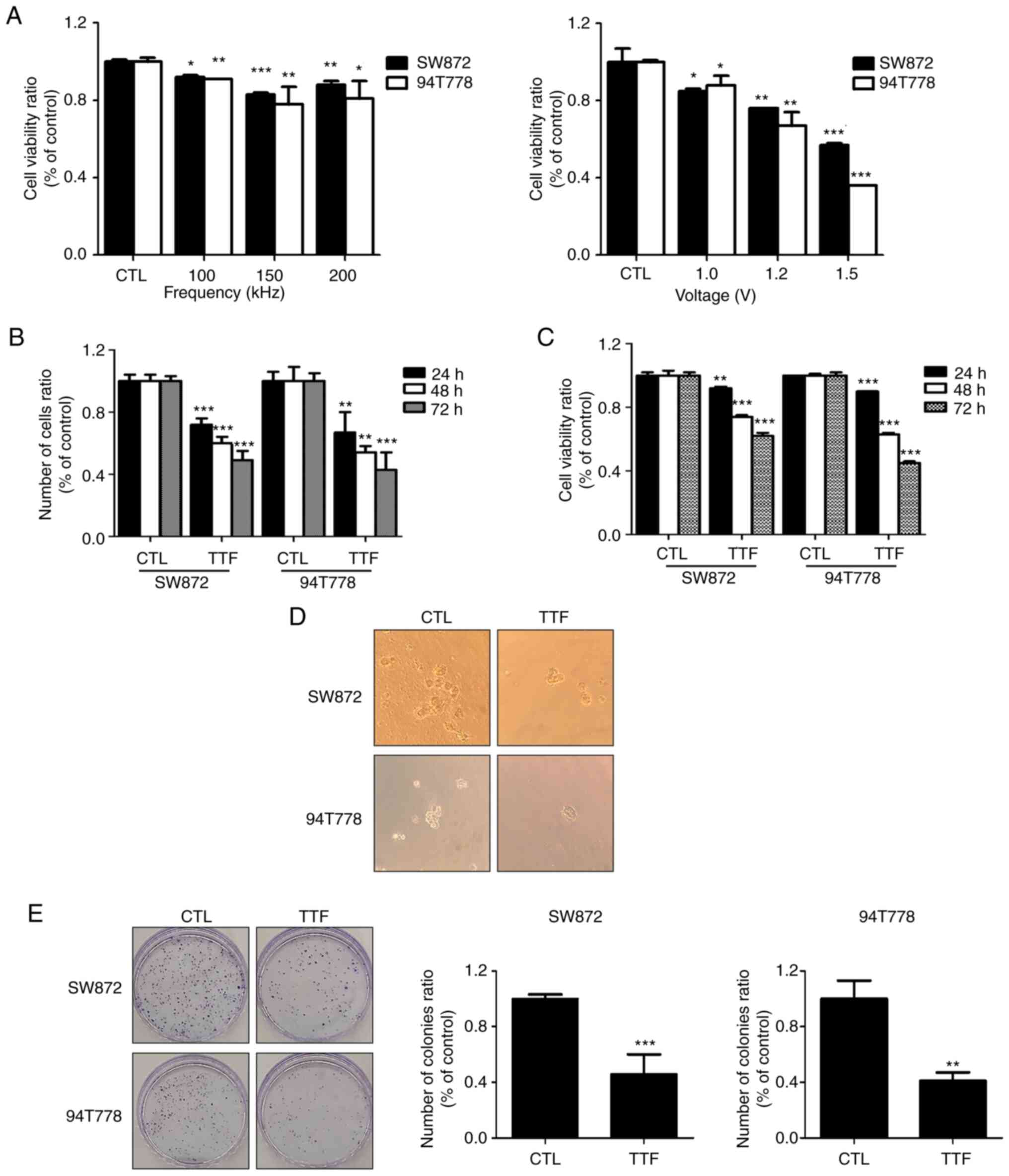
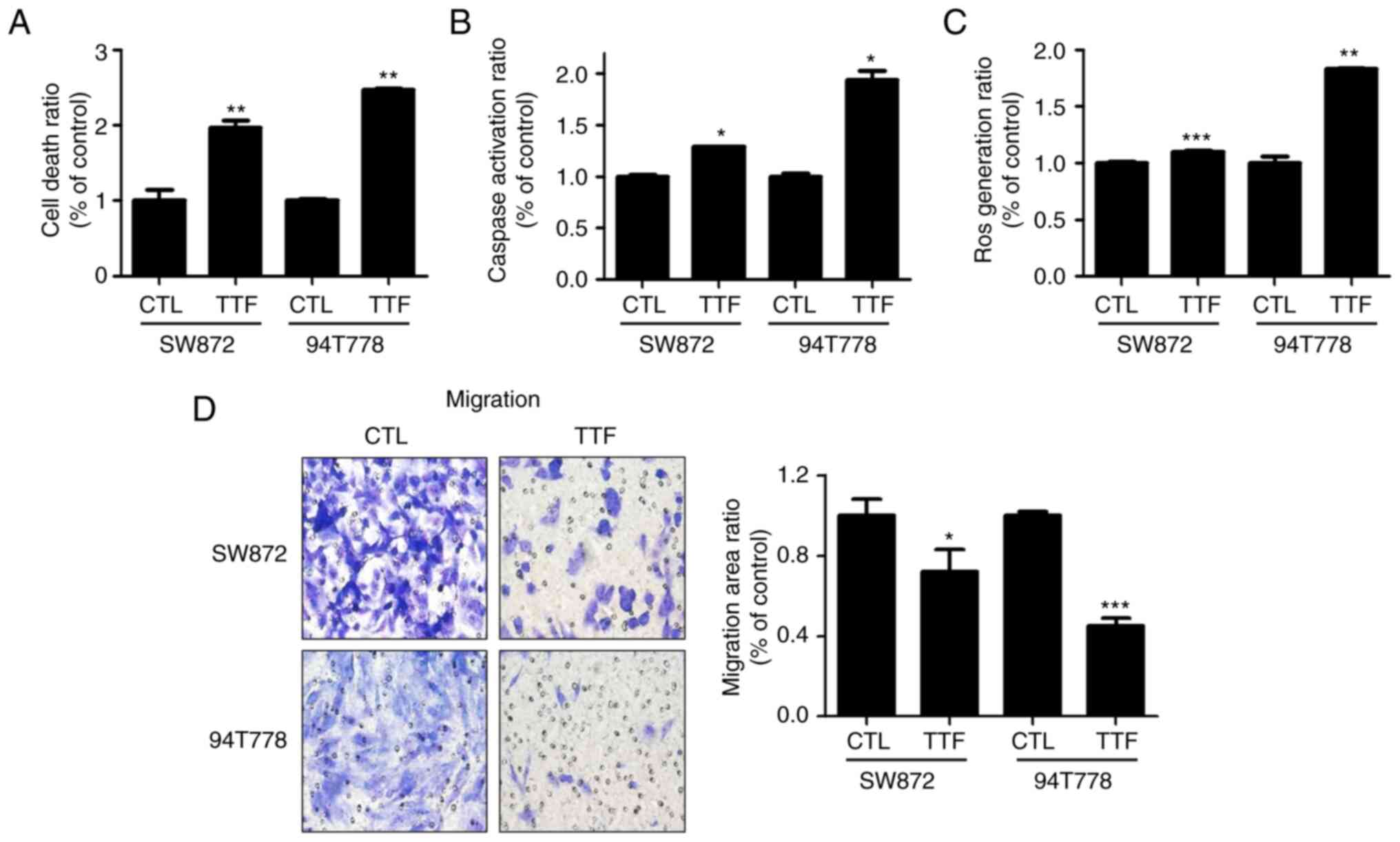
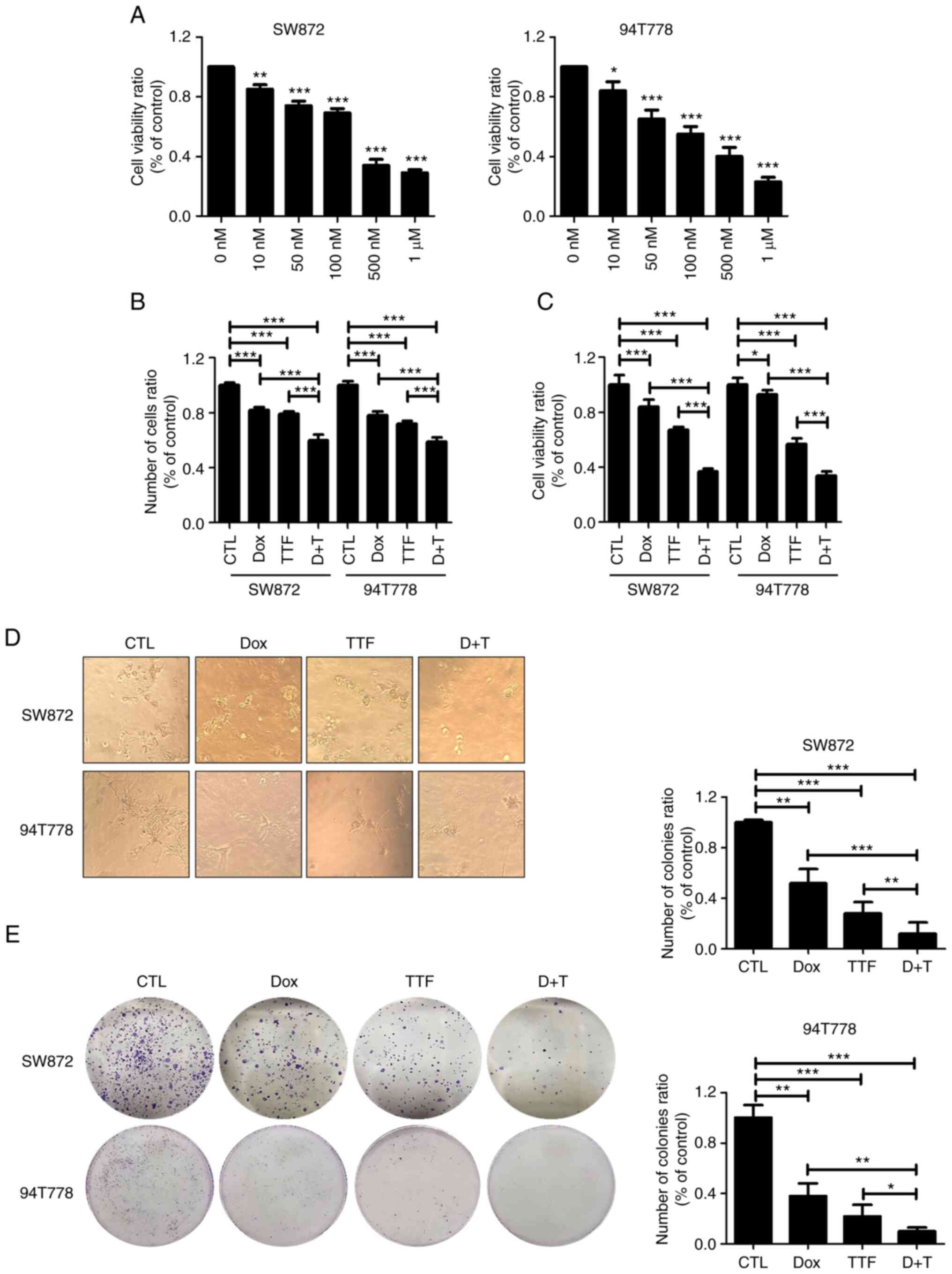
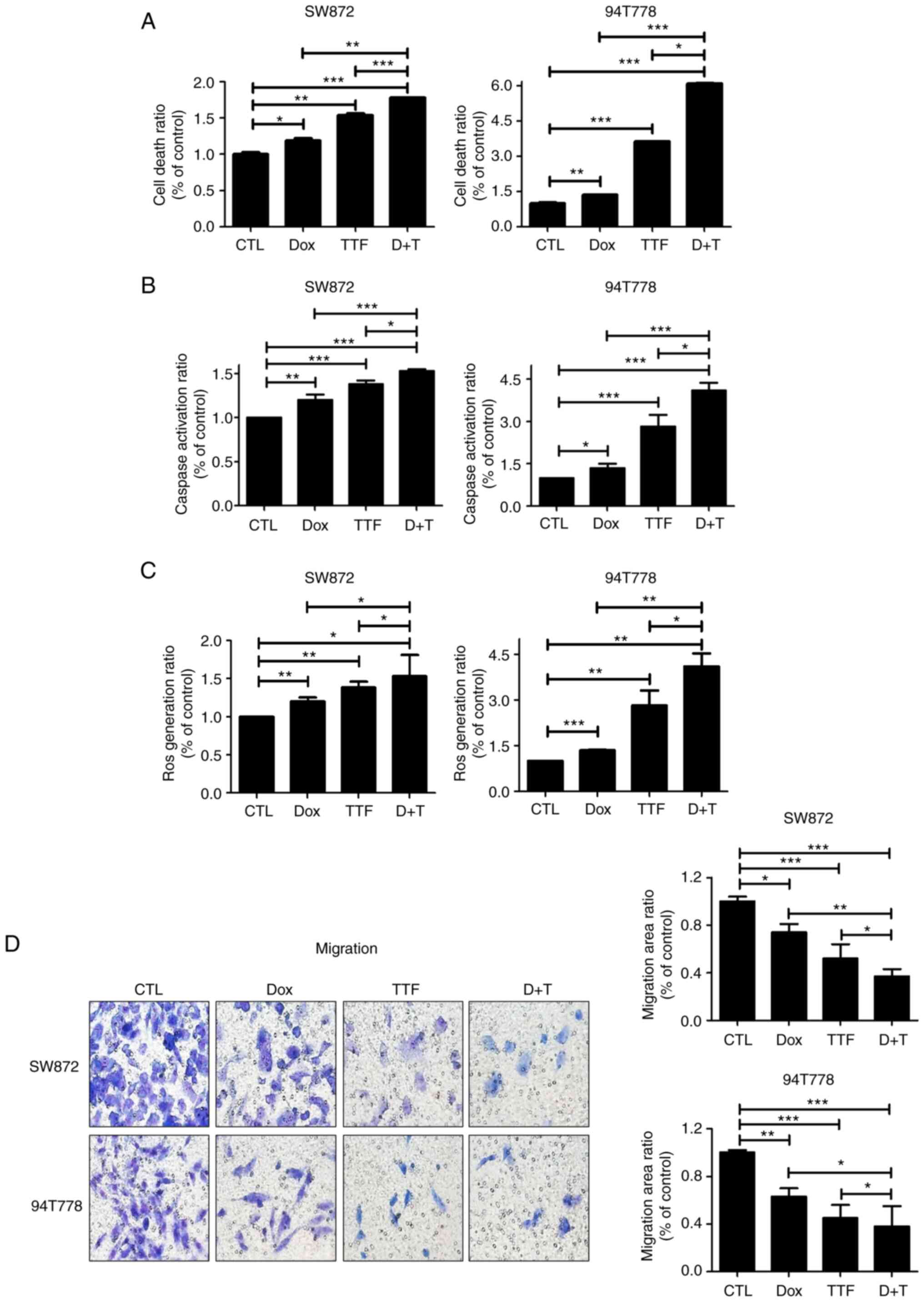
|
1
|
Conyers R, Young S and Thomas DM:
Liposarcoma: Molecular genetics and therapeutics. Sarcoma.
2011(483154)2011.PubMed/NCBI View Article : Google Scholar
|
|
2
|
Crago AM and Singer S: Clinical and
molecular approaches to well differentiated and dedifferentiated
liposarcoma. Curr Opin Oncol. 23:373–378. 2011.PubMed/NCBI View Article : Google Scholar
|
|
3
|
