Introduction
Neonatal sepsis is one of the most common critical
diseases in pediatrics, with a higher incidence, which can be
caused by a variety of pathogenic bacteria infections and often
accompanied by systemic inflammatory response syndrome (SIRS). In
the neonatal period, especially in premature infants, when
infection occurs, blood circulation is invaded by bacteria that
reproduces and produces toxins, resulting in systemic reactions
(1,2). Because of insidious onset, there are no
specific clinical symptoms in child patients during early stage;
given that the rapid progress of the disease will lead to secondary
organ damage, there is a need for early treatment, which is a huge
difficulty and challenge in clinical practice; the lack of early
treatment easily leads to death of children (3,4). Vitamin
D is an essential nutrient; vitamin D deficiency can easily cause
rickets and chondropathy. In recent years, studies by Chiesa et
al (5), El-Mazary et al
(6) and Cetinkaya et al
(7) have shown that vitamin D plays
an important role in immunoregulation. In this study, 150 neonates
with sepsis and 100 healthy newborns were selected, and the levels
of T-lymphocyte subsets, tumor necrosis factor-α (TNF-α),
interleukin-1 (IL-1) and calcitonin (CT) were detected, so as to
investigate the influence of vitamin D on apoptosis of T-lymphocyte
subsets, the effect on inflammatory factors and its possible
mechanism.
Patients and methods
Objects of study
One hundred and fifty neonatal patients with sepsis
admitted to Ruian People's Hospital (Wenzhou, China) from October
2015 to May 2017 were selected and randomly divided into
observation group (n=75) and treatment control group (n=75), while
100 healthy newborns during the same period were enrolled as
healthy control group. All the patients in the observation group
and the treatment control group had positive blood culture results,
and the imaging findings were in accordance with the positive
standard diagnosis. Inclusion criteria: Observation group and
treatment control group, patients aged <28 days; patients with
sepsis. Exclusion criteria: Patients with immune dysfunction;
patients with immune system disease; children with incomplete data;
children had administration with vitamin D; patients with liver or
kidney dysfunction; patients who gave up treatment. Normal control
group: Neonatal without sepsis.
The parents of the patients or their family members
signed the informed consent, and this study was approved by the
Ethics Committee of Ruian People's Hospital (Wenzhou, China).
Treatment methods
We initially collected 333 patients with neonatal
sepsis from October 2015 to May 2017 in Ruian People's Hospital,
while 183 patients were excluded according to exclusion criteria.
Therefore, only 150 patients with neonatal sepsis were included.
They were randomly divided into observation (n=75) and treatment
control group (n=75). Both treatment control and observation group
received fluid resuscitation and routine anti-infection treatment.
Based on it, the observation group was treated by intramuscular
injection with 0.25 µg/per child vitamin D (8,9); when
the condition was improved and remained stable, the dosage of
medication could be reduced until withdrawal (Fig. 1).
Detection methods and evaluation
Observation indicators: Cluster of differentiation 8
(CD8+) T-cells, CD4+ T-cells, TNF-α, IL-1 and
CT before treatment and at 72 h after treatment. T-lymphocyte
subsets, CD8+ T-cells and CD4+ T-cells were
detected by FACSCalibur flow cytometer (BD Biosciences, San Jose,
CA, USA). TNF-α, IL-1 and CT were determined by enzyme-linked
immunosorbent assay (ELISA), and the kits were provided by Jingmei
Biotech Co., Ltd. (Shanghai, China). Serum 25(OH)D was detected by
Roche Diagnostics GmbH (Mannheim, Germany) Cobas e411 automatic
electrochemiluminescence immunoassay analyzer. Baseline levels were
detected in the healthy control group.
Statistical analysis
Data were processed SPSS 22.0 (IBM Corp., Armonk,
NY, USA). Data conforming to normal distribution are expressed as
mean ± standard deviation and compared by t-test, and nonparametric
rank sum test was used for those meeting abnormal distribution. The
Chi-square test was adopted for the comparison of rate. P<0.05
was considered to indicate a statistically significant
difference.
