Introduction
Among tumors involving the female reproductive
system, ovarian cancer (OC) currently has the highest mortality
rate. According to statistics, there were 22,240 newly diagnosed
ovarian cancer cases and 14,070 ovarian cancer mortalities in the
United States in 2018 (1). Clear
cell carcinoma is a subtype of OC, which accounts for 5-25% of all
cases (2,3). Previous studies have reported that
ovarian clear cell carcinoma (OCCC) is relatively insensitive
compared with conventional chemotherapeutics for OC, including
paclitaxel and platinum-based drugs, demonstrates a poor response
to second-line chemotherapeutics, and there is also a lack of
specific targeted chemotherapeutics for OCCC (4-6).
Thus, the prognosis of OCCC is considered unfavorable compared with
other subtypes of OC, such as serous and mucinous, whereby the
5-year survival rate is only 27% (7). Studying the occurrence and
developmental mechanisms of OCCC has a significant value for
targeted treatment and improving prognosis.
Annexin IV (ANXA4), a member of the annexin family,
can reversibly bind with membrane phospholipids in a calcium
ion-dependent form and participate in regulating the biological
behavior of tumor cells, including proliferation, apoptosis,
invasion and metastasis (8-10).
A previous study demonstrated that ANXA4 expression is
significantly upregulated in OCCC compared with ovarian serous
carcinoma (11). Thus, it is
speculated that ANXA4 is a specific marker of OCCC; however, the
molecular mechanism by which ANXA4 regulates the biological
behavior of OCCC remains unknown. In 2010, Jeon et al
(12) reported that ANXA4 can
enter the nucleus of the cervical cancer Hela cell line and
influence the transcription of target genes associated with nuclear
factor-κ-light-chain-enhancer of activated B cells (NF-κB) p50.
NF-κB is retained in the cytoplasm at a resting state by binding to
inhibitor of κB (IκB), the inhibitory protein of NF-κB. When cells
are stimulated by exogenous factors, such as inflammatory factors,
oncogenes and viral infections, IκB is degraded via
phosphorylation, thus releasing NF-κB, which then enters the
nucleus to function as a transcriptional factor, and induces the
expression of target genes, including B-cell lymphoma-2 (Bcl-2)
family, c-Jun N-terminal kinase, Cyclin D1, tumor necrosis factor
and interleukin-1 (13-15). Consequently, the antiapoptotic
activity of tumor cells is enhanced and tumor growth is promoted
(16,17).
This raises two questions, the first is whether
ANXA4 interacts with NF-κB p50 in OCCC, and if so, how does this
interaction influence the biological behavior of OCCC? Masuishi
et al (18) reported that
high ANXA4 expression in OCCC is regulated by wild-type p53.
Wild-type p53, a key factor in regulating cell-cycle arrest, cell
aging and apoptosis, is the most effective tumor-inhibiting gene,
whereby exogenously-acquired wild-type p53 can inhibit autophagy,
reverse drug resistance and promote apoptosis of tumor cells
(19).
The present study aimed to investigate the
expression levels of p53, ANXA4 and NF-κB p50 in several subtypes
of epithelial ovarian cancer tissues, and assess the association
between their expression and OCCC clinicopathological
characteristics, and prognosis. The structural association and
interaction between ANXA4 and NF-κB p50 were assessed via
co-immunoprecipitation, confocal laser scanning micros-copy and
western blot analyses. In addition, a protein-protein interaction
(PPI) network of ANXA4, NF-κB p50 and p53 proteins was constructed
and enrichment analysis was performed to determine the underlying
molecular mechanism of ANXA4 in OCCC. The results demonstrated that
p53 and ANXA4/NF-κB p50 complexes activated the NF-κB signaling
pathway, increased the expression levels of Cyclin D1 and Bcl-2,
promoted proliferation and inhibited apoptosis of OCCC cells.
Materials and methods
Ethical approval
The patient samples used in the present study were
fully encoded to protect patient confidentially. All procedures
involving human participants were approved by the Ethics Committee
of Shengjing Hospital of China Medical University (Shenyang, China;
approval no. 2013PS66K). The ethics committee waived the need for
patient consent in this retrospective study, as the patient
information was withheld.
Specimen samples
A total of 151 ovarian paraffin specimens with
complete clinical and pathological data were collected from the
Department of Gynecology at Shengjing Hospital of China Medical
University between January 2003 and December 2012. For
immunohistochemistry, resected ovarian tissues were immediately
frozen in liquid nitrogen and stored at −80°C until subsequent
experimentation. Frozen sections (5 µm) were cut on a
cryostat, and control sections were stained with hematoxylin and
eosin (H&E) for 5 min at room temperature. Among these, 86
cases were OCCC, 30 cases were serous OC, 17 cases were mucinous
OC, eight cases were intrauterine membrane-like OC and 10 cases
were normal ovarian tissue (from ovariectomized patients with
squamous cervical cancer during the same period). All tissue
sections were blindly assessed by two pathologists from Shengjing
Hospital of China Medical University (Shenyang, China) to obtain a
definitive diagnosis.
The mean age of the OCCC group was 52.7 years (age
range, 29-73 years), while the mean age of the other OC subtypes
group and normal ovarian group was 50.2 years (age range, 16-77
years), and 49.4 years (age range, 46-57 years), respectively. No
statistically significant differences were observed between the age
of patients with OCCC, other subtypes of OC and normal ovarian
tissues. Cancer staging was determined according to the 2014
International Federation of Gynecology and Obstetrics (FIGO)
staging system (20). Among the
86 cases of OCCC, 51 were in stage I, eight were in stage II, 23
were in stage III and four cases were in stage IV. In addition,
fours cases refused chemotherapy, 11 patients were lost to
follow-up and the remaining 71 patients were given 6-8 cycles of
paclitaxel plus platinum systemic chemotherapy following surgery,
according to the National Comprehensive Cancer Network (NCCN)
guidelines (21) and underwent
regular follow-ups; there were 52 cases of sensitivity to
chemotherapy and 19 cases of chemo-therapy resistance. The outcomes
for the 71 patients included 25 deceased patients, 1 patient who
survived with recurrence and 45 patients who survived without
tumors. None of the patients with ovarian cancer underwent
chemotherapy prior to surgery. The NCCN criteria states that
(1) the chemotherapy resistant
group included patients who had a clinical response to the initial
paclitaxel and carboplatin (TC) chemotherapy, but had cancer
recurrence during the late stage of chemotherapy or within 6 months
after completion of chemotherapy, (2) and the chemotherapy sensitive group
included patients with clinical remission over 12 months after
completion of chemotherapy with TC. Factors diagnostic for ovarian
cancer relapse include: i) Continuously increased CA125 levels, ii)
Solid lesions identified by gynecological examination, iii) Tumors
identified by imaging, iv) Ascites and v) An intestinal obstruction
of unknown etiology. Follow-up time was calculated from the date of
surgery to the date of progression, death, and last visit or
contact with the patient. Overall survival (OS) was defined as the
time interval between the date of surgery and the date of
death.
