6
(12).
Spinal glia such as astrocytes and microglia, which
are central nervous system (CNS)-specific tissue macrophages, have
been demonstrated to have central roles in chronic pain development
and maintenance (13–15). For instance, spinal glia activation
has been implicated in the behavioral hypersensitivity induced by
spinal injury, peripheral nerve injury as well as contact with
formalin, zymosan and complete Freund's adjuvant (CFA) (16–21). In
addition, fluorocitrate- or minocycline-induced destruction of
glial function significantly attenuated mechanical allodynia and
thermal hyperalgesia in CFA-induced monoarthritic (MA) rats
(19–22). Therefore, activated glial cells are
suggested to be involved in joint inflammation-induced behavioral
hypersensitivity.
Similar to that of neurons, the membrane of glial
cells expresses α2ARs in the CNS (23–25), but
the function of α2ARs in glial cells has remained largely elusive.
The expression of glial fibrillary acid protein (GFAP) has been
demonstrated to be functionally linked to α2ARs in astrocytes
(26). However, the molecular
mechanism underlying the anti-nociceptive effects of DEX on
astrocytes has not been fully uncovered.
The present study aimed to investigate whether
repeated intrathecal injection with DEX has anti-nociceptive
effects through inhibition of TLR4 expression, NF-κB activation and
pro-inflammatory cytokine production in rats with CFA-induced ankle
joint MA.
Materials and methods
Reagents
DEX was purchased from Jiangsu Hengrui Medicine
(Lianyungang, China). VonFrey hairs were purchased from Stoelting
(Wood Dale, IL, USA). BRL44408, CFA, rabbit anti-NF-κB p65 primary
antibody (cat. no. SAB4502610), rabbit anti-α2AR primary antibody
(cat. no. A271) and mouse anti-GFAP primary antibody (cat. no.
SAB1405864) were purchased from Sigma-Aldrich (Merck KGaA,
Darmstadt, Germany). Donkey anti-rabbit immunoglobulin G (IgG; cat.
no. 711-005-152) and donkey anti-mouse IgG (cat. no. 715-005-150)
were purchased from Jackson ImmunoResearch (West Grove, PA, USA).
Rabbit anti-TLR4 primary antibody (cat. no. ab13556) was purchased
from Abcam (Cambridge, MA, USA). Rabbit anti NF-κB p65 antibody
(cat. no. 8242) and rabbit anti GAPDH antibody (cat. no. 2118) were
purchased from Cell Signaling Technology, Inc. (Danvers, MA, USA).
Horseradish peroxidase-conjugated donkey anti-mouse secondary
antibody (cat. no. sc-2318) was purchased from Santa Cruz
Biotechnology, Inc. (Dallas, TX, USA). A bicinchoninic acid (BCA)
kit, polyvinylidene difluoride (PVDF) membrane, goat anti-rabbit
secondary antibody (cat. no. 65-6120) and Super Signal West Pico
Chemiluminescent Substrate kit (cat. no. 34080) were purchased from
Thermo Fisher Scientific, Inc. (Waltham, MA, USA).
Animals
The present study was approved by the Animal Care
and Use Committee of Fudan University (Shanghai, China), and all
animal experiments were consistent with the National Institutes of
Health Guide for the Care and Use of Laboratory Animals. A total of
40 adult Sprague Dawley rats (male; weight, 200–250 g; 3 months
old) were purchased from the Experimental Animal Center of Fudan
University (Shanghai, China). These rats were housed in a
temperature- (22±2°C) and light-controlled (12-h light/dark cycle)
room with free access to food and water.
The rats were randomly divided into 5 groups of 8
animals each: The Sham group [intra-articular injection with 50 µl
sterile saline and intrathecal (i.t.) injection with 20 µl sterile
saline for 3 consecutive days], the CFA group (intra-articular
injection with 50 µl CFA to induce MA), the DEX group
[intra-articular injection with 50 µl CFA and intrathecal injection
with DEX (2.5 µg/20 µl) for 3 consecutive days post-MA], the B
group [intra-articular injection with 50 µl CFA and pre-treatment
with BRL44408 (15 µg/20 µl) for 30 min prior to i.t. injection with
DEX (2.5 µg/20 µl) for 3 consecutive days post-MA], the
epigallocatechin gallate (EGCG) group (intra-articular injection
with 50 µl CFA and intrathecal injection with 30 µg EGCG for 3
consecutive days post-MA). The Sham, DEX, B and EGCG groups were
treated by i.t. injection on 3 consecutive days after MA, and the
pain test was performed 1 h after the injection.
Drug administration
DEX (2.5 µg/20 µl) and BRL44408 (15 µg/20 µl)
(27) were diluted in normal saline
(NS, 0.9% NaCl). Lumbar puncture (LP) injection was performed as
previously described (28). Under
inhalation anesthesia using isoflurane (2% in oxygen; Abbott
Pharmaceutical Co. Ltd., Lake Bluff, IL, USA), the rats were
injected at the L5-6 interspace. The occurrence of an instantaneous
and rapid tail-flick indicated a successful puncture. Control
animals received an equivalent volume of sterile saline. Animals
then recovered in their cage prior to analgesic testing.
Induction of MA
Isoflurane was used to anesthetize the rats, and an
iodine tincture and 75% alcohol were used to sterilize the skin.
The fossa of the lateral malleolus of the fibula was located, and a
28-gauge needle was inserted from the gap between the tibiofibular
and tarsus bone into the articular cavity. CFA (50 µl) was injected
into the articular cavity. The Sham MA group was injected with
sterile saline.
Von Frey test for mechanical
allodynia
Mechanical allodynia was assessed by examining the
paw withdrawal mechanical threshold (PWMT) in response to a
calibrated series of Von Frey hairs (29). Each filament was applied five times,
each application lasted for 2 sec, and there was a 30-sec interval
between each application. A positive response to a filament was
indicated by the withdrawal of a hind paw upon application of a
particular hair for at least 3 out of 5 consecutive applications.
The PWT was defined as the smallest value of the hair force in
grams that elicited positive responses.
Hargreaves' test for thermal
hyperalgesia
Thermal hyperalgesia was studied by examining the
paw withdrawal thermal latency (PWTL) in response to a radiant heat
source. Rats in each group were put on an elevated glass platform.
A radiant heat source (Model 336; IITC Life Science, Woodland Hill,
CA, USA) was then applied to the plantar surface of the hind paw
through the glass plate. The time from onset of radiant heat
application to withdrawal of the hind paw was measured, and both
hind paws were tested independently with a 10-min interval between
trials (19,30).
Immunohistochemistry
Rats were given urethane (2 g/kg, intraperitoneally)
and perfused with 4% paraformaldehyde in 0.1 mol/l phosphate buffer
through the ascending aorta. Subsequently, the L4-5 segments of the
spinal cord were fixed in 4% paraformaldehyde at 4°C for 4 h, which
were then immersed in sucrose (10–30%) for 24 h at 4°C. Sections
(35 µm) were cut, which were then blocked in PBS with 10% donkey
serum (cat. no. ab7475; Abcam) and 0.3% Triton X-100 at room
temperature (RT) for 2 h. Subsequently, the sections were incubated
overnight at 4°C with rabbit anti-NF-κB p65 (1:200 dilution) or
mouse anti-GFAP (1:2,000 dilution) primary antibody in PBS with 1%
donkey serum and 0.3% Triton X-100. After washing with PBS for 3
times, the sections were incubated in donkey anti-rabbit IgG (1:200
dilution) for NF-κB p65 or donkey anti-mouse IgG (1:200 dilution)
for GFAP for 2 h at 4°C. The sections were observed with a
fluorescence microscope (Leica Microsystems, Wetzlar Germany).