Malik AT, Alexander JH, Mayerson JL, Khan
SN and Scharschmidt TJ: Is Surgical resection of the primary site
associated with an improved overall survival for patients with
primary malignant bone tumors who have metastatic disease at
presentation? Clin Orthop Relat Res. 478:2284–2295. 2020.PubMed/NCBI View Article : Google Scholar
|
|
4
|
Singer S, Socci ND, Ambrosini G, Sambol E,
Decarolis P, Wu Y, O'Connor R, Maki R, Viale A, Sander C, et al:
Gene expression profiling of liposarcoma identifies distinct
biological types/subtypes and potential therapeutic targets in
well-differentiated and dedifferentiated liposarcoma. Cancer Res.
67:6626–6636. 2007.PubMed/NCBI View Article : Google Scholar
|
|
5
|
Haddox CL and Riedel RF: Recent advances
in the understanding and management of liposarcoma. Fac Rev.
10(1)2021.PubMed/NCBI View
Article : Google Scholar
|
|
6
|
Thway K: Well-differentiated liposarcoma
and dedifferentiated liposarcoma: An updated review. Semin Diagn
Pathol. 36:112–121. 2019.PubMed/NCBI View Article : Google Scholar
|
|
7
|
Kirson ED, Gurvich Z, Schneiderman R,
Dekel E, Itzhaki A, Wasserman Y, Schatzberger R and Palti Y:
Disruption of cancer cell replication by alternating electric
fields. Cancer Res. 64:3288–3295. 2004.PubMed/NCBI View Article : Google Scholar
|
|
8
|
Fonkem E and Wong ET: NovoTTF-100A: A new
treatment modality for recurrent glioblastoma. Expert Rev
Neurother. 12:895–899. 2012.PubMed/NCBI View Article : Google Scholar
|
|
9
|
Rominiyi O, Vanderlinden A, Clenton SJ,
Bridgewater C, Al-Tamimi Y and Collis SJ: Tumour treating fields
therapy for glioblastoma: Current advances and future directions.
