Introduction
Breast cancer is the leading cause of cancer death
among women worldwide, accounting for 23% of the total cancer cases
and 14% of the cancer deaths in 2008 (1). Despite significant improvements in
detecting and treating early breast cancer, an estimated 75–80% of
patients with advanced disease develop bone metastasis (2). The pathologic complications of bone
metastasis can have devastating effects and patients experience
debilitating skeletal-related events (SREs), including pathological
fractures, hypercalcaemia of malignancy and spinal cord
compression. SREs are accompanied by severe bone pain and loss of
mobility, which eventually leads to reduction of quality of life
and survival (3–5).
Osteoprotegerin (OPG) is a secreted member of the
TNF receptor superfamily and plays an important role in bone
remodelling and osteoclastogenesis (6,7).
Osteoblasts and stromal cells express receptor activator of NF-κB
ligand (RANKL), which binds to its receptor RANK on pre-osteoclasts
and stimulates their differentiation and maturation into functional
osteoclasts (6). OPG, produced by
osteoblasts and other cell types, binds to RANKL and prevents the
association between RANKL and RANK, thereby inhibiting osteoclast
activation and function. Mice lacking OPG exhibit severe
osteoporosis (8), whereas
transgenic mice overexpressing OPG develop osteopetrosis (6). Several lines of experimental evidence
demonstrated that systemic administration of recombinant OPG
inhibits tumor growth in bone by inhibiting osteoclast function and
prevents bone loss in animal models of experimental bone metastasis
(9,10). Similarly, Corey et al
demonstrated that OPG produced locally by prostate cancer cells had
similar anti-osteolytic and anti-metastatic effects (11). However, contrary to these findings,
Fisher et al reported that local overexpression of OPG by
MCF-7 breast cancer cells co-expressing parathyroid hormone-related
protein enhanced tumor growth in bone and increased osteolysis
(12). Moreover, there is evidence
showing that high circulating levels of OPG in the serum of
patients with prostate cancer appear to be predictive of increased
bone metastases and increased osteolysis (13,14).
Taken together these findings indicate that OPG plays a significant
but perhaps context specific role in bone metastases, with evidence
supporting an anti-osteoclastogenic and tumor inhibiting action,
while in certain other situations it appears to stimulate
osteolysis and tumor growth. These apparently conflicting
observations suggest the need for additional research to delineate
the role of OPG in bone malignancies.
In this study we investigated the biological effects
of inhibiting bone resorption and bone remodelling on the behaviour
of breast cancer cells in bone. Specifically, we examined whether
OPG, when secreted locally by breast cancer cells in bone, can
inhibit osteolysis and tumor growth within the bone. Our data
demonstrate that overexpression of OPG by breast cancer cells
diminished intraosseus tumor growth and protected the bone from
breast cancer-induced osteolysis. However, despite the bone
protection, OPG overexpression led to a significant increase in the
incidence and severity of pulmonary metastasis. Taken together, our
data demonstrate that pharmacologic inhibition of bone remodelling
and bone resorption may in some cases affect the behaviour of
cancer cells within the bone microenvironment and their likelihood
of spreading and establishing metastasis elsewhere in the body.
Materials and methods
Cells and reagents
The MDA-MB-231 derivative cell line, MDA-MB-231-TXSA
was kindly provided by Dr Toshiyuki Yoneda (formerly at University
of Texas Health Sciences Centre, San Antonio, TX). Cells were
cultured in Dulbecco’s modified Eagle’s medium (DMEM, Gibco, Cat.
No. 12430-054), supplemented with 2 mM glutamine, 100 IU/ml
penicillin, 160 μg/ml gentamicin, HEPES (20 mM) and 10% fetal
bovine serum (Invitrogen, Cat. No. 11995-073), in a 5%
CO2-containing humidified atmosphere. The
MB-231-TXSA-TGL human breast cancer cell line has been tested and
authenticated by CellBank Australia (Wentworthville, NSW,
Australia) using short tandem repeat (STR) profiling (Report No.
13-163). The generation of luciferase-tagged
MDA-MB-231-TXSA-TGL-p-RUF and p-OPG overexpressing human breast
cancer cells were described previously (15).
In vitro osteoclast assays
Human peripheral blood mononuclear cells (PBMCs)
from healthy donors were isolated from buffy coats acquired from
the Australian Red Cross Blood Service. The cells were diluted in
Hank’s balanced salt solution (HBSS) and separated by gradient
centrifugation with Lymphoprep (Axis Shield, Cat. No. 1114547).
