Introduction
Total joint arthroplasty is a cost-effective
operative procedure for treating dysfunctional weight bearing
joints, and relieves pain and restores joint function (1). However, aseptic loosening secondary
to wear particle-associated osteolysis is an important cause of
complication following hip and knee replacement surgery.
Furthermore, revision surgery is associated with significant cost
and potential morbidity (2,3).
Titanium (Ti) alloy particles are common wear debris
particles in periprosthetic tissue. They stimulate the release and
phagocytic activity of monocytes and macrophages around
periprosthetic tissue and generate the release of a host of
proinflammatory cytokines, including tumor necrosis factor-α
(TNF-α), metalloproteinases (MMPs), interleukin-1β (IL-1β),
interleukin-6 (IL-6), transforming growth factor-β (TGF-β), colony
stimulating factor-1 (CSF-1) and macrophage-CSF (M-CSF) (4). The resulting inflammatory reaction
is widely considered to play a key role in the formation of
osteolysis induced by wear debris and finally results in aseptic
loosening of the replaced joint (4).
The presence of wear particles from prosthetic
implants may also trigger a histiocytic response at the
synovial-like membrane which is present at the interface between
the bone and the implant. This involves the activation and release
of macrophages, fibroblasts, potential proinflammatory cytokines
and MMPs, all of which promote bone absorption and osteolysis in
periprosthetic tissue (5).
MMPs are a family of proteolytic enzymes that have
been reported to be involved in the degradation of the
extracellular matrix of various tissues such as bone and have been
shown to induce bone resorption (6). MMP-1, -2, -3, -9, -10, -12 and -13
and membrane type 1 (MT-1)-MMP have all been implicated in
osteolysis associated with aseptic loosening of joint prostheses
(7–10). Matrix metalloproteinase-9 (MMP-9)
is regarded as a gelatinase B, type IV collagenase and is highly
expressed not only during the early stages of osteoclast
development, but also in mature osteoclasts which resorb bone
(11). Previous studies have
demonstrated that MMP-9 activity has an important function in the
pathogenesis of dental pulp inflammation (12). A high expression of MMP-9 has also
been discovered in human osteoclastomas and osteoclasts in Paget’s
disease (13). Activation of
MMP-9 activity has been demonstrated to be restricted to
macrophages in the pseudomembrane of the bone-implant interface
(14). Therefore, it constitutes
a potential drug target in this indication, and several studies
have demonstrated that the inhibition of MMP-9 resulted in a
reduced bone resorption in response to wear particles (15,16).
Other studies have demonstrated that the receptor
activator of nuclear factor-κB ligand (RANKL) and the receptor
activator of nuclear factor-κB (RANK) play an important role in
regulating wear debris-induced osteoclastogenesis (17,18). Both TNF-α and IL-1β have been
demonstrated to be regulated by the expression and activity of the
RANK/RANKL in histiocytic tissue around loosened implants (19,20). However, the possible interaction
between MMP-9 and RANK/RANKL signaling in the development of
inflammatory osteolysis is not well characterized.
The present study using a murine osteolysis model
was undertaken to examine the hypothesis that amelioration of Ti
particle-induced inflammatory osteolysis by MMP-9 inhibition is
mediated by the downregulation of RANK/RANKL, MMP-9 and TNF-α.
Materials and methods
Titanium alloy particles
Ti particles (diameter 0.1–20 μm) were
obtained from Alfa Aesar Company (Fig. 1). The particles were washed 3
times in 70% ethanol solution to remove bound endotoxin and were
sterilized in 10 mM phosphate-buffered saline (PBS) at pH 7.4. They
were then suspended in sterile 10 mM PBS at a concentration of 15
mg/ml and stored at 4°C until needed. The suspension contained 5%
(weight/volume) particles.
Murine osteolysis model
Female BALB/c mice, aged 8–10 weeks were purchased
from the Shanghai Laboratory Animal Centre (Shanghai, China). The
mice were randomly assigned to 3 experimental groups with 15
animals in each group: a control group (A), a Ti group (B) and a Ti
plus tetracycline group (C).
All mice weighed 20–25 g at the start of the
experiment. Previously described techniques were used to establish
air pouches (21). Briefly, the
dorsal area of each mouse (2×2 cm) was shaved and sterilized and 2
ml of sterile air was injected subcutaneously to establish an air
pouch. To maintain the pouch, 0.5 ml of sterile air was injected
into the pouch each day for 6 days.
After 6 days, mice with established air pouches were
anaesthetized by i.p. injection of pentobarbital (50 mg/kg). A
0.5-cm incision overlying the pouch was performed and a section of
calvarial bone (∼0.4×0.25 cm) from a genetically identical donor
mouse was inserted into the pouch. The Ti alloy particle suspension
(0.3 ml) was injected into established air pouches of the mice
(except those from group A) to provoke inflammatory osteolysis. The
pouch layers and the skin incision were then closed using 4-0
prolene sutures. Tetracycline 50 mg/kg was administered by i.p.
injection to group C and 0.1 ml sterile PBS was administered by
i.p. injection to groups A and B. Injections continued daily until
the mice were sacrificed 14 days after bone-Ti implantation. Pouch
membranes with the intact bone implants were harvested for
histological and molecular analysis.
The Institutional Animal Care and Use Committee of
the Shanghai Jiaotong University approved all animal
procedures.
Histological evaluation
Tissue samples were fixed in 4% polyoxymethylene (pH
7.4) for 24 h, after decalcification in 10%
ethylenediaminetetraacetic acid (EDTA). The specimens were
processed for dehydration in graded alcohol, cleared in dimethyl
benzene and embedded in paraffin. Tissue sections (6 μm)
were stained with hematoxylin and eosin (H&E) to evaluate pouch
membrane inflammation and implant bone erosion. The stained
sections were examined under a light microscope (Olympus DP70;
Olympus Optical Co., Tokyo, Japan) and digital photomicrographs
were captured and analyzed using a computerized image analysis
system with Image-Pro Plus software, version 6.0 (Media
Cybernetics, Silver Spring, MD, USA). Pouch membrane thickness and
the total numbers of infiltrated cells were measured using digital
image analysis.
Four separate sections/specimen were analyzed in a
blinded fashion. Pouch membrane thickness was measured at 6 points
on each section and 6 random 100-μm longitudinal pouch areas
were selected to count total cells (cells/mm2) based
upon nucleus counts.