Results
Clinical data of the three groups
There were 75 patients in the observation group,
including 39 males and 36 females, aged 1–10 days, with an average
of 4.71±1.59 days; there were 75 cases in the treatment control
group, including 37 males and 38 females, aged 1–10 days, with an
average of 4.45±1.42 days; there were 100 patients in the healthy
control group, including 50 males and 50 females, aged 1–10 days,
with an average of 4.67±1.62 days. The sex, age, mode of birth and
other general data of the three groups were comparable, the
differences were not statistically significant (P>0.05)
(Table I).
 | Table I.Comparison of clinical features in the
three groups. |
Table I.
Comparison of clinical features in the
three groups.
| Factor | Observation
group | Treatment control
group | Healthy control
group | Statistic | P-value |
|---|
| Case | 75 | 75 | 100 |
|
|
| Age (days) | 4.71±1.59 | 4.45±1.42 | 4.67±1.62 | t=0.79 | 0.53 |
| Sex (n) |
|
|
|
χ2=0.189 | 0.979 |
| Male | 39 | 37 | 50 |
|
|
|
Female | 36 | 38 | 50 |
|
|
| Apgar score |
| 1
min | 8.3±0.8 | 8.4±0.8 | 8.2±0.7 | t=0.86 | 0.48 |
| 5
min | 9.4±0.6 | 9.5±0.5 | 9.6±0.4 | t=0.88 | 0.47 |
| Mode of birth
(n) |
|
|
|
χ2=0.202 | 0.912 |
| Vaginal
delivery | 40 | 30 | 40 |
|
|
|