Isolated cells (2.5×105 cells/well) were then plated in
minimal essential medium (aMEM, Sigma-Aldrich, Cat. No. M4526),
supplemented with 10% fetal calf serum, L-glutamine (2 mM), HEPES
(20 mM), recombinant human M-CSF (25 ng/ml; Millipore, Cat. No.
GF053), 1α,25(OH)2vitamin D3 (10 nM; Wako Industries,
Cat. No. 031-14281) and dexamethasone (10 nM; Hospira, Cat. No.
483356) into osteologic slides (BD Biosciences, Cat. No. 354609),
for bone resorption assays, or directly into 96-well plates for
tartrate resistant acid phosphatase (TRAP) staining. The following
day, media from each well was removed and replaced with fresh media
supplemented with recombinant human RANKL (50 ng/ml; Millipore,
Cat. No. GF091), in the presence or absence of 10% conditioned
media from MDA-MB-231-TXSA-TGL-p-RUF and p-OPG-overexpressing
cells. Conditioned media (CM) was replaced every 3 days. Cells were
fixed on Day 7 and stained histochemically for TRAP (Sigma-Aldrich,
386-A), and TRAP+ve cells were visualized by light microscopy. To
assess bone resorption, osteologic slides were stained with Von
Kossa stain and resorption pits were counted using a light
microscope.
Animals
Five week old female athymic nude mice (Institute of
Medical and Veterinary Services Division, Gilles Plains, SA,
Australia) were acclimatized to the animal housing facility for a
minimum period of 1 week prior to the commencement of
experimentation. The general physical wellbeing and weight of
animals were monitored continuously throughout the experiments. All
mice were housed under pathogen free conditions and all
experimental procedures on animals were carried out with strict
adherence to the rules and guidelines for the ethical use of
animals in research and were approved by the Animal Ethics
Committees of the University of Adelaide and the Institute of
Medical and Veterinary Science, Adelaide, SA, Australia.
Intratibial injection model
MDA-MB-231-TXSA-TGL-p-RUF and p-OPG overexpressing
human breast cancer cells were cultured in Dulbecco’s modified
Eagle’s medium (DMEM), supplemented with 2 mM glutamine, 100 IU/ml
penicillin, 160 μg/ml gentamicin, HEPES (20 mM) and 10% fetal
bovine serum in a 5% CO2-containing humidified
atmosphere, until they reached 70–80% confluency. Adherent cells
were removed from flasks with 2 mM EDTA and resuspended in 1X PBS
at 0.5×105 cells/10 μl and kept on ice in an Eppendorf
tube. Mice (n=10/cell line) were anaesthetised by Isoflurane
(Faulding Pharmaceuticals, SA, Australia), the left tibia was
cleaned with 70% ethanol and a 27-gauge needle coupled to a
Hamilton syringe was inserted through the tibial plateau with the
knee flexed and 0.5×105 cells resuspended in 10 μl of
PBS were injected into the marrow space. Tumor volume was monitored
regularly and mice were imaged on a weekly basis by bioluminescence
imaging (BLI) (see below). Mice were humanely sacrificed 4 weeks
after cancer cell transplantation, due to high tumor load and lungs
and tibiae were removed for assessment of tumor burden and bone
volume, respectively.
Preparation of blood serum and detection
of OPG by ELISA
Blood was collected from all the mice at Day 18
(tail bleeds), and at the time of termination of the experiment, to
determine the OPG concentration in the blood serum of the mice.
Blood was collected in MiniCollect tubes (0.8 ml LH Lithium Hep
Sep) and serum was separated and transferred into fresh Eppendorf
tubes and stored at −80°C until assay. The concentration of OPG in
blood serum collected from all animals was determined using a
commercial ELISA kit as per the manufacturer’s instructions
(Immunodiagnostik AG, Cat. No. KB 1011).
Bioluminescence imaging (BLI) of tumor
growth
Non-invasive, whole body imaging for assessment of
tumor growth was performed once weekly using the IVIS 100 Imaging
system (Xenogen, Alameda, CA). Mice were injected i.p. with 100 μl
of the D-Luciferin solution at a final dose of 3 mg/20 g mouse body
weight (Biosynth, Cat. No. L-82220) and then gas-anaesthetized with
Isoflurane (Faulding Pharmaceuticals). Images were acquired for
0.5–30 sec (images are shown at 1 sec) from the side angle and the
photon emission transmitted from mice was captured and quantitated
in photons/sec/cm2/sr using Xenogen Living image (Igor
Pro version 2.5) software.