Br J Cancer. 124:697–709. 2021.PubMed/NCBI View Article : Google Scholar
|
|
10
|
Giladi M, Schneiderman RS, Voloshin T,
Porat Y, Munster M, Blat R, Sherbo S, Bomzon Z, Urman N, Itzhaki A,
et al: Mitotic spindle disruption by alternating electric fields
leads to improper chromosome segregation and mitotic catastrophe in
cancer cells. Sci Rep. 5(18046)2015.PubMed/NCBI View Article : Google Scholar
|
|
11
|
Kirson ED, Dbaly V, Tovarys F, Vymazal J,
Soustiel JF, Itzhaki A, Mordechovich D, Steinberg-Shapira S,
Gurvich Z, Schneiderman R, et al: Alternating electric fields
arrest cell proliferation in animal tumor models and human brain
tumors. Proc Natl Acad Sci USA. 104:10152–10157. 2007.PubMed/NCBI View Article : Google Scholar
|
|
12
|
Gera N, Yang A, Holtzman TS, Lee SX, Wong
ET and Swanson KD: Tumor treating fields perturb the localization
of septins and cause aberrant mitotic exit. PLoS One.
10(e0125269)2015.PubMed/NCBI View Article : Google Scholar
|
|
13
|
Karanam NK and Story MD: An overview of
potential novel mechanisms of action underlying Tumor Treating
Fields-induced cancer cell death and their clinical implications.
Int J Radiat Biol. 97:1044–1054. 2021.PubMed/NCBI View Article : Google Scholar
|
|
14
|
Kim EH, Kim YH, Song HS, Jeong YK, Lee JY,
Sung J, Yoo SH and Yoon M: Biological effect of an alternating
electric field on cell proliferation and synergistic antimitotic
effect in combination with ionizing radiation. Oncotarget.
7:62267–62279. 2016.PubMed/NCBI View Article : Google Scholar
|
|
15
|
Lee WS, Seo SJ, Chung HK, Park JW, Kim JK
and Kim EH: Tumor-treating fields as a proton beam-sensitizer for
glioblastoma therapy. Am J Cancer Res. 11:4582–4594.
2021.PubMed/NCBI
|
|
16
|
Fabian D, Guillermo Prieto Eibl MDP,
Alnahhas I, Sebastian N, Giglio P, Puduvalli V, Gonzalez J and
Palmer JD: Treatment of Glioblastoma (GBM) with the addition of
tumor-treating fields (TTF): A review. Cancers (Basel).
11(174)2019.PubMed/NCBI View Article : Google Scholar
|
|
17
|
Carrieri FA, Smack C, Siddiqui I,
Kleinberg LR and Tran PT: Tumor treating fields: At the crossroads
between physics and biology for cancer treatment. Front Oncol.
10(575992)2020.PubMed/NCBI View Article : Google Scholar
|
|
18
|
Hottinger AF, Pacheco P and Stupp R: Tumor
treating fields: A novel treatment modality and its use in brain
tumors. Neuro Oncol. 18:1338–1349. 2016.PubMed/NCBI View Article : Google Scholar
|
|
19
|
Pless M, Droege C, von Moos R, Salzberg M
and Betticher D: A phase I/II trial of Tumor Treating Fields
(TTFields) therapy in combination with pemetrexed for advanced
non-small cell lung cancer. Lung Cancer. 81:445–450.
2013.PubMed/NCBI View Article : Google Scholar
|
|
20
|
Jo Y, Hwang SG, Jin YB, Sung J, Jeong YK,
Baek JH, Cho JM, Kim EH and Yoon M: Selective toxicity of tumor
treating fields to melanoma: An in vitro and in vivo study. Cell
Death Discov. 4(46)2018.PubMed/NCBI View Article : Google Scholar
|
|
21
|
Mulita F, Verras GI, Liolis E,
Tchabashvili L, Kehagias D, Kaplanis C, Perdikaris I and Kehagias
I: Recurrent retroperitoneal liposarcoma: A case report and
literature review. Clin Case Rep. 9(e04717)2021.PubMed/NCBI View Article : Google Scholar
|
|
22
|
Spalek MJ, Kozak K, Czarnecka AM, Bartnik
E, Borkowska A and Rutkowski P: Neoadjuvant treatment options in
soft tissue sarcomas. Cancers (Basel). 12(2061)2020.PubMed/NCBI View Article : Google Scholar
|
|
23
|
Pasquali S and Gronchi A: Neoadjuvant
chemotherapy in soft tissue sarcomas: Latest evidence and clinical
implications. Ther Adv Med Oncol. 9:415–429. 2017.PubMed/NCBI View Article : Google Scholar
|
|
24
|
Romanenko A, Grassellino A, Crawford AC,
Sergatskov DA and Melnychuk O: Ultra-high quality factors in
superconducting niobium cavities in ambient magnetic fields up to
190 mG. Appl Phys Lett. 105(234103)2014.
|
|
25
|
Liu C, Zhu Y, Lou W, Cui Y, Evans CP and
Gao AC: Inhibition of constitutively active Stat3 reverses
enzalutamide resistance in LNCaP derivative prostate cancer cells.