Expression of tartrate-resistant acid
phosphatase (TRAP)
TRAP staining was used to identify the presence of
osteoclast-like cells in the pouch tissue. Histochemical TRAP
staining using a commercial kit (Sigma, St. Louis, MO, USA) was
performed on pouches implanted with calvarium. Frozen sections (6
μm) were prepared and incubated in xylene for 30 sec and
washed with ethanol. Sections were incubated at 37°C for 1 h in 100
mM acetate buffer (pH 5.2) containing 0.5 mM naphthol AS-BI
phosphoric acid, 2.2 mM Fast Garnet GBC and 8 mM sodium tartrate.
The sections were then washed in several changes of distilled
water, followed by counterstaining with a hematoxylin solution. The
presence of dark purple staining granules in the cytoplasm was
determined as the specific criterion for TRAP-positive cells.
Positive TRAP localization was quantified by pixel area count and
reported as the percentage of the total implanted bone area in the
pouch tissue.
Immunohistochemical staining for MMP-9,
TNF-α, RANK and RANKL
Tissue sections were deparaffinized in xylene and
rehydrated in ethanol. The rinsed sections were immersed in antigen
retrieval buffer of EDTA in a water bath at 97°C for 30 min. The
sections were allowed to cool to room temperature before
preincubation for 10 min with 0.3% (vol/vol) hydrogen peroxide in
methanol to suppress the activity of endogenous peroxidase. The
sections were blocked with 1.5% normal goat serum for 30 min. They
were then incubated at 4°C overnight with the primary antibodies:
rabbit anti-MMP-9 (Abcam, USA), mouse anti-TNF-α, mouse anti-RANK-α
and mouse anti-RANKL-α. The sections were rinsed and incubated for
30 min with biotin-conjugated secondary antibody (Sigma) at 37°C.
Streptavidin-horseradish peroxidase conjugate (SA-HRP) was applied
to sections for 30 min at 37°C. 3,3′-Diaminobenzidine
tetrahydrochloride (DAB) was added for color development with
counterstaining using hematoxylin. The digital photomicrographs
were analyzed using the Image-Pro image analysis software
package.
Gene expression of MMP-9, TNF-α, RANK and
RANKL
Total RNA was extracted from pouch tissue using
TRIzol reagent (Invitrogen Life Technologies, Carlsbad, CA, USA)
according to the manufacturer’s instructions. cDNA was synthesized
from the total RNA using a SYBR PrimeScript™ reverse
transcription-polymerase chain reaction (RT-PCR) kit (Takara Bio,
Inc., Shiga, Japan). Quantitative real-time RT-PCR was performed to
evaluate the relative levels of mRNA for MMP-9, TNF-α, RANK and
RANKL, using the SYBR PrimeScript™ RT-PCR kit and an ABI 7500
real-time thermal cycler (Applied Biosystems, Carlsbad, CA, USA)
according to the manufacturer’s instructions. Gene specific primers
for MMP-9, TNF-α, RANK and RANKL were designed using Primer 5.0
software (Premier Biosoft International, Palo Alto, CA, USA). The
housekeeping gene, glyceraldehyde-3-phosphate dehydrogenase (GAPDH)
was used as an internal control. The cycling program involved
preliminary denaturation at 95°C for 2 min, followed by 40 cycles
of denaturation at 95°C for 15 sec and annealing at 60°C for 1
min.
A comparative threshold cycle (Ct) method with
arithmetic formulae was used to determine the relative level of
gene expression following PCR amplification. Subtracting the Ct of
the housekeeping gene from the Ct of the target gene yielded the
ΔCt for each group (control and experimental groups) which was
entered into the equation 2−ΔCt. The gene activity in
the control group was arbitrarily assigned as 1 to serve as a
reference. The primers used in this study were as shown in Table I.
 | Table IGene-specific primers for MMP-9,
TNF-α, RANK, RANKL and GAPDH. |
Table I
Gene-specific primers for MMP-9,
TNF-α, RANK, RANKL and GAPDH.
| Genes | Reverse primer
sequence | Forward primer
sequence | Size (base
pair) |
|---|
| MMP-9 |
5′-TACTGGAAGATGTCGTGTGAG-3′ |
5′-GGCGTGTCTGGAGATTCG-3′ | 112 |
| TNF-α |
5′-CAGGTCACTGTGTCCCAGCATCT-3′ |
5′-GAGTCCGGGCAGGTCTACTTT-3′ | 235 |
| RANK |
5′-GCGAGGTCTGGCTGACATAC-3′ |
5′-CTGCCTCTGGGAACGTGACT-3′ | 108 |
| RANKL |
5′-CTGCGTTTTCATGGAGTCTCA-3′ |
5′-CAGCATCGCTCTGGTTCCTGTA-3′ | 107 |
| GAPDH |
5′-TGCTGTTGAAGTCGCAGGAC-3′ |
5′-CCAATGTGTCCGTCGTGGAT-3′ | 153 |
Statistical analysis
Statistical analyses were carried out using the SPSS
statistical package, version 11.0 (SPSS, Inc., Chicago, IL, USA).
Values are expressed as mean ± standard deviation (SD). Differences
between groups were analyzed using analysis of variance. Values of
P<0.05 were considered statistically significant.