Immunohistochemistry (IHC)
The ovarian tissue samples for immunohistochemical
analyses were fixed in 4% paraformaldehyde for 48 h at room
temperature and embedded in paraffin, according to conventional
histological method (22). The
paraffin-embedded tissue samples were cut into 5-µm thick
sections and the immunohistochemical expression of ANXA4, NF-κB p50
and wild-type p53 in tissue sections were determined using Strept
Actividin-Biotin Complex (SABC) IHC staining (Boster Biological
Technology). The tissue sections were deparaffinized in xylene at
room temperature and rehydrated in a descending ethanol series
(100, 100, 95, 90 and 75%). Deparaffinized sections were incubated
with 3% H2O2 for 10 min at room temperature
to inhibit endog-enous peroxidase activity, blocked with 10% normal
goat serum (cat. no. AR0009; Boster Biological Technology) for 30
min at 37°C, and subsequently incubated with the following primary
antibodies: Mouse anti-human ANXA4 (1:200; cat. no. sc-46693),
mouse anti-human NF-κB p50 (1:200; cat. no. sc-8414) and mouse
anti-human wild-type p53 (1:50; cat. no. sc-126) for 2 h at 37°C
(all purchased from Santa Cruz Biotechnology, Inc.). The slides
were washed three times with phosphate buffered saline (PBS,
Beyotime Institute of Biotechnology) and incubated with horseradish
peroxidase-linked anti-mouse immunoglobulin G secondary antibody
(1:2,000; cat. no. sc-2005; Santa Cruz Biotechnology, Inc.) for 1 h
at room temperature. The slides were subsequently stained using the
diaminobenzidine substrate kit (cat. no. SK-4100; Vector
Laboratories, Inc.). The nuclei were stained with hematoxylin for 4
min at room temperature, and the slides were observed under an
inverted light microscope (Olympus Corporation; magnifications ×200
and ×400). Tissue sections were blindly assessed by two
patholo-gists from Shengjing Hospital of China Medical University
(Shenyang, China). IHC results were interpreted, as previously
described (22). Briefly, 0=no
staining, 1=light yellow, 2=brownish yellow and 3=dark brown;
positive cells were also counted under ×400 magnification and
scored (0=positive rate <5%; 1=5-25%; 2=26-50%; 3=51-75% and
4=>75%). The final staining score was determined by multiplying
the two values: 0-2 was considered negative (-), 3-4 was (+), 5-8
was (++), and 9-12 was (+++). The symbols '-' and '+' represent low
expression, and '++' and '+++' represent high expression.
Cell culture and transfection
Human OCCC derived RMG-1 [donated by Professor
Iwamori Masao of Tokyo University (Tokyo, Japan)] (23) and ES-2 (purchased from American
Type Culture Collection) cell lines were cultured in RPMI-1640
medium or Macoy 5A supplemented with 10% fetal bovine serum (FBS),
respectively, at 37°C in 5% CO2 (all purchased from
HyClone; GE Healthcare Life Sciences).
The human OCCC cells (ES-2 cells and RMG-1 cells)
were plated into six-well plates at a density of 1.5×105
cells/well. After incubation at constant temperature (37°C, 5%
CO2), cells were transfected once they adhered to the
plates. Cells were starved of serum for 1 h prior to transfection.
The transfection mixture consisted of serum-free medium, 2.5
µg/ml ANXA4 overexpression vector (pc-ANXA4-GFP) or short
hairpin (sh)-ANXA4-GFP or corresponding empty control vectors (all
purchased from Shanghai GenePharma Co., Ltd.) and Lipofectamine
2000 (cat. no. L3000150; Invitrogen, Thermo Fisher Scientific,
Inc.). The transfection procedure was performed base on the
manufacturer's protocol of Lipofectamine 2000 transfection reagent.
The following stable cell lines were generated: ES-2-A4-O, ES-2
cells transfected with pc-ANXA4-GFP plasmids and RMG-1-A4-I, RMG-1
cells transfected with Sh-ANXA4-GFP plasmids. Transfection
efficiency was measured using western blot analysis. Subsequent
experiments with transfected cells were performed following
transfection for 24 h.
Co-immunoprecipitation
ES-2 cells and RMG-1 cells were lysed using RIPA
buffer (cat. no. sc-24948, Santa Cruz Biotechnology, Inc.) (50 Mm
Tris–HCl (pH 7.2), 150 mM NaCl, 1% (v/v) Triton X-100, 1% (w/v)
sodium deoxycholate, and 0.1% (w/v) SDS and protease inhibitors).
Cell lysates were incubated with the following primary antibodies
at a concentration of 5 µg/ml: Rabbit anti-human ANXA4
antibody (cat. no. ab153883) and rabbit anti-human NF-κB p50
antibody (cat. no. ab264235; both purchased from Abcam), and
Protein A/G plus-agarose (cat. no. sc-2003; Santa Cruz
Biotechnology, Inc.) overnight at 4°C. Normal rabbit IgG (5
µg/ml; cat. no. ab172730; Abcam) was used as the negative
control. The immunoprecipitated proteins were separated via 10%
SDS-PAGE gels and detected using rabbit anti-human ANXA4 antibody
(1:1,000; cat. no. ab153883; Abcam) and rabbit anti-human NF-κB p50
antibody (1:1,000; cat. no. ab264235; Abcam) overnight at 4°C.
Protein bands were visualized using enhanced chemiluminescence
(ECL) reagent (Amersham; Cytiva).
Confocal laser scanning microscopy
Monolayered cell slides were prepared using ES-2
cells. Cells were fixed with 100% methanol for 45 min at room
temperature and stained with the following primary antibodies:
Mouse anti-human ANXA4 antibody (1:100; cat. no. sc-46693; Santa
Cruz Biotechnology, Inc.) and rabbit anti-human NF-κB p50 antibody
(1:100; cat. no. ab264235; Abcam) overnight at 4°C. Following the
primary incubation, cells were incubated with secondary antibodies
FITC green fluorescence-labeled mouse IgG (1:50; cat. no. sc-2010;
Santa Cruz Biotechnology, Inc.) and TRITC red fluorescence-labeled
rabbit IgG (1:50; cat. no. sc-2492; Santa Cruz Biotechnology, Inc.)
at room temperature for 1 h. Cells were washed three times with PBS
and cell nuclei were stained using DAPI (cat. no. sc-3598, Santa
Cruz Biotechnology, Inc.) at a final concentration of 5
µg/ml for 5 min at 37°C. Fluorescent images were visualized
under by a laser confocal microscope (C1-SI; Nikon, Corporation,
magnifications ×200 and ×400). PBS was used as the negative control
instead of the primary antibodies.
Western blotting
Briefly, the cells (ES-2-A4-O, ES-2, RMG-1-A4-I, and
RMG-1 cells) were washed twice with ice-cold PBS, and total protein
was extracted on ice using radioimmunoprecipitation assay lysis
buffer (cat. no. P0013B; Beyotime Institute of Biotechnology).
Lysates were cleared by centrifugation (15 min at 13,000 × g, 4°C).
The protein content was measured using the protein assay BCA kit
(Beyotime Institute of Biotechnology). Following quantitative
analysis, equal amounts of protein (50 µg) were resolved
with 10% SDS-PAGE and electrotransferred onto polyvinylidene
difluoride membranes. The membranes were blocked with Tris-buffered
saline Tween [25 mM Tris-HCl, 150 mM NaCl (pH 7.5), and 0.1%
Tween-20] containing 5% non-fat milk at room temperature for 2 h
and incubated overnight at 4°C with the following primary
antibodies: Mouse anti-human ANXA4 (1:1,000; cat. no. sc-46693;
Santa Cruz Biotechnology, Inc.), rabbit anti-human NF-κB p50
(1:200; cat. no. ab264235; Abcam), mouse anti-human Bcl-2 (1:1,000;
cat. no. 15071; Cell Signaling Technology, CST, Co., USA) and
rabbit anti-human Cyclin D1 (1:1,000; cat. no. 55506; Cell
signaling technology) in TTBS/1% non-fat milk. Following three
washes with TTBS, the membranes were incubated with the appropriate
horse-radish peroxidase-linked IgG (goat anti-mouse IgG-HRP, cat.
no. sc-2005, 1:2,000 and goat anti-rabbit IgG-HRP, cat. no.
sc-2004, 1:2,000; Santa Cruz Biotechnology, Inc.) at room
temperature for 1 h, and visualization using an ECL (Pierce; Thermo
Fisher Scientific, Inc.) detection system. The western blots shown
are representative of at least 3 independent experiments. The
protein bands were visualized using the Molecular Imager system
GDS8000b (Analytik Jena AG). Total protein levels were normalized
to GAPDH (1:2,000; cat. no. sc-47724; Santa Cruz Biotechnology,
Inc.) expression on the same membrane, and the bands were
quantified using ImageJ software v1.8.0 (National Institutes of
Health).
