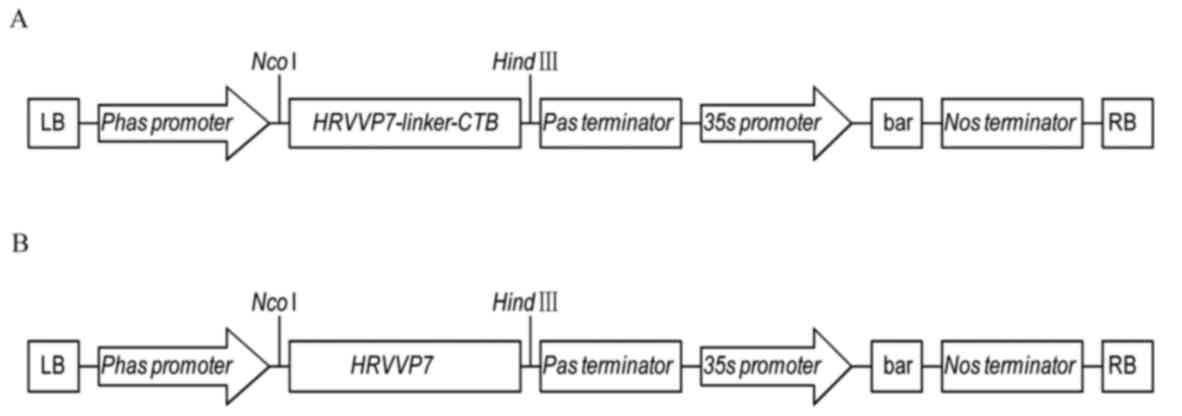
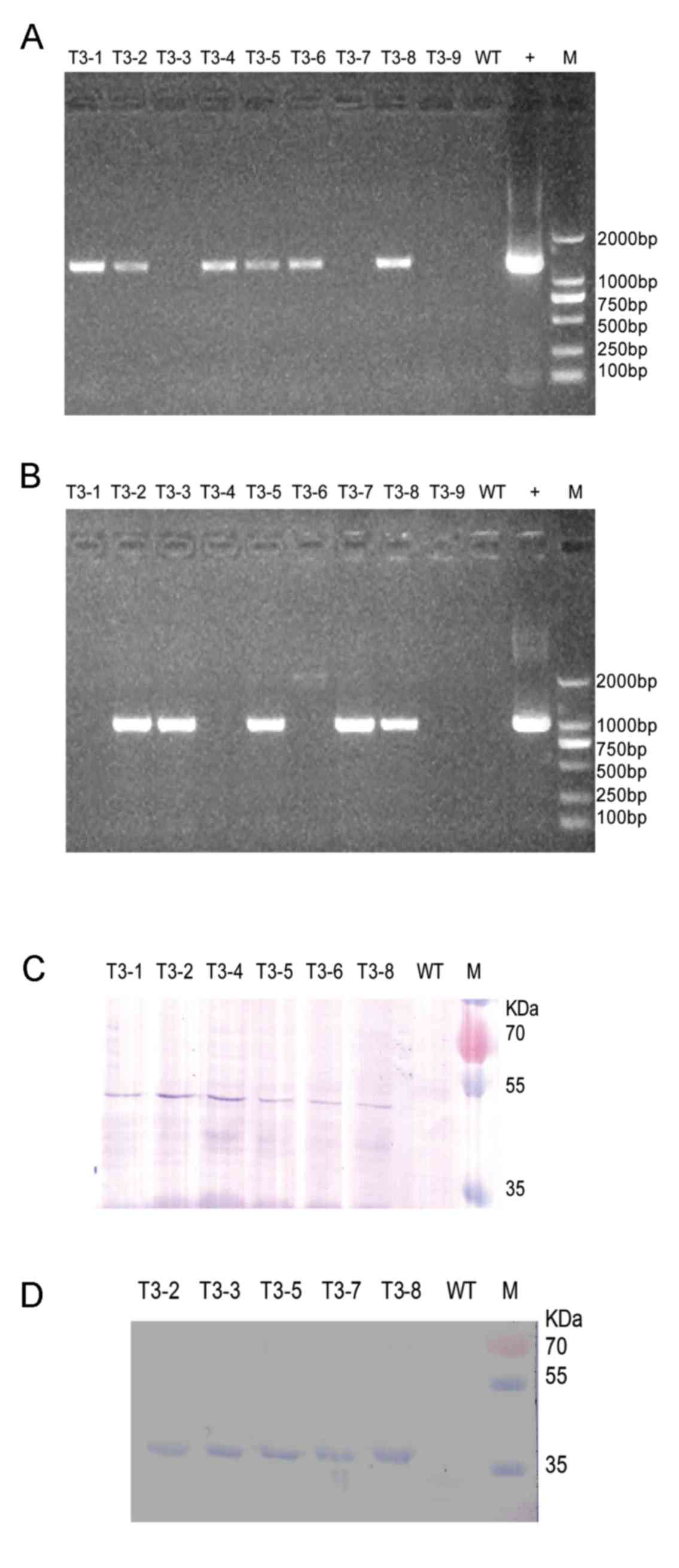
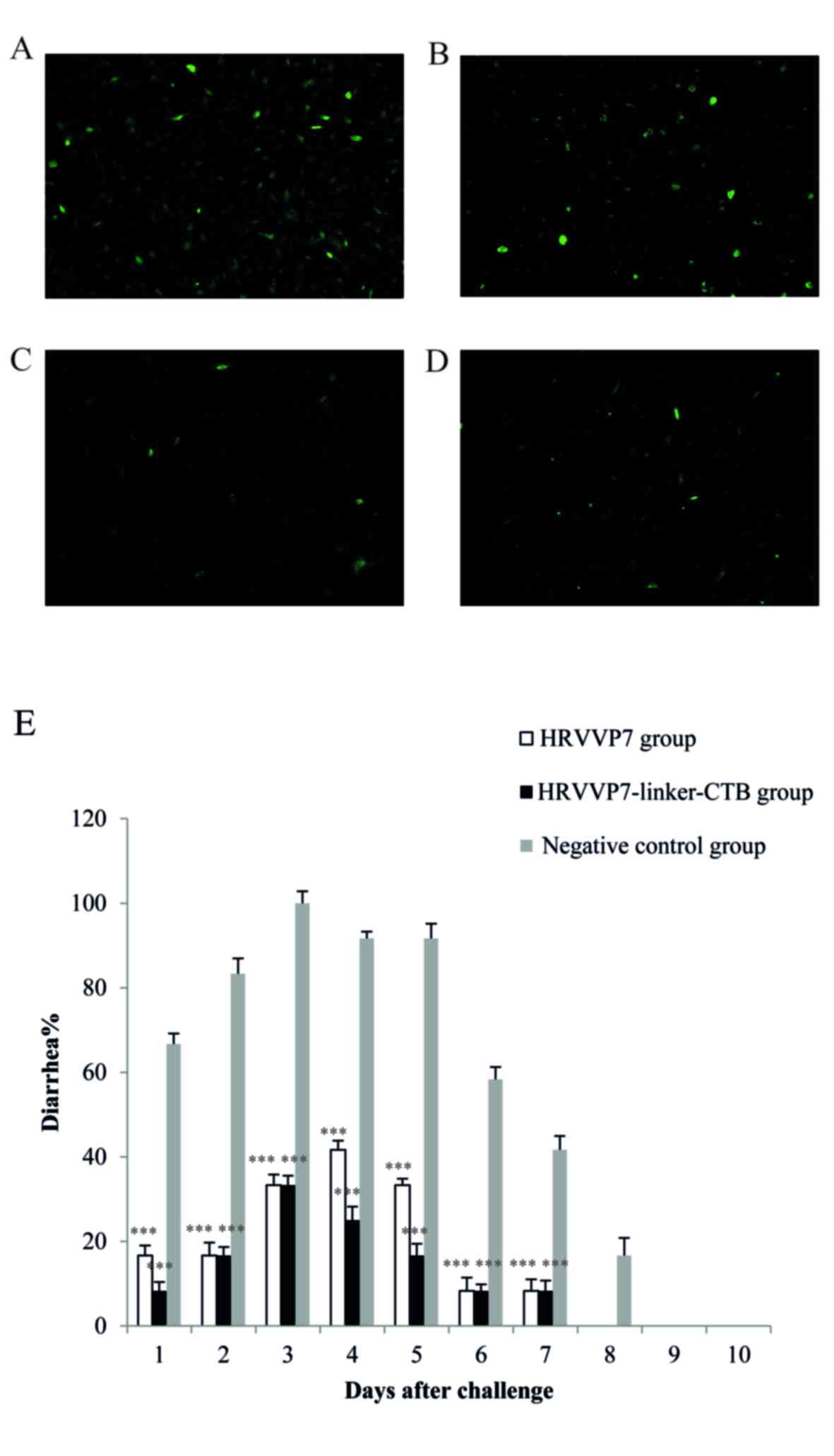
|
1
|
Parashar UD, Gibson CJ, Bresee JS and
Glass RI: Rotavirus and severe childhood diarrhea. Emerg Infect
Dis. 12:304–306. 2006. View Article : Google Scholar : PubMed/NCBI
|
|
2
|
Parashar UD, Bresee JS and Glass RI: The
global burden of diarrhoeal disease in children. Bull World Health
Organ. 81:2362003.PubMed/NCBI
|
|
3
|
Charles MD, Holman RC, Curns AT, Parashar
UD, Glass RI and Bresee JS: Hospitalizations associated with
rotavirus gastroenteritis in the United States, 1993–2002. Pediatr
Infect Dis J. 25:489–493. 2006. View Article : Google Scholar : PubMed/NCBI
|
|
4
|
Madhi SA, Cunliffe NA, Steele D, Witte D,