Micro-computed (μCT) tomography
analysis
Limbs for μCT analysis were surgically resected and
scanned using the SkyScan-1174 high-resolution μCT Scanner
(Skyscan, Belgium). For μCT scanning, the tibiae were placed
vertically in tightly fitting plastic tubes. The μCT Scanner was
operated at 50 kV, 800 μA, 0.4 rotation step, 0.25 mm aluminium
filter and scan resolution of 7.78 μm/pixel. The cross sections
were reconstructed using a cone-beam algorithm (software NRecon,
Skyscan). Files were then imported into CTAn software (Skyscan) for
3D analysis and 3D image generation. Using the 2D images obtained
from the μCT scan, the growth plate was identified and 400 sections
were selected starting from the growth plate/tibial interface and
moving down the tibia. All images were viewed and edited using
CTvol visualisation software. Histograms, representing bone volume
(mm3), total and trabecular, were generated from
tumor-bearing tibiae and compared to the contralateral non-tumor
bearing tibiae. Tumor burdens, measured in mm3, were
determined using the Skyscan software.
Histology
Tibiae were fixed in 10% (v/v) buffered formalin (24
h at 4°C), followed by 2–4 weeks of decalcification in 0.5 M
EDTA/0.5% paraformaldehyde in PBS, pH 8.0 at 4°C. Complete
decalcification of the tibiae was confirmed by radiography and
tibiae were then paraffin embedded. Five micron longitudinal
sections were prepared and stained with H&E. Additional
sections were used for osteoclast-specific tartrate-resistant acid
phosphatase 5 (ACP5/TRAP) staining, following the manufacturer’s
protocol (Sigma-Aldrich, 386-A). Analysis was performed on an
Olympus CX41 microscope and photo-images were taken using the
Nanozoomer Digital Pathology (NDP-Hamamatsu). Tumor area measured
in mm2 was assessed using the Nanozoomer software. Lungs
were also fixed in 10% (v/v) buffered formalin and were then
paraffin- embedded and sectioned at 5 μm at three different levels,
followed by H&E staining. Total lung area and metastatic foci
area were measured in mm2 using the Nanozoomer
software.
Data analysis and statistics
Experiments were performed in triplicate, and data
are presented as mean ± SE. All statistical analysis was performed
using SigmaStat for Windows version 3.0 (Systat Software, Inc.,
Port Richmond, CA), using the unpaired Student’s t-test.
Comparisons among groups were assessed using a one-way ANOVA test.
In all cases, p<0.05 was considered statistically
significant.
Results
OPG produced by MDA-MB-231-TXSA-p-OPG
cells is biologically active
To determine whether the OPG secreted by the
transfected breast cancer cells was biologically active, we
performed in vitro osteoclast formation assays in the
presence of condition media from control and OPG overexpressing
cell lines. Consistent with the role of OPG in inhibiting
osteoclast differentiation and bone resorption, we found that when
PBMCs were cultured with the receptor activator of nuclear factor
κB ligand (RANKL), CM (10%) from p-OPG-overexpressing cells, but
not from p-RUF empty vector transfected cells, dramatically
inhibited the formation of TRAP+ multinucleated cells
(Fig. 1A). Further, bone
resorption by mature osteoclasts (OCL) derived from PBMCs was
almost totally abolished by the transfected cell CM (Fig. 1B).
OPG overexpressed by breast cancer cells
does not affect total tumor burden in vivo
To evaluate the effect of OPG overexpression by
breast cancer cells on tumor growth we used an animal model in
which the empty vector or OPG-overexpressing MDA-MB-231-TXSA breast
cancer cells were transplanted directly into the tibial marrow
cavity of female athymic nude mice. Bioluminescence imaging
provided sensitive real-time in vivo assessment of breast
cancer growth in bone. Transfected cells were co-transduced with a
triple-fusion protein reporter construct encoding herpes simplex
virus thymidine kinase (TK), green fluorescent protein (GFP) and
firefly luciferase (Luc), as described previously (15,16).
Development of breast cancer-induced bone destruction was assessed
using high-resolution μCT analysis. Weekly imaging showed that all
animals inoculated with the empty vector transfected cells showed
an increase of mean photon emission, indicating an increase in
tumor burden, which was clearly evident from Day 14 onwards
(Fig. 2A and B). Similar results
were found with mice bearing OPG-overexpressing tumors, with BLI
showing no significant differences between the two groups. Bloods
were collected from all animals at different time points during the
experiment and the levels of circulating OPG in the mouse serum was
measured by ELISA (Fig. 2C).
