|
1
|
McCulloch K, Litherland GJ and Rai TS:
Cellular senescence in osteoarthritis pathology. Aging Cell.
16:210–218. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
2
|
Loeser RF: Aging and osteoarthritis: The
role of chondrocyte senescence and aging changes in the cartilage
matrix. Osteoarthritis Cartilage. 17:971–979. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
3
|
Singh P, Marcu KB, Goldring MB and Otero
M: Phenotypic instability of chondrocytes in osteoarthritis: On a
path to hypertrophy. Ann N Y Acad Sci. 1442:17–34. 2019. View Article : Google Scholar
|
|
4
|
Greene MA and Loeser RF: Aging-related
inflammation in osteoarthritis. Osteoarthritis Cartilage.
23:1966–1971. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
5
|
Liu T, Yang Q, Zhang X, Qin R, Shan W,
Zhang H and Chen X: Quercetin alleviates kidney fibrosis by
reducing renal tubular epithelial cell senescence through the
SIRT1/PINK1/mitophagy axis. Life Sci. 257:1181162020. View Article : Google Scholar : PubMed/NCBI
|
|
6
|
Schön MP: Adaptive and innate immunity in
psoriasis and other inflammatory disorders. Front Immunol.
10:17642019. View Article : Google Scholar : PubMed/NCBI
|
|
7
|
Neogi T and Zhang Y: Epidemiology of
osteoarthritis. Rheum Dis Clin North Am. 39:1–19. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
8
|