Kirsten M, Louw C, Ngwira B, Victor JC, Gillard PH, Cheuvart BB, et
al: Effect of human rotavirus vaccine on severe diarrhea in African
infants. N Engl J Med. 362:289–298. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
5
|
Richardson V, Hernandez-Pichardo J,
Quintanar-Solares M, Esparza-Aguilar M, Johnson B, Gomez-Altamirano
CM, Parashar U and Patel M: Effect of rotavirus vaccination on
death from childhood diarrhea in Mexico. N Engl J Med. 362:299–305.
2010. View Article : Google Scholar : PubMed/NCBI
|
|
6
|
Greenberg HB, Valdesuso J, van Wyke K,
Midthun K, Walsh M, McAuliffe V, Wyatt RG, Kalica AR, Flores J and
Hoshino Y: Production and preliminary characterization of
monoclonal antibodies directed at two surface proteins of rhesus
rotavirus. J Virol. 47:267–275. 1983.PubMed/NCBI
|
|
7
|
Andrew ME, Boyle DB, Coupar BE, Reddy D,
Bellamy AR and Both GW: Vaccinia-rotavirus VP7 recombinants protect
mice against rotavirus-induced diarrhoea. Vaccine. 10:185–191.
1992. View Article : Google Scholar : PubMed/NCBI
|
|
8
|
Dyall-Smith ML, Lazdins I, Tregear GW and
Holmes IH: Location of the major antigenic sites involved in
rotavirus serotype-specific neutralization. Proc Natl Acad Sci USA.
83:3465–3468. 1986. View Article : Google Scholar : PubMed/NCBI
|
|
9
|
Offit PA, Boyle DB, Both GW, Hill NL,
Svoboda YM, Cunningham SL, Jenkins RJ and McCrae MA: Outer capsid
glycoprotein vp7 is recognized by cross-reactive,
rotavirus-specific, cytotoxic T lymphocytes. Virology. 184:563–568.