Results
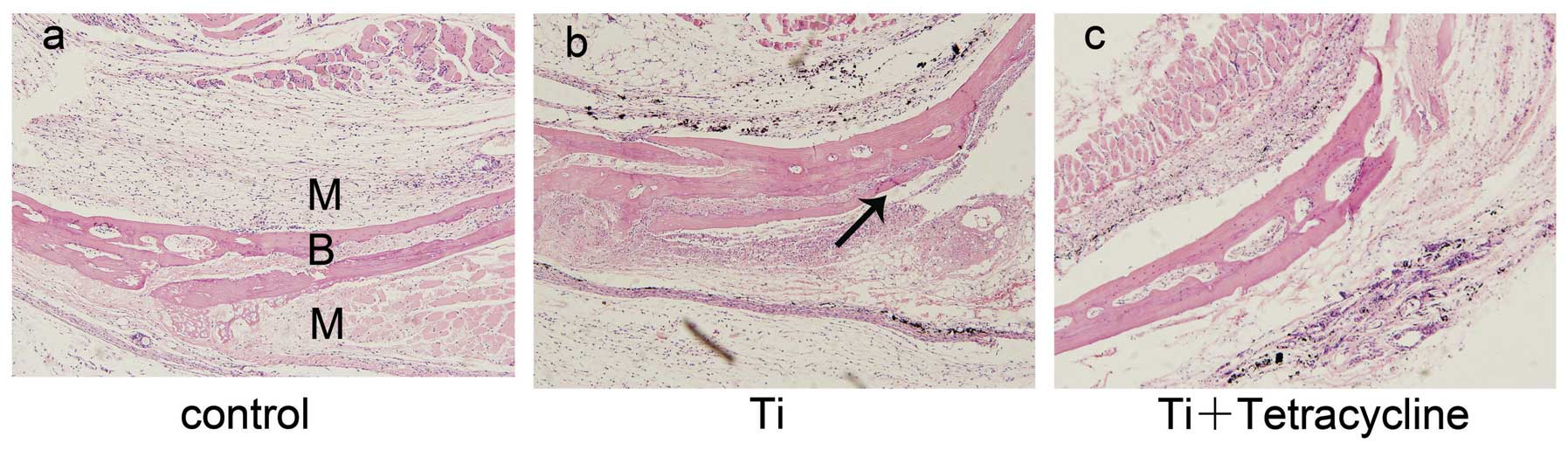
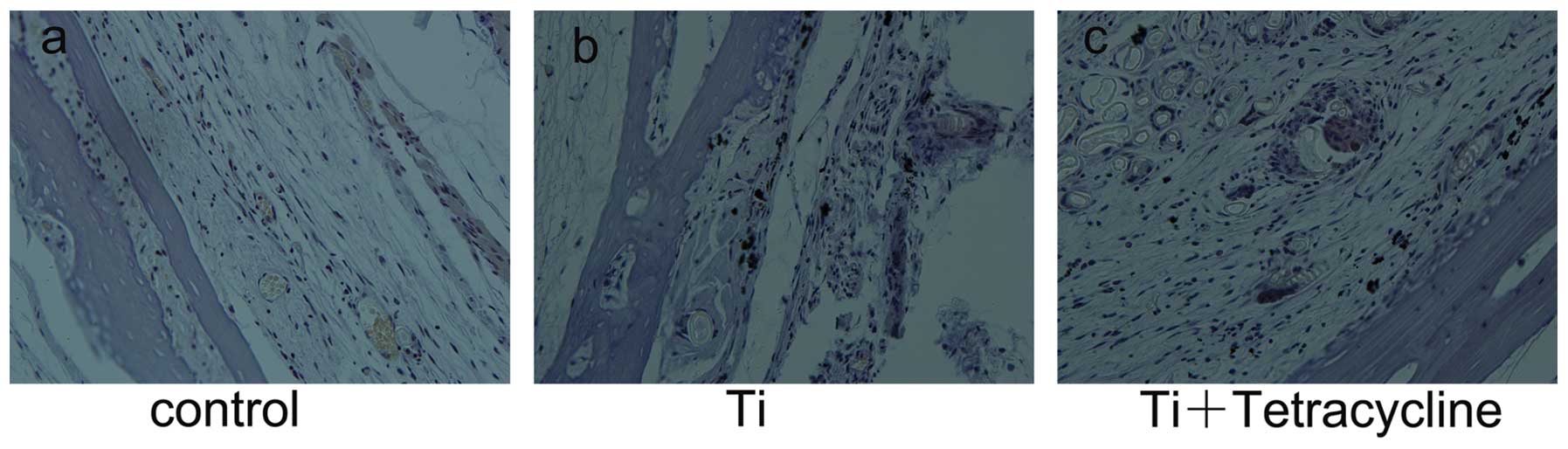
Morphological appearance of
H&E-stained sections
Air pouch thickness and cellular infiltration were
assessed on H&E-stained sections (22). Biomaterial particles stimulate
tissue inflammation, which is characterized by increased cellular
infiltration and pouch membrane thickness (23). As shown in Fig. 2, membrane thickness and frequency
of infiltrated cells were significantly increased in the Ti group
compared with the control group (P<0.05). These effects were
significantly alleviated by injection with tetracycline
(P<0.05). Bone morphology remained intact in the control group
(Fig. 2a), However, bone erosion
was observed in pouches stimulated with Ti particles, particularly
at the bone-membrane interface where erosion pits were observed
(Fig. 2b). Exposure to
tetracycline reduced the inflammatory osteolysis response in this
murine osteolysis model (Fig.
2c).
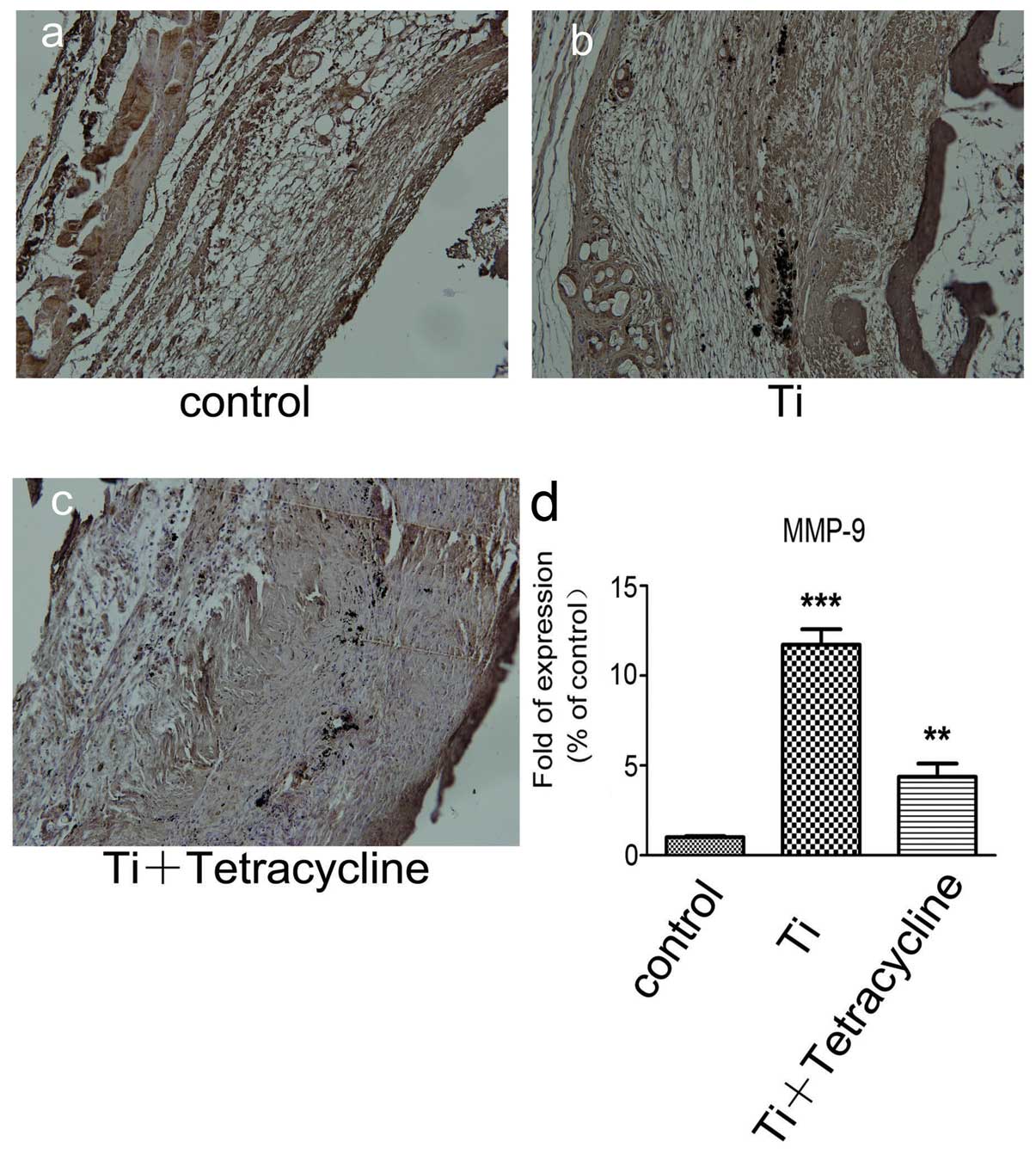
Effect of tetracycline on MMP-9
expression
Staining for MMP-9 in the Ti plus tetracycline group
was more intense compared to the control group, but less intense
compared to the Ti group (Fig.