Uterine-incision delivery | 35 | 45 | 60 | t=7.34 | 0.0051 |
| Birth season |
|
|
|
χ2=0.208 | 0.909 |
|
Spring | 19 | 17 | 24 |
|
|
|
Summer | 17 | 19 | 26 |
|
|
|
Autumn | 18 | 17 | 23 |
|
|
|
Winter | 21 | 22 | 27 |
|
|
Test results of vitamin D in each
group
There were statistically significant differences in
comparisons of 25(OH)D among three groups. Serum 25(OH)D level in
the observation group was higher than that in the treatment control
group (P<0.05), but the levels in the two groups were remarkably
lower than that in the healthy control group, and the differences
were statistically significant (P<0.05) (Table II).
 | Table II.Comparison of 25(OH)D levels among
three groups. |
Table II.
Comparison of 25(OH)D levels among
three groups.
| Groups | Case | 25(OH)D (mg/l) |
|---|
| Observation | 76 | 22.52±5.56 |
| Treatment
control | 74 | 14.85±6.14 |
| Healthy
control | 100 | 26.38±6.56 |
| F-value |
| 56.55 |
| P-value |
| <0.05 |
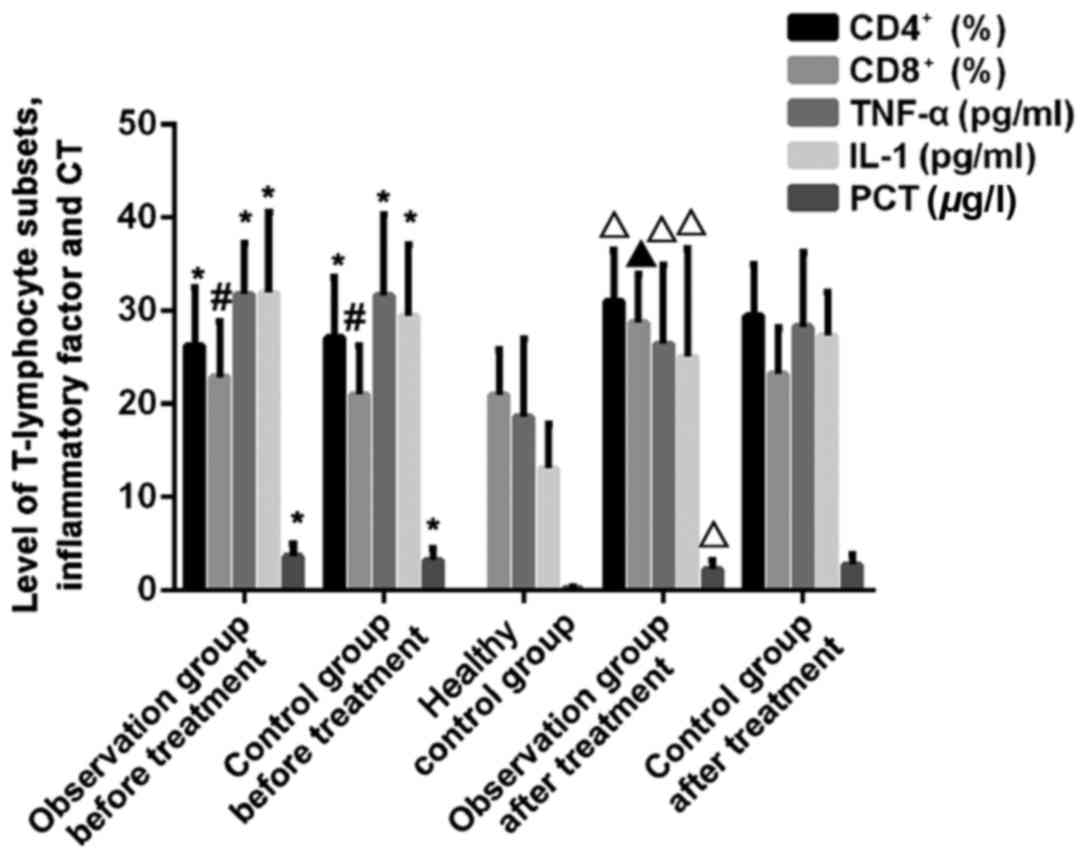
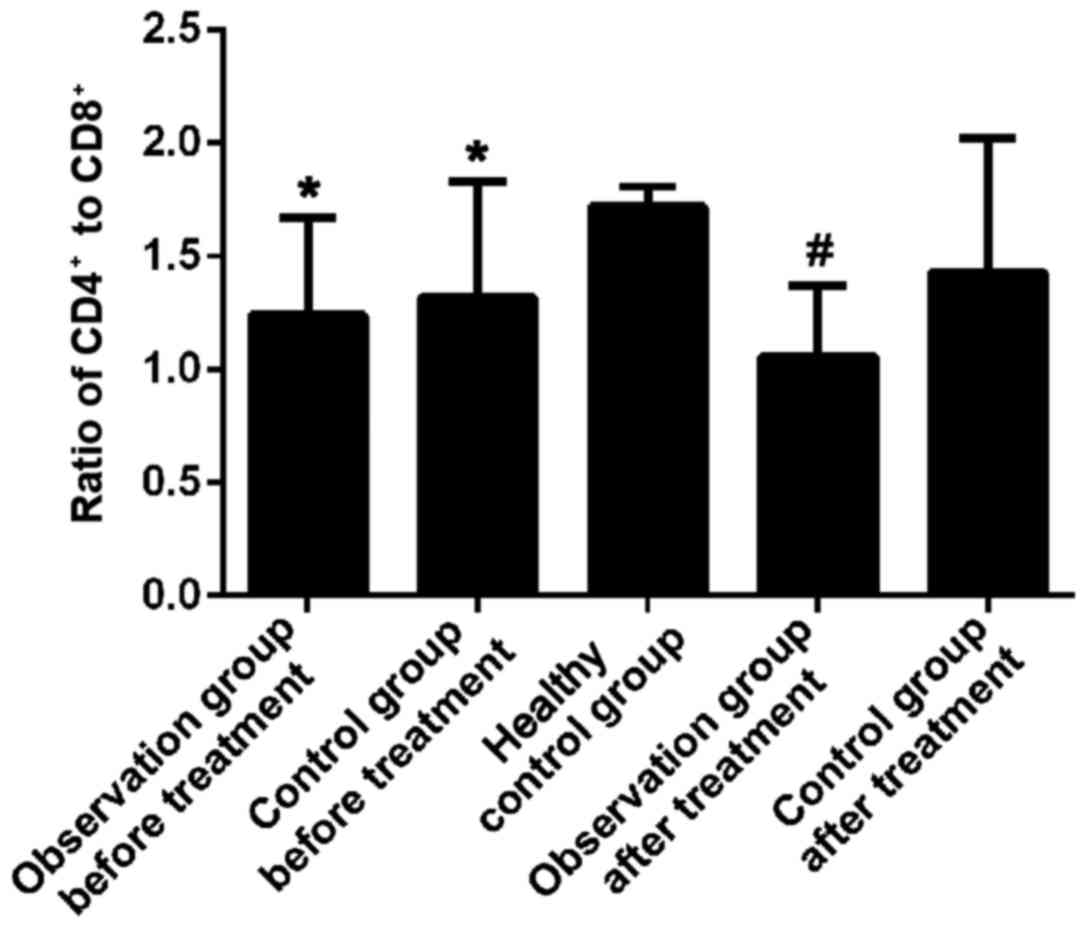
Comparison of T-lymphocyte subsets,
inflammatory factor and CT in three groups before and after
treatment
The difference in comparison of CD4+
T-cells between sepsis patients and healthy control group had