1991. View Article : Google Scholar : PubMed/NCBI
|
|
10
|
Łucka M, Kowalczyk T, Szemraj J and
Sakowicz T: Plants as an alternative source of therapeutic
proteins. Postepy Hig Med Dosw (Online). 69:362–373. 2015.(In
Polish). View Article : Google Scholar : PubMed/NCBI
|
|
11
|
Yusibov V and Streatfield SJ:
Plant-produced microbial vaccines: Alexander V. Karasev, editor:
Current topics in microbiology and immunology 2009; v. 332. Hum
Vaccin. 6:pii: 12006. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
12
|
Arntzen CJ: High-tech herbal medicine:
Plant-based vaccines. Nat Biotechnol. 15:221–222. 1997. View Article : Google Scholar : PubMed/NCBI
|
|
13
|
Dong JL, Liang BG, Jin YS, Zhang WJ and
Wang T: Oral immunization with pBsVP6-transgenic alfalfa protects
mice against rotavirus infection. Virology. 339:153–163. 2005.
View Article : Google Scholar : PubMed/NCBI
|
|
14
|
Zhou B, Zhang Y, Wang X, Dong J, Wang B,
Han C, Yu J and Li D: Oral administration of plant-based rotavirus
VP6 induces antigen-specific IgAs, IgGs and passive protection in
mice. Vaccine. 28:6021–6027. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
15
|
Tacket CO, Mason HS, Losonsky G, Clements
JD, Levine MM and Arntzen CJ: Immunogenicity in humans of a
recombinant bacterial antigen delivered in a transgenic potato. Nat
Med. 4:607–609. 1998. View Article : Google Scholar : PubMed/NCBI
|
|
16
|
Arakawa T, Yu J and Langridge W: Synthesis
of a cholera toxin B subunit-rotavirus NSP4 fusion protein in
potato. Plant Cell Rep. 20:343–348. 2001. View Article : Google Scholar
|
|
17
|
Yu J and Langridge WH: A plant-based
multicomponent vaccine protects mice from enteric diseases. Nat
Biotechnol. 19:548–552. 2001. View
Article : Google Scholar : PubMed/NCBI
|
|
18
|
Kim TG and Langridge W: Assembly of
cholera toxin B subunit full-length rotavirus NSP4 fusion protein
oligomers in transgenic potato. Plant Cell Rep. 21:884–890.
2003.PubMed/NCBI
|
|
19
|
Choi NW, Estes MK and Langridge WH:
Synthesis of a ricin toxin B subunit-rotavirus VP7 fusion protein
in potato. Mol Biotechnol. 32:117–127. 2006. View Article : Google Scholar : PubMed/NCBI
|
|
20
|
Pêra FF, Mutepfa DL, Khan AM, Els JH,
Mbewana S, van Dijk AA, Rybicki EP and Hitzeroth II: Engineering
and expression of a human rotavirus candidate vaccine in Nicotiana
benthamiana. Virol J. 12:2052015. View Article : Google Scholar : PubMed/NCBI
|
|
21
|
Mason HS, Tacket CO, Richter LJ and
Arntzen CJ: Subunit vaccines produced and delivered in transgenic
plants as ‘edible vaccines’. Res Immunol. 149:71–74. 1998.
View Article : Google Scholar
|
|
22
|
Yang J, Guan L, Guo Y, Du L, Wang F, Wang
Y, Zhen L, Wang Q, Zou D, Chen W, et al: Expression of biologically
recombinant human acidic fibroblast growth factor in Arabidopsis
thaliana seeds via oleosin fusion technology. Gene. 566:89–94.
2015. View Article : Google Scholar : PubMed/NCBI
|
|
23
|
Mikschofsky H, König P, Keil GM, Hammer M,
Schirrmeier H and Broer I: Cholera toxin B (CTB) is functional as
an adjuvant for cytoplasmatic proteins if directed to the
endoplasmatic reticulum (ER), but not to the cytoplasm of plants.
