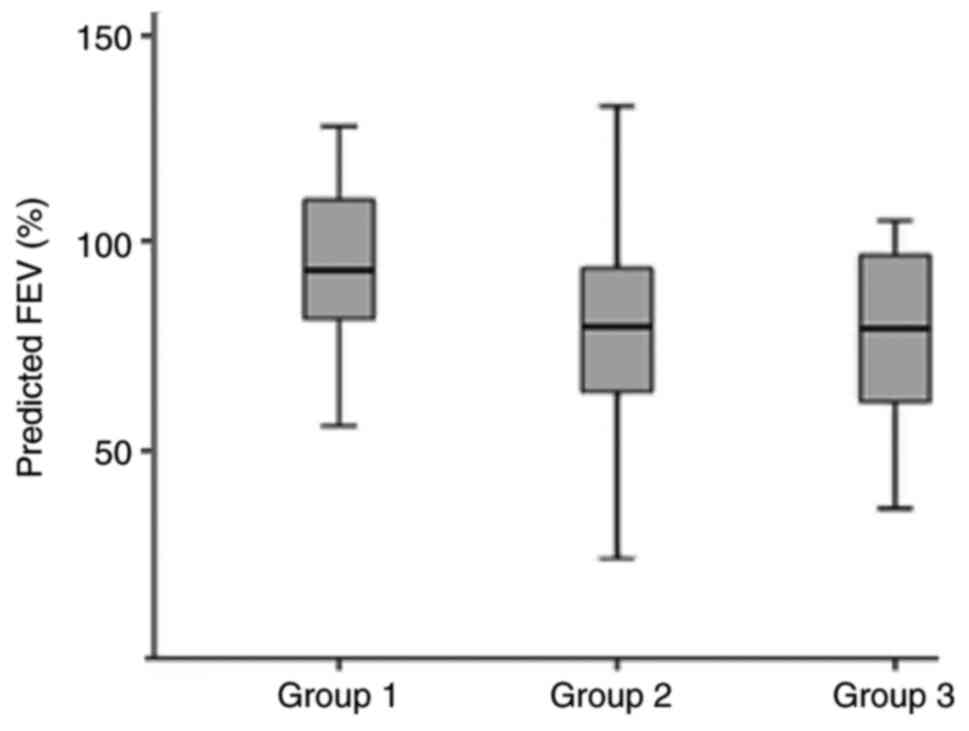
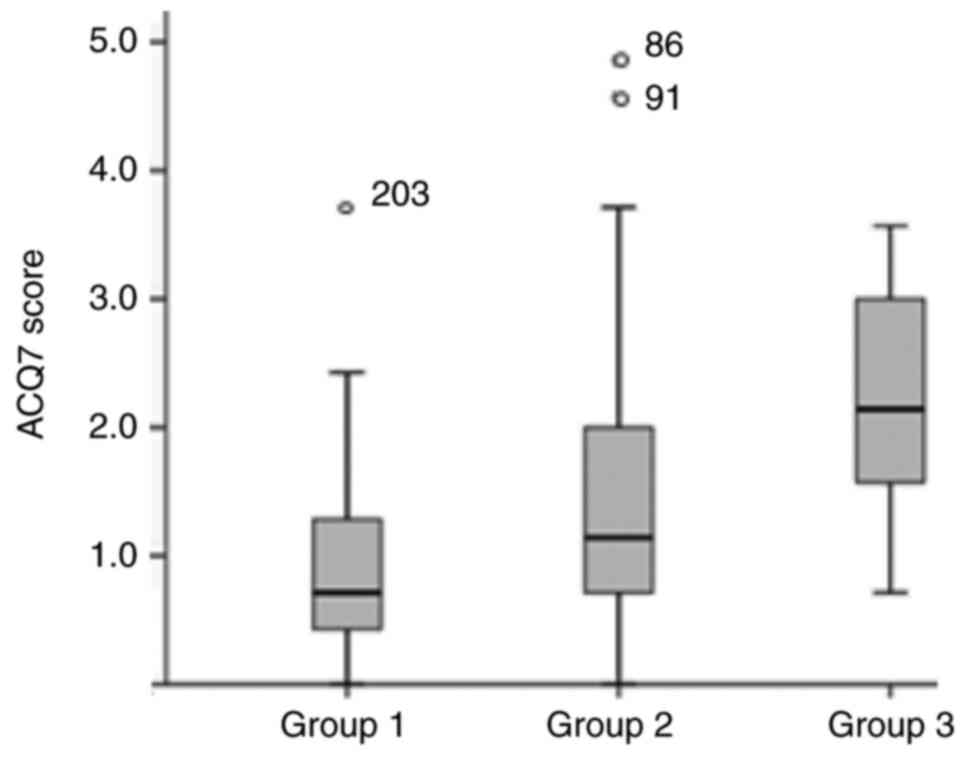
|
1
|
Global Initiative for Asthma: 2021 GINA
Report, Global Strategy for Asthma Management and Prevention. GINA,
Fontana, 2021. https://ginasthma.org/gina-reports.
|
|
2
|
Chung KF, Wenzel SE, Brozek JL, Bush A,
Castro M, Sterk PJ, Adcock IM, Bateman ED, Bel EH, Bleecker ER, et
al: International ERS/ATS guidelines on definition, evaluation and
treatment of severe asthma. Eur Respir J. 43:343–373.