statistical significance before treatment (P<0.01), and it
showed a statistically significant difference after treatment with
vitamin D for 72 h between the observation group and the treatment
control group (P<0.05). The difference in comparison of
CD8+ T-cells between sepsis patients and healthy people
had no statistical significance before treatment (P>0.05), but
it showed a statistically significant difference after treatment
for 72 h between the observation group and the treatment control
group (P<0.01). After treatment for 72 h, CD4+
T-cells were increased, and the ratio of CD4+ to
CD8+ was close to 1. The differences in comparisons of
inflammatory factor levels between children with sepsis and healthy
control group were statistically significant before treatment
(P<0.01). The differences in comparisons of TNF-α, IL-1 and CT
levels between the observation group and the treatment control
group were statistically significant after treatment with vitamin D
for 72 h (P<0.05) (Figs. 2 and
3).
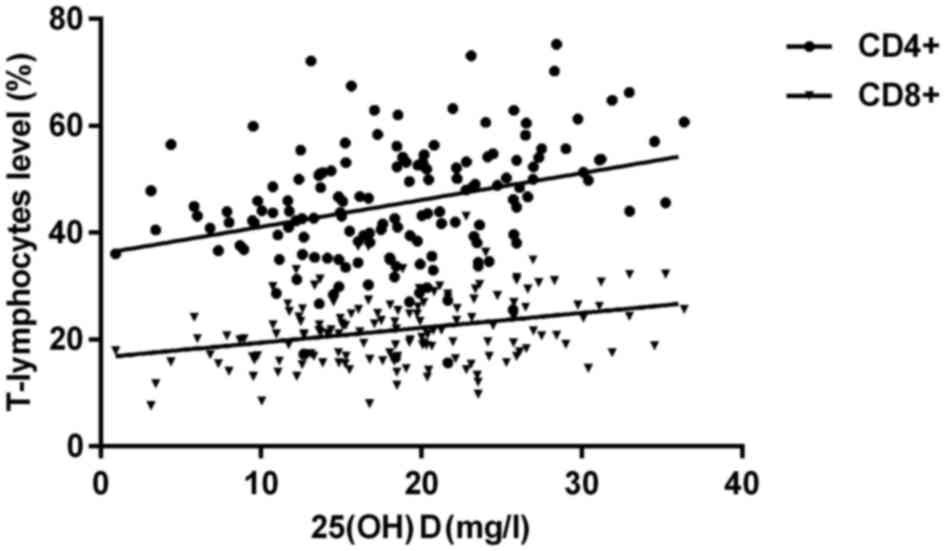
Logistic regression analysis of
vitamin D, CD4+ T-lymphocytes and CD8+
T-lymphocytes
There was a positive correlation between vitamin D
and CD4+ T-lymphocytes and CD8+ T-lymphocytes
(r=0.712, P=0.001; r=0.685, P=0.003) (Fig. 4).
