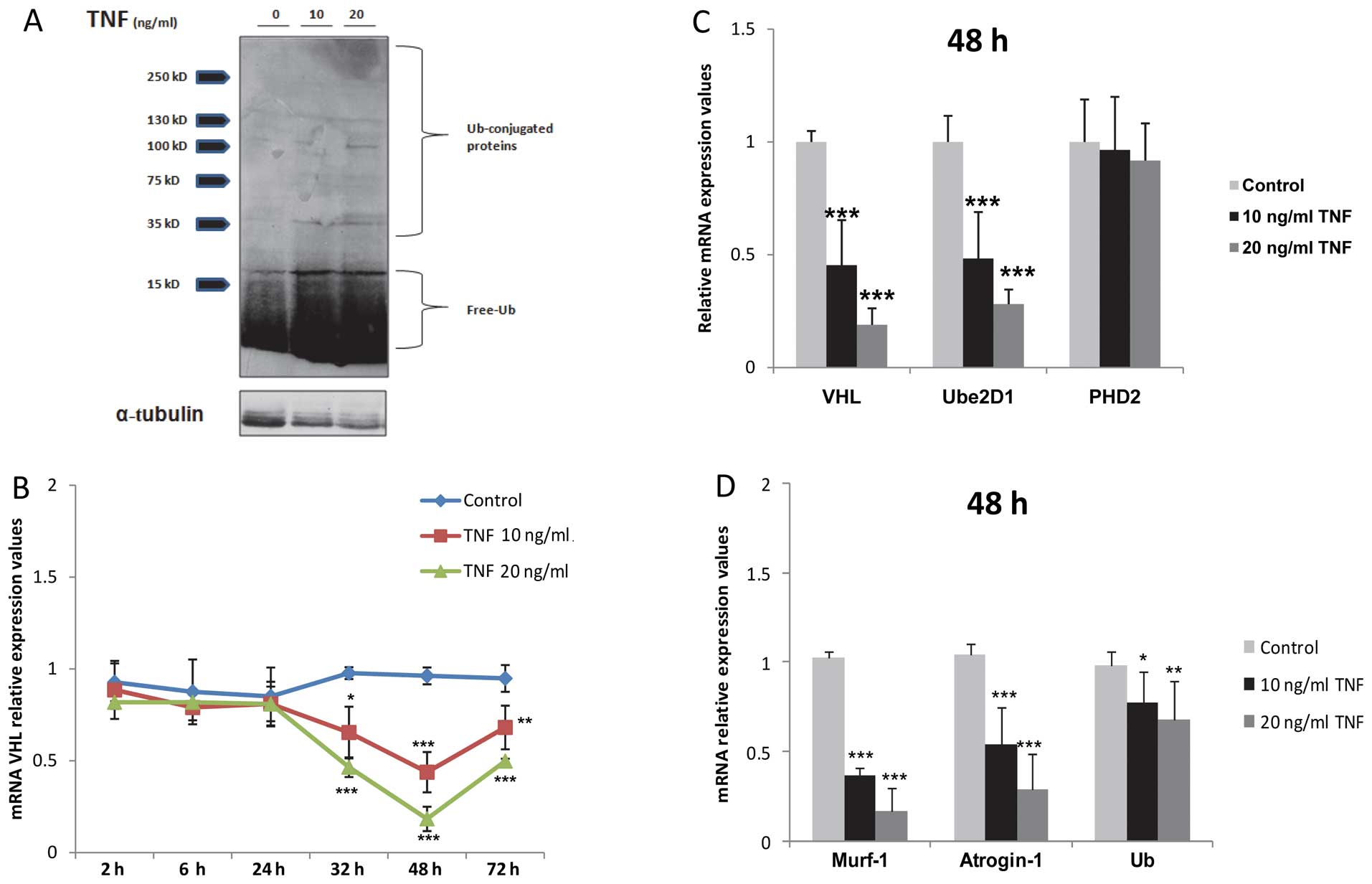
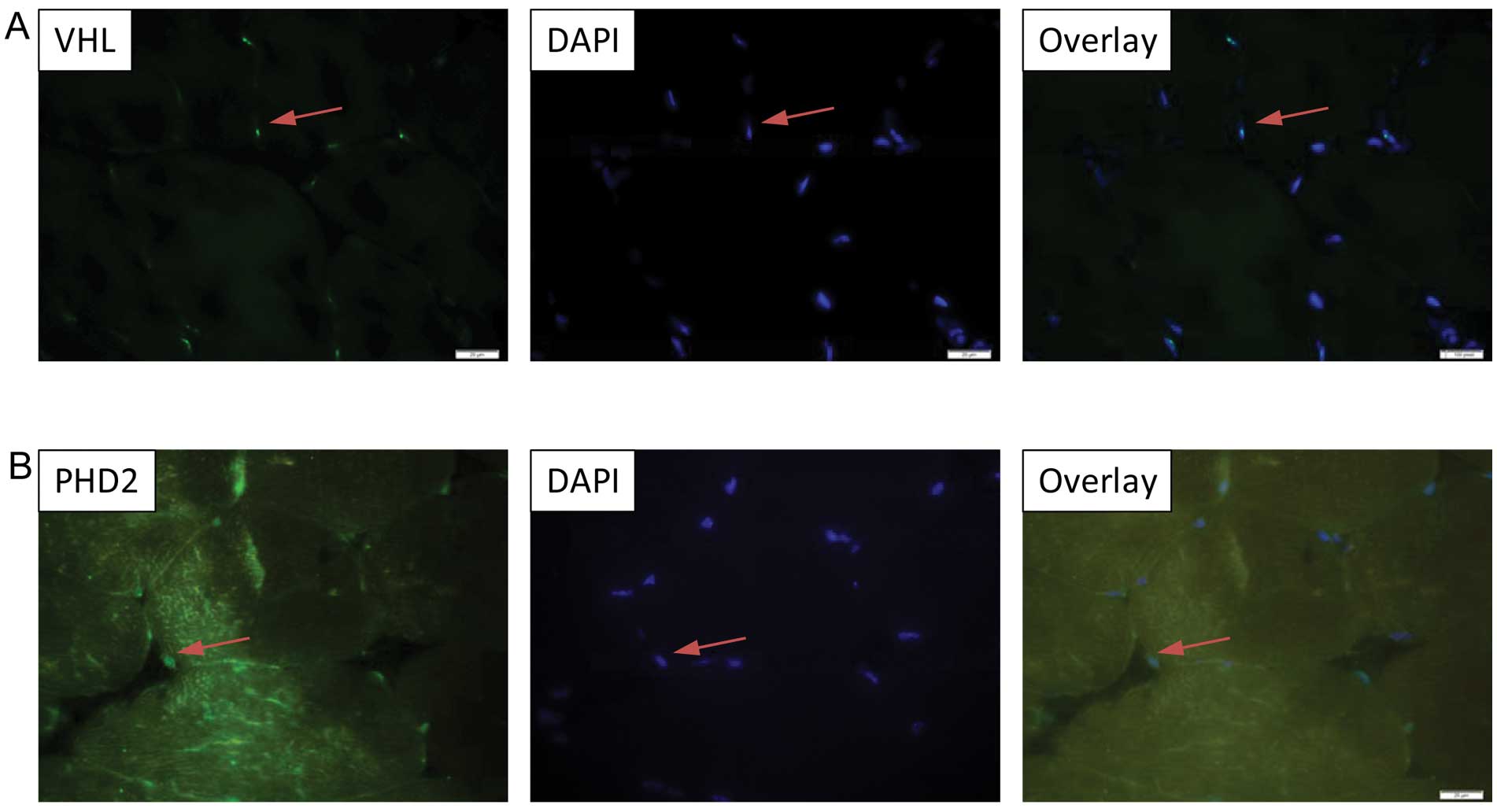
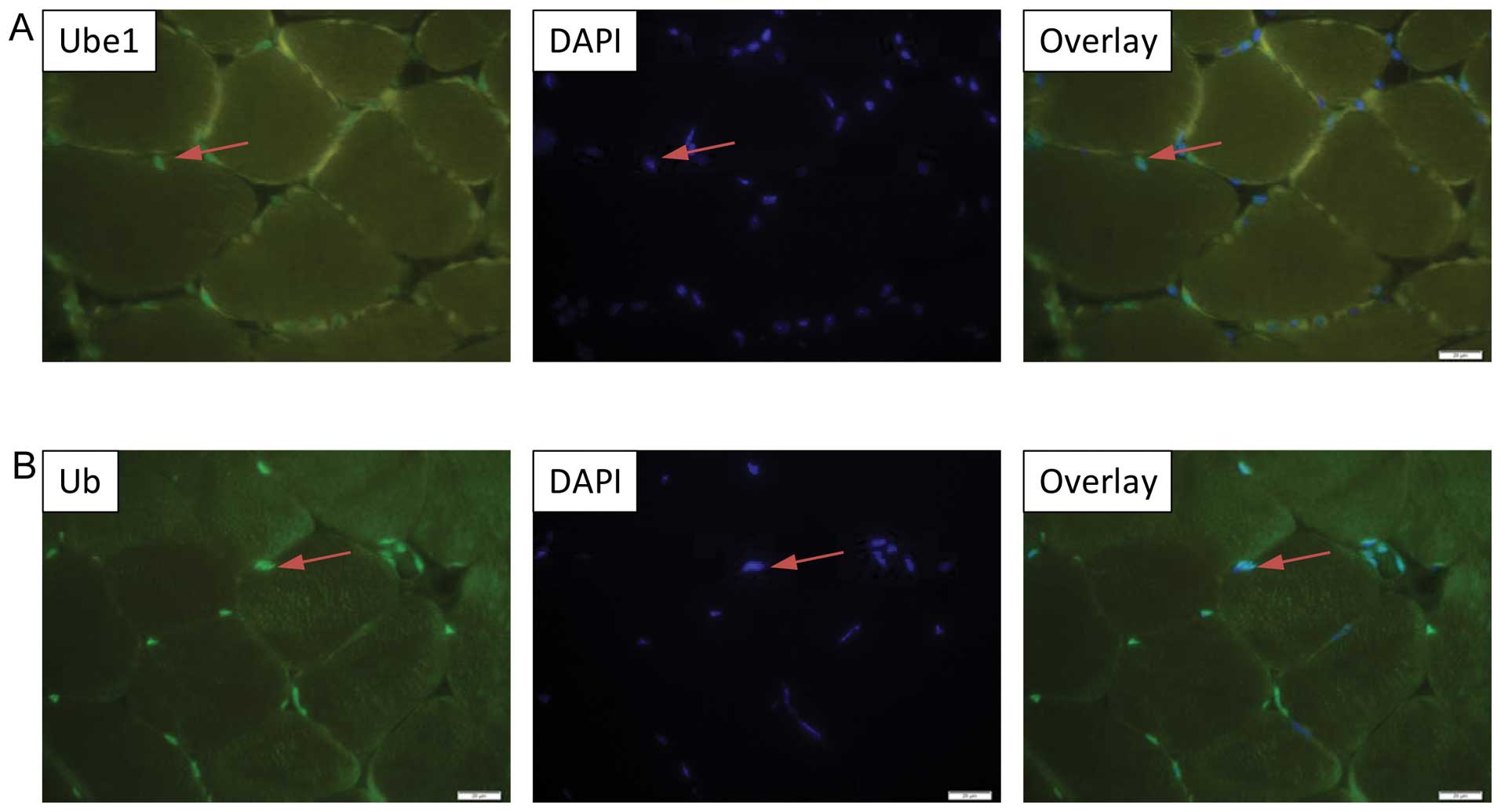
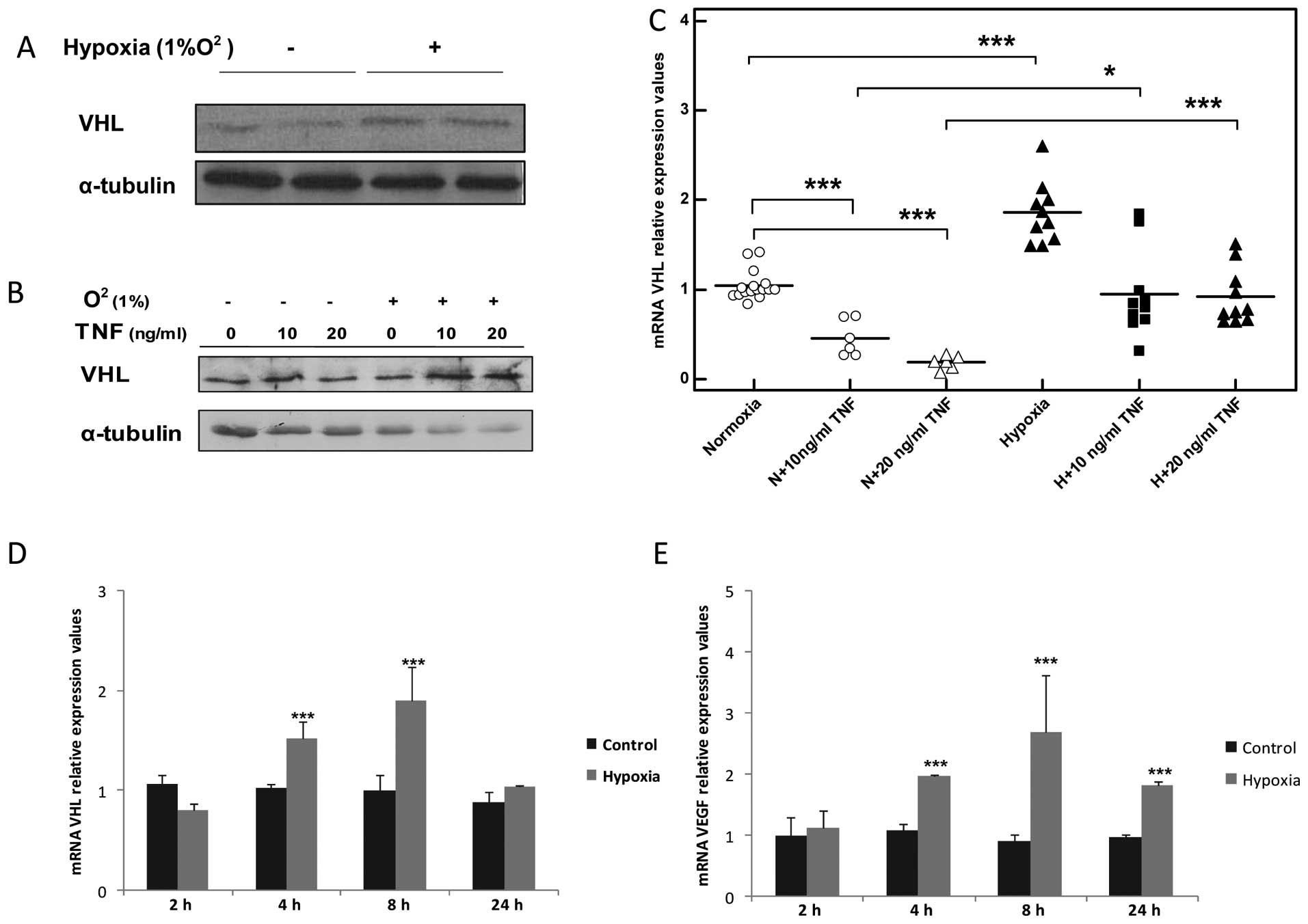
|
1
|
Jatta K, Eliason G, Portela-Gomes GM, et
al: Overexpression of von Hippel-Lindau protein in skeletal muscles
of patients with chronic obstructive pulmonary disease. J Clin
Pathol. 62:70–76. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
2
|
Jobin J, Maltais F, Doyon JF, et al:
Chronic obstructive pulmonary disease: capillarity and fiber-type
characteristics of skeletal muscle. J Cardiopulm Rehabil.