Immunocytochemistry
Monolayered cell slides were prepared using ES-2,
ES-2-A4-O, RMG-1 and RMG-1-A4-I cells, while they were in the
exponential growth phase. The expression of ANXA4 and NF-κB p50 in
the cells was detected according to the instructions provided with
the SABC kit (cat. no. SA1054; Boster Biological Technology Co.,
Ltd.). Briefly, the cells were fixed with 4% paraformaldehyde for
30 min at room tempera-ture, and the nonspecific antigens were
blocked with 10% normal goat serum (cat. no. AR0009; Boster
Biological Technology Co., Ltd.) for 30 min at room temperature.
The cells were incubated with primary antibodies overnight at 4°C.
Following rinsing with PBS three times, the cells were incubated
with secondary antibody (horseradish peroxidase-conjugated goat
anti-rabbit immunoglobulin G; 1:500; cat. no. sc-2004; Santa Cruz
Biotechnology, Inc.) for 30 min at room temperature. The following
primary antibodies were used: Rabbit anti-human ANXA4 (1:100; cat.
no. ab153883; Abcam) and rabbit anti-human NF-κB p50 (1:300; cat.
no. ab264235; Abcam). The primary antibody was replaced by PBS for
the negative control.
Treatment with NF-κB pathway
inhibitor
The NF-κB pathway inhibitor, BAY 11-7082
(Sigma-Aldrich; Merck KGaA) was added to cells at a concentration
of 5 µmol/ml, and proteins were collected after 24 h.
Growth inhibition assay
Cell viability was assessed via the MTT assay.
Briefly, 6×103 cells were seeded into 96-well plates and
cultured in RPMI-1640 medium or Macoy 5A supplemented with 10% FBS,
respectively, at 37°C in 5% CO2 overnight. After 24 h,
the cells were treated with serum-free media supplemented with BAY
11-7082 (Sigma-Aldrich; Merck KGaA). A total of four cell lines
were assessed: ES-2, ES-2-A4-O, ES-2-A4-O+BAY 11-2078 and ES-2+BAY
11-2078. The 0 point was achieved after 6 h of cell culture, and
optical density (OD) was measured at the 0 point after 24, 48 and
72 h. Following the different durations of treatment, the cells
were incubated with 20 µl MTT (5 mg/ml; Sigma-Aldrich; Merck
KGaA) for 4 h at 37°C. The supernatant was removed, the purple
formazan crystals were dissolved using 150 µl dimethyl
sulfoxide (DMSO) and viability was subsequently analyzed at a
wavelength of 570 nm using a 96-well multiscanner autoreader
(Bio-Rad Laboratories, Inc.).
Flow cytometric analysis of
apoptosis
Cell apoptosis was assessed via flow cytometric
analysis of the ES-2, ES-2-A4-O and ES-2-A4-O+BAY 11-2078 cell
lines. Cells were double stained with allophycocyanin (APC) annexin
V conjugate (Annexin V-APC) and propidium iodide (PI), using the
Annexin V-APC/PI kit (Nanjing KeyGen Biotech Co., Ltd.) for 10 min
at room temperature away from light, according to the
manufacturer's protocol. Blanks were run as controls for both PI
and Annexin V-APC staining, for each group. Apoptotic cells were
subsequently analyzed using a flow cytometer (BD FACSCanto II; BD
Biosciences) and Cell Quest program (version 6.0; FACScan; BD
Biosciences). All experiments were performed in triplicate.
PPI network of ANXA4, NF-κB p50 and
p53
The PPI network was constructed using the cBioPortal
database (http://www.cbioportal.org) and
visualized using Cytoscape software version 3.7.2 (24). And confidence score ≥0.4 was set
as the cut-off criterion.
Functional and signaling pathway
enrichment analysis
Gene Ontology (GO) and Kyoto Encyclopedia of Genes
and Genomes (KEGG; http://www.kegg.jp) analyses were
performed for functional annotation and pathway assessment, using
the Database for Annotation Visualization and Integrated Discovery
(DAVID) (http://david.abcc.ncifcrf.gov). The human genome was
selected as the background parameter. P<0.05 and a count ≥2 were
set as the thresholds to indicate a statistically significant
difference.
Statistical analysis
Statistical analysis was performed using SPSS 17.0
software (SPSS, Inc.). Data are presented as the mean ± standard
deviation. The χ2 test was used to assess the positive
ratio rates. Pearson's χ2 test was used to determine the
association between ANXA4 and NF-κB p50 (or mutant p53). Survival
analysis was performed using Kaplan-Meier curves, and significant
differences among different clinico-pathological variants and
immunomarkers were determined using the log-rank test. Cox
proportional hazards regression model was used to control for
confounding variables. Unpaired Student's t-test was performed to
compare differences between two groups, while one-way analysis of
variance and Least Significant Difference or Bonferroni post hoc
tests were used to compare differences between multiple groups. All
the experiments were performed in triplicate. P<0.05 was
considered to indicate a statistically significant difference.
Results
Nuclear expression of ANXA4 and NF-κB p50
in different ovarian tissues
The positive expression of ANXA4 was observed
throughout the cell membrane, cytoplasm and nucleus in different
ovarian tissues. The positive nuclear expression of ANXA4 in OCCC
was 77.9%, which was significantly higher compared with the three
subtypes (serous, mucinous and endometrioid) of OC (27.3%) and
normal ovarian tissues (0%). The strong positive ('++' and '+++'
represent strong positive expression) nuclear expression of ANXA4
in OCCC was 38.3% (33/86), which was markedly greater compared with
the three subtypes of OC (9.1%, 5/55) and normal ovarian tissues
(0%) (Fig. 1 and Table I).
 | Table INuclear expression of ANXA4 and NF-κB
p50 in different ovarian tissues. |
Table I
Nuclear expression of ANXA4 and NF-κB
p50 in different ovarian tissues.
| Characteristic | Number of cases,
n | ANXA4
| P-value | NF-κB p50
| P-value |
|---|
| − | + | ++ | +++ | Positive n (%) | Strong positive n
(%) | − | + | ++ | +++ | Positive n (%) | Strong positive n
(%) |
|---|
| Normal | 10 | 10 | 0 | 0 | 0 | 0 (0b) | 0 (0b) | <0.001 | 10 | 0 | 0 | 0 | 0 (0d) | 0 (0d) | <0.001 |
| OCCC | 86 | 19 | 34 | 15 | 18 | 67 (77.9a) | 33 (38.3a) | <0.001 | 2 | 34 | 32 | 18 | 84 (97.7c) | 50 (58.1c) | <0.001 |
| Other subtypes | 55 | 40 | 10 | 2 | 3 | 15 (27.3) | 5 (9.1) | | 19 | 23 | 8 | 5 | 36 (65.5) | 13 (23.6) | |
| Serous | 30 | 25 | 4 | 0 | 1 | 5 (8.3) | 1 (3.3) | | 10 | 14 | 4 | 2 | 20 (66.7) | 6 (20.0) | |
| Mucinous | 17 | 9 | 5 | 1 | 2 | 8 (47.1) | 3 (17.6) | | 8 | 6 | 2 | 1 | 9 (52.9) | 3 (17.6) | |
| Endometrioid | 8 | 6 | 1 | 1 | 0 | 2 (25.0) | 1 (12.5) | | 1 | 3 | 2 | 2 | 7 (87.5) | 4 (50.0) | |
NF-κB p50 was predominantly expressed in the nucleus
and the cytoplasm. The positive nuclear expression of NF-κB p50 in
OCCC was 97.7%, which was significantly higher compared with the
three subtypes of OC (65.5%) and normal ovarian tissues (0%). The
strong positive nuclear expression of NF-κB p50 in OCCC was 58.1%
(50/86), which was markedly greater compared with the three
subtypes of OC (23.6%, 13/55) and normal ovarian tissues (0%)
(Fig. 1 and Table I).