Immunofluorescence
We chose the CFA group at 3 days to detect the
double immunofluorescence. Spinal sections were incubated with a
mixture of rabbit anti-TLR4 (1:500 dilution) and mouse anti-GFAP
(1:2,000 dilution), rabbit anti-α2AR (1:2,000 dilution) and mouse
anti-GFAP (1:2,000 dilution) overnight at 4°C, followed by a
mixture of donkey anti-rabbit IgG (1:200 dilution) for TLR4 and
α2AR and donkey anti-mouse IgG (1:200 dilution) for GFAP for 1 h at
RT. The sections were observed with a fluorescence microscope
(Leica Microsystems).
Western blot analysis
The L4-5 segments of spinal cords were dissected and
split into left and right halves from the ventral midline, which
were then cut into the dorsal and ventral horn at the level of the
central canal. The dorsal horn was then lysed with ice-cold lysis
buffer. Proteins (50 µg) in the supernatants were quantified by
using a BCA kit and separated by 12% SDS-PAGE. Proteins were
transferred onto a PVDF membrane, which was then incubated with PBS
containing 5% milk overnight at 4°C. Subsequently, the membrane was
incubated with mouse anti-GFAP (1:2,000 dilution), rabbit anti-TLR4
(1:1,000 dilution), rabbit anti NF-κB p65 (1:1,000 dilution) and
GAPDH (1:20,000 dilution) antibodies overnight at 4°C, and then
with horseradish peroxidase-conjugate donkey anti-mouse secondary
antibody (1:2,000 dilution) and goat anti-rabbit secondary antibody
(1:4,000 dilution) for 2 h at RT. Signals on the membrane were
visualized using the Super Signal West Pico Chemiluminescent
Substrate kit and exposed to X-ray films (Bio-Rad Laboratories,
Inc., Hercules, CA, USA) for 1–10 min as follows: GFAP 5 min, TLR4
8 min, NF-κB p65 10 min and GAPDH 3 min. The optical densities of
bands were analyzed by Image-Pro plus software 6.0 (Media
Cybernetics, Inc., Rockville, MD, USA) and presented as the density
ratio vs. GAPDH.
ELISA
ELISA was performed to examine the levels of IL-1β,
IL-6 and TNF-α in the L4-5 dorsal horns of the MA side using the
Rat IL-1β (cat. no. ab100768), IL-6 (cat. no. ab100772) and TNF-α
(cat. no. ab100785) ELISA kits, according to the manufacturer's
protocols.
Co-immunoprecipitation
Protein G Sepharose (10 µl) was washed with lysate
buffer for 3 times, followed by centrifugation at 3,000 × g for 3
min once. A total of 20 µg sample protein preparation from the
western blot experiment was added to the Protein G Sepharose and
incubated overnight at 4°C with gentle shaking. Protein from
monoarthritic rats of 3 days as the positive control, the normal
IgG protein as the negative control. Next, after centrifugation at
3,000 × g for 3 min at 4°C, the supernatant was carefully removed
and the Protein G Sepharose was washed with 1 ml lysate buffer for
3 times. A total of 6 µl sample buffer was added, followed by
boiling at 70°C. Finally, the samples were subjected to western
blot analysis.
Statistical analysis
We used the Sigmastat 3.1 (Systat software, Inc.,
San Jose, CA, USA) for statistical analysis. Values are expressed
as the mean ± standard error of the mean. One-way analysis of
variance (ANOVA) was used to analyze the pre-MA baseline and
pre-drug treatment measures. Two-way ANOVA (treatment time)
followed by the Holm-Sidak test was used to analyze the post-drug
time course measures for hyperalgesia. Immunohistochemical and
western blot analysis results were analyzed by Student's t-test
when comparing two groups or one-way ANOVA followed by the
Holm-Sidak test when comparing >2 groups. P<0.05 was
considered to indicate a statistically significant difference.
Results
Effects of DEX treatment on GFAP
expression as well as mechanical and thermal hypersensitivity in
rats with CFA-induced MA
As presented in Fig. 1A
and B, the measures of PWMT and PWTL in either hind paw in the
sham group did not differ prior to and after intra-articular saline
injection; however, unilateral intra-articular injection with CFA
caused significant mechanical or thermal hyperalgesia, which peaked
at 3 days after CFA injection. Mechanical and thermal hyperalgesia
significantly increased in the CFA group compared with the control
group (P<0.01; Fig. 1A and
B).
 | Figure 1.Expression of GFAP and the
development of mechanical and thermal hypersensitivity in
CFA-induced MA. Effects of i.t. injection with DEX (2.5 µg) on (A)
paw withdrawal mechanical threshold and (B) paw withdrawal thermal
latency of the ipsilateral hind paws prior to the behavioral test
on day 1–3 after surgery. **P<0.05, SHAM vs. CFA;
#P<0.01, ##P<0.05, CFA vs. DEX;
£P<0.01, ££P<0.05, CFA vs. EGCG;
&&P<0.05, B vs. DEX; $$P<0.05,
B vs. EGCG. (C and D) Western blot analysis of GFAP in the spinal
dorsal horn of rats in the Sham, CFA, DEX, EGCG and B groups.
Effects of intrathecal injection with DEX, BRL44408 and EGCG over 3
consecutive days on the GFAP levels in the spinal dorsal horn
induced by MA on (C) day 3 and (D) day 7. GAPDH served as loading
control. **P<0.05, SHAM vs. CFA; #P<0.01, DEX vs.
CFA; £P<0.01, DEX vs. B;
&&P<0.05, EGCG vs. CFA;
$P<0.01, $$P<0.05, EGCG vs. B. Groups:
Sham, i.a. injection with 50 µl sterile saline and i.t. injection
with 20 µl sterile saline for 3 consecutive days; CFA, i.a.
injection with 50 µl CFA; DEX, i.a. injection with 50 µl CFA and
i.t. injection with DEX (2.5 µg/20 µl) for 3 consecutive days; B,
i.a. injection with 50 µl CFA and pre-treatment with BRL44408 (15
µg/20 µl) 30 min prior to i.t. injection with DEX 2.5 (µg/20 µl)
for 3 consecutive days; EGCG, i.a. injection with 50 µl CFA and
i.t. injection with EGCG 30 µg for 3 consecutive days. CFA,
complete Freund's adjuvant; MA, monoarthritis; DEX,
dexmedetomidine; i.t., intrathecal; i.a., intra-articular; EGCG,
epigallocatechin gallate; B, BRL44408; GFAP, glial fibrillary acid
protein. |
Furthermore, the effects of repeated injection with
DEX on PWMT or PWTL different time-points after intra-articular
injection with CFA were determined. DEX or sterile saline were
given i.t. for 3 consecutive days after MA. Compared with that in
the CFA group, the PWMT in the DEX group was significantly
increased at 1, 3, 5 and 7 days (P<0.01; Fig. 1A). Furthermore, the PWTL was enhanced
in the DEX group at 1, 3, 5 and 7 days (P<0.01), as well as in
the EGCG group compared with that in the CFA group (Fig. 1B). In comparison with that in the B
group, the PWMT and PWTL in the DEX and in the EGCG group were
significantly enhanced at 1, 3, 5 and 7 days (P<0.01; Fig. 1A and B), which indicated that
pre-treatment with BRL44408 for 3 consecutive days abrogated the
beneficial effect of DEX.