18:432–437. 1998. View Article : Google Scholar : PubMed/NCBI
|
|
3
|
Prior SJ, McKenzie MJ, Joseph LJ, et al:
Reduced skeletal muscle capillarization and glucose intolerance.
Microcirculation. 16:203–212. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
4
|
Kivela R, Silvennoinen M, Touvra AM, Lehti
TM, Kainulainen H and Vihko V: Effects of experimental type 1
diabetes and exercise training on angiogenic gene expression and
capillarization in skeletal muscle. FASEB J. 20:1570–1572. 2006.
View Article : Google Scholar : PubMed/NCBI
|
|
5
|
Gouzi F, Prefaut C, Abdellaoui A, et al:
Blunted muscle angiogenic training-response in COPD patients versus
sedentary controls. Eur Respir J. 41:806–814. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
6
|
Gagnon P, Lemire BB, Dube A, et al:
Preserved function and reduced angiogenesis potential of the
quadriceps in patients with mild COPD. Respir Res. 15:42014.
View Article : Google Scholar : PubMed/NCBI
|
|
7
|
Gan WQ, Man SF, Senthilselvan A and Sin
DD: Association between chronic obstructive pulmonary disease and
systemic inflammation: a systematic review and a meta-analysis.
Thorax. 59:574–580. 2004. View Article : Google Scholar : PubMed/NCBI
|
|
8
|
Petersen AM, Penkowa M, Iversen M, et al:
Elevated levels of IL-18 in plasma and skeletal muscle in chronic
obstructive pulmonary disease. Lung. 185:161–171. 2007. View Article : Google Scholar : PubMed/NCBI
|
|
9
|
Van Helvoort HA, Heijdra YF, Thijs HM,
Vina J, Wanten GJ and Dekhuijzen PN: Exercise-induced systemic
effects in muscle-wasted patients with COPD. Med Sci Sports Exerc.
38:1543–1552. 2006.
|
|
10
|
Deans C and Wigmore SJ: Systemic
inflammation, cachexia and prognosis in patients with cancer. Curr
Opin Clin Nutr Metab Care. 8:265–269. 2005. View Article : Google Scholar : PubMed/NCBI
|
|
11
|
Morley JE, Thomas DR and Wilson MM:
Cachexia: pathophysiology and clinical relevance. Am J Clin Nutr.
83:735–743. 2006.PubMed/NCBI
|
|
12
|
Delano MJ and Moldawer LL: The origins of
cachexia in acute and chronic inflammatory diseases. Nutr Clin
Pract. 21:68–81. 2006. View Article : Google Scholar : PubMed/NCBI
|
|
13
|
Piehl-Aulin K, Jones I, Lindvall B,
Magnuson A and Abdel-Halim SM: Increased serum inflammatory markers
in the absence of clinical and skeletal muscle inflammation in
patients with chronic obstructive pulmonary disease. Respiration.
78:191–196. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
14
|
Wagner PD: Possible mechanisms underlying
the development of cachexia in COPD. Eur Respir J. 31:492–501.
2008. View Article : Google Scholar : PubMed/NCBI
|
|
15
|
Garcia-Rio F, Miravitlles M, Soriano JB,
et al: Systemic inflammation in chronic obstructive pulmonary
disease: a population-based study. Respir Res. 11:632010.
View Article : Google Scholar : PubMed/NCBI
|
|
16
|
Pinto-Plata V, Casanova C, Mullerova H, et
al: Inflammatory and repair serum biomarker pattern: association to
clinical outcomes in COPD. Respir Res. 13:712012. View Article : Google Scholar : PubMed/NCBI
|
|
17
|
Tanni SE, Pelegrino NR, Angeleli AY,
Correa C and Godoy I: Smoking status and tumor necrosis
factor-alpha mediated systemic inflammation in COPD patients. J
Inflamm (Lond). 7:292010. View Article : Google Scholar : PubMed/NCBI
|
|
18
|
Eagan TM, Gabazza EC, D’Alessandro-Gabazza
C, et al: TNF-alpha is associated with loss of lean body mass only
in already cachectic COPD patients. Respir Res. 13:482012.