3a–c). Staining for MMP-9 was predominantly observed in the
inflammatory cells surrounding the Ti particles.
Real-time RT-PCR assay indicated that MMP-9 gene
transcription was significantly higher in the Ti plus tetracycline
group compared to the control group (P<0.001) but was less
marked compared to the Ti group. There was also a significant
difference in the gene transcripts of MMP-9 between the Ti and Ti
plus tetracycline groups (Fig.
3d). Real-time RT-PCR analysis also demonstrated that Ti
particles stimulated a significant increase in the gene expression
of inflammatory mediators in air pouch tissues, while tetracycline
treatment significantly attenuated these responses.
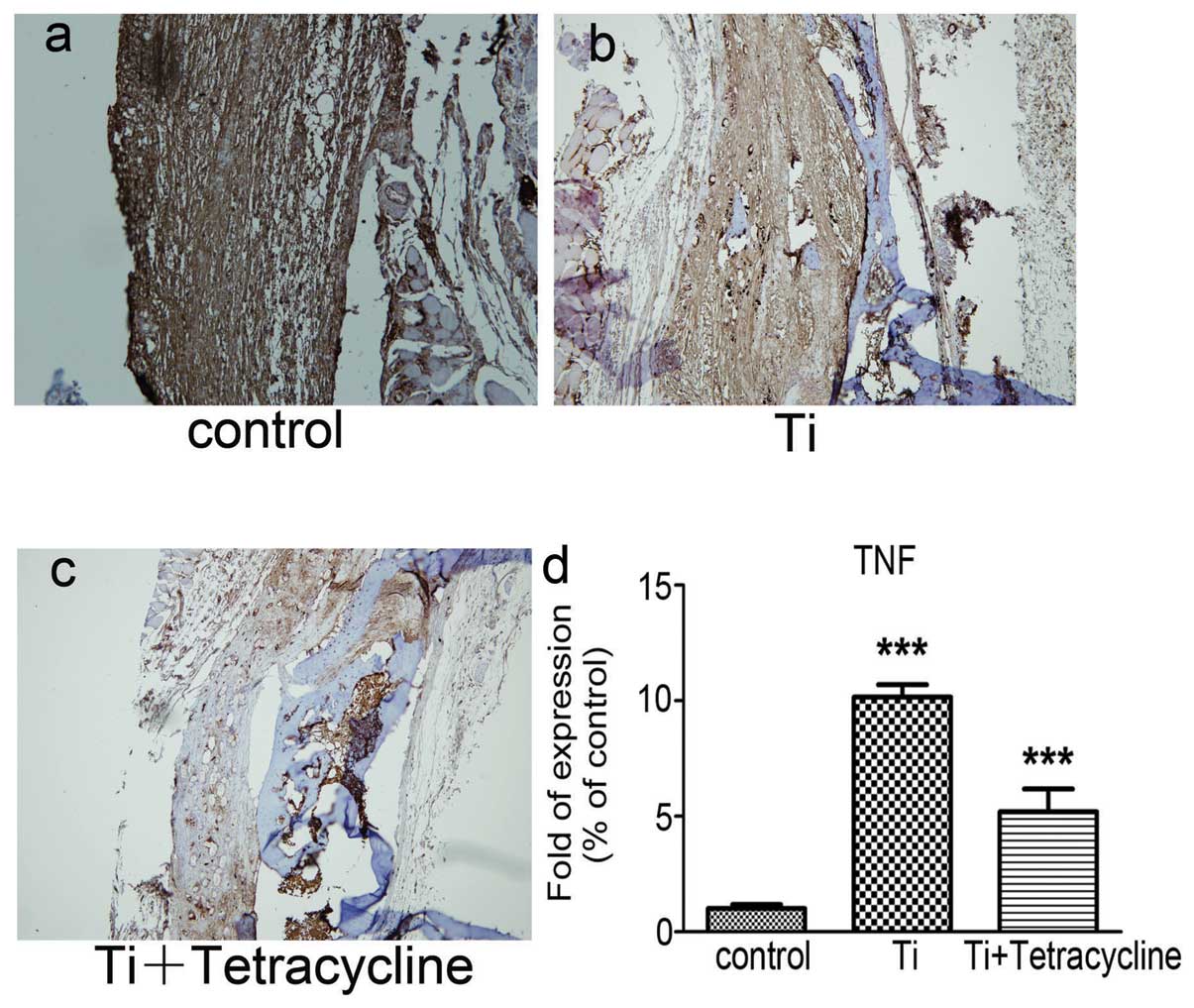
Effect of tetracycline on TNF-α
expression
Staining for TNF-α in the Ti group was significantly
stronger compared to the control group (Fig. 4a–c). In the Ti plus tetracycline
group there was evidence of significant suppression of the Ti
particle-induced increase in TNF-α (P<0.05).
The gene levels of TNF-α determined by real-time RT
PCR are displayed in Fig. 4d. The
Ti particles significantly increased TNF-α gene expression compared
to the control group (P<0.001). In addition, the number of gene
copies of TNF-α was significantly reduced in the Ti plus
tetracycline group (P<0.001).