Cox regression analysis
We analyzed the possibility of vitamin D,
CD4+ T-lymphocytes and CD8+ T-lymphocytes as
the risk factors for sepsis. The median vitamin D level before
treatment was 15.82 mg/l and the median percentage of
CD4+ T-lymphocytes was 26.58%, and the median percentage
of CD8+ T-lymphocytes was 21.75%. Patients were divided
into high and low levels according to the median level of
CD8+ T-lymphocytes. The results showed that they may be
risk factors for sepsis (Table
III).
 | Table III.Single factor COX regression analysis
of the occurrence of sepsis. |
Table III.
Single factor COX regression analysis
of the occurrence of sepsis.
| Single factor | HR | 95% CI | P-value |
|---|
| Vitamin D (low vs
high) | 1.412 | 1.124–1.903 | 0.025 |
| CD4 +T lymphocyte
(low vs high) | 1.315 | 1.033–2.013 | 0.031 |
| CD8 +T lymphocyte
(low vs high) | 0.512 | 0.235–1.059 | 0.024 |
| Gender (male vs
female) | 0.854 | 0.244–2.252 | 0.745 |
| Labor (natural
labor vs C-section) | 0.832 | 0.376–1.864 | 0.672 |
| Premature birth
(yes vs no) | 1.114 | 0.813–1.324 | 0.181 |
The outcomes of patients in different
groups
The mortality rate of sepsis children with vitamin D
level <15.82 mg/l is 35.2%. The mortality rate of sepsis
children with vitamin D level >15.82 mg/l is 11.3%. The
difference is statistically significant (P<0.05). The AUC of the
ROC curve analysis of vitamin D levels predicted the prognosis of
children with sepsis was 0.743, which had a good predictive value
(P<0.05) (Fig. 5).
Discussion
Neonatal sepsis is a common clinical syndrome in
newborns, with high morbidity and mortality. As the disease is
relatively insidious, the condition is usually in a state of danger
when it is found, involving complex immune dysfunction, abnormal
coagulation and other aspects; systemic immune response easily
occurs under the induction of infection in neonates with
deficiencies in immune function (10). Admittedly, the disease can be
controlled to a certain extent by adopting antibiotics, but at the
same time, the bacteria died from antibiotics can release large
amounts of endotoxin, resulting in the rapid increase in endotoxin
levels in patients, thus activating immune response; on the
contrary, it will aggravate the disease (11). The studies by Yang et al
(12) and Cizmeci et al
(13) have revealed that vitamin D
has an important regulatory role in the immune system, which can
inhibit cell proliferation and maturation, and regulate
inflammatory cytokines. Xiao et al (14) and Nzegwu et al (15) investigated the relationship between
vitamin D and sepsis. By detecting apoptosis of T-lymphocyte
subsets and inflammatory factors, this study aimed to investigate
the effect of vitamin D on apoptosis of T-lymphocyte subsets in
neonatal sepsis.
Compared to other studies, the objects of this study
were selected in strict accordance with inclusion and exclusion
criteria, ensuring the reliability of the study. At present, there
is little research on the immunological regulation mechanism of
vitamin D on neonatal sepsis; however, this study investigated the
possible immune regulation mechanism of vitamin D on neonatal
sepsis, displaying better innovation.