Agarwal S, Misra R and Aggarwal A:
Interleukin 17 levels are increased in juvenile idiopathic
arthritis synovial fluid and induce synovial fibroblasts to produce
proinflammatory cytokines and matrix metalloproteinases. J
Rheumatol. 35:515–519. 2008.PubMed/NCBI
|
|
9
|
Robert M and Miossec P: IL-17 in
rheumatoid arthritis and precision medicine: From synovitis
expression to circulating bioactive levels. Front Med (Lausanne).
5:3642019. View Article : Google Scholar
|
|
10
|
Wojdasiewicz P, Poniatowski ŁA and
Szukiewicz D: The role of inflammatory and anti-inflammatory
cytokines in the pathogenesis of osteoarthritis. Mediators Inflamm.
2014:5614592014. View Article : Google Scholar : PubMed/NCBI
|
|
11
|
Honorati MC, Bovara M, Cattini L,
Piacentini A and Facchini A: Contribution of interleukin 17 to
human cartilage degradation and synovial inflammation in
osteoarthritis. Osteoarthritis Cartilage. 10:799–807. 2002.
View Article : Google Scholar : PubMed/NCBI
|
|
12
|
Lee YH and Bae SC: Associations between
circulating IL-17 levels and rheumatoid arthritis and between IL-17
gene polymorphisms and disease susceptibility: A meta-analysis.
Postgrad Med J. 93:465–471. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
13
|
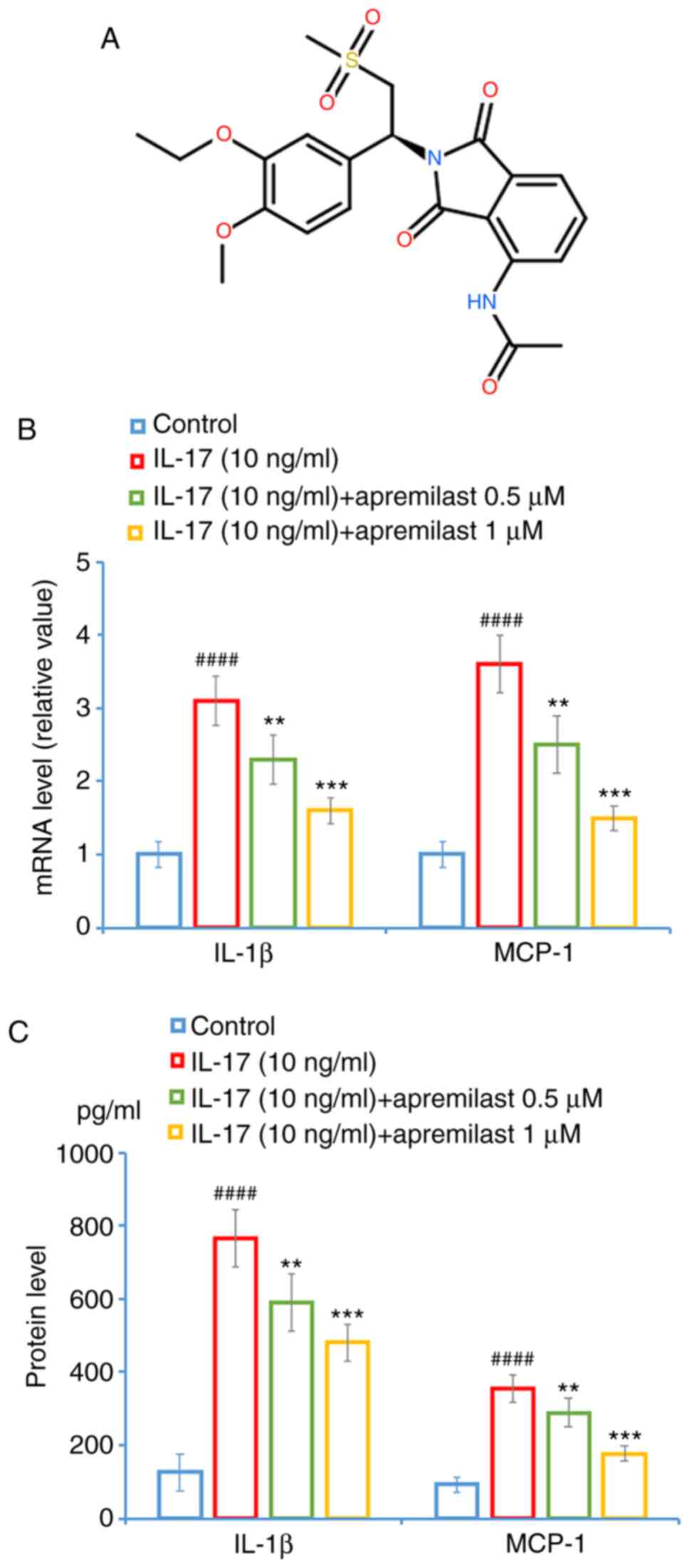
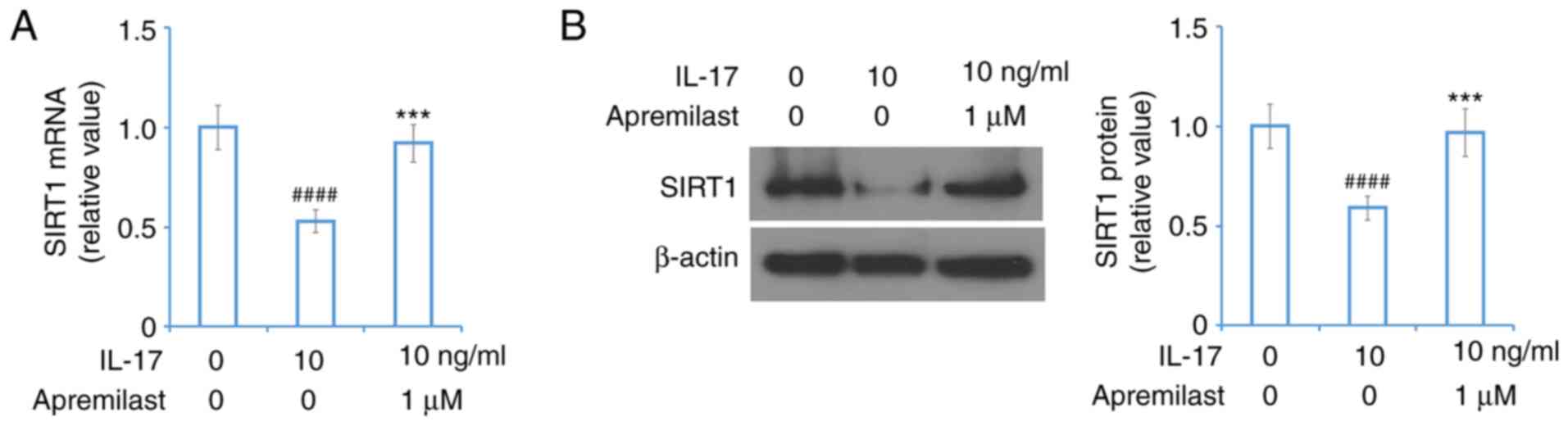
Otezla® (package insert).
