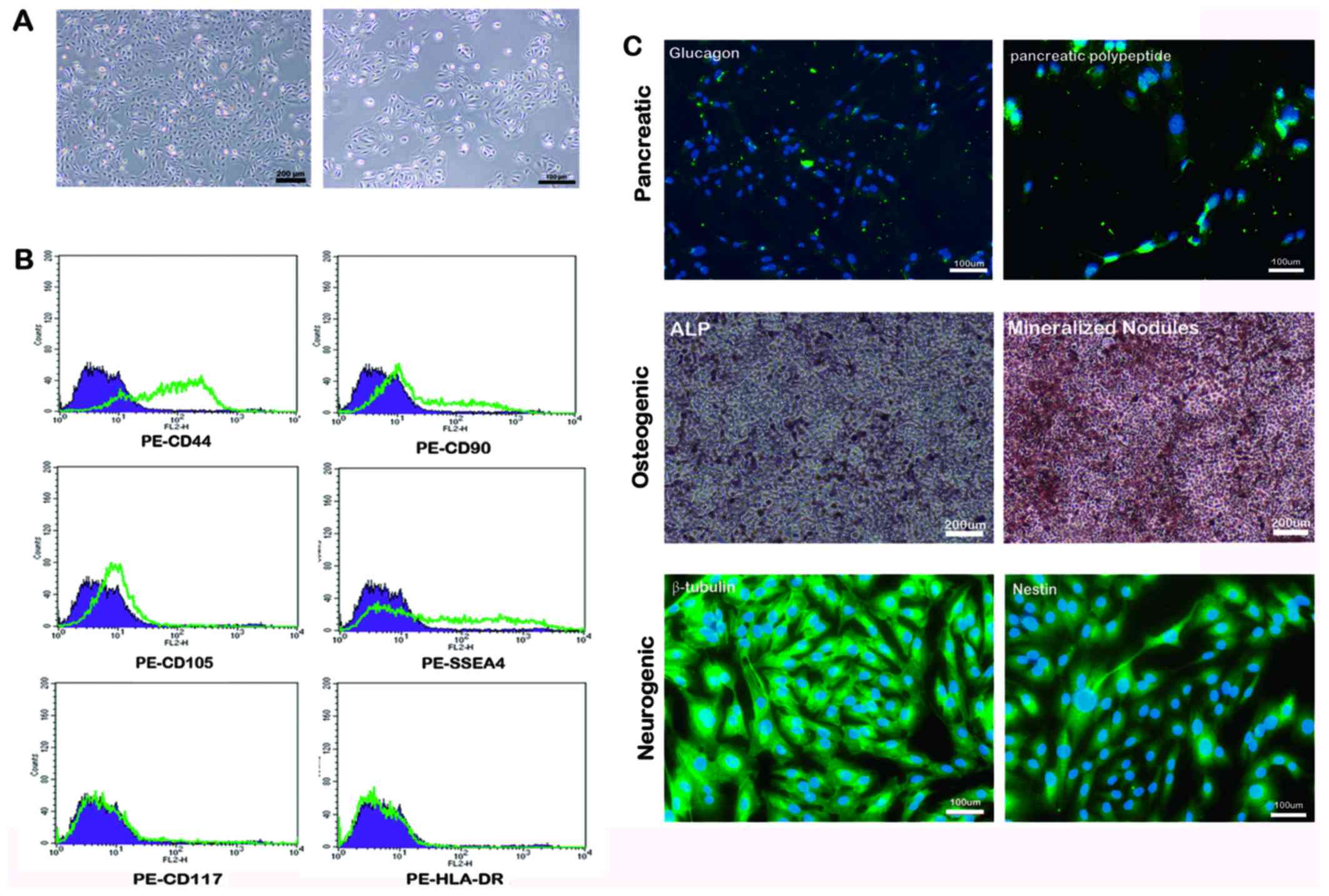
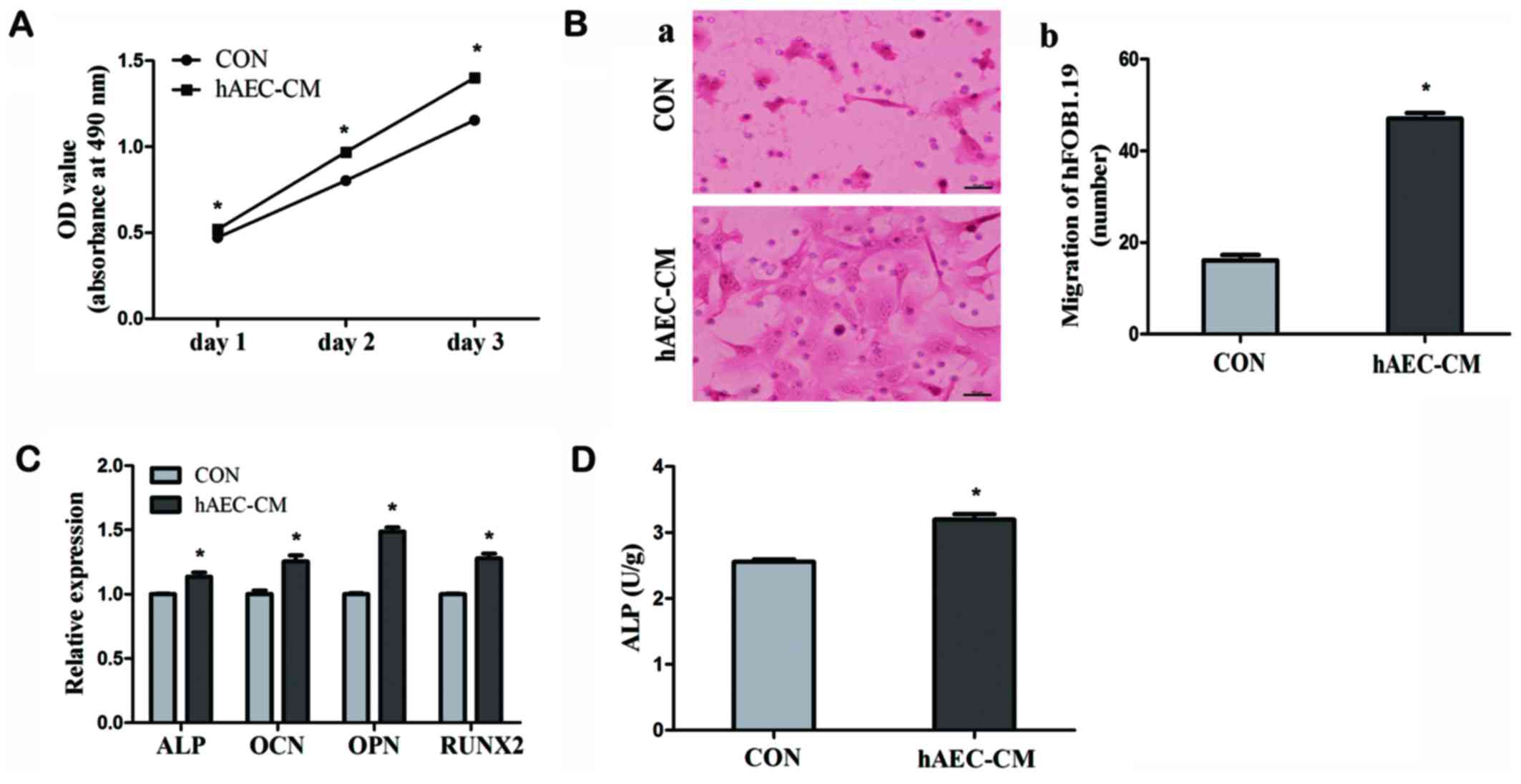
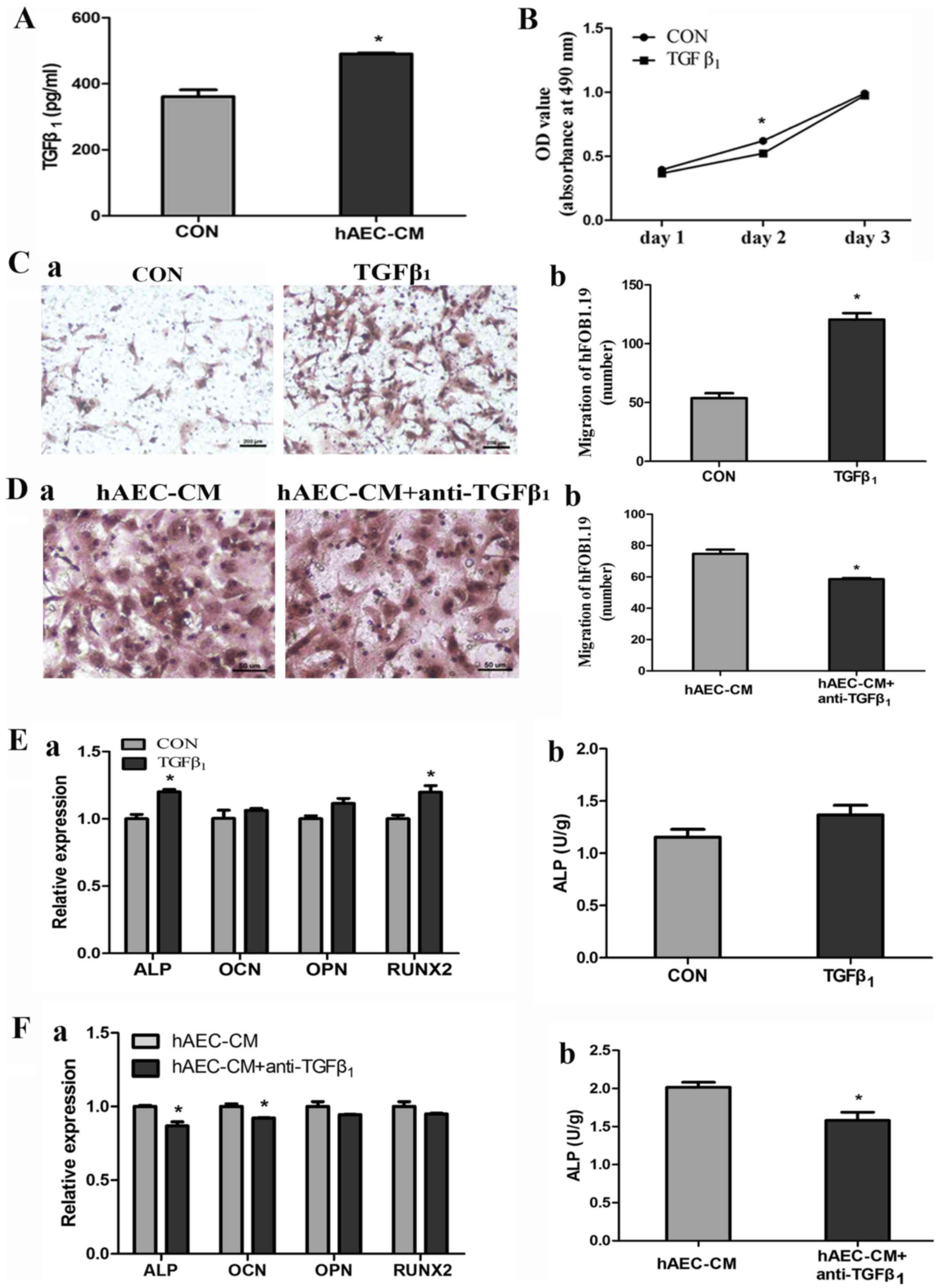
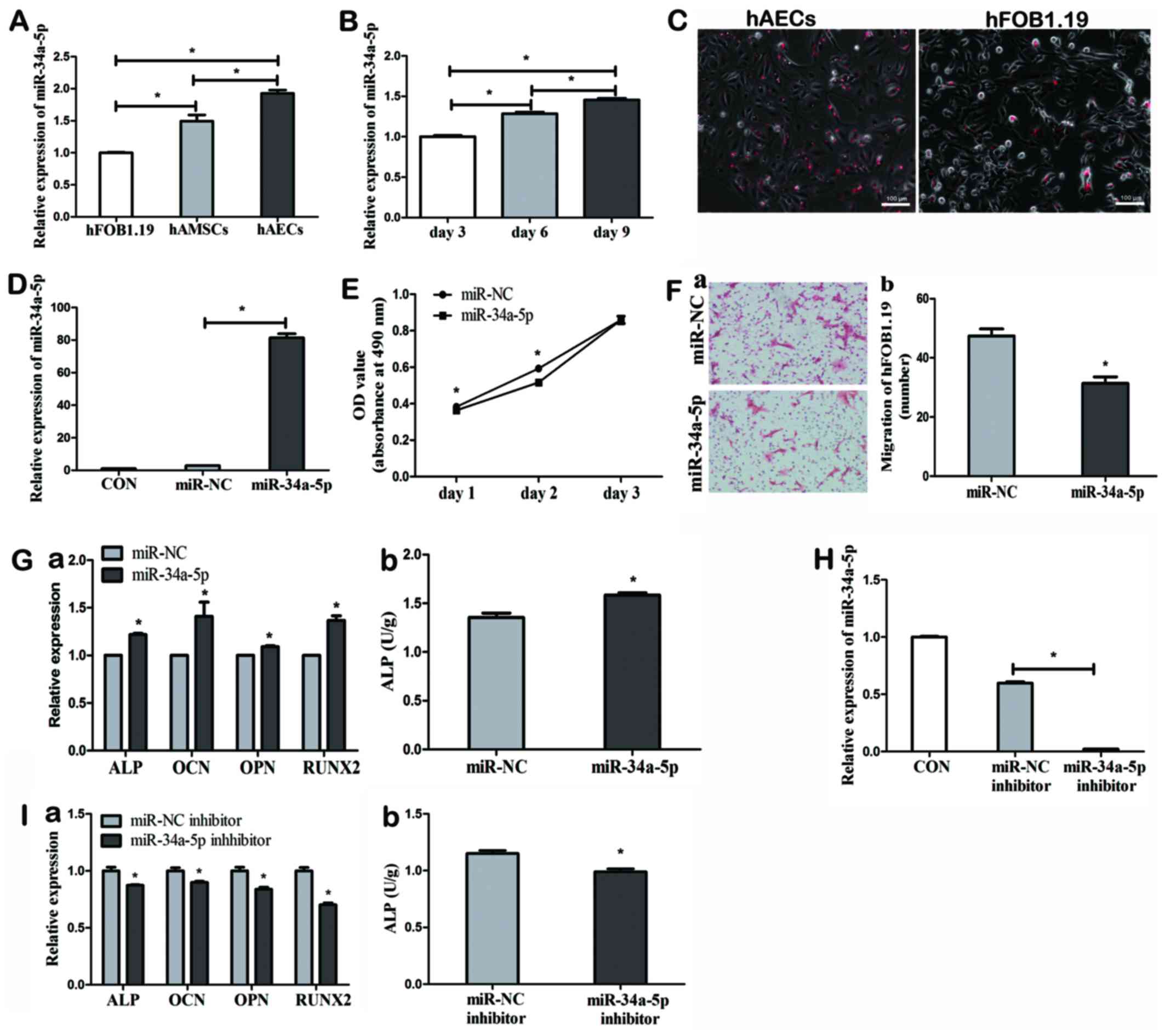
|
1
|
Grayson WL, Bunnell BA, Martin E, Frazier
T, Hung BP and Gimble JM: Stromal cells and stem cells in clinical
bone regeneration. Nat Rev Endocrinol. 11:140–150. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
2
|
Miki T, Lehmann T, Cai H, Stolz DB and
Strom SC: Stem cell characteristics of amniotic epithelial cells.
Stem Cells. 23:1549–1559. 2005. View Article : Google Scholar : PubMed/NCBI
|
|
3
|
Bilic G, Zeisberger SM, Mallik AS,
Zimmermann R and Zisch AH: Comparative characterization of cultured
human term amnion epithelial and mesenchymal stromal cells for
application in cell therapy. Cell Transplant. 17:955–968. 2008.