Following CFA-induced MA, the protein expression of
GFAP was detected by western blot analysis at 3 and 7 days. The
results demonstrated that the expression of GFAP was significantly
increased in the ipsilateral spinal dorsal horn of the CFA at 3 and
7 days, which was suppressed by intrathecal injection with DEX and
EGCG for 3 consecutive days, and pre-treatment with BRL44408 for 3
consecutive days abrogated the effect of DEX (Fig. 1C and D). These findings suggested
that the upregulation of GFAP may be associated with the pain
threshold.
TLR4 expression and NF-κB p65 activity
in the spinal dorsal horn
Following CFA-induced MA, western blot analysis was
performed to examine the protein levels of TLR4 and NF-κB p65 at 3
and 7 days. The results indicated that, when compared with the sham
group, the TLR4 levels were significantly increased in the
ipsilateral spinal dorsal horn of the CFA group at 3 (P=0.0001) and
7 (P=0.00021) days; when compared with the CFA group at 3 and 7
days, the TLR4 levels were suppressed by intrathecal injection with
DEX (P=0.0007 and P=0.0015, respectively) and EGCG (P3
d=0.00082 and P7 d=0.0003) for 3 consecutive days;
when compared with the B group at 3 day, the TLR4 levels were
suppressed by intrathecal injection with DEX (P=0.0001) and EGCG
(P=0.00022). Furthermore, pretreatment with BRL44408 for 3
consecutive days abrogated the effect of DEX (Fig. 2A). Furthermore, when compared with
the sham group, NF-κB p65 levels were significantly increased in
the ipsilateral spinal dorsal horn of the CFA and B groups at 3 and
7 days, which was suppressed by intrathecal injection with DEX and
EGCG for 3 consecutive days, and pretreatment with BRL44408 for 3
consecutive days abrogated the effect of DEX (Fig. 2B). These findings suggested that the
upregulation of TLR4 and NF-κB p65 may also be associated with the
pain thresholds.
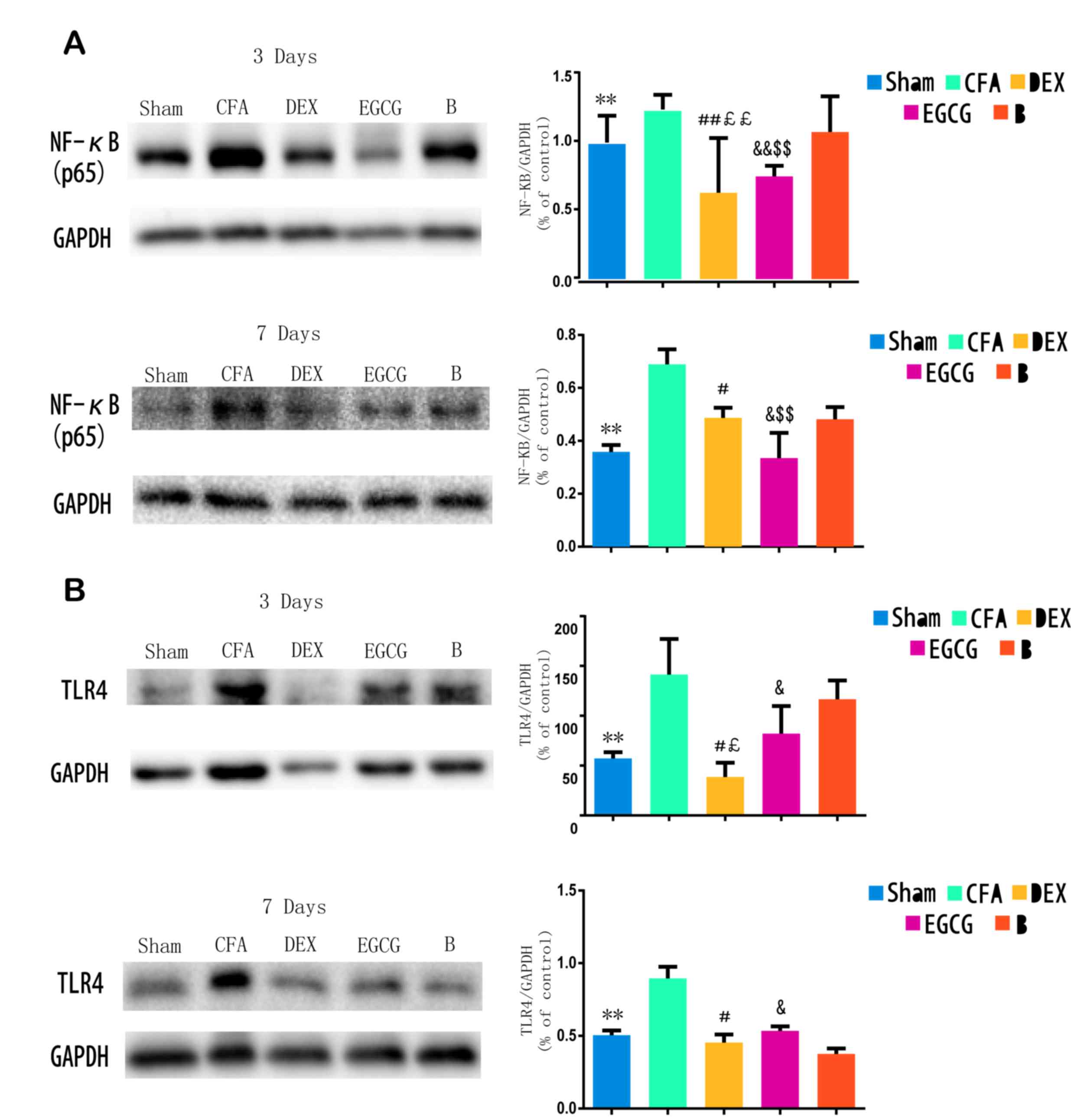 | Figure 2.Western blot analysis of TLR4
expression and NF-κB p65 activity in spinal dorsal horn of Sham,
CFA, DEX, EGCG and B groups. (A and B) Effects of intrathecal
injection with DEX, BRL44408 and EGCG for 3 consecutive days on the
(A) NF-κB p65 and (B) TLR4 levels in the spinal dorsal horn induced
by MA on days 3 and 7 after MA, respectively. GAPDH served as
loading control. **P<0.05, SHAM vs. CFA; #P<0.01
and ##P<0.05, DEX vs. CFA; £P<0.01 and
££P<0.05, DEX vs. B; &P<0.01 and
&&P<0.05, EGCG vs. CFA; $P<0.01
and $$P<0.05, EGCG vs. B. Sham, i.a. injection with
50 µl sterile saline and i.t. injection with 20 µl sterile saline
for 3 consecutive days; CFA, i.a. injection with 50 µl CFA; DEX,
i.a. injection with 50 µl CFA and i.t. injection with DEX (2.5
µg/20 µl) for 3 consecutive days; B, i.a. injection with 50 µl CFA
and pre-treatment with BRL44408 (15 µg/20 µl) 30 min prior to i.t.