Celgene Corporation; Summit, NJ:
|
|
14
|
Lanna C, Cesaroni GM, Mazzilli S, Vollono
L, Gaziano R, Marino D, Bianchi L and Campione E: Apremilast as a
target therapy for nail psoriasis: A real-life observational study
proving its efficacy in restoring the nail unit. J Dermatolog
Treat. Aug 3–2020.Epub ahead of print. View Article : Google Scholar : PubMed/NCBI
|
|
15
|
Schafer PH, Adams M, Horan G, Truzzi F,
Marconi A and Pincelli C: Apremilast normalizes gene expression of
inflammatory mediators in human keratinocytes and reduces
Antigen-Induced Atopic Dermatitis in mice. Drugs R D. 19:329–338.
2019. View Article : Google Scholar : PubMed/NCBI
|
|
16
|
Mavropoulos A, Zafiriou E, Simopoulou T,
Brotis AG, Liaskos C, Roussaki-Schulze A, Katsiari CG, Bogdanos DP
and Sakkas LI: Apremilast increases IL-10-producing regulatory B
cells and decreases proinflammatory T cells and innate cells in
psoriatic arthritis and psoriasis. Rheumatology (Oxford).
58:2240–2250. 2019. View Article : Google Scholar
|
|
17
|
Livak KJ and Schmittgen TD: Analysis of
relative gene expression data using real-time quantitative PCR and
the 2(-Delta Delta C(T)) method. Methods. 25:402–408. 2001.
View Article : Google Scholar
|
|
18
|
Sarubbo F, Esteban S, Miralles A and
Moranta D: Effects of resveratrol and other polyphenols on Sirt1:
Relevance to brain function during aging. Curr Neuropharmacol.
16:126–136. 2018. View Article : Google Scholar :
|
|
19
|
Hwang HS and Kim HA: Chondrocyte apoptosis
in the pathogenesis of osteoarthritis. Int J Mol Sci.
16:26035–26054. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
20
|
Toh WS, Brittberg M, Farr J, Foldager CB,
Gomoll AH, Hui JH, Richardson JB, Roberts S and Spector M: Cellular
senescence in aging and osteoarthritis. Acta Orthop. 87(Sup363):
6–14. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
21
|
Musumeci G, Szychlinska MA and Mobasheri
A: Age-related degeneration of articular cartilage in the
pathogenesis of osteoarthritis: Molecular markers of senescent
chondrocytes. Histol Histopathol. 30:1–12. 2015.
|
|
22
|
Malemud CJ: Cytokines as therapeutic
targets for osteoarthritis. BioDrugs. 18:23–35. 2004. View Article : Google Scholar : PubMed/NCBI
|
|
23
|
Wang M, Liu L, Zhang CS, Liao Z, Jing X,
Fishers M, Zhao L, Xu X and Li B: Mechanism of traditional Chinese
medicine in treating knee osteoarthritis. J Pain Res. 13:1421–1429.
2020. View Article : Google Scholar : PubMed/NCBI
|
|
24
|
Miller RE, Miller RJ and Malfait AM:
Osteoarthritis joint pain: The cytokine connection. Cytokine.
70:185–193. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
25
|
Ashraf S, Cha BH, Kim JS, Ahn J, Han I,
Park H and Lee SH: Regulation of senescence associated signaling
mechanisms in chondrocytes for cartilage tissue regeneration.
Osteoarthritis Cartilage. 24:196–205. 2016. View Article : Google Scholar
|
|
26
|
Lepetsos P and Papavassiliou AG:
ROS/oxidative stress signaling in osteoarthritis. Biochim Biophys
Acta. 1862:576–591. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
27
|
Drevet S, Gavazzi G, Grange L, Dupuy C and
Lardy B: Reactive oxygen species and NADPH oxidase 4 involvement in
osteoarthritis. Exp Gerontol. 111:107–117. 2018. View Article : Google Scholar : PubMed/NCBI
|
|
28
|
Zahan OM, Serban O, Gherman C and Fodor D:
The evaluation of oxidative stress in osteoarthritis. Med Pharm
Rep. 93:12–22. 2020.PubMed/NCBI
|
|
29
|
Li YS, Xiao WF and Luo W: Cellular aging
towards osteoarthritis. Mech Ageing Dev. 162:80–84. 2017.