Association between the nuclear
expression of ANXA4 and NF-κB p50, and the clinicopathological
characteristics of OCCC
As presented in Table
II, the strong positive nuclear expression of ANXA4 in FIGO
advanced stage OCCC (III-IV stage) was 55.6%, which was
significantly higher compared with FIGO early stage OCCC (I-II
stage; 30.5%; P=0.0266). The strong positive nuclear expression of
ANXA4 in the chemotherapeutics-resistant group was 68.4%, which was
significantly higher compared with the chemotherapeutics-sensitive
group (30.8%; P=0.0043). No significant difference in the positive
nuclear expression of ANXA4 was observed between the lymph node
metastasis groups (60.0% vs. 33.8%).
 | Table IIAssociation between the nuclear
expression of ANXA4 and NF-κB p50 and the clinicopathological
characteristics of ovarian clear cell carcinoma. |
Table II
Association between the nuclear
expression of ANXA4 and NF-κB p50 and the clinicopathological
characteristics of ovarian clear cell carcinoma.
| Characteristic | Number of cases,
n | ANXA4 expression
| P-value | NF-κB p50
expression
| P-value |
|---|
| Low, n | High, n | Low, n | High, n |
|---|
| Surgical stage | | | | | | | |
| I-II | 59 | 41 | 18 | 0.0266 | 29 | 30 | 0.0427 |
| III-IV | 27 | 12 | 15 | | 7 | 20 | |
| Lymph node
metastasis | | | | | | | |
| Yes | 10 | 4 | 6 | 0.2064 | 2 | 8 | 0.2241 |
| No | 74 | 49 | 25 | | 34 | 40 | |
|
Chemosensitivity | | | | | | | |
| Sensitive | 52 | 36 | 16 | 0.0043 | 27 | 25 | 0.0064 |
| Resistant | 19 | 6 | 13 | | 3 | 16 | |
Similarly, the strong positive nuclear expression of
NF-κB p50 in FIGO advanced stage OCCC was 74.1%, which was
significantly higher compared with FIGO early stage OCCC (50.8%;
P=0.0427). In addition, the strong positive nuclear expression of
NF-κB p50 in the chemotherapeutics-resistant group was 84.2%, which
was significantly higher compared with the
chemotherapeutics-sensitive group (48.1%; P=0.0064). No significant
difference in the strong positive nuclear expression of NF-κB p50
was observed between the lymph node metastasis groups (80.0 vs.
54.1%) (Table II).
High nuclear expression of ANXA4 and
NF-κB p50 in OCCC predicts a poor prognosis
A total of 86 patients with OCCC were followed up
(36-108 months). By December 2015, 75 cases completed follow-up,
while 11 cases were lost to follow-up (because the patients and
their families refused contact during the follow-up.). As presented
in Fig. 2, in the high ANXA4
nuclear expression group (n=29), 14 cases died and the mean
survival time was 39.1 months, while in the low ANXA4 nuclear
expression group (n=46), 10 cases died and the mean survival time
was 48.1 months. In the high NF-κB p50 nuclear expression group
(n=43), 20 cases died and the mean survival time was 40 months,
while in the low NF-κB p50 nuclear expression group (n=32), four
cases died and the mean survival time was 50.8 months.
Survival rates of patients with OCCC were evaluated
via Kaplan-Meier analysis, which indicated that the prognosis was
significantly unfavorable in the high ANXA4 and NF-κB p50 nuclear
expression groups compared with the respectively low ANXA4 and
NF-κB p50 nuclear expression groups (P=0.011 and P=0.003; Fig. 2).
Multivariate analysis of OCCC prognosis was
performed using the Cox proportional hazards regression model. The
FIGO stage, residual tumor size, lymph node metastasis status, age
and expression levels of ANXA4 and NF-κB p50 were included in the
dependent variable analysis. The results demonstrated that the
advanced FIGO stage, and high expression levels of ANXA4 and NF-κB
p50 were independent risk factors for the prognosis of patients
with OCCC (P=0.007, P=0.042 and P=0.015; Table III).
 | Table IIICox regression analysis of overall
survival of ovarian clear cell carcinoma. |
Table III
Cox regression analysis of overall
survival of ovarian clear cell carcinoma.
| Variable | P-value | Hazard ratio (95%
CI) |
|---|
| ANXA4 (high vs.
low) | 0.042 | 2.367
(1.030-5.438) |
| NF-κB p50 (high vs.
low) | 0.015 | 3.894
(1.302-11.644) |
| Surgical stage
(III-IV vs. I-II) | 0.007 | 3.783
(1.432-9.991) |
Nuclear expression of ANXA4, NF-κB p50
and mutant p53 in OCCC
A previous study reported that ANXA4 expression in
OCCC is closely associated with p53 (18). Thus, the present study assessed
mutant p53 expression in OCCC. The results demonstrated that mutant
p53 was primarily localized in the cell nucleus. However, as
presented in Fig. 3, mutant p53
expression was rarely seen in OCCC tissues, and the expression
levels were very low. In contrast to mutant p53, the expression
levels of ANXA4 and NF-κB p50 were generally enhanced in OCCC
tissues.
Analysis of the correlation between ANXA4
and NF-κB p50 nuclear expression in OCCC
Of the 86 OCCC tissue samples, high ANXA4 and NF-κB
p50 nuclear co-expression was observed in 24 cases, while low ANXA4
and NF-κB p50 nuclear co-expression was observed in 28 cases. ANXA4
and NF-κB p50 expression levels exhibited a linear correlation
(χ2=5.95, P=0.015; Table
IV).
 | Table IVCorrelation between ANXA4 and NF-κB
p50 expression in ovarian clear cell carcinoma. |
Table IV
Correlation between ANXA4 and NF-κB
p50 expression in ovarian clear cell carcinoma.
| NF-κB p50
expression | ANXA4 expression
| Total, n | P-value | χ2 |
|---|
| High | Low |
|---|
| High | 24 | 26 | 50 | 0.015 | 5.95 |
| Low | 8 | 28 | 36 | | |
| Total | 32 | 54 | 86 | | |
Analysis of the association between
mutant p53 and ANXA4 expression in OCCC
Of the 86 OCCC tissue samples, 16 cases had positive
p53 expression and 70 cases had negative p53 expression. Mutant p53
expression was observed in two cases with high ANXA4 expression and
14 cases with low ANXA4 expression. No mutant p53 expression was
exhibited in 31 cases with high ANXA4 expression and 39 cases with
low ANXA4 expression. A linear association was observed between p53
and ANXA4 expression levels (χ2=5.56; P=0.018; Table V).
 | Table VAssociation between ANXA4 and mutant
p53 expression in ovarian clear cell carcinoma. |
Table V
Association between ANXA4 and mutant
p53 expression in ovarian clear cell carcinoma.
| Mutant p53
expression | ANXA4 expression
| Total, n | P-value | χ2 |
|---|
| Positive | Negative |
|---|
| Negative | 31 | 39 | 70 | 0.018 | 5.56 |
| Positive | 2 | 14 | 16 | | |
| Total | 33 | 53 | 86 | | |
ANXA4 and NF-κB p50 are co-expressed in
OCCC
Co-immunoprecipitation analysis demonstrated that
there was an interaction between ANXA4 and NF-κB p50 expression in
human OCCC derived RMG-1 and ES-2 cell lines (Fig. 4A). Laser scanning confocal
microscopy confirmed the co-localization of ANXA4 and NF-κB p50 in
OCCC cells (Fig. 4B). Western
blot and immunocytochemical analyses demonstrated that both ANXA4
and NF-κB p50 expression in established ES-2-A4-O cells increased
compared with ES-2 cells following transfection of ANXA4 gene in
ES-2 cells. Furthermore, NF-κB p50 expression (when the ANXA4 gene
was silenced in RMG-1 cells), and the expression levels of ANXA4
and NF-κB p50 significantly decreased in RMG-1-A4-I cells compared
with RMG-1 cells, following transfection (Fig. 4C and D). Taken together, these
results suggest that ANXA4 and NF-κB p50 are interacting proteins,
and their interaction may be enhanced by upregulating ANXA4
expression.
PPI network construction and module
selection
As presented in Fig.
5, the PPI network of the differentially expressed genes
consisted of 50 nodes and 219 edges. ANXA4, NFκB1 (the precursor of
NF-κB p50), and TP53 were demonstrated to regulate each other,
whilst forming complex regulatory associations with other genes,
such as MYC and BCL2L1.