View Article : Google Scholar : PubMed/NCBI
|
|
19
|
Caron MA, Morissette MC, Theriault ME,
Nikota JK, Stampfli MR and Debigare R: Alterations in skeletal
muscle cell homeostasis in a mouse model of cigarette smoke
exposure. PLoS One. 8:e664332013. View Article : Google Scholar : PubMed/NCBI
|
|
20
|
Tang K, Wagner PD and Breen EC:
TNF-alpha-mediated reduction in PGC-1alpha may impair skeletal
muscle function after cigarette smoke exposure. J Cell Physiol.
222:320–327. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
21
|
Gosker HR, Langen RC, Bracke KR, et al:
Extrapulmonary manifestations of chronic obstructive pulmonary
disease in a mouse model of chronic cigarette smoke exposure. Am J
Respir Cell Mol Biol. 40:710–716. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
22
|
Langen RC, Schols AM, Kelders MC, van der
Velden JL, Wouters EF and Janssen-Heininger YM: Muscle wasting and
impaired muscle regeneration in a murine model of chronic pulmonary
inflammation. Am J Respir Cell Mol Biol. 35:689–696. 2006.
View Article : Google Scholar : PubMed/NCBI
|
|
23
|
Garcia-Martinez C, Agell N, Llovera M,
Lopez-Soriano FJ and Argiles JM: Tumour necrosis factor-alpha
increases the ubiquitinization of rat skeletal muscle proteins.
FEBS Lett. 323:211–214. 1993. View Article : Google Scholar : PubMed/NCBI
|
|
24
|
Garcia-Martinez C, Llovera M, Agell N,
Lopez-Soriano FJ and Argiles JM: Ubiquitin gene expression in
skeletal muscle is increased during sepsis: involvement of
TNF-alpha but not IL-1. Biochem Biophys Res Commun. 217:839–844.
1995. View Article : Google Scholar : PubMed/NCBI
|
|
25
|
Langen RC, Van Der Velden JL, Schols AM,
Kelders MC, Wouters EF and Janssen-Heininger YM: Tumor necrosis
factor-alpha inhibits myogenic differentiation through MyoD protein
destabilization. FASEB J. 18:227–237. 2004. View Article : Google Scholar : PubMed/NCBI
|
|
26
|
Frater-Schroder M, Risau W, Hallmann R,
Gautschi P and Bohlen P: Tumor necrosis factor type alpha, a potent
inhibitor of endothelial cell growth in vitro, is angiogenic in
vivo. Proc Natl Acad Sci USA. 84:5277–5281. 1987. View Article : Google Scholar : PubMed/NCBI
|
|
27
|
Cao PR, Kim HJ and Lecker SH:
Ubiquitin-protein ligases in muscle wasting. Int J Biochem Cell
Biol. 37:2088–2097. 2005. View Article : Google Scholar : PubMed/NCBI
|
|
28
|
Lecker SH, Solomon V, Price SR, Kwon YT,
Mitch WE and Goldberg AL: Ubiquitin conjugation by the N-end rule
pathway and mRNAs for its components increase in muscles of
diabetic rats. J Clin Invest. 104:1411–1420. 1999. View Article : Google Scholar : PubMed/NCBI
|
|
29
|
Combaret L, Adegoke OA, Bedard N, Baracos
V, Attaix D and Wing SS: USP19 is a ubiquitin-specific protease
regulated in rat skeletal muscle during catabolic states. Am J
Physiol Endocrinol Metab. 288:E693–E700. 2005. View Article : Google Scholar : PubMed/NCBI
|
|
30
|
Basic VT, Tadele E, Elmabsout AA, et al:
Exposure to cigarette smoke induces overexpression of von
Hippel-Lindau tumor suppressor in mouse skeletal muscle. Am J
Physiol Lung Cell Mol Physiol. 303:L519–L527. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
31
|
Semenza GL: Regulation of oxygen
homeostasis by hypoxia-inducible factor 1. Physiology (Bethesda).