Prostate. 74:201–209. 2014.PubMed/NCBI View Article : Google Scholar
|
|
26
|
Ji WO, Lee MH, Kim GH and Kim EH:
Quantitation of the ROS production in plasma and radiation
treatments of biotargets. Sci Rep. 9(19837)2019.PubMed/NCBI View Article : Google Scholar
|
|
27
|
Kim EH, Song HS, Yoo SH and Yoon M: Tumor
treating fields inhibit glioblastoma cell migration, invasion and
angiogenesis. Oncotarget. 7:65125–65136. 2016.PubMed/NCBI View Article : Google Scholar
|
|
28
|
Wenger C, Miranda PC, Salvador R,
Thielscher A, Bomzon Z, Giladi M, Mrugala MM and Korshoej AR: A
review on tumor-treating fields (TTFields): Clinical implications
inferred from computational modeling. IEEE Rev Biomed Eng.
11:195–207. 2018.PubMed/NCBI View Article : Google Scholar
|
|
29
|
Bernhardt M, Angerer B, Buss M and
Struppler A: Neural observer based spasticity quantification during
therapeutic muscle stimulation. Conf Proc IEEE Eng Med Biol Soc.
2006:4897–4900. 2006.PubMed/NCBI View Article : Google Scholar
|
|
30
|
Alesanco A, Garcia J, Serrano P, Ramos L
and Istepanian RH: On the guarantee of reconstruction quality in
ECG wavelet codecs. Conf Proc IEEE Eng Med Biol Soc.
2006:6461–6464. 2006.PubMed/NCBI View Article : Google Scholar
|
|
31
|
Sies H and Jones DP: Reactive oxygen
species (ROS) as pleiotropic physiological signalling agents. Nat
Rev Mol Cell Biol. 21:363–383. 2020.PubMed/NCBI View Article : Google Scholar
|
|
32
|
McGovern Y, Zhou CD and Jones RL: Systemic
therapy in metastatic or unresectable
well-differentiated/dedifferentiated liposarcoma. Front Oncol.
7(292)2017.PubMed/NCBI View Article : Google Scholar
|
|
33
|
Codenotti S, Vezzoli M, Monti E and
Fanzani A: Focus on the role of Caveolin and Cavin protein families
in liposarcoma. Differentiation. 94:21–26. 2017.PubMed/NCBI View Article : Google Scholar
|
|
34
|
Jo VY and Fletcher CD: WHO classification
of soft tissue tumours: An update based on the 2013 (4th) edition.
Pathology. 46:95–104. 2014.PubMed/NCBI View Article : Google Scholar
|
|
35
|
Casali PG, Abecassis N, Aro HT, Bauer S,
Biagini R, Bielack S, Bonvalot S, Boukovinas I, Bovee JVMG,
Brodowicz T, et al: Soft tissue and visceral sarcomas: ESMO-EURACAN
Clinical Practice Guidelines for diagnosis, treatment and
follow-up. Ann Oncol. 29 (Suppl 4):iv268–iv269. 2018.PubMed/NCBI View Article : Google Scholar
|
|
36
|
Judson I, Verweij J, Gelderblom H,
Hartmann JT, Schöffski P, Blay JY, Kerst JM, Sufliarsky J, Whelan
J, Hohenberger P, et al: Doxorubicin alone versus intensified
doxorubicin plus ifosfamide for first-line treatment of advanced or
metastatic soft-tissue sarcoma: A randomised controlled phase 3
trial. Lancet Oncol. 15:415–423. 2014.PubMed/NCBI View Article : Google Scholar
|
|
37
|
Demetri GD, von Mehren M, Jones RL,
Hensley ML, Schuetze SM, Staddon A, Milhem M, Elias A, Ganjoo K,
Tawbi H, et al: Efficacy and safety of trabectedin or dacarbazine
for metastatic liposarcoma or leiomyosarcoma after failure of
conventional chemotherapy: Results of a phase III randomized
multicenter clinical trial. J Clin Oncol. 34:786–793.