Function and signaling pathway enrichment
analysis
Function and pathway enrichment analysis of related
genes in the PPI network was performed using DAVID database. The
results demonstrated that pathways with related genes were enriched
in 'NF-κB signaling pathway', 'apoptosis', 'cell cycle', 'PI3K/Akt
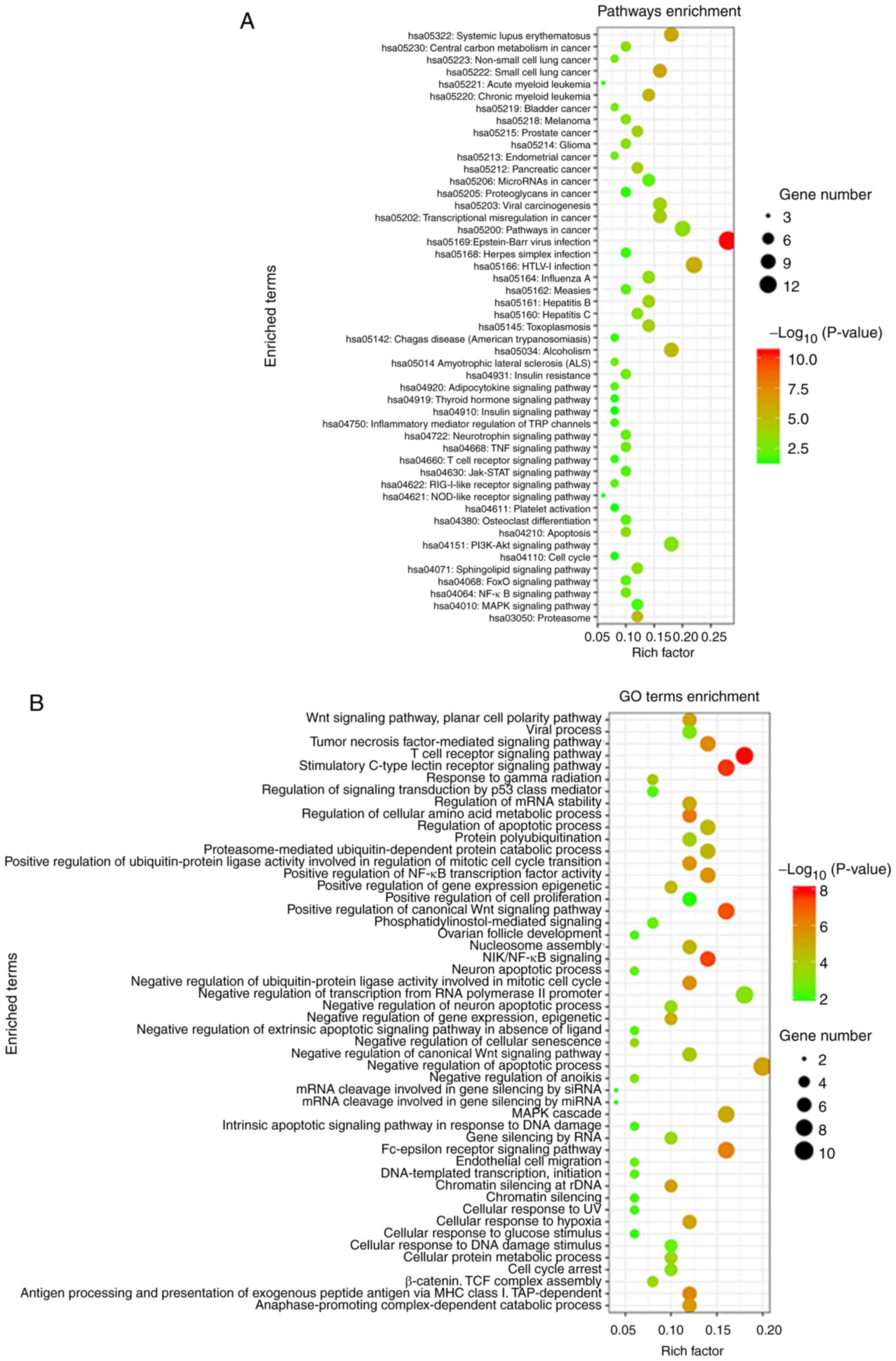
signaling pathway' and 'MAPK signaling pathway' (Fig. 6A). These genes are predominantly
involved in 'biological processes', including 'negative regulation
of apoptotic process', 'regulation of cell proliferation', 'cell
cycle arrest' and 'NIK/NF-κB signaling' (Fig. 6B).
Interaction between ANXA4 and NF-κB p50
promotes cell proliferation and inhibits cell apoptosis
The expression of Cyclin D1 and Bcl-2 (downstream
molecules of the NF-κB signaling pathway) in ES-2 cells was
detected via western blotting after the ES-2 cells were transfected
with the ANXA4 gene and added to the NF-κB inhibitor, respectively.
The results demonstrated that the expression levels of both Cyclin
D1 and Bcl-2 increased following transfection of the ANXA4 gene;
however, these levels decreased after 24 h, following addition of
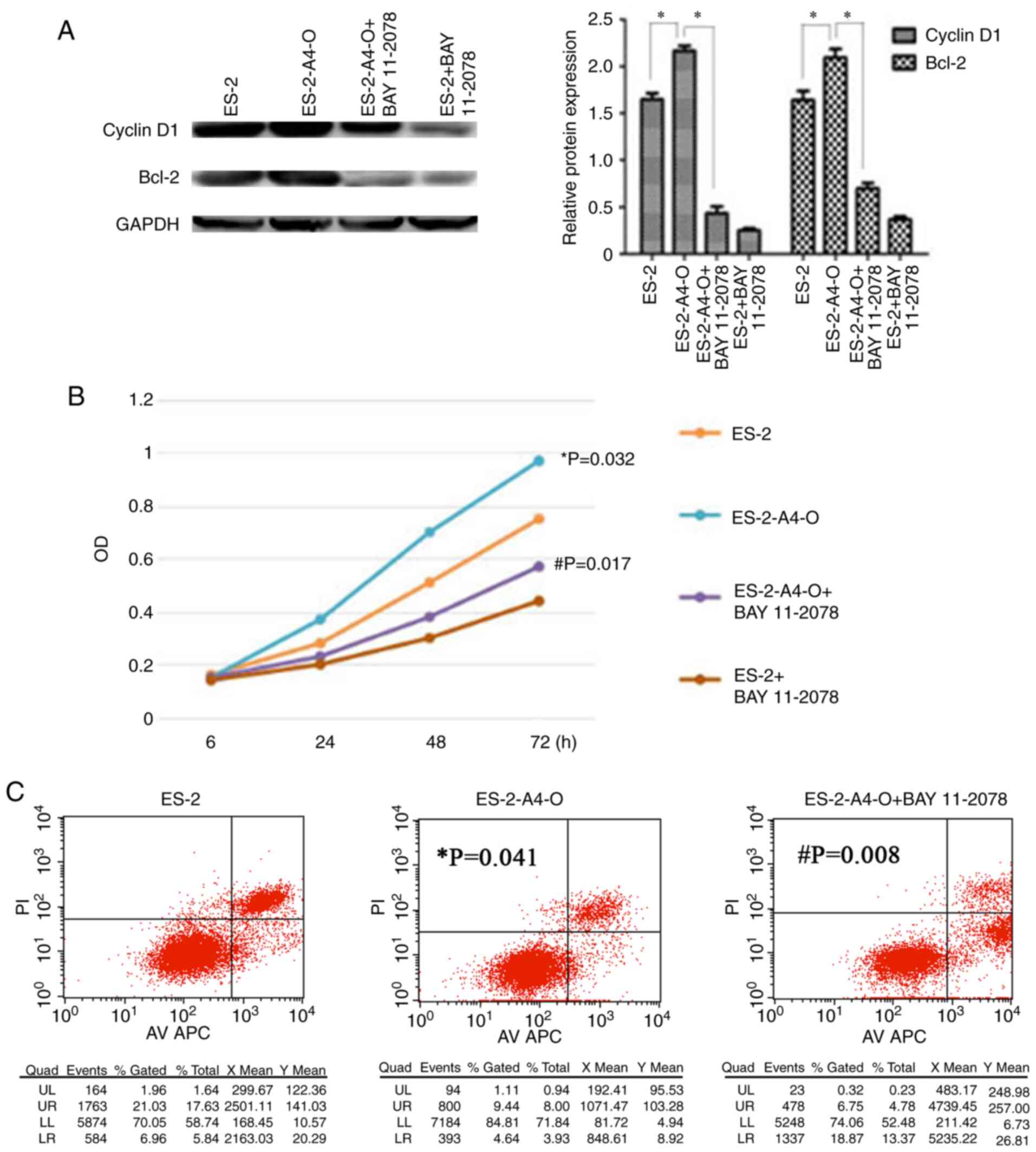
the NF-κB inhibitor (Fig. 7A).