Circulating OPG levels in animals bearing empty vector transfected
cells were approximately 0.2 pmol/l, when measured on Day 18-post
cancer cell transplantation. Mice bearing OPG-overexpressing tumors
showed 50-fold higher serum OPG levels of 10 pmol/l in mice bearing
OPG-overexpressing tumors. By Day 28, the levels of OPG in mice
bearing OPG-overexpressing tumors further increased to 45 pmol/l,
indicating that these cells maintained high level expression of OPG
over the entire period of the study. Mice in both groups were
humanely sacrificed on Day 28 due to the high tumor load in the
tibiae.
OPG overexpression inhibits
cancer-induced osteolysis
To assess the effects of OPG overexpression on bone,
high-resolution μCT analysis was performed on isolated tibiae at
the end of the study. Reconstructed 3-D μCT images of
representative empty vector transfected tumor-bearing tibiae
demonstrated extensive osteolysis when compared to the
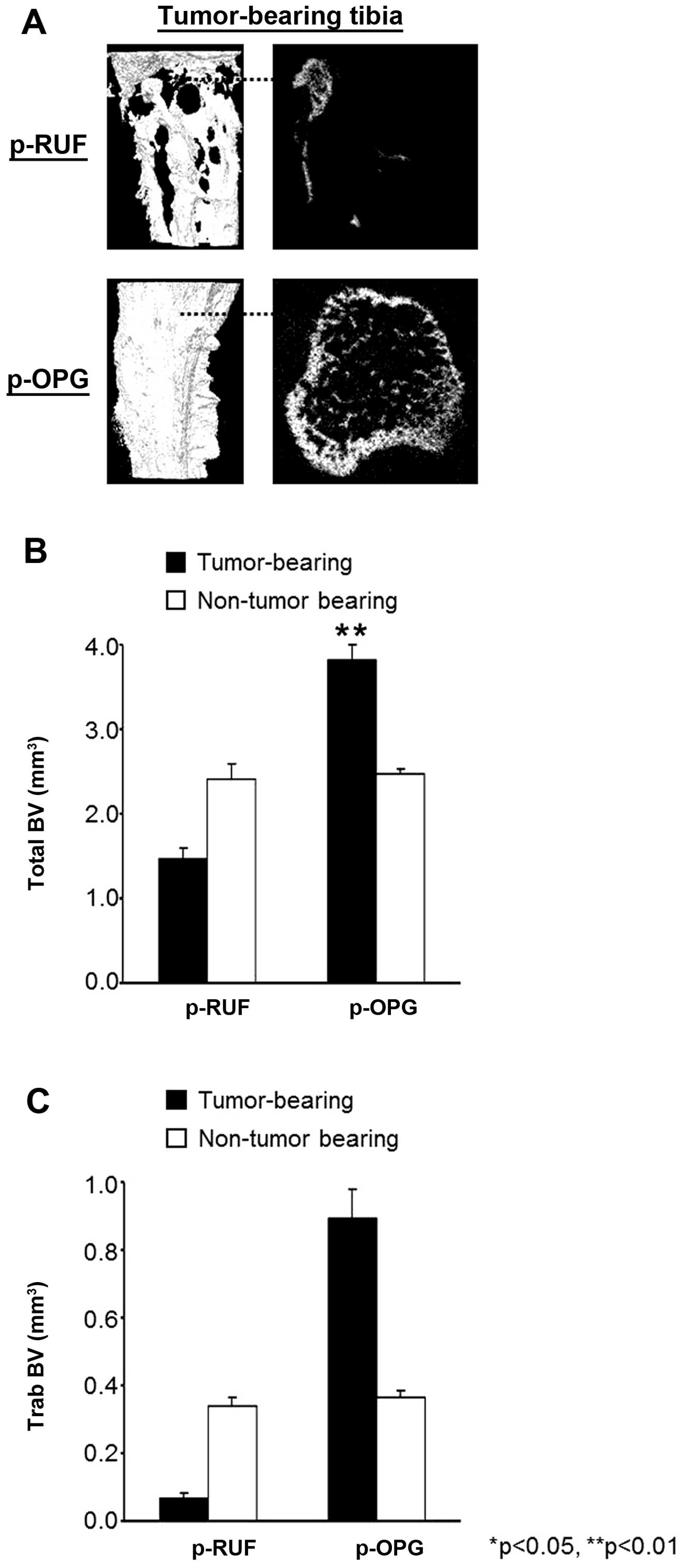
contralateral non-tumor bearing tibiae (Fig. 3A). In contrast, and consistent with
the role of OPG in inhibiting osteoclastic bone resorption, all
animals inoculated with the p-OPG transfected cells showed
preservation of the integrity of bone around the tumors and
protection from tumor-induced osteolysis, as shown in Fig. 3A. To quantify the total bone volume
(BV), we compared the tumor-bearing tibiae with the contralateral
non-tumor bearing tibiae of all the animals in each group in a
region beginning at the growth plate and extending downwards
400×7.8 μm slices, which encompassed all of the cancer lesions. As
seen in Fig. 3B, the amount of
bone lost in the tibiae of mice inoculated with empty vector
transfected cells exceeded 40% in the tumor-bearing tibiae when
compared to the contralateral non-tumor bearing tibiae. In
contrast, OPG overexpression by cancer cells resulted in remarkable
protection from breast cancer-induced osteolysis, translating to a
significant further increase (55%) in BV in the tibiae bearing
OPG-overexpressing tumors when compared to the contralateral tibia.