injection with DEX 2.5 (µg/20 µl) for 3 consecutive days; EGCG,
i.a. injection with 50 µl CFA and i.t. injection with EGCG 30 µg
for 3 consecutive days. i.t., intrathecal; i.a., intra-articular;
TLR, Toll-like receptor; NF, nuclear factor; CFA, complete Freund's
adjuvant; MA, monoarthritis; DEX, dexmedetomidine; EGCG,
epigallocatechin gallate; B, BRL44408. |
Immunohistochemical analysis of NF-κB
p65 in the spinal dorsal horn
After the pain test at 3 days after MA, 3 rats in
each group were sacrificed and immunohistochemistry was performed
to examine the expression of NF-κB p65. As indicated in Fig. 3, NF-κB p65-positive cells were
distributed in the spinal dorsal horn. In addition, the CFA group
contained a markedly higher amount of positive staining for NF-κB
p65 in the spinal dorsal horn compared with that in the sham group,
while treatment with DEX and EGCG significantly reduced the
expression of NF-κB p65 at 3 days after MA (Fig. 3).
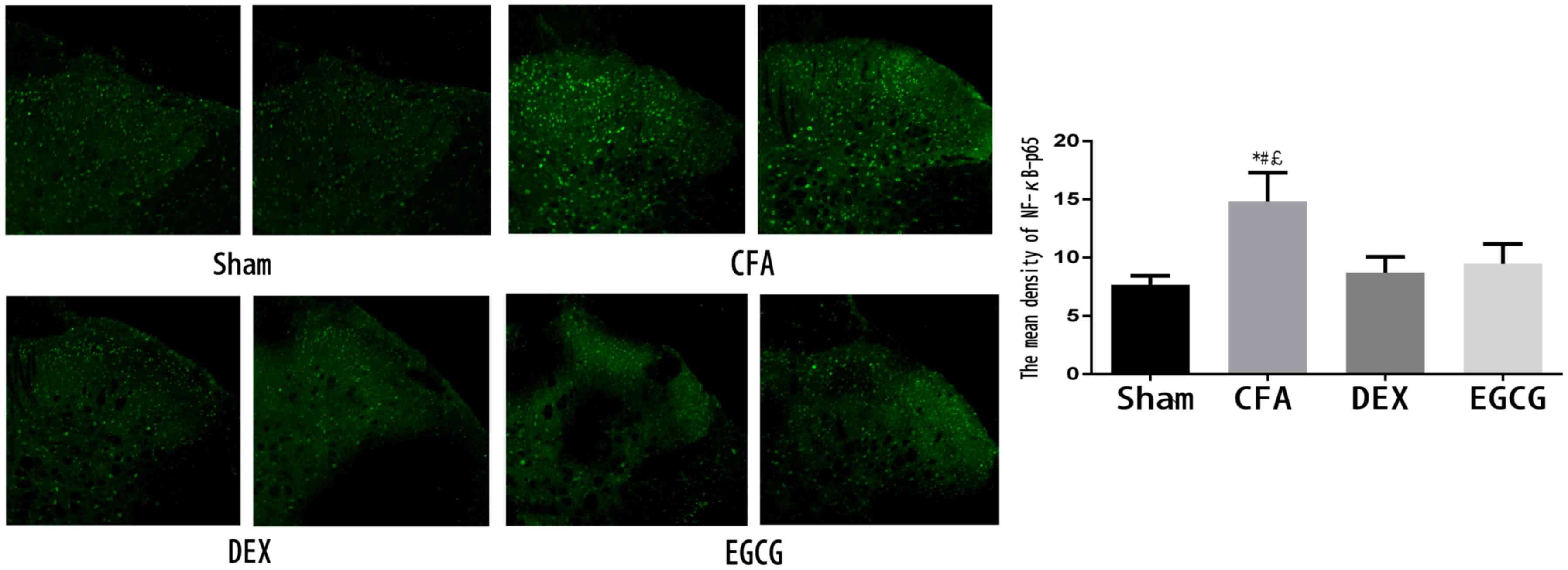 | Figure 3.Activation of NF-κB p65 in the spinal
dorsal horn on day 3 after MA in the Sham, CFA, DEX and EGCG
groups. Immunohistochemistry revealed markedly decreased of NF-κB
p65 in the DEX and EGCG groups on day 3 after MA. *P<0.01, CFA
vs. SHAM; #P<0.01, CFA vs. DEX;
£P<0.01, CFA vs. EGCG. Magnification, ×20. Sham, i.a.
injection with 50 µl sterile saline and i.t. injection with 20 µl
sterile saline for 3 consecutive days; CFA, i.a. injection with 50
µl CFA; DEX, i.a. injection with 50 µl CFA and i.t. injection with
DEX (2.5 µg/20 µl) for 3 consecutive days; EGCG, i.a. injection
with 50 µl CFA and i.t. injection with EGCG 30 µg for 3 consecutive
days. i.t., intrathecal; i.a., intra-articular; NF, nuclear factor;
CFA, complete Freund's adjuvant; MA, monoarthritis; DEX,
dexmedetomidine; EGCG, epigallocatechin gallate. |
Levels of IL-1β, IL-6 and TNF-α in the
spinal dorsal horn
The contents of IL-1β, TNF-α and IL-6 were
determined by ELISA. The results demonstrated that, when compared
with the Sham group, the contents of IL-1β (P3 d=0.006
and P7 d=0.005), TNF-α (P3 d=0.002 and
P7 d=0.012) and IL-6 (P3 d=0.0024 and P7
d=0.0022) increased significantly in the CFA group. When
compared with the CFA and B groups, the expression of IL-1β was
markedly decreased in the DEX (vs. CFA, P3 d=0.0001 and
P7 d=0.00022; vs. B, P3 d=0.0001 and P7
d=0.0003) group at 3 and 7 days, as well as in the EGCG (vs.
CFA, P3 d=0.0007 and P7 d=0.0003; vs. B,
P3 d=0.0001 and P7 d=0.015) group at 3 and 7
days, (Fig. 4A). The expression of
TNF-α was significantly decreased in the DEX (vs. CFA, P3
d=0.0002 and P7 d=0.00034; vs. B, P3
d=0.00016 and P7 d=0.0009) group at 3 and 7 days,
as well as in the EGCG (vs. CFA, P3 d=0.00012 and
P7 d=0.015; vs. B, P3 d=0.00026 and P7
d=0.03) group at 3 and 7 days, when compared with the CFA and
B group at 3 and 7 days (Fig. 4B).