As known, T-lymphocytes mainly include
CD4+ T-cells, CD8+ T-cells, which play
important roles in the organism (16,17).
Moreover, the size of CD4+ T-cells is closely related to
immunosuppression (18). In this
study, the difference in comparison of CD4+ T-cells was
statistically significant after treatment with vitamin D for 72 h
betweent the observation group and the treatment control group.
After treatment with vitamin D for 72 h, the amounts of
CD4+ T-cells and CD8+ T-cells were increased,
and the increase of CD4+ T-cells was more remarkable. We
analyzed the correlation between vitamin D and CD4+
T-lymphocytes and CD8+ T-lymphatic level. The results of
logistics regression analysis showed that the correlation
coefficient was 0.712, which showed that vitamin D may be able to
regulate the levels of CD4+ T-lymphocyte and
CD8+ T-lymphocytes. Chen et al (19) found that vitamin D can regulate the
proliferation of CD8+ T cells in patients with
gastroenteritis and promote the development of gastrointestinal
inflammation. Cantorna et al (20) also found that vitamin D can regulate
T-lymphocyte levels in mouse. Therefore, we sepeculate that vitamin
D is associated with the prognosis of neonatal sepsis. Thus, the
role of vitamin D in neonatal sepsis may be achieved by reducing
apoptosis in T cells via inhibiting excessive immune response
(21). Under the action of
inflammatory factors, nuclear factor (NF-κB) is activated, which
induces inflammatory cell infiltration, thus leading to immune
dysfunction (22,23). We will further test this hypothesis
through in vitro experiment in our future studies. This
study indicated that the differences in comparison of TNF-α, IL-1
and CT levels between the observation group and the treatment
control group were statistically significant before treatment, and
after treatment with vitamin D for 72 h, the levels of these
indicators in the observation group were lower than those in the
treatment control group. The high levels of inflammatory factors
were obviously ameliorated, which reduced damage to the body to a
certain extent. We also analyzed the prognosis of children with
sepsis. We divided the children into low level group (≥15.82 mg/l)
and high level group (<15.82 mg/l) according to the level of
vitamin D. There is significant difference in mortality rate
between two groups of children. The AUC was 0.743, indicating that
vitamin D may have good prognostic value in children with
sepsis.
In this study, given that there may be some bias
because of the limited sample size, and the inappropriate dose of
vitamin D may cause hypercalcemia (24), the dosage, time and mode of
medication and other specific issues still need to be further
investigated by experts worldwide. Vitamin D may be a risk factor
for sepsis, but more research and larger samples are needed to
verify whether it is an independent factor. More studies are needed
to investigate the applications value of vitamin D in the diagnosis
of sepsis. It is hoped that individualized treatment of neonatal
sepsis can be carried out from the perspective of combined
treatment, so as to reduce the morbidity and mortality of neonatal
sepsis in China.
In conclusion, vitamin D can enhance the immune
function of neonates with sepsis by regulating the levels of
inflammatory factors and T-lymphocyte subsets.
Acknowledgements
Not applicable.
Funding
This study was supported by Wenzhou Science and
Technology Bureau Project (Y20150128), Wenzhou Health Bureau
Project (2015B37) and Ruian Science &Technology Bureau Project
(MS2017006).
Availability of data and materials
The datasets used and/or analyzed during the present
study are available from the corresponding author on reasonable
request.
Authors' contributions
GZ and WJ conceived and designed the study. GZ, MP
and ZL were responsible for the collection and analysis of the
data. WX and WJ interpreted the data and drafted the manuscript. GZ
revised the manuscript critically for important intellectual
content. All authors read and approved the final study.
Ethics approval and consent to
participate
The study was approved by the Ethics Committee of
Ruian People's Hospital (Wenzhou, China). Signed informed consents
were obtained from the parents of the child patients.