The effect of OPG on trabecular bone volume (TbBV) was more
pronounced (Fig. 3C). Mice
inoculated with the empty vector transfected cells showed a
dramatic loss of their trabecular bone (>80%) when compared to
the contralateral leg and to the tibiae of tumors containing
OPG-overexpressing cells. In contrast, mice bearing
OPG-overexpressing tumors showed a 141% gain in their TbBV when
compared to the contralateral right tibiae bars ± SEM,
*p<0.05.
OPG secreted by breast cancer cells
maintains skeletal integrity but alters the intra- and
extra-medullary tumor distribution
Tumor burden was evaluated using bioluminescence and
showed no significant differences in the average tumor signal
between the mice bearing p-OPG tumors when compared to the mice
with p-RUF tumors. However, a detailed histological examination of
the tibiae, using high resolution imaging, showed that the
distribution of the tumor in the bone was different in animals with
OPG-overexpressing tumors compared to animals bearing empty vector
transfected tumors. In mice bearing empty vector transfected
tumors, there was persistent growth of cancer cells within the bone
marrow cavity, representing 76.4% of the overall tumor burden,
which penetrated the cortical bone and invaded the surrounding soft
tissue. In contrast, histological sections of tibiae with
OPG-overexpressing tumors showed that the intra-osseous tumor
burden was significantly decreased, accounting only for 3% of the
overall tumor burden. In these tibiae, cancer cells were almost
undetectable within the bone marrow space and this was also
associated with a dramatic increase in trabecular bone density and
a concomitant decrease in bone marrow volume (Fig. 4A). Importantly, OPG-overexpressing
cancer cells escaped the marrow cavity and continued to grow in the
extra-medullary space, accounting for 96.8% of the total tumor
burden. Fig. 4B shows the intra
and extra-osseous tumor burden in the tibiae, expressed as an
average tumor area per group. The observed inhibition of osteolysis
by OPG was due to the suppression of osteoclastic bone resorption
since OPG released by tumor cells significantly decreased the
number of osteoclasts lining the bone surface. Fig. 4C shows that TRAP+
osteoclasts were abundant and attached to the bone surfaces in
tumor lesions from the vector only transfected cells. In contrast,
there was almost complete absence of TRAP+ osteoclasts
in tibiae from animals inoculated with p-OPG transfected cells,
likely accounting for the protective effect of OPG overexpression
on bone destruction and also confirming the biological activity of
OPG in vivo (Fig. 4D).