Collectively, these results indicate that the interaction between
ANXA4 and NF-κB p50 may activate the NF-κB signaling pathway, and
promote Cyclin D1 and Bcl-2 expression.
The changes in the capacity of cell proliferation
and apoptosis before and after transfection of the ANXA4 gene, and
addition of the NF-κB inhibitor were further assessed via the MTT
assay and flow cytometric analysis. The results demonstrated that
the proliferation capacity increased (ES-2 compared with ES-2-A4-O,
P=0.032) and cell apoptosis decreased (ES-2 compared with
ES-2-A4-O, P=0.041) following transfection of the ANXA4 gene;
however, this effect was significantly reversed after 24 h,
following addition of the NF-κB inhibitor, BAY 11-2078 (5
µmol/ml; Fig. 7B and C;
ES-2-A4-O compared with ES-2-A4-O + BAY 11-2078, P=0.017 and
P=0.008, respectively). Taken together, these results suggest that
the interaction between ANXA4 and NF-κB p50 may activate the NF-κB
signaling pathway, whilst promoting the proliferation and
inhibiting the apoptosis of OCCC cells.
Discussion
OCCC is a type of ovarian epithelial carcinoma that
currently lacks effective therapies due to lack of knowledge
regarding the occurrence and developmental mechanisms. Its
prognosis is unfavorable compared with other subtypes, such as
serous and mucinous, and the 5-year survival rate is only 27%
(25,26). A previous study has reported that
ANXA4 is highly expressed in OCCC, while weakly expressed or even
absent in other subtypes of ovarian epithelial carcinoma, thus,
ANXA4 is considered a specific marker of OCCC (11). Subsequent studies have revealed
that in the cervical cancer Hela cell line, ANXA4 can enter into
the nucleus and influence the transcription of target genes and
those associated with NF-κB p50 (12). In order to determine the
interaction between ANXA4 and NF-κB p50 in different pathological
types of OC, the nuclear expression of ANXA4 and NF-κB p50 was
assessed in different subtypes of ovarian epithelial carcinoma and
normal ovarian tissues via IHC analysis. The results demonstrated
that the positive nuclear expression (77.9, 97.7%) and strong
positive expression (38.3, 58.1%) of ANXA4 and NF-κB p50 were both
significantly higher in OCCC tissues compared with ANXA4 expression
in other subtypes of ovarian epithelial carcinoma (27.3, 65.5%;
9.1, 23.6%) and normal ovarian tissues (0, 0%; 0, 0%). The nuclear
expression of ANXA4 and NF-κB p50 demonstrated a linear correlation
in OCCC tissues, and high ANXA4 and NF-κB p50 expression levels
were associated with an advanced clinical stage and
chemotherapeutic resistance of OCCC. Cox proportional hazards
regression analysis demonstrated that high expression levels of
both ANXA4 and NF-κB p50 were independent risk factors for the poor
prognosis of OCCC. Collectively, these results suggest that ANXA4
and NF-κB p50 may promote malignant progression, thus resulting in
poor prognosis of patients with OCCC.
The structural association between ANXA4 and NF-κB
p50 in OCCC cell lines was assessed via co-immunoprecipitation, and
the results suggested that ANXA4 and NF-κB p50 were structurally
associated and their nuclear expression was co-localized. This
confirms the interaction between ANXA4 and NF-κB p50 in OCCC cell
nuclei.
In mammals, five members of the NF-κB family have
been identified, namely NF-κB1 (NF-κB p50/p105), NF-κB2 (p52), RelA
(p65), RelB and C-Rel. In humans, NF-κB is predominantly expressed
in the cytoplasm as a p65/p50 heterodimer and forms a
NF-κB/IκB-inactive trimer with IκB to hinder nuclear entry, thus
preventing DNA-binding of NF-κB (27,28). However, IκB is phosphorylated by
IκB kinase (IKK), under the stimulation of some factors, such as
IL-1β and LPS, and subsequently degraded to activate NF-κB and its
activation product, NF-κB p50 translocates to the nucleus from the
cytoplasm, and binds with a DNA-specific κB sequence to promote
transcription and expression of a series of genes, including
cytokines and growth factors, in different phases of several types
of disease (29-31). Previous studies have suggested
that overexpression of NF-κB as a nuclear transcription factor in
tumor cells affects the progression of tumors mainly by the
following mechanisms, upregulating cyclins or activating the
expression of target genes and promoting cell proliferation, and
inhibiting transduction of cell death signals and protecting cells
from the induction of stimulating factors and the resultant
apoptosis mechanisms (32,33).
Thus, this raises the question, does the interaction between ANXA4
and NF-κB p50 in OCCC cells influence the proliferation and
apoptosis of tumor cells?
First, the present study established the ANXA4
gene-expressing and ANXA4 gene-silencing cell lines; secondly,
NF-κB p50 expression was detected both before and after
transfection, followed by western blotting and immunocytochemistry
analyses. The results demonstrated that NF-κB p50 expression
changed over ANXA4, and suggested that upregulating ANXA4 may
enhance its binding with NF-κB p50. The ANXA4 gene-transfected OCCC
cell lines were additionally treated with the NF-κB inhibitor, and
the changes in the capacity of cell proliferation and apoptosis
were subsequently assessed. The results demonstrated that the
proliferation capacity of ANXA4 gene-transfected cells increased
and cell apoptosis decreased, while the proliferation capacity of
ANXA4 gene-transfected cells treated with the NF-κB inhibitor
decreased and cell apoptosis increased. Collectively, these results
suggest that ANXA4 bound to NF-κB p50 may activate the NF-κB
signaling pathway, promote proliferation and inhibit apoptosis of
OCCC cells. The expression levels of the target molecules
associated with cell proliferation and apoptosis, Cyclin D1 and
Bcl-2, downstream of the NF-κB signaling pathway (34) were detected both before and after
transfection of the ANXA4 gene and addition of the NF-κB inhibitor,
respectively. The results demonstrated that the expression levels
of Cyclin D1 and Bcl-2 increased following transfection of the
ANXA4 gene, and significantly decreased following addition of the
NF-κB inhibitor. This confirmed that ANXA4 bound to NF-κB p50
promoted the expression of NF-κB downstream target molecules,
Cyclin D1 and Bcl-2, and thus facilitated cell proliferation and
inhibited cell apoptosis.
Previous studies have reported that ANXA4 is
particularly expressed in pathological tumor subtypes, for example,
its expression is significantly higher in thyroid follicular
carcinoma and medullary carcinoma compared with other subtypes of
thyroid cancer, and also higher in salivary pleomorphic tumor and
Warthin tumor compared with other subtypes of salivary tumor
(35,36). Thus, ANXA4 is considered a marker
to distinguish different subtypes of thyroid cancer and salivary
tumor. Similarly, in OC, ANXA4 is highly expressed in the
pathological subtypes compared with ovarian serous carcinoma, and
is significantly upregulated in OCCC (37). The results of the present study
demonstrated that ANXA4 can bind to NF-κB p50 in OCCC cells, thus
promoting cell proliferation and inhibiting cell apoptosis.