2014.PubMed/NCBI View Article : Google Scholar
|
|
3
|
Holguin F, Cardet JC, Chung KF, Diver S,
Ferreira DS, Fitzpatrick A, Gaga M, Kellermeyer L, Khurana S,
Knight S, et al: Management of severe asthma: A European
respiratory society/American thoracic society guideline. Eur Respir
J. 55(1900588)2020.PubMed/NCBI View Article : Google Scholar
|
|
4
|
Selroos O, Kupczyk M, Kuna P, Łacwik P,
Bousquet J, Brennan D, Palkonen S, Contreras J, FitzGerald M,
Hedlin G, et al: National and regional asthma programmes in Europe.
Eur Respir Rev. 24:474–483. 2015.PubMed/NCBI View Article : Google Scholar
|
|
5
|
MacDonald KM, Kavati A, Ortiz B, Alhossan
A, Lee CS and Abraham I: Short- and long-term real-world
effectiveness of omalizumab in severe allergic asthma: Systematic
review of 42 studies published 2008-2018. Expert Rev Clin Immunol.
15:553–569. 2019.PubMed/NCBI View Article : Google Scholar
|
|
6
|
Pelaia C, Calabrese C, Terracciano R, de
Blasio F, Vatrella A and Pelaia G: Omalizumab, the first available
antibody for biological treatment of severe asthma: More than a
decade of real-life effectiveness. Ther Adv Respir Dis.
12(1753466618810192)2018.PubMed/NCBI View Article : Google Scholar
|
|
7
|
Deeks ED: Mepolizumab: A review in
eosinophilic asthma. BioDrugs. 30:361–370. 2016.PubMed/NCBI View Article : Google Scholar
|
|
8
|
Harvey ES, Langton D, Katelaris C, Stevens
S, Farah CS, Gillman A, Harrington J, Hew M, Kritikos V,
Radhakrishna N, et al: Mepolizumab effectiveness and identification
of super-responders in severe asthma. Eur Respir J.
55(1902420)2020.PubMed/NCBI View Article : Google Scholar
|
|
9
|
Khatri S, Moore W, Gibson PG, Leigh R,
Bourdin A, Maspero J, Baros M, Buhl R, Howarth P, Alberts F, et al:
Assessment of the long-term safety of mepolizumab and durability of
clinical response in patients with severe eosinophilic asthma. J
Allergy Clin Immunol. 143:1742–1751.e7. 2019.PubMed/NCBI View Article : Google Scholar
|
|
10
|
Hekking PW, Wener RR, Amelink M,
Zwinderman AH, Bouvy ML and Bel EH: The prevalence of severe
refractory asthma. J Allergy Clin Immunol. 135:896–902.
2015.PubMed/NCBI View Article : Google Scholar
|
|
11
|
von Bülow A, Kriegbaum M, Backer V and
Porsbjerg C: The prevalence of severe asthma and low asthma control
among Danish adults. J Allergy Clin Immunol Pract. 2:759–767.
2014.PubMed/NCBI View Article : Google Scholar
|
|
12
|
ISAR Study Group. International severe
asthma registry: Mission statement. Chest. 157:805–814.
2020.PubMed/NCBI View Article : Google Scholar
|
|
13
|
Zervas E, Loukides S, Kostikas K, Bakakaos
P, Tzortzaki E and Gaga M: On Behalf of Asthma Working Group of
Hellenic Thoracic Society. Asthma and asthma-like symptoms in
Greece. The Greece asthma national prevalence survey. Eur Respir J.
40 (Suppl 56)(P3936)2012.
|
|
14
|
Kourlaba G, Bakakos P, Loukides S,
Vellopoulou K, Solakidi A and Maniadakis N: The self-reported
prevalence and disease burden of asthma in Greece. J Asthma.
56:478–497. 2019.PubMed/NCBI View Article : Google Scholar
|
|
15
|
Juniper EF, O'byrne PM, Guyatt GH, Ferrie
PJ and King DR: Development and validation of a questionnaire to
measure asthma control. Eur Respir J. 14:902–907. 1999.PubMed/NCBI View Article : Google Scholar
|
|
16
|
Khusial RJ, Honkoop PJ, van der Meer V,
Snoeck-Stroband JB and Sont JK: Validation of online asthma control
questionnaire and asthma quality of life questionnaire. ERJ Open
Res. 6:00289–2019. 2020.PubMed/NCBI View Article : Google Scholar
|
|
17
|
Zervas E, Samitas K, Papaioannou AI,
Bakakos P, Loukides S and Gaga M: An algorithmic approach for the
treatment of severe uncontrolled asthma. ERJ Open Res.
4:00125–2017. 2018.PubMed/NCBI View Article : Google Scholar
|
|
18
|
Papi A, Saetta M and Fabbri L: Severe
asthma: Phenotyping to endotyping or vice versa? Eur Respir J.