2016.PubMed/NCBI View Article : Google Scholar
|
|
38
|
Schoffski P, Chawla S, Maki RG, Italiano
A, Gelderblom H, Choy E, Grignani G, Camargo V, Bauer S, Rha SY, et
al: Eribulin versus dacarbazine in previously treated patients with
advanced liposarcoma or leiomyosarcoma: A randomised, open-label,
multicentre, phase 3 trial. Lancet. 387:1629–1637. 2016.PubMed/NCBI View Article : Google Scholar
|
|
39
|
Samuels BL, Chawla SP, Somaiah N, Staddon
AP, Skubitz KM, Milhem MM, Kaiser PE, Portnoy DC, Priebat DA,
Walker MS and Stepanski EJ: Results of a prospective phase 2 study
of pazopanib in patients with advanced intermediate-grade or
high-grade liposarcoma. Cancer. 123:4640–4647. 2017.PubMed/NCBI View Article : Google Scholar
|
|
40
|
Oki E, Makiyama A, Miyamoto Y, Kotaka M,
Kawanaka H, Miwa K, Kabashima A, Noguchi T, Yuge K, Kashiwada T, et
al: Trifluridine/tipiracil plus bevacizumab as a first-line
treatment for elderly patients with metastatic colorectal cancer
(KSCC1602): A multicenter phase II trial. Cancer Med. 10:454–461.
2021.PubMed/NCBI View Article : Google Scholar
|
|
41
|
Tchekmedyian V, Sherman EJ, Dunn L, Tran
C, Baxi S, Katabi N, Antonescu CR, Ostrovnaya I, Haque SS, Pfister
DG and Ho AL: Phase II study of lenvatinib in patients with
progressive, recurrent or metastatic adenoid cystic carcinoma. J
Clin Oncol. 37:1529–1537. 2019.PubMed/NCBI View Article : Google Scholar
|
|
42
|
Dickson MA, Schwartz GK, Keohan ML,
D'Angelo SP, Gounder MM, Chi P, Antonescu CR, Landa J, Qin LX,
Crago AM, et al: Progression-Free survival among patients with
well-differentiated or dedifferentiated liposarcoma treated with
CDK4 inhibitor palbociclib: A phase 2 clinical trial. JAMA Oncol.
2:937–940. 2016.PubMed/NCBI View Article : Google Scholar
|
|
43
|
Chawla SP, Staddon AP, Baker LH, Schuetze
SM, Tolcher AW, D'Amato GZ, Blay JY, Mita MM, Sankhala KK, Berk L,
et al: Phase II study of the mammalian target of rapamycin
inhibitor ridaforolimus in patients with advanced bone and soft
tissue sarcomas. J Clin Oncol. 30:78–84. 2012.PubMed/NCBI View Article : Google Scholar
|
|
44
|
Demetri GD, Fletcher CD, Mueller E, Sarraf
P, Naujoks R, Campbell N, Spiegelman BM and Singer S: Induction of
solid tumor differentiation by the peroxisome
proliferator-activated receptor-gamma ligand troglitazone in
patients with liposarcoma. Proc Natl Acad Sci USA. 96:3951–3956.
1999.PubMed/NCBI View Article : Google Scholar
|
|
45
|
U.S. National Library of Medicine: Ph II
Cabazitaxel DD Liposarcoma. ClinicalTrials.gov, 2013. Accessed August 1,
2013.
|
|
46
|
Keung EZ, Lazar AJ, Torres KE, Wang WL,
Cormier JN, Ashleigh Guadagnolo B, Bishop AJ, Lin H, Hunt KK, Bird
J, et al: Phase II study of neoadjuvant checkpoint blockade in
patients with surgically resectable undifferentiated pleomorphic
sarcoma and dedifferentiated liposarcoma. BMC Cancer.
18(913)2018.PubMed/NCBI View Article : Google Scholar
|
|
47
|
Dizon DS, Krilov L, Cohen E, Gangadhar T,
Ganz PA, Hensing TA, Hunger S, Krishnamurthi SS, Lassman AB,
Markham MJ, et al: Clinical cancer advances 2016: Annual report on
progress against cancer from the American society of clinical
oncology. J Clin Oncol. 34:987–1011. 2016.PubMed/NCBI View Article : Google Scholar
|
|
48
|
Stupp R, Taillibert S, Kanner A, Read W,
Steinberg D, Lhermitte B, Toms S, Idbaih A, Ahluwalia MS, Fink K,
et al: Effect of tumor-treating fields plus maintenance
temozolomide vs. maintenance temozolomide alone on survival in
patients with glioblastoma: A randomized clinical trial. JAMA.