A previous study has confirmed that the particularly
high ANXA4 expression in OCCC is regulated by wild-type p53
(38). p53 is classified into
wild-type and mutant type. Reports have suggested that p53 is
mostly wild-type rather than mutant type in OCCC, which was
confirmed by the results of the present study, demonstrating that
mutant p53 was expressed at lows levels in OCCC. Wild-type p53 can
bind with the upstream transcription initiation site of the ANXA4
gene and positively regulate ANXA4 expression in cells. Following
transfection of wild-type p53 siRNA, both wild-type p53 and ANXA4
expression simultaneously decrease; however, this effect is absent
in mutant type p53 (39,40). The results of the present study
also confirmed that ANXA4 is weakly expressed in OCCC tissues that
express mutant p53, while highly expressed in OCCC tissues without
mutant p53 expression. The expression of ANXA4 and mutant p53
exhibited a negatively linear correlation. It is well-known that
wild-type p53, a key factor in regulating cell-cycle arrest, cell
aging and apoptosis, is the most effective tumor-inhibiting gene.
Exogenously acquired wild-type p53 can inhibit autophagy, reverse
drug resistance and promote apoptosis of tumor cells (41,42). However, the molecular mechanism
underlying the role of ANXA4 on tumor progression remains unclear.
A previous report has demonstrated that NF-κB inhibits the
transcription of p53-dependent genes, while the NF-κB inhibitor
suppresses the cytoplasmic-to-nuclear shift of NF-κB, thus
inhibiting the transcriptional activity of NF-κB and increasing the
expression of p53 downstream target molecules, including p21, in
cells (43). The results of the
present study demonstrated that ANXA4 and NF-κB p50 are
overexpressed and interact with one another in OCCC tissues and
cells. Bioinformatics analysis indicated that ANXA4, NFκB1 (a
precursor of NF-κB p50) and TP53 regulate each other and form
complex regulatory associations with other genes, further
activating the NF-κB signaling pathway, apoptosis, cell cycle, and
the PI3K/Akt and MAPK signaling pathways, thus participating in
cell proliferation, apoptosis and other biological processes such
as cell adhesion; for all that, the mechanism of ANXA4 complex
action is still warranted more in-depth and concentrated
investigations.
In conclusion, the results of the present study
demonstrated that wild -type p53 activates ANXA4 transcription and
promotes its expression, which subsequently binds to NF-κB p50 and
enters the nuclei of cells, activating the NF-κB signaling pathway.
The complex biological regulation among wild-type p53, ANXA4 and
NF-κB p50 increases the expression of Cyclin D1 and Bcl-2, thus
facilitating cell proliferation and inhibiting cell apoptosis,
while promoting the malignant progression of tumors. Taken
together, the results of the present study suggest that ANXA4/NF-κB
p50 complexes play a critical role in tumorigenesis of OCCC, and
thus may provide novel ideas for early detection and surveillance
for OCCC treatment.
Acknowledgments
Not applicable.
Funding
The present study was funded by The National Natural
Science Foundation of China (grant nos. 81172491, 81101527, 8147243
and 81672590) and the Outstanding Scientific Fund of Shengjing
Hospital (grant no. 201804).
Availability of data and materials
All data generated or analyzed during this study are
included in this published article.
Authors' contributions
BL designed the present study. JL and HW performed
most of the experiments, and MZ and LD participated in the
experiments. MZ and XZ performed the statistical analyses. All
authors read and approved the final manuscript.
Ethics approval and consent to
participate
Samples were fully encoded to protect patient
confidentially. All procedures performed involving human
participants were in accordance with the ethical standards of the
Ethical Committee of Shengjing Hospital of China Medical University
(Shenyang, China, approval no. 2013PS66K). The ethics committee
waived the need for patient consent, as the patient information was
withheld in this retrospective study.
Patient consent for publication
Not applicable.
Competing interests
The authors declare that they have no competing
interests.
References
|
1
|
Siegel RL, Miller KD and Jemal A: Cancer
statistics, 2018. CA Cancer J Clin. 68:7–30. 2018. View Article : Google Scholar : PubMed/NCBI
|
|
2
|
Bamias A, Psaltopoulou T, Sotiropoulou M,
Haidopoulos D, Lianos E, Bournakis E, Papadimitriou C, Rodolakis A,
Vlahos G and Dimopoulos MA: Mucinous but not clear cell histology
is associated with inferior survival in patients with advanced
stage ovarian carcinoma treated with platinum-paclitaxel
chemotherapy. Cancer. 116:1462–1468. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
3
|
Matsuzaki S, Yoshino K, Ueda Y, Matsuzaki
S, Kakuda M, Okazawa A, Egawa-Takata T, Kobayashi E and Kimura T:
Potential targets for ovarian clear cell carcinoma: A review of
updates and future perspectives. Cancer Cell Int. 15:1172015.
View Article : Google Scholar : PubMed/NCBI
|
|
4
|
Crotzer DR, Sun CC, Coleman RL, Wolf JK,
Levenback CF and Gershenson DM: Lack of effective systemic therapy
for recurrent clear cell carcinoma of the ovary. Gynecol Oncol.
105:404–408. 2007. View Article : Google Scholar : PubMed/NCBI
|
|
5
|
Sugiyama T, Kamura T, Kigawa J, Terakawa
N, Kikuchi Y, Kita T, Suzuki M, Sato I and Taguchi K: Clinical
characteristics of clear cell carcinoma of the ovary: A distinct
histologic type with poor prognosis and resistance to
platinum-based chemotherapy. Cancer. 88:2584–2589. 2000. View Article : Google Scholar : PubMed/NCBI
|
|
6
|
Itamochi H, Kigawa J and Terakawa N:
Mechanisms of chemo-resistance and poor prognosis in ovarian clear
cell carcinoma. Cancer Sci. 99:653–658. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
7
|
Smith RA, Manassaram-Baptiste D, Brooks D,
Doroshenk M, Fedewa S, Saslow D, Brawley OW and Wender R: Cancer
screening in the United States, 2015: A review of current American
cancer society guidelines and current issues in cancer screening.
CA Cancer J Clin. 65:30–54. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
8
|
Lin LL, Huang HC and Juan HF: Revealing
the molecular mechanism of gastric cáncer marker annexin A4
incancer cell proliferation using exon arrays. PLoS One.
7:e446152012. View Article : Google Scholar
|
|
9
|
Huang HL, Yao HS, Wang Y, Wang WJ, Hu ZQ
and Jin KZ: Proteomic identification of tumor biomarkers associated
with primary gallbladder cancer. World J Gastroenterol.
20:5511–5518. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
10
|
Liu YY, Ge C, Tian H, Jiang JY, Zhao FY,
Li H, Chen TY, Yao M and Li JJ: The transcription factor Ikaros
inhibits cell proliferation by downregulating ANXA4 expression in
hepatocellular carcinoma. Am J Cancer Res. 7:1285–1297.
2017.PubMed/NCBI
|
|
11
|
Wang H, Deng L, Cai M, Zhuang H, Zhu L,
Hao Y, Gao J, Liu J, Li X and Lin B: Annexin A4 fucosylation
enhances its interaction with NF-κB p50 and promotes tumor
progression of ovarian clear cell carcinoma. Oncotarget.
8:108093–108107. 2016. View Article : Google Scholar
|
|
12
|
Jeon YJ, Kim DH, Jung H, Chung SJ, Chi SW,
Cho S, Lee SC, Park BC, Park SG and Bae KH: Annexin A4 interacts
with the NF-kappaB p50 subunit and modulates NF-kappaB
transcriptional activity in a Ca2+-dependent manner.
Cell Mol Life Sci. 67:2271–2281. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
13
|
Bolshakova A, Magnusson KE, Pinaev G and
Petukhova O: Functional compartmentalisation of NF-κB-associated
proteins in A431 cells. Cell Biol Int. 37:387–396. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
14
|
Fang Y, Chai Z, Wang D, Kuang T, Wu W and
Lou W: DNA-PKcs deficiency sensitizes the human hepatoma HepG2
cells to cisplatin and 5-fluorouracil through suppression of the
PI3K/Akt/NF-κB pathway. Mol Cell Biochem. 399:269–278. 2015.