OPG secreted by breast cancer cells
promotes pulmonary metastasis
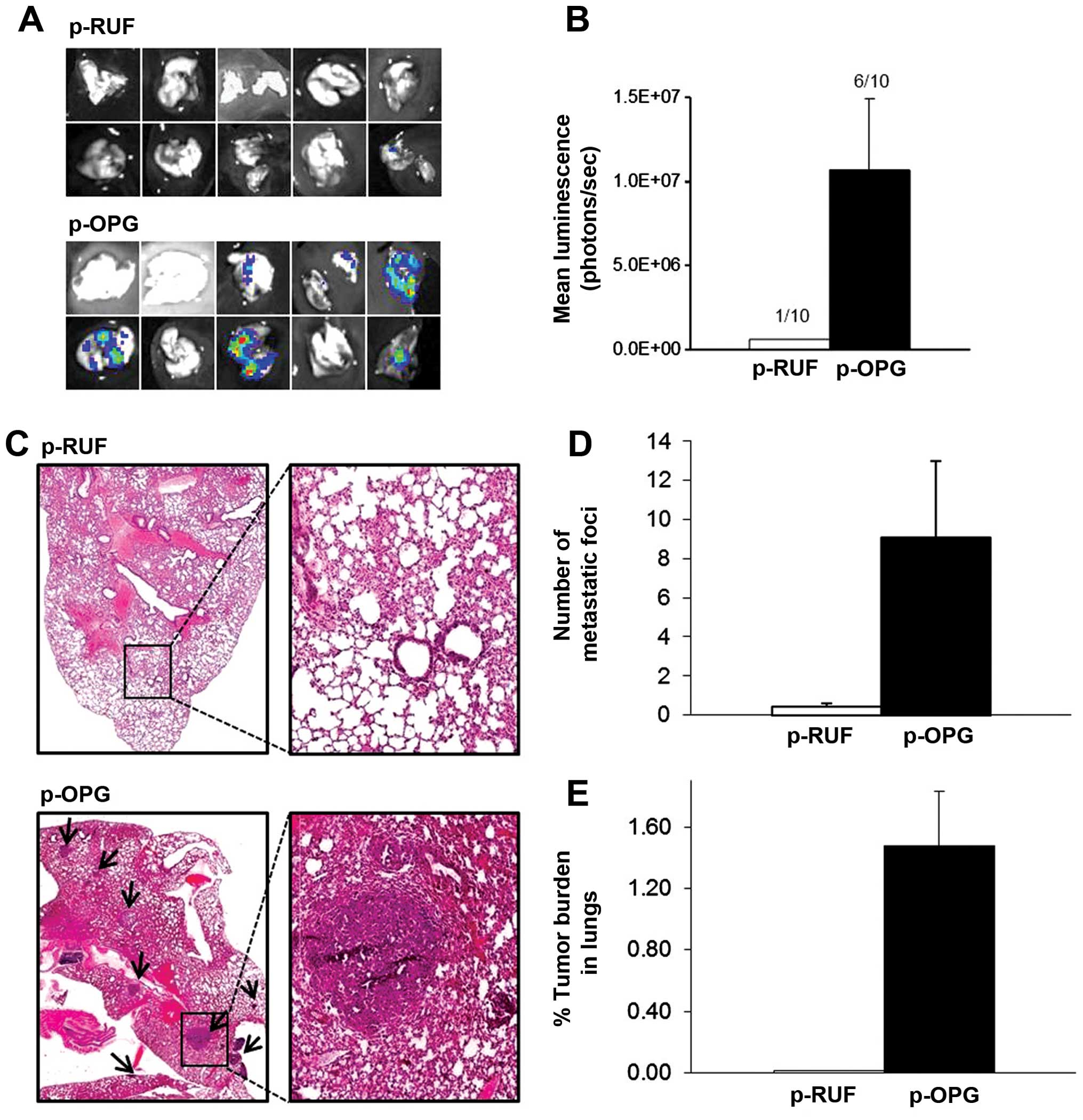
While OPG overexpression protected the bone from
cancer-induced bone destruction, and dramatically reduced
intra-osseous tumor mass, lung metastases increased significantly
in OPG-overexpressing animals. At the end of the experiment the
lungs from each group of animals were excised and lung metastases,
as a function of photon counts per second, were quantified ex
vivo using BLI. Six of ten animals in the OPG-overexpressing
group had detectable bioluminescence signal in the lungs compared
with one in ten from the group of mice inoculated with empty vector
transfected cells (Fig. 5A). The
mean luciferase activity in lungs from mice bearing
OPG-overexpressing tumors was significantly higher than that of the
animals bearing empty vector transfected tumors (Fig. 5B). Histological assessment of the
lungs corroborated these findings in that multiple pulmonary
macrometastases were detected in the lungs of animals with OPG
expressing tumors that were also positive with BLI (Fig. 5C). The number of metastatic foci in
the lungs was significantly higher in these mice when compared to
the animals bearing empty vector transfected cancer cells (Fig. 5D). Tumor area was also calculated
from histological sections and expressed as an average tumor area
per group in absolute units (mm2). The percentage of
tumor burden of the lung area was approximately 105-fold higher in
the mice with OPG-overexpressing tumors compared to the p-RUF mice
(Fig. 5E).