Compared with that in the CFA group, the content of IL-6 was
significantly decreased in the DEX (P3 d=0.0007 and
P7 d=0.0022) and EGCG (P3 d=0.0001 and
P7 d=0.018) groups at 3 and 7 days; furthermore, IL-6 in
the B group at 3 (P=0.0004) and 7 (P=0.012) days was significantly
higher than that in the DEX group (Fig.
4C).
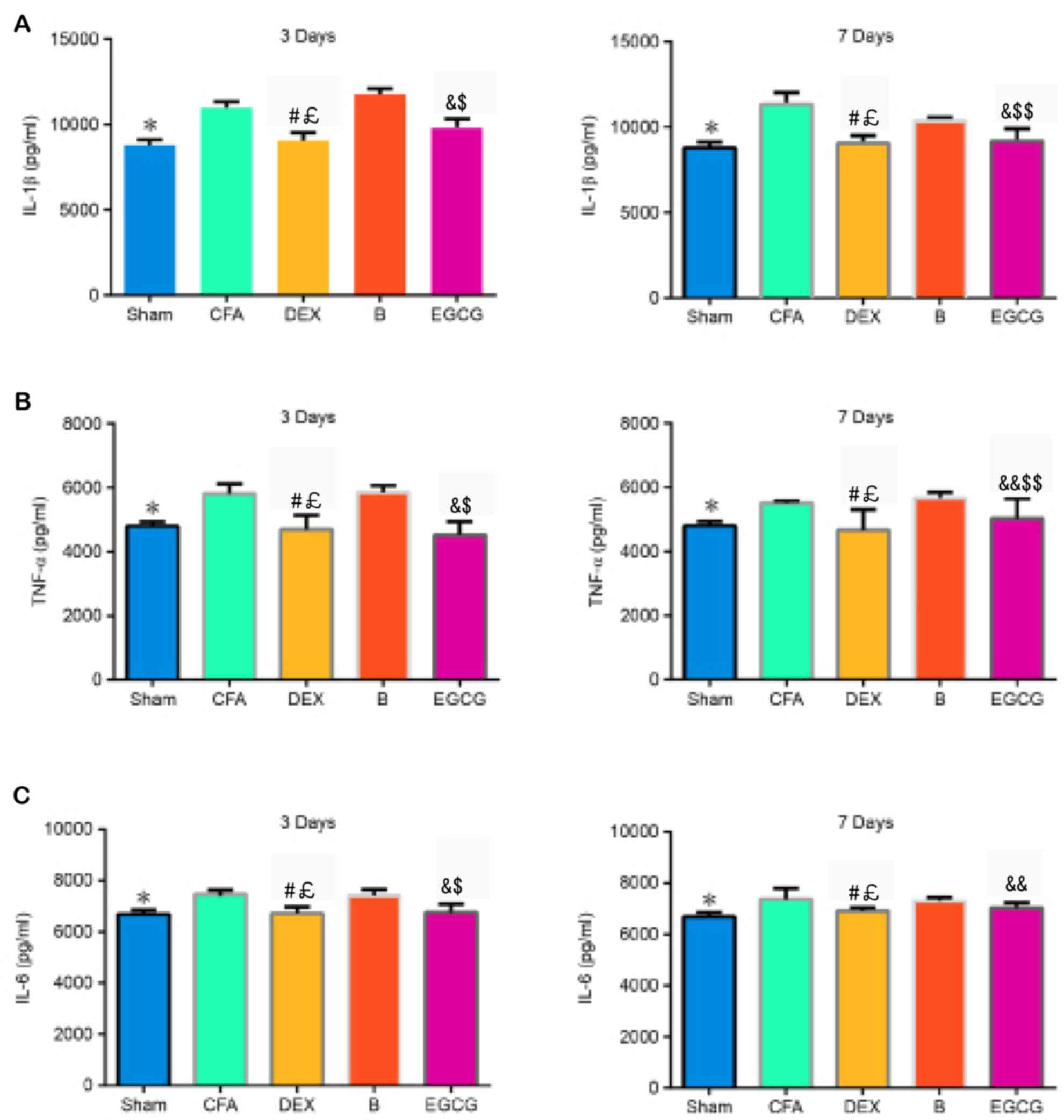 | Figure 4.Effects of intrathecal administration
of DEX, BRL44408 or EGCG on the expression of (A) IL-1β, (B) TNF-α
and (C) IL-6 in the L4-5 dorsal horns of the MA side of MA rats.
Values are expressed as the mean ± standard deviation. *P<0.01,
SHAM vs. CFA; #P<0.01, DEX vs. CFA;
£P<0.01, DEX vs. B; &P<0.01,
&&P<0.05, EGCG vs. CFA;
$P<0.01, $$P<0.05, EGCG vs. B. Groups:
Sham, i.a. injection with 50 µl sterile saline and i.t. injection
with 20 µl sterile saline for 3 consecutive days; CFA, i.a.
injection with 50 µl CFA; DEX, i.a. injection with 50 µl CFA and
i.t. injection with DEX (2.5 µg/20 µl) for 3 consecutive days; B,
i.a. injection with 50 µl CFA and pre-treatment with BRL44408 (15
µg/20 µl) 30 min prior to i.t. injection with DEX 2.5 (µg/20 µl)
for 3 consecutive days; EGCG, i.a. injection with 50 µl CFA and
i.t. injection with EGCG 30 µg for 3 consecutive days. i.t.,
intrathecal; i.a., intra-articular; CFA, complete Freund's
adjuvant; MA, monoarthritis; DEX, dexmedetomidine; EGCG,
epigallocatechin gallate; B, BRL44408; IL, interleukin; TNF, tumor
necrosis factor. |
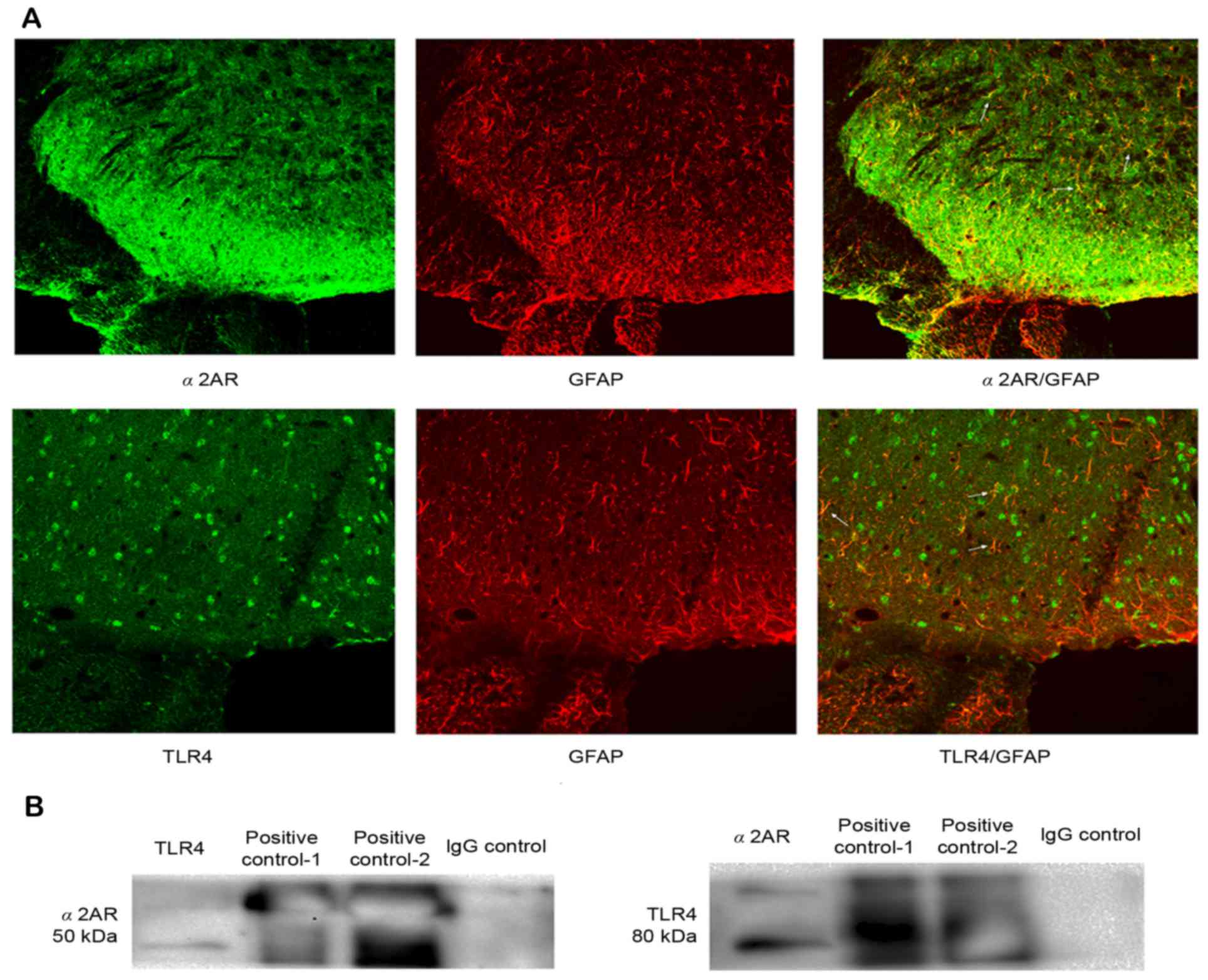
Double immunofluorescence of GFAP and
TLR4 and α2AR with GFAP in the spinal dorsal horn, and the
interaction between α2AR and TLR4
We chose the CFA group at 3 days to detect the
double immunofluorescence. TLR4 and α2AR immunoreactivity
co-localized with GFAP, indicating that TLR4 and α2AR were
expressed in the same cell of the spinal dorsal horn (Fig. 5A). The above results demonstrated
that i.t. administration of DEX for 3 consecutive days not only
attenuated hyperalgesia, but also inhibited TLR4 expression and
NF-κB p65 activity in the spinal cord. Therefore, it was speculated
that α2AR possibly interacts with TLR4 via a certain unknown
mechanism. Therefore, the present study first detected whether α2AR
and TLR4 were expressed in the same cell of the spinal dorsal horn.
Immunofluorescence confirmed that α2AR and TLR4 were significantly
co-expressed in astrocytes of the spinal dorsal horn, which
provided possible histological evidence for the interaction between
α2AR and TLR4. Furthermore, the interaction between α2AR and TLR4
was assessed through co-immunoprecipitation. The results indicated
that after immunoprecipitation, TLR4 was detected with the specific
antibody for α2AR, and α2AR was also detected with the specific
antibody for TLR4, but these proteins were not detected with normal
IgG as a negative control. These results indicated that α2AR may
interact with TLR4 in the spinal dorsal horn at a certain level
(Fig. 5B).
Discussion
The present study investigated the anti-nociceptive