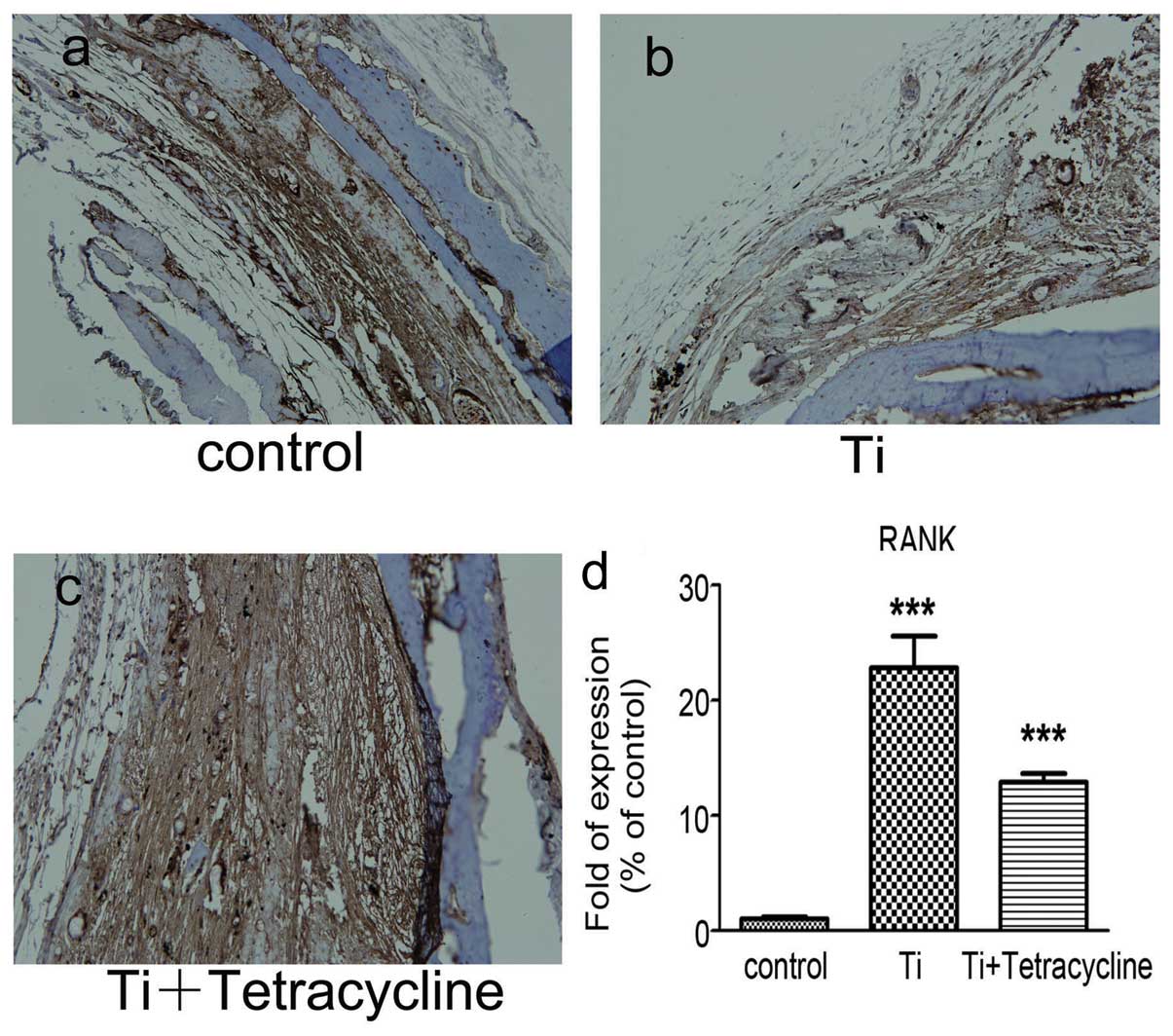
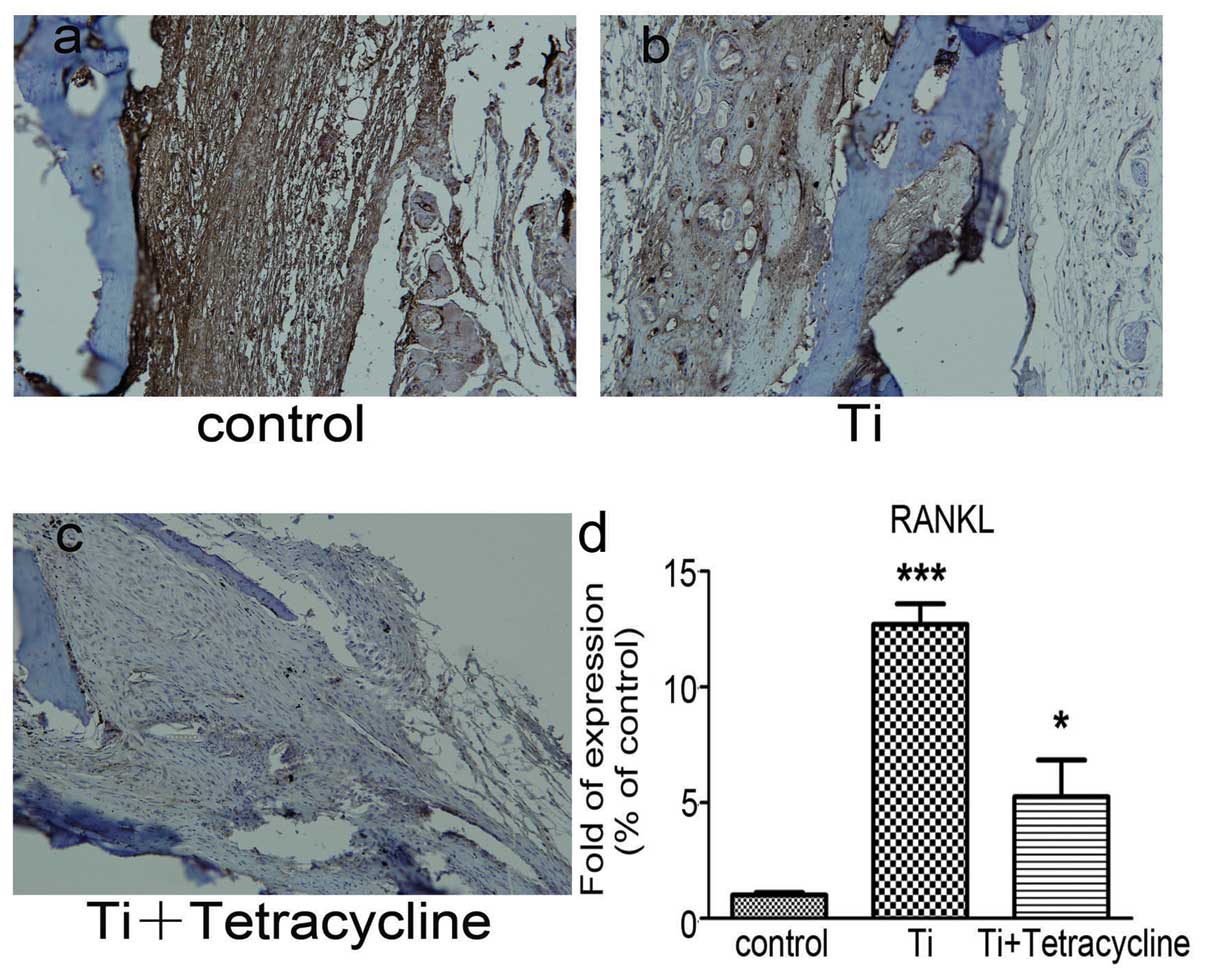
Effect of tetracycline on RANK and RANKL
expression
Immunohistochemical staining, as depicted in
Figs. 5a–c and 6a–c, demonstrated increases in staining
for both RANK and RANKL in the Ti group compared to the control
group. RANKL staining was discovered at the interface between the
pouch membranes and the implanted bone tissue and was significantly
decreased in the tetracycline-treated group (P<0.05).