Discussion
In this study we investigated the biological effect
of inhibiting bone resorption and bone remodelling on the behaviour
of breast cancer cells within the bone microenvironment and tested
the hypothesis that inhibition of bone resorption per se may
augment breast cancer metastasis from bone to other tissues. For
this, we used a well-established xenogeneic murine model of
osteolytic breast cancer, in which human breast cancer cells were
transplanted directly into the tibial marrow cavity of female
athymic mice. This in vivo model mimics the late stages of
bone metastasis and is ideally suited for monitoring the effects of
antiresorptive agents on breast cancer growth in the bone and also
on cancer-induced bone destruction. Inhibition of bone resorption
was achieved using two approaches. We engineered MDA-MB-231-TXSA
breast cancer cells to overexpress native full length human OPG
and, following their intratibial transplantation, tested the
effects of high doses of OPG when produced and secreted locally by
breast cancer cells in the bone microenvironment.
Our results show that OPG overexpression
significantly reduced intra-osseous tumor burden and protected the
bone from cancer-induced osteolysis. These effects were due to the
actions of OPG on osteoclastic bone resorption as demonstrated by
the near complete absence of TRAP+ osteoclasts lining
the bone surface. However, despite such marked bone protection, OPG
overexpression failed to reduce the total tumor burden and breast
cancer cells persisted to grow unaffected in the extra-medullary
space. Importantly, inhibition of bone resorption by OPG was
associated with an increased tendency of breast cancer cells to
metastasize to the lungs, since OPG overexpression by the breast
cancer cells demonstrated a significant increase in the incidence
of pulmonary metastasis, when compared to the control animals.
The data presented here are in line with our
previously published data which demonstrated that inhibiting bone
resorption by systemic administration of clinically relevant doses
of the antiresorptive agent zoledronic acid (ZOL) had a significant
protective effect on osteosarcoma (OS)-induced bone destruction but
did not inhibit the total tumor burden or reduce lung metastases,
and in some cases even promoted lung metastases (16). This observation was further
validated by another independent study in which we showed that
osteoclast inhibition by ZOL treatment increased lung metastases in
mice with intra-femorally transplanted OS cells, while fulvestrant
treatment lead to increases in osteoclast numbers and decreased the
number of lung metastases (17).
In a human setting, we conducted a transcriptomic screen of patient
OS biopsies and found that expression of TRAP is significantly
downregulated in primary OS compared with non-malignant bone
(17). Moreover, within the OS
patient cohort, patients who subsequently developed pulmonary
metastasis had significantly lower TRAP expression and osteoclast
numbers in their biopsies than those patients who did not develop
metastasis suggesting that the metastatic potential of OS is
determined in primary tumor development and that loss of
osteoclasts and consequently alterations in bone remodelling in the
primary lesion may enhance OS metastasis. Importantly, using the
same breast cancer model as used here we have recently demonstrated
that systemic administration of conventional clinical relevant
doses of ZOL had no effect on tumor growth in bone but similarly
promoted lung metastases in mice (18). However, metronomic doses of ZOL
(i.e., lower doses given more frequently on a prolonged schedule)
significantly reduced tumor burden in the tibiae of mice and
reduced lung and liver metastasis when compared to the conventional
treatment, suggesting that a metronomic dosing regimen may be more
beneficial in the clinical setting.
Several clinical trials have shown the ability of
oral and intravenous bisphosphonates to reduce the incidence and
frequency of SREs and skeletal morbidity and prevent bone loss in
patients with breast cancer bone metastasis when administrated
alone or in combination with adjuvant therapy (5,19–24).
However, results from the recent AZURE (4) trial, in which over 3000 patients with
stage II/III breast cancer were randomized to receive standard
therapy (chemotherapy, endocrine therapy, radiation) or standard
therapy plus 4 mg of ZOL showed no significant difference between
patients receiving ZOL in addition to adjuvant therapy compared to
patients receiving only standard therapy, in terms of recurrence of
breast cancer or on overall survival. However, a subgroup analysis
that evaluated the patients receiving ZOL plus adjuvant therapy by
their menopausal status, showed a significant positive effect on
both recurrence and survival but only in postmenopausal women
compared to patients who were pre- and peri-menopausal.
Importantly, in pre- or peri-menopausal women there was an
increased risk of extraskeletal metastasis. The data from the AZURE
study suggest that the reproductive hormones play a role in the
bone remodeling process and on the behaviour of cancer cells in the
bone microenvironment, possibly contributing to cancer cell escape
from bone to distant sites and implies that the hormonal
environment influences the zolendronic effect on the metastatic
potential of the cancer cells.
Taken together, the results of our study suggest
that therapeutic agents that inhibit osteoclastic bone resorption
and bone remodeling may in certain instances potentially cause more
harm than good by promoting extraskeletal metastasis in patients
with already established bone metastases, especially in an
environment with pre-menopausal levels of reproductive hormones.