View Article : Google Scholar : PubMed/NCBI
|
|
4
|
Magatti M, Caruso M, De Munari S, Vertua
E, De D, Manuelpillai U and Parolini O: Human amniotic
membrane-derived, esenchymal and epithelial cells exert different
effects on monocyte-derived dendritic cell differentiation and
function. Cell Transplant. 24:1733–1752. 2015. View Article : Google Scholar
|
|
5
|
Wolbank S, Peterbauer A, Fahrner M,
Hennerbichler S, van Griensven M, Stadler G, Redl H and Gabriel C:
Dose-dependent immunomodulatory effect of human stem cells from
amniotic membrane: A comparison with human mesenchymal stem cells
from adipose tissue. Tissue Eng. 13:1173–1183. 2007. View Article : Google Scholar : PubMed/NCBI
|
|
6
|
Zhou Q, Liu XY, Ruan YX, Wang L, Jiang MM,
Wu J and Chen J: Construction of corneal epithelium with human
amniotic epithelial cells and repair of limbal deficiency in rabbit
models. Hum Cell. 28:22–36. 2015. View Article : Google Scholar
|
|
7
|
Vosdoganes P, Wallace EM, Chan ST, Acharya
R, Moss TJ and Lim R: Human amnion epithelial cells repair
established lung injury. Cell Transplant. 22:1337–1349. 2013.
View Article : Google Scholar
|
|
8
|
Vosdoganes P, Lim R, Koulaeva E, Chan ST,
Acharya R, Moss TJ and Wallace EM: Human amnion epithelial cells
modulate hyperoxia-induced neonatal lung injury in mice.
Cytotherapy. 15:1021–1029. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
9
|
Si J, Dai J, Zhang J, Liu S, Gu J, Shi J,
Shen SG and Guo L: Comparative investigation of human amniotic
epithelial cells and mesenchymal stem cells for application in bone
tissue engineering. Stem Cells Int. 2015:5657322015. View Article : Google Scholar : PubMed/NCBI
|
|
10
|
Jiawen S, Jianjun Z, Jiewen D, Dedong Y,
Hongbo Y, Jun S, Xudong W, Shen SG and Lihe G: Osteogenic
differentiation of human amniotic epithelial cells and its
application in alveolar defect restoration. Stem Cells Transl Med.
3:1504–1513. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
11
|
Dittmer J and Leyh B: Paracrine effects of
stem cells in wound healing and cancer progression (Review). Int J
Oncol. 44:1789–1798. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
12
|
Baraniak PR and McDevitt TC: Stem cell
paracrine actions and tissue regeneration. Regen Med. 5:121–143.
2010. View Article : Google Scholar :
|
|
13
|
Yao X, Guo Y, Wang Q, Xu M, Zhang Q, Li T
and Lai D: The Paracrine effect of transplanted human amniotic
epithelial cells on ovarian function improvement in a mouse model
of chemotherapy-induced primary ovarian insufficiency. Stem Cells
Int. 4148923:2016. View Article : Google Scholar
|
|
14
|
Ando Y, Matsubara K, Ishikawa J, Fujio M,
Shohara R, Hibi H, Ueda M and Yamamoto A: Stem cell-conditioned
medium accelerates distraction osteogenesis through multiple
regenerative mechanisms. Bone. 61:82–90. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
15
|
Wang Y, Ma J, Du Y, Miao J and Chen N:
Human amnion-derived mesenchymal stem cells protect human bone
marrow mesenchymal stem cells against oxidative stress-mediated
dysfunction via ERK1/2 MAPK signaling. Mol Cells. 39:186–194. 2016.
View Article : Google Scholar : PubMed/NCBI
|
|
16
|
van der Pol E, Böing AN, Harrison P, Sturk
A and Nieuwland R: Classification, functions, and clinical
relevance of extracellular vesicles. Pharmacol Rev. 64:676–705.
2012. View Article : Google Scholar : PubMed/NCBI
|
|
17
|
Desrochers LM, Antonyak MA and Cerione RA:
Extracellular vesicles: Satellites of information transfer in
cancer and stem cell biology. Dev Cell. 37:301–309. 2016.
View Article : Google Scholar : PubMed/NCBI
|
|
18
|
Davis LA, Dienelt A and zur Nieden I:
Absorption-based assays for the analysis of osteogenic and
chondrogenic yield. Methods Mol Biol. 690:255–272. 2011. View Article : Google Scholar
|
|
19
|
An JH, Park H, Song JA, Ki KH, Yang JY,
Choi HJ, Cho SW, Kim SW, Kim SY, Yoo JJ, et al: Transplantation of
human umbilical cord blood-derived mesenchymal stem cells or their
conditioned medium prevents bone loss in ovariectomized nude mice.