Real-time RT-PCR demonstrated that the Ti particles
significantly (P<0.05) increased the expression of both RANK and
RANKL gene transcripts in comparison with the control group
(Figs. 5d and 6d). Exposure to tetracycline
significantly reduced (P<0.001) the increases in RANK and RANKL
gene transcripts compared with the Ti group.
Effect of tetracycline on
Ti-particle-induced osteoclastogenesis
Paraffin sectioned calvaria-implanted pouches were
stained to identify osteoclast-like cells. A typical TRAP-stained
photomicrograph exhibiting an accumulation of dark purple
TRAP-positive cells at the interface between the implanted calvaria
and the pouch membrane is displayed in Fig. 7. Relatively few TRAP-positive
cells were present in the control or Ti plus tetracycline
groups.
Discussion
Wear debris-induced osteolysis and aseptic loosening
is the most common complication of total joint replacement surgery
(2). During the 10-year period
following joint replacement, up to 20% of patients experience
radiographic loosening of the joint (24). From the data available to date,
persistent debris-induced inflammation and osteoclastogenesis may
be critical factors involved in the etiology of aseptic implant
loosening (25,26). Failed implants are often
surrounded by a synovial membrane-like interface, known as the
interfacial membrane, which plays a significant role in tissue
destruction and causes characteristic chronic inflammation in the
peri-implant area (27,28). The wear debris stimulus stimulates
the gathering of fibroblasts, macrophages, and foreign body giant
cells in interfacial membranes where they produce multiple
proinflammatory cytokines such as TNF-α and IL-6 (29,30). In addition, the abundance of MMPs
produced by these cells facilitates bone resorption around implants
resulting in aseptic loosening (31).
MMP-9 is produced by multinucleated giant cells in
response to wear debris at the interface membranes where it may
facilitate orthoclastic bone resorption by degradation of
extracellular matrix macromolecules present around and on the
surface of the bone trabeculae (32). Previous studies have demonstrated
that receptor activator of NF-κB (RANK) and RANK ligand (RANKL) are
key factors that regulate bone turnover and are present in tissues
near the site of peri-prosthetic osteolysis (18). However, whether MMP-9 acts as an
upstream regulator of inflammation and bone resorption-induced
RANK/RANKL signaling during inflammatory osteoclastogenesis remains
ambiguous.
The present study was undertaken in a murine
osteolysis model to examine the hypothesis that inhibition of Ti
particle-induced inflammatory osteoclastogenesis by treatment with
an MMP-9 inhibitor occurs through the downregulation of RANK/RANKL,
MMP-9 and TNF-α. Immunohistochemistry and real-time RT-PCR for
proinflammatory cytokines demonstrated that bone implantation and
Ti particle stimulation of air pouches significantly increased
protein and gene expression of MMP-9 and TNF-α, resulting in a
significant inflammatory reaction (33–35). We also demonstrated that TNF-α is
produced in response to wear particles and that it affects
downstream processes resulting in osteolysis (24,36). We used tetracycline, as a MMP-9
inhibitor on the basis that it has been observed to have a role in
the treatment of several ophthalmologic diseases by affecting the
expression of MMP-9 and TNF-α (37). Additionally, the tetracycline
derivative, doxycycline, reduces MMP-9-mediated inflammation and
the downstream effects of inflammation (16,38,39). This provides a potentially
important therapeutic strategy for the management of debris-induced
inflammation.
In our study, real-time RT-PCR displayed that the
number of gene copies of MMP-9 was significantly reduced by
exposure to tetracycline. In addition, immunohistochemical studies
suggested that tetracycline impeded MMP-9 expression in Ti particle
pouches. These findings suggest that tetracycline suppresses the
activation and expression of MMP-9 in Ti particle-induced
inflammatory osteolysis.
Data from our H&E-stained sections revealed that
the presence of Ti particles increased cellular infiltration and
pouch membrane thickness by stimulating tissue inflammation.
However, membrane thickness and the frequency of cell infiltration
were both significantly reduced in animals treated with
tetracycline. In addition, we were able to identify TRAP-positive
osteoclasts, by TRAP staining and discovered that tetracycline
reduced the number of TRAP-positive cells in Ti particle-induced
pouches. These findings suggest that tetracycline has the potential
to decrease bone resorption in the murine air pouch osteolysis
model.
The expression of both RANK and RANKL genes in our
mouse model of peri-prosthesis tissue was well defined. Other
studies have demonstrated that Ti particles significantly increased
RANK/RANKL gene expression in murine pouch tissues (17,40) and it has been proposed that
activation of the RANK/RANKL pathway may play a prominent role in
the bone loss associated with aseptic loosening (40). In our study, tetracycline, an
MMP-9 inhibitor, significantly reduced the expression of RANK and
RANKL. MMP-9 has numerous effects on osteoclast and osteoblast
function (41–43), and previous research has
demonstrated that wear debris increases the expression of MMP-9 and
TNF-α, both in vivo and in vitro (44). These findings are consistent with
our results.
TNF-α is considered to stimulate osteoclastogenesis
through the upregulation of RANKL production in osteoblasts and
through the stimulation of RANK expression in osteoclasts (45). This is in accordance with the
observation that RANK and RANKL are downstream signaling molecules
of the MMP-9 pathway. In our study, Ti particles increased the gene
expression of RANK and RANKL in comparison with the control group,
and tetracycline significantly decreased the expression of RANK and
RANKL and reduced the number of TRAP-positive cells, compared with
the control group. These findings suggest that tetracycline
suppresses the expression of the RANK and RANKL signaling pathways
in the presence of Ti particle-induced inflammatory osteolysis.