24:97–106. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
32
|
Li YP, Schwartz RJ, Waddell ID, Holloway
BR and Reid MB: Skeletal muscle myocytes undergo protein loss and
reactive oxygen-mediated NF-kappaB activation in response to tumor
necrosis factor alpha. FASEB J. 12:871–880. 1998.PubMed/NCBI
|
|
33
|
Adams V, Mangner N, Gasch A, et al:
Induction of MuRF1 is essential for TNF-alpha-induced loss of
muscle function in mice. J Mol Biol. 384:48–59. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
34
|
Pijet B, Pijet M, Litwiniuk A, Gajewska M,
Pajak B and Orzechowski A: TNF-alpha and IFN-s-dependent muscle
decay is linked to NF-kappaB- and STAT-1alpha-stimulated Atrogin1
and MuRF1 genes in C2C12 myotubes. Mediators Inflamm.
2013:1714372013. View Article : Google Scholar : PubMed/NCBI
|
|
35
|
Tong X, Buelow K, Guha A, Rausch R and Yin
L: USP2a protein deubiquitinates and stabilizes the circadian
protein CRY1 in response to inflammatory signals. J Biol Chem.
287:25280–25291. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
36
|
Shukla R, Yue J, Siouda M, et al:
Proinflammatory cytokine TNF-alpha increases the stability of
hepatitis B virus X protein through NF-kappaB signaling.
Carcinogenesis. 32:978–985. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
37
|
Alvarez B, Quinn LS, Busquets S,
Lopez-Soriano FJ and Argiles JM: Direct effects of tumor necrosis
factor alpha (TNF-alpha) on murine skeletal muscle cell lines.
Bimodal effects on protein metabolism. Eur Cytokine Netw.
12:399–410. 2001.PubMed/NCBI
|
|
38
|
Plaisance I, Morandi C, Murigande C and
Brink M: TNF-alpha increases protein content in C2C12 and primary
myotubes by enhancing protein translation via the TNF-R1, PI3K, and
MEK. Am J Physiol Endocrinol Metab. 294:E241–E250. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
39
|
Bhatnagar S, Panguluri SK, Gupta SK,
Dahiya S, Lundy RF and Kumar A: Tumor necrosis factor-alpha
regulates distinct molecular pathways and gene networks in cultured
skeletal muscle cells. PLoS One. 5:e132622010. View Article : Google Scholar : PubMed/NCBI
|
|
40
|
Zhou J, Schmid T and Brune B: Tumor
necrosis factor-alpha causes accumulation of a ubiquitinated form
of hypoxia inducible factor-1alpha through a nuclear
factor-kappaB-dependent pathway. Mol Biol Cell. 14:2216–2225. 2003.
View Article : Google Scholar
|
|
41
|
Terasaki H, Kase S, Shirasawa M, et al:
TNF-alpha decreases VEGF secretion in highly polarized RPE cells
but increases it in non-polarized RPE cells related to crosstalk
between JNK and NF-kappaB pathways. PLoS One. 8:e699942013.
View Article : Google Scholar : PubMed/NCBI
|
|
42
|
Turcotte S, Desrosiers RR and Beliveau R:
Hypoxia upregulates von Hippel-Lindau tumor-suppressor protein
through RhoA-dependent activity in renal cell carcinoma. Am J
Physiol Renal Physiol, United States. F338–FP348. 2004. View Article : Google Scholar : PubMed/NCBI
|
|
43
|
Karhausen J, Kong T, Narravula S and
Colgan SP: Induction of the von Hippel-Lindau tumor suppressor gene
by late hypoxia limits HIF-1 expression. J Cell Biochem.
95:1264–1275. 2005. View Article : Google Scholar : PubMed/NCBI
|