View Article : Google Scholar
|
|
15
|
Basso F, Rocchetti F, Rodriguez S,
Nesterova M, Cormier F, Stratakis CA, Ragazzon B, Bertherat J and
Rizk-Rabin M: Comparison of the effects of PRKAR1A and PRKAR2B
depletion on signaling pathways, cell growth, and cell cycle
control of adrenocortical cells. Horm Metab Res. 46:883–888. 2014.
View Article : Google Scholar : PubMed/NCBI
|
|
16
|
Naugler WE and Karin M: NF-kappa B and
cancer-identifying targets and mechanisms. Curr Opin Genet Dev.
18:19–26. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
17
|
Luo JL, Kamata H and Karin M: IKK/NF-kappa
B signaling: Balancing life and death-a new approach to cancer
therapy. J Clin Invest. 115:2625–2632. 2005. View Article : Google Scholar : PubMed/NCBI
|
|
18
|
Masuishi Y, Arakawa N, Kawasaki H, Miyagi
E, Hirahara F and Hirano H: Wild-Type p53 enhances annexin IV gene
expression in ovarian clear cell adenocarcinoma. FEBS J.
278:1470–1483. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
19
|
Bykov VJ, Eriksson SE, Bianchi J and Wiman
KG: Targeting mutant p53 for efficient cancer therapy. Nat Rev
Cancer. 18:89–102. 2018. View Article : Google Scholar
|
|
20
|
Pereira A, Pérez-Medina T, Magrina JF,
Magtibay PM, Rodríguez-Tapia A, Peregrin I, Mendizabal E and
Ortiz-Quintana L: International federation of gynecology and
obstetrics staging classification for cancer of the ovary,
fallopian tube, and peritoneum: Estimation of survival in patients
with node-positive epithelial ovarian cancer. Int J Gynecol Cancer.
25:49–54. 2015. View Article : Google Scholar
|
|
21
|
Daly MB, Pilarski R, Berry M, Buys SS,
Farmer M, Friedman S, Garber JE, Kauff ND, Khan S, Klein C, et al:
NCCN guidelines insights: Genetic/Familial high-risk assessment:
Breast and ovarian, version 2.2017. J Natl Compr Canc Netw.
15:9–20. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
22
|
Wang H, Tan M, Zhang S, Li X, Gao J, Zhang
D, Hao Y, Gao S, Liu J and Lin B: Expression and significance of
CD44, CD47 and c-met in ovarian clear cell carcinoma. Int J Mol
Sci. 16:3391–3404. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
23
|
Iwamori M, Tanaka K, Kubushiro K, Lin B,
Kiguchi K, Ishiwata I, Tsukazaki K and Nozawa S: Alterations in the
glyolipid compo-sition and cellular properties of ovarian
carcinoma-derived RMG-1 cells on transfection of the α1,
2-fucosyltransferase gene. Cancer Sci. 96:26–30. 2005. View Article : Google Scholar : PubMed/NCBI
|
|
24
|
Shannon P, Markiel A, Ozier O, Baliga NS,
Wang JT, Ramage D, Amin N, Schwikowski B and Ideker T: Cytoscape: A
software environment for integrated models of biomolecular
interaction networks. Genom Res. 13:2498–2504. 2003. View Article : Google Scholar
|
|
25
|
Wade J, Armon S and McNally OM: Ovarian
clear cell carcinoma recurrence presenting as subcutaneous nodules.
Eur J Gynaecol Oncol. 37:575–577. 2016.PubMed/NCBI
|
|
26
|
Elvin JA, Chura J, Gay LM and Markman M:
Comprehensive genomic profiling (CGP) of ovarian clear cell
carcinomas (OCCC) identifies clinically relevant genomic
alterations (CRGA) and targeted therapy options. Gynecol Oncol Rep.
20:62–66. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
27
|
Dolcet X, Llobet D, Pallares J and
Matias-Guiu X: NF-κB in development and progression of human
cancer. Virchows Arch. 446:475–482. 2005. View Article : Google Scholar : PubMed/NCBI
|
|
28
|
DiDonato JA, Mercurio F and Karin M: NF-κB
and the link between inflammation and cancer. Immunol Rev.
246:379–400. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
29
|
Chariot A: The NF-kappaB-independent
functions of IKK subunits in immunity and cancer. Trends Cell Biol.
19:404–413. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
30
|
Rao P, Hayden MS, Long M, Scott ML, West
AP, Zhang D, Oeckinghaus A, Lynch C, Hoffmann A, Baltimore D and
Ghosh S: IkappaBbeta acts to inhibit and activate gene expres-sion
during the inflammatory response. Nature. 466:1115–1119. 2010.
View Article : Google Scholar : PubMed/NCBI
|
|
31
|
Wan F and Lenardo MJ: The nuclear
signaling of NF-kappaB: Current knowledge, new insights, and future
perspectives. Cell Res. 20:24–33. 2010. View Article : Google Scholar
|
|
32
|
Patel M, Horgan PG, McMillan DC and
Edwards J: NF-κB path-ways in the development and progression of
colorectal cancer. Transl Res. 197:43–56. 2018. View Article : Google Scholar : PubMed/NCBI
|
|
33
|
Perkins ND: The diverse and complex roles
of NF-κB subunits in cancer. Nat Rev Cancer. 12:121–132. 2012.
View Article : Google Scholar : PubMed/NCBI
|
|
34
|
Tilborghs S, Corthouts J, Verhoeven Y,
Arias D, Rolfo C, Trinh XB and van Dam PA: The role of nuclear
factor-kappa B signaling in human cervical cancer. Crit Rev Oncol
Hematol. 120:141–150. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
35
|
Wei B, Guo C, Liu S and Sun MZ: Annexin A4
and cancer. Clin Chim Acta. 447:72–78. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
36
|
Yao H, Sun C, Hu Z and Wang W: The role of
annexin A4 in cancer. Front Biosci (Landmark Ed). 21:949–957. 2016.
View Article : Google Scholar
|
|
37
|
Kim A, Serada S, Enomoto T and Naka T:
Targeting annexin A4 to counteract chemoresistance in clear cell
carcinoma of the ovary. Expert Opin Ther Targets. 14:963–971. 2010.
View Article : Google Scholar : PubMed/NCBI
|
|
38
|
Hatoum D, Yagoub D, Ahadi A, Nassif NT and
McGowan EM: Annexin/S100A protein family regulation through
p14ARF-p53 activation: A role in cell survival and predicting
treatment outcomes in breast cancer. PLoS One. 12:e01699252017.
View Article : Google Scholar : PubMed/NCBI
|
|
39
|
Perego P, Giarola M, Righetti SC, Supino
R, Caserini C, Delia D, Pierotti MA, Miyashita T, Reed JC and
Zunino F: Association between cisplatin resistance and mutation of
p53 gene and reduced bax expression in ovarian carcinoma cell
systems. Cancer Res. 56:556–562. 1996.PubMed/NCBI
|
|
40
|
Matsuo Y, Tashiro H, Yanai H, Moriya T and
Katabuchi H: Clinicopathological heterogeneity in ovarian clear
cell adeno-carcinoma: A study on individual therapy practice. Med
Mol Morphol. 48:146–154. 2015. View Article : Google Scholar
|
|
41
|
Shi J, Xiao H, Li J, Zhang J, Li Y, Zhang
J, Wang X, Bai X, Tao K, Hu D and Guan H: Wild-Type p53-modulated
autophagy and autophagic fibroblast apoptosis inhibit hypertrophic
scar formation. Lab Invest. 98:1423–1437. 2018. View Article : Google Scholar : PubMed/NCBI
|
|
42
|
Galmarini CM, Kamath K, Vanier-Viornery A,
Hervieu V, Peiller E, Falette N, Puisieux A, Jordan AM and Dumontet
C: Drug resistance associated with loss of p53 involves extensive
alterations in microtubule composition and dynamics. Br J Cancer.
88:1793–1799. 2003. View Article : Google Scholar : PubMed/NCBI
|
|
43
|
Meylan E, Dooley AL, Feldser DM, Shen L,
Turk E, Ouyang C and Jacks T: Requirement for NF-κB signaling in a
mouse model of lung adenocarcinoma. Nature. 462:104–107. 2009.
View Article : Google Scholar : PubMed/NCBI
|