However, a previous study demonstrated that RANKL signals through
TRAF6 and that NFATc1 is a downstream effector of RANKL signaling
to modulate MMP-9 gene expression during osteoclast differentiation
(46). There may, therefore, be
an interrelationship between MMP-9 and RANK/RANKL signaling in the
development of inflammatory osteolysis, suggested by the fact that
treatment with an MMP-9 inhibitor significantly reduced the levels
of RANK/RANKL gene expression in the current study.
We used a murine model in this study, as it provides
a sensitive and cost-effective way to investigate the responses of
particle-induced inflammatory osteolysis in aseptic loosening and
offers advantages over previously reported models (47). We used air pouches implanted with
genetically identical donor mouse calvarial bone to gain insight
into changes in bone resorption and to provide a model of chronic
inflammation observed in inflammatory osteolysis. Collectively, our
results of the murine osteolysis model suggest that MMP-9 plays a
critical role in the process of wear debris-induced inflammatory
osteolysis. MMP-9 inhibition by tetracycline impeded the expression
of RANK/RANKL in the development of Ti-particle-induced
inflammatory osteolysis, indicating that MMP-9 inhibition is a
potential therapeutic target for the prevention of wear
debris-induced inflammatory osteolysis. The optimal clinical
protocol for tetracycline in this type of inflammation has yet to
be determined through further research.
Acknowledgements
This study was supported by the
Interdisciplinary (Engineering-Medical) Research Fund of Shanghai
Jiao Tong University (grant no. YG2011MS30), the Shanghai Municipal
Health Bureau Science Fund for Young Scholars (grant no.
2010QJ036A) and the National Natural Science Foundation of China
(grant no. 81171688).
References
|
1.
|
SM HorowitzSB DotyJM LaneAH
BursteinStudies of the mechanism by which the mechanical failure of
polymethylmethacrylate leads to bone resorptionJ Bone Joint Surg
Am7580281319938314821
|
|
2.
|
WH HarrisWear and periprosthetic
osteolysis: the problemClin Orthop Relat
Res6670200110.1097/00003086-200112000-00007
|
|
3.
|
E InghamJ FisherThe role of macrophages in
osteolysis of total joint
replacementBiomaterials2612711286200510.1016/j.biomaterials.2004.04.03515475057
|
|
4.
|
G HoltC MurnaghanJ ReillyRM MeekThe
biology of aseptic osteolysisClin Orthop Relat
Res460240252200717620815
|
|
5.
|
F von KnochC WedemeyerA HeckeleiPromotion
of bone formation by simvastatin in polyethylene particle-induced
osteolysisBiomaterials2657835789200515869791
|
|
6.
|
X PengK TaoT ChengJ ZhuX ZhangEfficient
inhibition of wear debris-induced inflammation by locally delivered
siRNABiochem Biophys Res
Commun377532537200810.1016/j.bbrc.2008.10.02618930023
|
|
7.
|
M TakagiYT KonttinenS
SantavirtaExtracellular matrix metalloproteinases around loose
total hip prosthesesActa Orthop
Scand65281286199410.3109/174536794089954548042479
|
|
8.
|
B NawrockiM PoletteH BurletP BirembautJJ
AdnetExpression of gelatinase A and its activator MT1-MMP in the
inflammatory periprosthetic response to polyethyleneJ Bone Miner
Res14288294199910.1359/jbmr.1999.14.2.2889933484
|
|
9.
|
I TakeiM TakagiS SantavirtaMatrix
metalloproteinases and tissue inhibitors of metalloproteinases in
joint fluid of the patients with loose artificial hip jointsJ
Biomed Mater
Res45175183199910.1002/(SICI)1097-4636(19990605)45:3%3C175::AID-JBM3%3E3.0.CO;2-910397973
|
|
10.
|
S WagnerH GollwitzerD WernickeR LangerKA
SiebenrockW HofstetterInterface membrane fibroblasts around
aseptically loosened endoprostheses express MMP-13J Orthop
Res26143152200810.1002/jor.2049417853491
|
|
11.
|
P ReponenC SahlbergC MunautI ThesleffK
TryggvasonHigh expression of 92-kD type IV collagenase (gelatinase
B) in the osteoclast lineage during mouse developmentJ Cell
Biol12410911102199410.1083/jcb.124.6.10918132709
|
|
12.
|
CH TsaiYJ ChenFM HuangYF SuYC ChangThe
upregulation of matrix metalloproteinase-9 in inflamed human dental
pulpsJ
Endod31860862200510.1097/01.don.0000164851.55389.4e16306818
|
|
13.
|
AL WucherpfennigYP LiWG
Stetler-StevensonAE RosenbergP StashenkoExpression of 92 kD type IV
collagenase/gelatinase B in human osteoclastsJ Bone Miner
Res9549556199410.1002/jbmr.56500904158030443
|
|
14.
|
PT De JongW TigchelaarCJ Van NoordenHM Van
der VisPolyethylene wear particles do not induce inflammation or
gelatinase (MMP-2 and MMP-9) activity in fibrous tissue interfaces
of loosening total hip arthroplastiesActa
Histochem113556563201120656340
|
|
15.
|
C ZhangTT TangWP RenXL ZhangKR
DaiInhibiting wear particle-induced osteolysis with doxycyclineActa
Pharmacol
Sin2816031610200710.1111/j.1745-7254.2007.00638.x17883947
|
|
16.
|
Y GuHM LeeT SorsaSR SimonLM
GolubDoxycycline inhibits mononuclear cell-mediated connective
tissue breakdownFEMS Immunol Med
Microbiol58218225201010.1111/j.1574-695X.2009.00625.x19909341
|
|
17.
|
D GengY XuH YangProtection against
titanium particle induced osteolysis by cannabinoid receptor 2
selective
antagonistBiomaterials3119962000201010.1016/j.biomaterials.2009.11.06920004468
|
|
18.
|
CA HoldingDM FindlayR StamenkovThe
correlation of RANK, RANKL and TNFalpha expression with bone loss
volume and polyethylene wear debris around hip
implantsBiomaterials2752125219200616806459
|
|
19.
|
C WedemeyerC NeuerburgA
PfeifferPolyethylene particle-induced bone resorption in
alpha-calcitonin gene-related peptide-deficient miceJ Bone Miner
Res2210111019200710.1359/jbmr.07040817419680
|
|
20.
|
SY YangB WuL MaytonProtective effects of
IL-1Ra or vIL-10 gene transfer on a murine model of wear
debris-induced osteolysisGene
Ther11483491200410.1038/sj.gt.330219214724688
|
|
21.