effects of DEX on inflammatory responses in CFA-induced MA rats. It
was demonstrated that intrathecal administration of 2.5 µg DEX for
3 consecutive days significantly attenuated mechanical and thermal
hyperalgesia, decreased the production of IL-1β, IL-6 and TNF-α,
and suppressed the activation of the TLR4/NF-κB p65 pathway in the
spinal tissues of rats with CFA-induced MA. All these effects were
markedly reversed by pre-treatment with BRL44408, a selective α2AR
antagonist.
α2AR is located diffusely in the nervous system,
including primary afferent nerves, spinal dorsal horn neurons and
the brainstem (31). As a highly
specific potent and selective α2AR agonist, DEX has been widely
used for sedation and analgesia in clinical therapy (2). The main site of action of α2AR
agonists, including DEX, is the spinal cord (32,33). DEX
exerts dose-dependent analgesic effects against inflammation as
well as neuropathic and post-operative pain. To further reveal the
effect of DEX on neuropathic pain, the present study investigated
the anti-nociceptive effects of DEX in rats with CFA-induced MA,
and found that repeated intrathecal administration of DEX for 3
consecutive days after CFA injection, starting from the early phase
of inflammation, significantly attenuated mechanical and thermal
hyperalgesia. Although the present and previous studies have
demonstrated the anti-nociceptive effects of DEX on CFA-induced MA
rats, the underlying molecular mechanisms still remain largely
elusive (2,34).
TLR4, a transmembrane receptor protein, functions as
a signaling transduction molecule through its extracellular
leucine-rich repeated domains and a cytoplasmic signaling domain.
It has been demonstrated that TLR4 not only has a key role in the
innate immune response, but also participates in the triggering of
adaptive immunity (35–38). While LPS is well recognized as a
major ligand of TLR4 through interaction with CD14, endogenous
ligands for TLR4 also likely exist. For instance, the association
of TLR4 with myeloid differentiation factor 88 (MYD88) has been
demonstrated to activate the TNF receptor- and IL-1
receptor-associated kinases, which further leads to inflammatory
responses (35,39).
NF-κB, a key nuclear transcription factor, generally
consists of the p50 and p65 subunits. It has been well-established
that NF-κB regulates the expression of pro-inflammatory cytokines,
and thus has an essential role in immune and inflammatory
responses. Furthermore, abnormal activation of the NF-κB signaling
has been implicated in the pathogenesis of acute and chronic
inflammatory diseases (40,41). In the inactive state, NF-κB is
located in the cytosol through binding to NF-κB inhibitory protein
(IκBα). Activation of NF-κB induces the release and degradation of
IκBα from the dimeric complex, followed by phosphorylation of NF-κB
p65 as well as its translocation into the nucleus (42). Once entering into the nucleus, NF-κB
initiates the gene transcription of pro-inflammatory cytokines,
including IL-1β, IL-6 and TNF-α (43).