View Article : Google Scholar : PubMed/NCBI
|
|
30
|
Takács-Buia L, Iordachel C, Efimov N,
Caloianu M, Montreuil J and Bratosin D: Pathogenesis of
osteoarthritis: Chondrocyte replicative senescence or apoptosis?
Cytometry B Clin Cytom. 74:356–362. 2008.PubMed/NCBI
|
|
31
|
Regulski MJ: Cellular Senescence: What,
why, and how. Wounds. 29:168–174. 2017.PubMed/NCBI
|
|
32
|
Dai SM, Shan ZZ, Nakamura H, Masuko-Hongo
K, Kato T, Nishioka K and Yudoh K: Catabolic stress induces
features of chondrocyte senescence through overexpression of
caveolin 1: Possible involvement of caveolin 1-induced
down-regulation of articular chondrocytes in the pathogenesis of
osteoarthritis. Arthritis Rheum. 54:818–831. 2006. View Article : Google Scholar : PubMed/NCBI
|
|
33
|
Guo Y, Fan W, Xie Y, Cao S, Wan H and Jin
B: SIRT1 is the target gene for
2,3,5,4′-tetrahydroxystilbene-2-O-β-D-Gluco side alleviating the
HUVEC senescence. Front Pharmacol. 11:5429022020. View Article : Google Scholar
|
|
34
|
Vaughan DE, Rai R, Khan SS, Eren M and
Ghosh AK: Plasminogen activator inhibitor-1 is a marker and a
mediator of senescence. Arterioscler Thromb Vasc Biol.
37:1446–1452. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
35
|
Kortlever RM, Higgins PJ and Bernards R:
Plasminogen activator inhibitor-1 is a critical downstream target
of p53 in the induction of replicative senescence. Nat Cell Biol.
8:877–884. 2006. View
Article : Google Scholar : PubMed/NCBI
|
|
36
|
Samarakoon R, Higgins SP, Higgins CE and
Higgins PJ: The TGF-β1/p53/PAI-1 signaling axis in vascular
senescence: Role of caveolin-1. Biomolecules. 9:3412019. View Article : Google Scholar
|
|
37
|
Li X: SIRT1 and energy metabolism. Acta
Biochim Biophys Sin (Shanghai). 45:51–60. 2013. View Article : Google Scholar
|
|
38
|
Takayama K, Ishida K, Matsushita T, Fujita
N, Hayashi S, Sasaki K, Tei K, Kubo S, Matsumoto T, Fujioka H, et
al: SIRT1 regulation of apoptosis of human chondrocytes. Arthritis
Rheum. 60:2731–2740. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
39
|
Dvir-Ginzberg M, Gagarina V, Lee EJ, Booth
R, Gabay O and Hall DJ: Tumor necrosis factor α-mediated cleavage
and inactivation of SirT1 in human osteoarthritic chondrocytes.
Arthritis Rheum. 63:2363–2373. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
40
|
Fujita N, Matsushita T, Ishida K, Kubo S,
Matsumoto T, Takayama K, Kurosaka M and Kuroda R: Potential
involvement of SIRT1 in the pathogenesis of osteoarthritis through
the modulation of chondrocyte gene expressions. J Orthop Res.
29:511–515. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
41
|
Gabay O, Zaal KJ, Sanchez C, Dvir-Ginzberg
M, Gagarina V, Song Y, He XH and McBurney MW: Sirt1-deficient mice
exhibit an altered cartilage phenotype. Joint Bone Spine.
80:613–620. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
42
|
Wei M and Duan D: Efficacy and safety of
monoclonal antibodies targeting interleukin-17 pathway for
inflammatory arthritis: A meta-analysis of randomized controlled
clinical trials. Drug Des Devel Ther. 10:2771–2777. 2016.
View Article : Google Scholar : PubMed/NCBI
|
|
43
|
Miossec P: Update on interleukin-17: A
role in the pathogenesis of inflammatory arthritis and implication
for clinical practice. RMD Open. 3:e0002842017. View Article : Google Scholar : PubMed/NCBI
|
|
44
|
Faust HJ, Zhang H, Han J, Wolf MT, Jeon
OH, Sadtler K, Peña AN, Chung L, Maestas DR Jr, Tam AJ, et al:
IL-17 and immunologically induced senescence regulate response to
injury in osteoarthritis. J Clin Invest. 130:5493–5507. 2020.
View Article : Google Scholar : PubMed/NCBI
|