49(1700053)2017.PubMed/NCBI View Article : Google Scholar
|
|
19
|
Vellopoulou K, Bakakos P, Loukides S,
Maniadakis N and Kourlaba G: The Economic burden of asthma in
Greece: A cross-sectional study. Appl Health Econ Health Policy.
17:629–640. 2019.PubMed/NCBI View Article : Google Scholar
|
|
20
|
Souliotis K, Kousoulakou H, Hillas G,
Bakakos P, Toumbis M, Loukides S and Vassilakopoulos T: Direct and
indirect costs of asthma management in Greece: An expert panel
approach. Front Public Health. 5(67)2017.PubMed/NCBI View Article : Google Scholar
|
|
21
|
Wenzel SE, Brillhart S and Nowack K: An
invisible disease: Severe asthma is more than just ‘bad asthma’.
Eur Respir J. 50(1701109)2017.PubMed/NCBI View Article : Google Scholar
|
|
22
|
van Bragt JJMH, Adcock IM, Bel EHD,
Braunstahl GJ, ten Brinke A, Busby J, Canonica GW, Cao H, Chung KF,
Csoma Z, et al: Characteristics and treatment regimens across ERS
SHARP severe asthma registries. Eur Respir J.
55(1901163)2020.PubMed/NCBI View Article : Google Scholar
|
|
23
|
Sá-Sousa A, Fonseca JA, Pereira AM,
Ferreira A, Arrobas A, Mendes A, Drummond M, Videira W, Costa T,
Farinha P, et al: The portuguese severe asthma registry:
Development, features, and data sharing policies. Biomed Res Int.
2018(1495039)2018.PubMed/NCBI View Article : Google Scholar
|
|
24
|
Taube C, Bramlage P, Hofer A and Anderson
D: Prevalence of oral corticosteroid use in the German severe
asthma population. ERJ Open Res. 5:00092–2019. 2019.PubMed/NCBI View Article : Google Scholar
|
|
25
|
Bousquet J, Brusselle G, Buhl R, Busse WW,
Cruz AA, Djukanovic R, Domingo C, Hanania NA, Humbert M, Menzies
Gow A, et al: Care pathways for the selection of a biologic in
severe asthma. Eur Respir J. 50(1701782)2017.PubMed/NCBI View Article : Google Scholar
|
|
26
|
AL-Jahdali H, Anwar A, AL-Harbi A,
Baharoon S, Halwani R, Al Shimemeri A and Al-Muhsen S: Factors
associated with patient visits to the emergency department for
asthma therapy. BMC Pulm Med. 12(80)2012.PubMed/NCBI View Article : Google Scholar
|
|
27
|
Stensen L, Porsbjerg C, Sverrild A, Nybo
Jensen B and Vibeke B: Managing asthma in the outpatient clinic: Is
the diagnosis of asthma confirmed objectively according to
guidelines? Eur Respir J. 40 (Suppl 56)(P2263)2012.
|
|
28
|
Assaf SM and Hanania NA: Biological
treatments for severe asthma. Curr Opin Allergy Clin Immunol.
19:379–386. 2019.PubMed/NCBI View Article : Google Scholar
|
|
29
|
McGregor MC, Krings JG, Nair P and Castro
M: Role of biologics in asthma. Am J Respir Crit Care Med.
199:433–445. 2019.PubMed/NCBI View Article : Google Scholar
|
|
30
|
Rabe KF: New biologics for severe asthma:
What patients, what agents, what results, at what cost? Am J Respir
Crit Care Med. 199:406–408. 2019.PubMed/NCBI View Article : Google Scholar
|
|
31
|
Pavord I, Bahmer T, Braido F, Cosío BG,
Humbert M, Idzko M and Adamek L: Severe T2-high asthma in the
biologics era: European experts' opinion. Eur Respir Rev.
28(190054)2019.PubMed/NCBI View Article : Google Scholar
|
|
32
|
Doroudchi A, Pathria M and Modena BD:
Asthma biologics: Comparing trial designs, patient cohorts and
study results. Ann Allergy Asthma Immunol. 124:44–56.
2020.PubMed/NCBI View Article : Google Scholar
|
|
33
|
Papaioannou AI, Fouka E, Papakosta D,
Papiris S and Loukides S: Switching between biologics in severe
asthma patients. When the first choice is not proven to be the
best. Clin Exp Allergy. 51:221–227. 2021.PubMed/NCBI View Article : Google Scholar
|
|
34
|
Pepper AN, Hanania NA, Humbert M and
Casale TB: How to assess effectiveness of biologics for asthma and
what steps to take when there is not benefit. J Allergy Clin
Immunol Pract. 9:1081–1088. 2021.PubMed/NCBI View Article : Google Scholar
|
|
35
|
Kallieri M, Zervas E, Katsoulis K, Fouka
E, Porpodis K, Samitas K, Papaioannou AI, Kipourou M, Gaki E,
Vittorakis S, et al: Mepolizumab in severe eosinophilic asthma: A
2-year follow-up in specialized asthma clinics in Greece: An
interim analysis. Int Arch Allergy Immunol. 181:613–617.
2020.PubMed/NCBI View Article : Google Scholar
|