Furthermore, TLR4 mainly activates the downstream
effector NF-κB through MYD88-dependent or -independent pathways. In
the present study, the MA model was established by CFA. The
findings indicated that TLR4 expression and NF-κB p65 activation
were significantly upregulated in the spinal cord of MA rats,
accompanied with mechanical and thermal hyperalgesia. Furthermore,
treatment with DEX significantly decreased the CFA-induced
upregulation of TLR4 expression and NF-κB p65 activation in rat
spinal cords, along with attenuated mechanical and thermal
hyperalgesia, as well as reduced production of pro-inflammatory
IL-1β, IL-6 and TNF-α. Since TLR4 is an essential upstream sensor
for NF-κB p65 activation, and NF-κB p65 activation increases the
production of these inflammatory cytokines, it is possible that DEX
reduces the production of these inflammatory cytokines by
inhibiting the activation of the TLR4/NF-κB p65 signaling pathway
in spinal cord of MA rats induced by CFA. These results suggested
that inhibition of TLR4/NF-κB p65 signaling is a probable mechanism
through which DEX inhibits the production of inflammatory cytokines
and thus exerts its anti-inflammatory effects, which contribute to
its anti-nociceptive effects in CFA-induced MA rats.
In conclusion, the present study demonstrated that
DEX attenuates mechanical and thermal hyperalgesia in rats with
CFA-induced MA, probably through decreasing the production of
pro-inflammatory cytokines. The anti-nociceptive effect of DEX may
be associated with downregulation of TLR4 and NF-κB signaling.
Furthermore, α2AR interacted with TLR4, which may have compromised
the activation of the TLR4 pathway. These properties are probably
associated with the anti-nociceptive effects of DEX in the CNS.
References
|
1
|
Schaible HG, Ebersberger A and Von Banchet
GS: Mechanisms of pain in arthritis. Ann N Y Acad Sci. 966:343–354.
2002. View Article : Google Scholar : PubMed/NCBI
|
|
2
|
Xu B, Zhang WS, Yang JL, Xu H, Deng XM and
Zhang YQ: Dexmedetomidine blocks thermal hyperalgesia and spinal
glial activation in rat model of monoarthritis. Acta Pharmacol Sin.
31:523–530. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
3
|
Virtanen R, Savola JM, Saano V and Nyman
L: Characterization of the selectivity, specificity and potency of
medetomidine as an alpha 2-adrenoceptor agonist. Eur J Pharmacol.
150:9–14. 1988. View Article : Google Scholar : PubMed/NCBI
|
|
4
|
Shimode N, Fukuoka T, Tanimoto M, Tashiro
C, Tokunaga A and Noguchi K: The effects of dexmedetomidine and
halothane on Fos expression in the spinal dorsal horn using a rat
postoperative pain model. Neurosci Lett. 343:45–48. 2003.
View Article : Google Scholar : PubMed/NCBI
|
|
5
|
Malmberg AB, Hedley LR, Jasper JR, Hunter
JC and Basbaum AI: Contribution of alpha(2) receptor subtypes to
nerve injury-induced pain and its regulation by dexmedetomidine. Br
J Pharmacol. 132:1827–1836. 2001. View Article : Google Scholar : PubMed/NCBI
|
|
6
|
Xu M, Kontinen VK and Kalso E: Effects of
radolmidine, a novel alpha2-adrenergic agonist compared with
dexmedetomidine in different pain models in the rat.
Anesthesiology. 93:473–481. 2000. View Article : Google Scholar : PubMed/NCBI
|
|
7
|
Lee HG, Choi JI, Kim YO and Yoon MH: The
role of alpha-2 adrenoceptor subtype in the antiallodynic effect of
intraplantar dexmedetomidine in a rat spinal nerve ligation model.
Neurosci Lett 557 Pt B. 1–122. 2013.
|
|
8
|
Paqueron X, Conklin D and Eisenach JC:
Plasticity in action of intrathecal clonidine to mechanical but not
thermal nociception after peripheral nerve injury. Anesthesiology.
99:199–204. 2003. View Article : Google Scholar : PubMed/NCBI
|
|
9
|
Pertovaara A and Wei H: Attenuation of
ascending nociceptive signals to the rostroventromedial medulla
induced by a novel alpha2-adrenoceptor agonist, MPV-2426, following
intrathecal application in neuropathic rats. Anesthesiology.
92:1082–1092. 2000. View Article : Google Scholar : PubMed/NCBI
|
|
10
|
O'Neill L: The Toll/interleukin-1 receptor
domain: A molecular switch for inflammation and host defence.
Biochem Soc Trans. 28:557–563. 2000. View Article : Google Scholar : PubMed/NCBI
|
|
11
|
Akira S and Takeda K: Toll-like receptor
signalling. Nat Rev Immunol. 4:499–511. 2004. View Article : Google Scholar : PubMed/NCBI
|
|
12
|
Baeuerle PA and Henkel T: Function and
activation of NF-kappa B in the immune system. Annu Rev Immunol.
12:141–179. 1994. View Article : Google Scholar : PubMed/NCBI
|
|
13
|
Watkins LR, Milligan ED and Maier SF:
Glial activation: A driving force for pathological pain. Trends
Neurosci. 24:450–455. 2001. View Article : Google Scholar : PubMed/NCBI
|
|
14
|
Tsuda M, Inoue K and Salter MW:
Neuropathic pain and spinal microglia: A big problem from molecules
in ‘small’ glia. Trends Neurosci. 28:101–107. 2005. View Article : Google Scholar : PubMed/NCBI
|
|
15
|
Cao H and Zhang YQ: Spinal glial
activation contributes to pathological pain states. Neurosci
Biobehav Rev. 32:972–983. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
16
|
Gwak YS, Kang J, Unabia GC and Hulsebosch
CE: Spatial and temporal activation of spinal glial cells: Role of
gliopathy in central neuropathic pain following spinal cord injury
in rats. Exp Neurol. 234:362–372. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
17
|
Milligan ED, Twining C, Chacur M,
Biedenkapp J, O'Connor K, Poole S, Tracey K, Martin D, Maier SF and
Watkins LR: Spinal glia and proinflammatory cytokines mediate
mirror-image neuropathic pain in rats. J Neurosci. 23:1026–1040.
2003.PubMed/NCBI
|
|
18
|
Ledeboer A, Sloane EM, Milligan ED, Frank
MG, Mahony JH, Maier SF and Watkins LR: Minocycline attenuates
mechanical allodynia and proinflammatory cytokine expression in rat
models of pain facilitation. Pain. 115:71–83. 2005. View Article : Google Scholar : PubMed/NCBI
|
|
19
|
Sun S, Cao H, Han M, Li TT, Pan HL, Zhao
ZQ and Zhang YQ: New evidence for the involvement of spinal
fractalkine receptor in pain facilitation and spinal glial
activation in rat model of monoarthritis. Pain. 129:64–75. 2007.
View Article : Google Scholar : PubMed/NCBI
|
|
20
|
Shan S, Qi-Liang MY, Hong C, Tingting L,
Mei H, Haili P, Yan-Qing W, Zhi-Qi Z and Yu-Qiu Z: Is functional
state of spinal microglia involved in the anti-allodynic and
anti-hyperalgesic effects of electroacupuncture in rat model of
monoarthritis? Neurobiol Dis. 26:558–568. 2007. View Article : Google Scholar : PubMed/NCBI
|
|
21
|
Sun S, Cao H, Han M, Li TT, Zhao ZQ and
Zhang YQ: Evidence for suppression of electroacupuncture on spinal
glial activation and behavioral hypersensitivity in a rat model of
monoarthritis. Brain Res Bull. 75:83–93. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
22
|
Sun S, Chen WL, Wang PF, Zhao ZQ and Zhang
YQ: Disruption of glial function enhances electroacupuncture
analgesia in arthritic rats. Exp Neurol. 198:294–302. 2006.