|
W RenSY YangPH WooleyA novel murine model
of orthopaedic wear-debris associated osteolysisScand J
Rheumatol33349357200410.1080/0300974041000594415513686
|
|
22.
|
X MaoX PanS ZhaoX PengT ChengX
ZhangProtection against titanium particle-induced inflammatory
osteolysis by the proteasome inhibitor bortezomib in
vivoInflammation3513781391201210.1007/s10753-012-9451-822391745
|
|
23.
|
PH WooleyR MorrenJ AndaryInflammatory
responses to orthopaedic biomaterials in the murine air
pouchBiomaterials23517526200210.1016/S0142-9612(01)00134-X11761173
|
|
24.
|
JD SamsJC MilbrandtJM FroelichAD
RainvilleDG AllanHospital outcome after emergent vs elective
revision total hip arthroplastyJ
Arthroplasty25826828201010.1016/j.arth.2010.01.09720378305
|
|
25.
|
NJ EpsteinBA WarmeJ SpanogleInterleukin-1
modulates periprosthetic tissue formation in an intramedullary
model of particle-induced inflammationJ Orthop
Res23501510200510.1016/j.orthres.2004.10.00415885468
|
|
26.
|
J WaddellKP PritzkerEL BoyntonIncreased
cytokine secretion in patients with failed implants compared with
patients with primary implantsClin Orthop Relat
Res434170176200510.1097/01.blo.0000155079.29604.d415864048
|
|
27.
|
RM AtkinsVG LangkamerMJ PerryCJ ElsonCM
CollinsBone-membrane interface in aseptic loosening of total joint
arthroplastiesJ
Arthroplasty12461464199710.1016/S0883-5403(97)90203-59195323
|
|
28.
|
SB GoodmanP HuieY SongCellular profile and
cytokine production at prosthetic interfaces. Study of tissues
retrieved from revised hip and knee replacementsJ Bone Joint Surg
Br80531539199810.1302/0301-620X.80B3.81589619952
|
|
29.
|
SR GoldringAL SchillerM RoelkeCM RourkeDA
O’NeilWH HarrisThe synovial-like membrane at the bone-cement
interface in loose total hip replacements and its proposed role in
bone lysisJ Bone Joint Surg Am6557558419836304106
|
|
30.
|
KJ KimHE RubashSC WilsonJA D’AntonioEJ
McClainA histologic and biochemical comparison of the interface
tissues in cementless and cemented hip prosthesesClin Orthop Relat
Res28714215219938448933
|
|
31.
|
GF MaA AliN VerzijlIncreased collagen
degradation around loosened total hip replacement implantsArthritis
Rheum5429282933200610.1002/art.2206416948130
|
|
32.
|
Y YokohamaT MatsumotoM HirakawaProduction
of matrix metalloproteinases at the bone-implant interface in loose
total hip replacementsLab Invest7389991119958558853
|
|
33.
|
F VelardD Laurent-MaquinJ BrauxThe effect
of zinc on hydroxyapatite-mediated activation of human
polymorphonuclear neutrophils and bone implant-associated acute
inflammationBiomaterials3120012009201010.1016/j.biomaterials.2009.11.066
|
|
34.
|
M TakagiY TamakiH HasegawaToll-like
receptors in the interface membrane around loosening total hip
replacement implantsJ Biomed Mater Res
A8110171026200710.1002/jbm.a.3123517415764
|
|
35.
|
P LaquerriereA Grandjean-LaquerriereS
Addadi-RebbahMMP-2, MMP-9 and their inhibitors TIMP-2 and TIMP-1
production by human monocytes in vitro in the presence of different
forms of hydroxyapatite
particlesBiomaterials2525152524200410.1016/j.biomaterials.2003.09.03414751736
|
|
36.
|
N TakiJM TatroR LoweVM GoldbergEM
GreenfieldComparison of the roles of IL-1, IL-6, and TNFalpha in
cell culture and murine models of aseptic
looseningBone4012761283200710.1016/j.bone.2006.12.05317236833
|
|
37.
|
TJ FedericiThe non-antibiotic properties
of tetracyclines: clinical potential in ophthalmic diseasePharmacol
Res64614623201110.1016/j.phrs.2011.06.01321843641
|
|
38.
|
AC LauTT DuongS ItoGJ WilsonRS
YeungInhibition of matrix metalloproteinase-9 activity improves
coronary outcome in an animal model of Kawasaki diseaseClin Exp
Immunol157300309200910.1111/j.1365-2249.2009.03949.x19604270
|
|
39.
|
S SamtaniJ AmaralMM CamposRN FarissSP
BecerraDoxycycline-mediated inhibition of choroidal
neovascularizationInvest Ophthalmol Vis
Sci5050985106200910.1167/iovs.08-317419516001
|
|
40.
|
WP RenDC MarkelR ZhangAssociation between
UHMWPE particle-induced inflammatory osteoclastogenesis and
expression of RANKL, VEGF, and Flt-1 in
vivoBiomaterials2751615169200610.1016/j.biomaterials.2006.04.00416814378
|
|
41.
|
JJ HyunHJ ChunB KeumEffect of omeprazole
on the expression of transcription factors in osteoclasts and
osteoblastsInt J Mol Med26877883201021042782
|
|
42.
|
W SriarjK AokiK OhyaY TakagiH
ShimokawaBovine dentine organic matrix down-regulates osteoclast
activityJ Bone Miner
Metab27315323200910.1007/s00774-009-0063-919296049
|
|
43.
|
SK TatJP PelletierF MineauJ CaronJ
Martel-PelletierStrontium ranelate inhibits key factors affecting
bone remodeling in human osteoarthritic subchondral bone
osteoblastsBone49559567201110.1016/j.bone.2011.06.00521700005
|
|
44.
|
MG ChoiHS KohD KluessEffects of titanium
particle size on osteoblast functions in vitro and in vivoProc Natl
Acad Sci USA10245784583200510.1073/pnas.050069310215755807
|
|
45.
|
S JuradoN Garcia-GiraltA Diez-PerezEffect
of IL-1beta, PGE(2), and TGF-beta1 on the expression of OPG and
RANKL in normal and osteoporotic primary human osteoblastsJ Cell
Biochem110304310201020225238
|
|
46.
|
K SundaramR NishimuraJ SennRF YoussefSD
LondonSV ReddyRANK ligand signaling modulates the matrix
metalloproteinase-9 gene expression during osteoclast
differentiationExp Cell
Res313168178200710.1016/j.yexcr.2006.10.00117084841
|
|
47.
|
X MaoX PanT ChengX ZhangTherapeutic
potential of the proteasome inhibitor bortezomib on titanium
particle-induced inflammation in a murine
modelInflammation35905912201210.1007/s10753-011-9392-721965047
|