Tissue Eng Part A. 19:685–696. 2013. View Article : Google Scholar :
|
|
20
|
Burke J, Kolhe R, Hunter M, Isales C,
Hamrick M and Fulzele S: Stem cell-derived exosomes: A potential
alternative therapeutic agent in orthopaedics. Stem Cells Int.
5802529:2016. View Article : Google Scholar : PubMed/NCBI
|
|
21
|
Furuta T, Miyaki S, Ishitobi H, Ogura T,
Kato Y, Kamei N, Miyado K, Higashi Y and Ochi M: Mesenchymal stem
cell-derived exosomes promote fracture healing in a mouse model.
Stem Cells Transl Med. 5:1620–1630. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
22
|
Osugi M, Katagiri W, Yoshimi R, Inukai T,
Hibi H and Ueda M: Conditioned media from mesenchymal stem cells
enhanced bone regeneration in rat calvarial bone defects. Tissue
Eng Part A. 18:1479–1489. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
23
|
Meirelles LS, Fontes AM, Covas DT and
Caplan AI: Mechanisms involved in the therapeutic properties of
mesenchymal stem cells. Cytokine Growth Factor Rev. 20:419–427.
2009. View Article : Google Scholar
|
|
24
|
Lawson C, Vicencio JM, Yellon DM and
Davidson SM: Microvesicles and exosomes: New players in metabolic
and cardiovascular disease. J Endocrinol. 228:R57–R71. 2016.
View Article : Google Scholar : PubMed/NCBI
|
|
25
|
Javelaud D and Mauviel A: Crosstalk
mechanisms between the mitogen-activated protein kinase pathways
and Smad signaling downstream of TGF-beta: Implications for
carcinogenesis. Oncogene. 24:5742–5750. 2005. View Article : Google Scholar : PubMed/NCBI
|
|
26
|
Kassem M, Kveiborg M and Eriksen EF:
Production and action of transforming growth factor-beta in human
osteoblast cultures: Dependence on cell differentiation and
modulation by calcitriol. Eur J Clin Invest. 30:429–437. 2000.
View Article : Google Scholar : PubMed/NCBI
|
|
27
|
Janssens K, ten Dijke P, Janssens S and
Van Hul W: Transforming growth factor-beta1 to the bone. Endocr
Rev. 26:743–774. 2005. View Article : Google Scholar : PubMed/NCBI
|
|
28
|
Ehnert S, Baur J, Schmitt A, Neumaier M,
Lucke M, Dooley S, Vester H, Wildemann B, Stöckle U and Nussler AK:
TGF-β1 as possible link between loss of bone mineral density and
chronic inflammation. PLoS One. 5:pp. e140732010, View Article : Google Scholar
|
|
29
|
Eichner A, Brock J, Heldin CH and
Souchelnytskyi S: Bone morphogenetic protein-7 (OP1) and
transforming growth factor-beta1 modulate
1,25(OH)2-vitamin D -induced differentiation of human
osteoblasts. Exp Cell Res. 275:132–142. 2002. View Article : Google Scholar : PubMed/NCBI
|
|
30
|
Fan C, Jia L, Zheng Y, Jin C, Liu Y, Liu H
and Zhou Y: MiR-34a promotes osteogenic differentiation of human
adipose-derived stem cells via the RBP2/NOTCH1/CYCLIN D1
coregulatory network. Stem Cell Reports. 7:236–248. 2016.
View Article : Google Scholar : PubMed/NCBI
|
|
31
|
Komori T: Glucocorticoid signaling and
bone biology. Horm Metab Res. 48:755–763. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
32
|
Yan K, Gao J, Yang T, Ma Q, Qiu X, Fan Q
and Ma B: MicroRNA-34a inhibits the proliferation and metastasis of
osteosarcoma cells both in vitro and in vivo. PLoS One. 7:pp.
e337782012, View Article : Google Scholar : PubMed/NCBI
|