318:2306–2316. 2017.PubMed/NCBI View Article : Google Scholar
|
|
49
|
Lee WS and Kim EH: Combination therapy of
Doxorubicin with TTFields and radiation: Newer approaches to combat
lung cancer. Am J Cancer Res. 12:2673–2685. 2022.PubMed/NCBI
|
|
50
|
Kim EH, Lee WS and Oh HK: Tumor-treating
fields in combination with sorafenib curtails the growth of
colorectal carcinoma by inactivating AKT/STAT3 signaling. Transl
Cancer Res. 11:2553–2561. 2022.PubMed/NCBI View Article : Google Scholar
|
|
51
|
Oh JY, Lee YJ and Kim EH: Tumor-Treating
fields inhibit the metastatic potential of osteosarcoma cells.
Technol Cancer Res Treat. 19(1533033820947481)2020.PubMed/NCBI View Article : Google Scholar
|
|
52
|
Jang Y, Lee WS, Sai S, Kim JY, Kim JK and
Kim EH: Tumor-treating fields in combination with sorafenib
restrain the proliferation of liver cancer in vitro. Oncol Lett.
24(338)2022.PubMed/NCBI View Article : Google Scholar
|
|
53
|
Park JI, Song KH, Jung SY, Ahn J, Hwang
SG, Kim J, Kim EH and Song JY: Tumor-Treating fields induce
RAW264.7 macrophage activation via NK-κB/MAPK signaling pathways.
Technol Cancer Res Treat. 18(1533033819868225)2019.PubMed/NCBI View Article : Google Scholar
|
|
54
|
Giladi M, Munster M, Schneiderman RS,
Voloshin T, Porat Y, Blat R, Zielinska-Chomej K, Hååg P, Bomzon Z,
Kirson ED, et al: Tumor treating fields (TTFields) delay DNA damage
repair following radiation treatment of glioma cells. Radiat Oncol.
12(206)2017.PubMed/NCBI View Article : Google Scholar
|
|
55
|
Tanzhu G, Chen L, Xiao G, Shi W, Peng H,
Chen D and Zhou R: The schemes, mechanisms and molecular pathway
changes of Tumor Treating Fields (TTFields) alone or in combination
with radiotherapy and chemotherapy. Cell Death Discov.
8(416)2022.PubMed/NCBI View Article : Google Scholar
|
|
56
|
Kim EH, Jo Y, Sai S, Park MJ, Kim JY, Kim
JS, Lee YJ, Cho JM, Kwak SY, Baek JH, et al: Tumor-treating fields
induce autophagy by blocking the Akt2/miR29b axis in glioblastoma
cells. Oncogene. 38:6630–6646. 2019.PubMed/NCBI View Article : Google Scholar
|
|
57
|
Chang E, Patel CB, Pohling C, Young C,
Song J, Flores TA, Zeng Y, Joubert LM, Arami H, Natarajan A, et al:
Tumor treating fields increases membrane permeability in
glioblastoma cells. Cell Death Discov. 4(113)2018.PubMed/NCBI View Article : Google Scholar
|
|
58
|
Voloshin T, Kaynan N, Davidi S, Porat Y,
Shteingauz A, Schneiderman RS, Zeevi E, Munster M, Blat R, Tempel
Brami C, et al: Tumor-treating fields (TTFields) induce immunogenic
cell death resulting in enhanced antitumor efficacy when combined
with anti-PD-1 therapy. Cancer Immunol Immunother. 69:1191–1204.
2020.PubMed/NCBI View Article : Google Scholar
|
|
59
|
Schneiderman RS, Voloshin T, Giladi M,
Porat Y, Munster M, Blat R, Sherbo S, Kirson ED, Weinberg U and
Palti Y: ATPS-25p53 Status dependence of tumor treating fields
(TTFIELDS) efficacy against glioma cancer cells. Neuro Oncol. 17
(Suppl 5)(v23)2015.
|
|
60
|
Lee YJ, Seo HW, Baek JH, Lim SH, Hwang SG
and Kim EH: Gene expression profiling of glioblastoma cell lines
depending on TP53 status after tumor-treating fields (TTFields)
treatment. Sci Rep. 10(12272)2020.PubMed/NCBI View Article : Google Scholar
|
|
61
|
Lee YJ, Cho JM, Sai S, Oh JY, Park JA, Oh
SJ, Park M, Kwon J, Shin US, Beak JH, et al: 5-Fluorouracil as a
Tumor-treating field-sensitizer in colon cancer therapy. Cancers
(Basel). 11(1999)2019.PubMed/NCBI View Article : Google Scholar
|