Consent for publication
Not applicable.
Competing interests
The authors declare that they have no competing
interests.
References
|
1
|
Kempker JA, Han JE, Tangpricha V, Ziegler
TR and Martin GS: Vitamin D and sepsis: An emerging relationship.
Dermatoendocrinol. 4:101–108. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
2
|
Herrmann M, Farrell CL, Pusceddu I,
Fabregat-Cabello N and Cavalier E: Assessment of vitamin D status -
a changing landscape. Clin Chem Lab Med. 55:3–26. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
3
|
Wang TJ, Zhang F, Richards JB, Kestenbaum
B, van Meurs JB, Berry D, Kiel DP, Streeten EA, Ohlsson C, Koller
DL, et al: Common genetic determinants of vitamin D insufficiency:
A genome-wide association study. Lancet. 376:180–188. 2010.
View Article : Google Scholar : PubMed/NCBI
|
|
4
|
Karras SN, Fakhoury H, Muscogiuri G, Grant
WB, van den Ouweland JM, Colao AM and Kotsa K: Maternal vitamin D
levels during pregnancy and neonatal health: Evidence to date and
clinical implications. Ther Adv Musculoskelet Dis. 8:124–135. 2016.
View Article : Google Scholar : PubMed/NCBI
|
|
5
|
Chiesa C, Natale F, Pascone R, Osborn JF,
Pacifico L, Bonci E and de Curtis M: C reactive protein and
procalcitonin: Reference intervals for preterm and term newborns
during the early neonatal period. Clin Chim Acta. 412:1053–1059.
2011. View Article : Google Scholar : PubMed/NCBI
|
|
6
|
El-Mazary AM, Abdel-Maaboud M, Mohamed M
and Nasef K: Vitamin D supplementation and the risk of infections
in fullterm infants. Correlations with the maternal serum vitamin
D. Arch Dis Child. 97:A2572012.
|
|
7
|
Cetinkaya M, Cekmez F, Buyukkale G,
Erener-Ercan T, Demir F, Tunc T, Aydın FN and Aydemir G: Lower
vitamin D levels are associated with increased risk of early-onset
neonatal sepsis in term infants. J Perinatol. 35:39–45. 2015.
View Article : Google Scholar : PubMed/NCBI
|
|
8
|
Kara Elitok G, Bulbul L, Zubarioglu U,
Kıray Bas E, Acar D, Uslu S and Bulbul A: How should we give
vitamin D supplementation? evaluation of the pediatricians'
knowledge in Turkey. Ital J Pediatr. 43:952017. View Article : Google Scholar : PubMed/NCBI
|
|
9
|
Huynh J, Lu T, Liew D, Doery JC, Tudball
R, Jona M, Bhamjee R and Rodda CP: Vitamin D in newborns. A
randomised controlled trial comparing daily and single oral bolus
vitamin D in infants. J Paediatr Child Health. 53:163–169. 2017.
View Article : Google Scholar : PubMed/NCBI
|
|
10
|
Onwuneme C, Blanco A, O'Neill A, Watson B
and Molloy EJ: Vitamin D enhances reactive oxygen intermediates
production in phagocytic cells in term and preterm infants. Pediatr
Res. 79:654–661. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
11
|
Han JE, Jones JL, Tangpricha V, Brown MA,
Brown LAS, Hao L, Hebbar G, Lee MJ, Liu S, Ziegler TR, et al: High
dose vitamin D administration in ventilated intensive care unit
patients: A pilot double blind randomized controlled trial. J Clin
Transl Endocrinol. 4:59–65. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
12
|
Yang LR, Li H, Yang TY, Zhang T and Zhao
RC: Relationship between vitamin D deficiency and early-onset
neonatal sepsis. Zhongguo Dang Dai Er Ke Za Zhi. 18:791–795.