View Article : Google Scholar : PubMed/NCBI
|
|
23
|
Mori K, Ozaki E, Zhang B, Yang L, Yokoyama
A, Takeda I, Maeda N, Sakanaka M and Tanaka J: Effects of
norepinephrine on rat cultured microglial cells that express
alpha1, alpha2, beta1 and beta2 adrenergic receptors.
Neuropharmacology. 43:1026–1034. 2002. View Article : Google Scholar : PubMed/NCBI
|
|
24
|
Peng L, Yu AC, Fung KY, Prévot V and Hertz
L: Alpha-adrenergic stimulation of ERK phosphorylation in
astrocytes is alpha(2)-specific and may be mediated by
transactivation. Brain Res. 978:65–71. 2003. View Article : Google Scholar : PubMed/NCBI
|
|
25
|
Heal DJ, Butler SA, Prow MR and Buckett
WR: Quantification of presynaptic alpha 2-adrenoceptors in rat
brain after short-term DSP-4 lesioning. Eur J Pharmacol. 249:37–41.
1993. View Article : Google Scholar : PubMed/NCBI
|
|
26
|
Conway EL, Gundlach AL and Craven JA:
Temporal changes in glial fibrillary acidic protein messenger RNA
and [3H]PK11195 binding in relation to imidazoline-I2-receptor and
alpha 2-adrenoceptor binding in the hippocampus following transient
global forebrain ischaemia in the rat. Neuroscience. 82:805–817.
1998. View Article : Google Scholar : PubMed/NCBI
|
|
27
|
Li SS, Zhang WS, Ji D, Zhou YL, Li H, Yang
JL, Xiong YC, Zhang YQ and Xu H: Involvement of spinal microglia
and interleukin-18 in the anti-nociceptive effect of
dexmedetomidine in rats subjected to CCI. Neurosci Lett. 560:21–25.
2014. View Article : Google Scholar : PubMed/NCBI
|
|
28
|
Xu JJ, Walla BC, Diaz MF, Fuller GN and
Gutstein HB: Intermittent lumbar puncture in rats: A novel method
for the experimental study of opioid tolerance. Anesth Analg.
103:714–720. 2006. View Article : Google Scholar : PubMed/NCBI
|
|
29
|
Chu YX, Zhang YQ and Zhao ZQ: Involvement
of microglia and interleukin-18 in the induction of long-term
potentiation of spinal nociceptive responses induced by tetanic
sciatic stimulation. Neurosci Bull. 28:49–60. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
30
|
Yang JL, Xu B, Li SS, Zhang WS, Xu H, Deng
XM and Zhang YQ: Gabapentin reduces CX3CL1 signaling and blocks
spinal microglial activation in monoarthritic rats. Mol Brain.
5:182012. View Article : Google Scholar : PubMed/NCBI
|
|
31
|
Unnerstall JR, Kopajtic TA and Kuhar MJ:
Distribution of alpha 2 agonist binding sites in the rat and human
central nervous system: Analysis of some functional, anatomic
correlates of the pharmacologic effects of clonidine and related
adrenergic agents. Brain Res. 319:69–101. 1984. View Article : Google Scholar : PubMed/NCBI
|
|
32
|
Liu L, Ji F, Liang J, He H, Fu Y and Cao
M: Inhibition by dexmedetomidine of the activation of spinal dorsal
horn glias and the intracellular ERK signaling pathway induced by
nerve injury. Brain Res. 1427:1–9. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
33
|
Buerkle H and Yaksh TL: Pharmacological
evidence for different alpha 2-adrenergic receptor sites mediating
analgesia and sedation in the rat. Br J Anaesth. 81:208–215. 1998.
View Article : Google Scholar : PubMed/NCBI
|
|
34
|
Xu B, Zhang WS, Yang JL, Lû N, Deng XM, Xu
H and Zhang YQ: Evidence for suppression of spinal glial activation
by dexmedetomidine in a rat model of monoarthritis. Clin Exp
Pharmacol Physiol. 37:e158–e166. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
35
|
Wu Y, Liu Y, Huang H, Zhu Y, Zhang Y, Lu F
and Zhou C, Huang L, Li X and Zhou C: Dexmedetomidine inhibits
inflammatory reaction in lung tissues of septic rats by suppressing
TLR4/NF-κB pathway. Mediators Inflamm. 2013:5621542013. View Article : Google Scholar : PubMed/NCBI
|
|
36
|
Bettoni I, Comelli F, Rossini C, Granucci
F, Giagnoni G, Peri F and Costa B: Glial TLR4 receptor as new
target to treat neuropathic pain: Efficacy of a new receptor
antagonist in a model of peripheral nerve injury in mice. Glia.
56:1312–1319. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
37
|
Saito O, Svensson CI, Buczynski MW, Wegner
K, Hua XY, Codeluppi S, Schaloske RH, Deems RA, Dennis EA and Yaksh
TL: Spinal glial TLR4-mediated nociception and production of
prostaglandin E(2) and TNF. Br J Pharmacol. 160:1754–1764. 2010.
View Article : Google Scholar : PubMed/NCBI
|
|
38
|
Wu GJ, Chen TL, Ueng YF and Chen RM:
Ketamine inhibits tumor necrosis factor-alpha and interleukin-6
gene expressions in lipopolysaccharide-stimulated macrophages
through suppression of toll-like receptor 4-mediated c-Jun
N-terminal kinase phosphorylation and activator protein-1
activation. Toxicol Appl Pharmacol. 228:105–113. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
39
|
Feng Y and Longmore GD: The LIM protein
Ajuba influences interleukin-1-induced NF-kappaB activation by
affecting the assembly and activity of the protein kinase
Czeta/p62/TRAF6 signaling complex. Mol Cell Biol. 25:4010–4022.
2005. View Article : Google Scholar : PubMed/NCBI
|
|
40
|
Wright JG and Christman JW: The role of
nuclear factor kappa B in the pathogenesis of pulmonary diseases:
Implications for therapy. Am J Respir Med. 2:211–219. 2003.
View Article : Google Scholar : PubMed/NCBI
|
|
41
|
Wu Y, Li W and Zhou C, Lu F, Gao T, Liu Y,
Cao J, Zhang Y, Zhang Y and Zhou C: Ketamine inhibits
lipopolysaccharide-induced astrocytes activation by suppressing
TLR4/NF-κB pathway. Cell Physiol Biochem. 30:609–617. 2012.
View Article : Google Scholar : PubMed/NCBI
|
|
42
|
Baeuerle PA and Baltimore D: I kappa B: A
specific inhibitor of the NF-kappa B transcription factor. Science.
242:540–546. 1988. View Article : Google Scholar : PubMed/NCBI
|
|
43
|
Grilli M, Chiu JJ and Lenardo MJ: NF-kappa
B and Rel: Participants in a multiform transcriptional regulatory
system. Int Rev Cytol. 143:1–62. 1993. View Article : Google Scholar : PubMed/NCBI
|