Plant Sci. 177:35–42. 2009. View Article : Google Scholar
|
|
24
|
Arêas AP, Oliveira ML, Miyaji EN, Leite
LC, Aires KA, Dias WO and Ho PL: Expression and characterization of
cholera toxin B-pneumococcal surface adhesin A fusion protein in
Escherichia coli: Ability of CTB-PsaA to induce humoral immune
response in mice. Biochem Biophys Res Commun. 321:192–196. 2004.
View Article : Google Scholar : PubMed/NCBI
|
|
25
|
Guo L, Yin R, Liu K, Lv X, Li Y, Duan X,
Chu Y, Xi T and Xing Y: Immunological features and efficacy of a
multi-epitope vaccine CTB-UE against H. pylori in BALB/c mice
model. Appl Microbiol Biotechnol. 98:3495–3507. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
26
|
Huston JS, Tai MS, McCartney J, Keck P and
Oppermann H: Antigen recognition and targeted delivery by the
single-chain Fv. Cell Biophys. 22:189–224. 1993. View Article : Google Scholar : PubMed/NCBI
|
|
27
|
Zhang X, Henriques R, Lin SS, Niu QW and
Chua NH: Agrobacterium-mediated transformation of Arabidopsis
thaliana using the floral dip method. Nat Protoc. 1:641–646. 2006.
View Article : Google Scholar : PubMed/NCBI
|
|
28
|
Weigel D and Glazebrook J: ArabidopsisA
Laboratory Manual. Cold Spring Harbor Laboratory Press 165;
2002
|
|
29
|
Bradford MM: A raoid and sensitive method
for the quantification of microgram quantities of protein utilizing
the principle of protein-bye-binding. Anal Biochem. 72:248–254.
1976. View Article : Google Scholar : PubMed/NCBI
|
|
30
|
Health Union: Guide for the care and use
of laboratory animals. NIH Publication No. 85–23. 1996.
|
|
31
|
Capes-Davis A, Theodosopoulos G, Atkin I,
Drexler HG, Kohara A, MacLeod RA, Masters JR, Nakamura Y, Reid YA,
Reddel RR and Freshney RI: Check your cultures! A list of
cross-contaminated or misidentified cell lines. Int J. Cancer.
127:1–8. 2010.
|
|
32
|
Mi K, Ou X, Guo L, Ye J, Wu J, Yi S, Niu
X, Sun X, Li H and Sun M: Comparative analysis of the
immunogenicity of monovalent and multivalent rotavirus immunogens.
PLoS One. 12:e01721562017. View Article : Google Scholar : PubMed/NCBI
|
|
33
|
Rojas-Mancilla E, Oyarce A, Verdugo V,
Morales-Verdejo C, Echeverria C, Velásquez F, Chnaiderman J,
Valiente-Echeverría F and Ramirez-Tagle R: The [Mo(6)Cl14]2-cluster
is biologically secure and has anti-rotavirus activity in vitro.
Molecules. 22:pii: E1108. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
34
|
O'Ryan M: Rotarix (RIX4414): An oral human
rotavirus vaccine. Expert Rev Vaccines. 6:11–19. 2007. View Article : Google Scholar : PubMed/NCBI
|
|
35
|
Offit P and Clark H: Protection against
rotavirus-induced gastroenteritis in a murine model by passively
acquired gastrointestinal but not circulating antibodies. J Virol.
54:58–64. 1985.PubMed/NCBI
|
|
36
|
Filgueira Pérez DM, Mozgovoj M,
Wigdorovitz A, Santos Dus MJ, Parreño V, Trono K, Fernandez FM,
Carrillo C, Babiuk LA, Morris TJ and Borca MV: Passive protection
to bovine rotavirus (BRV) infection induced by a BRV VP8* produced
in plants using a TMV-based vector. Arch Virol. 149:2337–2348.