2016.(In Chinese). PubMed/NCBI
|
|
13
|
Cizmeci MN, Kanburoglu MK, Akelma AZ,
Ayyildiz A, Kutukoglu I, Malli DD and Tatli MM: Cord-blood
25-hydroxyvitamin D levels and risk of early-onset neonatal sepsis:
A case-control study from a tertiary care center in Turkey. Eur J
Pediatr. 174:809–815. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
14
|
Xiao T, Chen LP, Liu H, Xie S, Luo Y and
Wu DC: The analysis of etiology and risk factors for 192 cases of
neonatal sepsis. BioMed Res Int. 2017:86170762017. View Article : Google Scholar : PubMed/NCBI
|
|
15
|
Nzegwu NI, Rychalsky MR, Nallu LA, Song X,
Deng Y, Natusch AM, Baltimore RS, Paci GR and Bizzarro MJ:
Implementation of an Antimicrobial Stewardship Program in a
Neonatal Intensive Care Unit. Infect Control Hosp Epidemiol.
38:1137–1143. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
16
|
Omran A, Maaroof A, Saleh MH and
Abdelwahab A: Salivary C-reactive protein, mean platelet volume and
neutrophil lymphocyte ratio as diagnostic markers for neonatal
sepsis. J Pediatr (Rio J). 94:82–87. 2018. View Article : Google Scholar : PubMed/NCBI
|
|
17
|
Stritzke A, Mohammad K, Shah PS, Ye XY,
Bhandari V, Akierman A, Harrison A, Bertelle V and Lodha A: Use and
timing of surfactant administration: Impact on neonatal outcomes in
extremely low gestational age infants born in Canadian Neonatal
Intensive Care Units. J Matern Fetal Neonatal Med. 31:1–8. 2017.
View Article : Google Scholar : PubMed/NCBI
|
|
18
|
McCarthy RA, McKenna MJ, Oyefeso O, Uduma
O, Murray BF, Brady JJ, Kilbane MT, Murphy JF, Twomey A, O' Donnell
CP, et al: Vitamin D nutritional status in preterm infants and
response to supplementation. Br J Nutr. 110:156–163. 2013.
View Article : Google Scholar : PubMed/NCBI
|
|
19
|
Chen J, Bruce D and Cantorna MT: Vitamin D
receptor expression controls proliferation of naïve
CD8+T cells and development of CD8 mediated
gastrointestinal inflammation. BMC Immunol. 15:62014. View Article : Google Scholar : PubMed/NCBI
|
|
20
|
Cantorna MT, Snyder L, Lin YD and Yang L:
Vitamin D and 1,25(OH)2D regulation of T cells. Nutrients.
7:3011–3021. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
21
|
Watkins RR, Lemonovich TL and Salata RA:
An update on the association of vitamin D deficiency with common
infectious diseases. Can J Physiol Pharmacol. 93:363–368. 2015.
View Article : Google Scholar : PubMed/NCBI
|
|
22
|
Barragan M, Good M and Kolls JK:
Regulation of dendritic cell function by vitamin D. Nutrients.
7:8127–8151. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
23
|
Bahrami A, Mazloum SR, Maghsoudi S,
Soleimani D, Khayyatzadeh SS, Arekhi S, Arya A, Mirmoosavi SJ,
Ferns GA, Bahrami-Taghanaki H, et al: High dose vitamin D
supplementation is associated with a reduction in depression score
among adolescent girls: A nine-week follow-up study. J Diet Suppl.
15:173–182. 2018. View Article : Google Scholar : PubMed/NCBI
|
|
24
|
Wang EW, Siu PM, Pang MY, Woo J, Collins
AR and Benzie IFF: Vitamin D deficiency, oxidative stress and
antioxidant status: Only weak association seen in the absence of
advanced age, obesity or pre-existing disease. Br J Nutr.
118:11–16. 2017. View Article : Google Scholar : PubMed/NCBI
|