2004. View Article : Google Scholar : PubMed/NCBI
|
|
37
|
Mason HS, Lam D and Arntzen CJ: Expression
of hepatitis B surface antigen in transgenic plants. Proc Natl Acad
Sci USA. 89:11745–11749. 1992. View Article : Google Scholar : PubMed/NCBI
|
|
38
|
Haq TA, Mason HS, Clements JD and Arntzen
CJ: Oral immunization with a recombinant bacterial antigen produced
in transgenic plants. Science. 268:714–716. 1995. View Article : Google Scholar : PubMed/NCBI
|
|
39
|
Mason HS, Ball JM, Shi JJ, Jiang X, Estes
MK and Arntzen CJ: Expression of Norwalk virus capsid protein in
transgenic tobacco and potato and its oral immunogenicity in mice.
Proc Natl Acad Sci USA. 93:5335–5340. 1996. View Article : Google Scholar : PubMed/NCBI
|
|
40
|
Hiatt A, Caffferkey R and Bowdish K:
Production of antibodies in transgenic plants. Nature. 342:76–78.
1989. View Article : Google Scholar : PubMed/NCBI
|
|
41
|
Ma JK, Hiatt A, Hein M, Vine ND, Wang F,
Stabila P, van Dolleweerd C, Mostov K and Lehner T: Generation and
assembly of secretory antibodies in plants. Science. 268:716–719.
1995. View Article : Google Scholar : PubMed/NCBI
|
|
42
|
Kapusta J, Modelska A, Figlerowicz M,
Pniewski T, Letellier M, Lisowa O, Yusibov V, Koprowski H,
Plucienniczak A and Legocki AB: A plant-derived edible vaccine
against hepatitis B virus. FASEB J. 13:1796–1799. 1999. View Article : Google Scholar : PubMed/NCBI
|
|
43
|
Fischer R, Stoger E, Schillberg S,
Christou P and Twyman RM: Plant-based production of
biopharmaceuticals. Curr Opin Plant Biol. 7:152–158. 2004.
View Article : Google Scholar : PubMed/NCBI
|
|
44
|
Mason HS, Warzecha H, Mor T and Arntzen
CJ: Edible plant vaccines: Applications for prophylactic and
therapeutic molecular medicine. Trends Mol Med. 8:324–329. 2002.
View Article : Google Scholar : PubMed/NCBI
|
|
45
|
Perez CA, Eichwald C, Burrone O and
Mendoza D: Rotavirus vp7 antigen produced by Lactococcus lactis
induces neutralizing antibodies in mice. J Appl Microbiol.
99:1158–1164. 2005. View Article : Google Scholar : PubMed/NCBI
|
|
46
|
Perez CA, Eichwald C, Burrone O and
Mendoza D: Rotavirus vp7 antigen produced by Lactococcus lactis
induces neutralizing antibodies in mice. J Appl Microbiol.
99:1158–1164. 2005. View Article : Google Scholar : PubMed/NCBI
|
|
47
|
Yuan LY, Liu Y, Li CH, Sun MS and Dai CB:
Expression in Escherichia coli and immunogenicity of rotavirus VP7.
Sheng Wu Gong Cheng Xue Bao. 17:145–149. 2001.(In Chinese).
PubMed/NCBI
|
|
48
|
Guo L, Liu K, Xu G, Li X, Tu J, Tang F,
Xing Y and Xi T: Prophylactic and therapeutic efficacy of the
epitope vaccine CTB-UA against Helicobacter pylori infection in a
BALB/c mice model. Appl Microbiol Biotechnol. 95:1437–1444. 2012.
View Article : Google Scholar : PubMed/NCBI
|
|
49
|
Wu YZ, Li JT, Mou ZR, Fei L, Ni B, Geng M,
Jia ZC, Zhou W, Zou LY and Tang Y: Oral immunization with rotavirus
VP7 expressed in transgenic potatoes induced high titers of mucosal
neutralizing IgA. Virology. 313:337–342. 2003. View Article : Google Scholar : PubMed/NCBI
|
|
50
|
George-Chandy A, Eriksson K, Lebens M,
Nordström I, Schön E and Holmgren J: Cholera toxin B subunit as a
carrier molecule promotes antigen presentation and increases CD40
and CD86 expression on antigen-presenting cells. Infect Immun.
69:5716–5725. 2001. View Article : Google Scholar : PubMed/NCBI
|