These important and apparently conflicting observations suggest the
need for additional research to better understand the role of
osteoclast inhibition in patients with bone metastasis based on
their hormonal status.
Acknowledgements
This study was supported by grants from the National
Health and Medical Research Council (NHMRC) of Australia ID#
627015, The Hospital Research Foundation, Adelaide, South
Australia, the National Breast Cancer Foundation and the Australian
Breast Cancer Research. The funders had no role in study design,
data collection and analysis, decision to publish or preparation of
the manuscript.
References
|
1
|
Jemal A, Bray F, Center MM, Ferlay J, Ward
E and Forman D: Global cancer statistics. CA Cancer J Clin.
61:69–90. 2011. View Article : Google Scholar
|
|
2
|
Siegel R, Ward E, Brawley O and Jemal A:
Cancer statistics, 2011: the impact of eliminating socioeconomic
and racial disparities on premature cancer deaths. CA Cancer J
Clin. 61:212–236. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
3
|
Coleman RE, Lipton A, Roodman GD, et al:
Metastasis and bone loss: advancing treatment and prevention.
Cancer Treat Rev. 36:615–620. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
4
|
Coleman RE, Marshall H, Cameron D, et al:
Breast-cancer adjuvant therapy with zoledronic acid. N Engl J Med.
365:1396–1405. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
5
|
Guise TA, Brufsky A and Coleman RE:
Understanding and optimizing bone health in breast cancer. Curr Med
Res Opin. 26(Suppl 3): S3–S20. 2010. View Article : Google Scholar
|
|
6
|
Simonet WS, Lacey DL, Dunstan CR, et al:
Osteoprotegerin: a novel secreted protein involved in the
regulation of bone density. Cell. 89:309–319. 1997. View Article : Google Scholar
|
|
7
|
Tsuda E, Goto M, Mochizuki S, et al:
Isolation of a novel cytokine from human fibroblasts that
specifically inhibits osteoclastogenesis. Biochem Biophys Res
Commun. 234:137–142. 1997. View Article : Google Scholar : PubMed/NCBI
|
|
8
|
Bucay N, Sarosi I, Dunstan CR, et al:
Osteoprotegerin-deficient mice develop early onset osteoporosis and
arterial calcification. Genes Dev. 12:1260–1268. 1998. View Article : Google Scholar : PubMed/NCBI
|
|
9
|
Buijs JT and van der Pluijm G: Osteotropic
cancers: from primary tumor to bone. Cancer Lett. 273:177–193.
2009. View Article : Google Scholar : PubMed/NCBI
|
|
10
|
Croucher PI, Shipman CM, Lippitt J, et al:
Osteoprotegerin inhibits the development of osteolytic bone disease
in multiple myeloma. Blood. 98:3534–3540. 2001. View Article : Google Scholar : PubMed/NCBI
|
|
11
|
Corey E, Brown LG, Kiefer JA, et al:
Osteoprotegerin in prostate cancer bone metastasis. Cancer Res.
65:1710–1718. 2005. View Article : Google Scholar : PubMed/NCBI
|
|
12
|
Fisher JL, Thomas-Mudge RJ, Elliott J, et
al: Osteoprotegerin overexpression by breast cancer cells enhances
orthotopic and osseous tumor growth and contrasts with that
delivered therapeutically. Cancer Res. 66:3620–3628. 2006.
View Article : Google Scholar
|
|
13
|
Brown JM, Vessella RL, Kostenuik PJ,
Dunstan CR, Lange PH and Corey E: Serum osteoprotegerin levels are
increased in patients with advanced prostate cancer. Clin Cancer
Res. 7:2977–2983. 2001.PubMed/NCBI
|
|
14
|
Chen G, Sircar K, Aprikian A, Potti A,
Goltzman D and Rabbani SA: Expression of RANKL/RANK/OPG in primary
and metastatic human prostate cancer as markers of disease stage
and functional regulation. Cancer. 107:289–298. 2006. View Article : Google Scholar : PubMed/NCBI
|
|
15
|
Zinonos I, Labrinidis A, Lee M, et al:
Anticancer efficacy of Apo2L/TRAIL is retained in the presence of
high and biologically active concentrations of osteoprotegerin in
vivo. J Bone Miner Res. 26:630–643. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
16
|
Labrinidis A, Diamond P, Martin S, et al:
Apo2L/TRAIL inhibits tumor growth and bone destruction in a murine
model of multiple myeloma. Clin Cancer Res. 15:1998–2009. 2009.
View Article : Google Scholar : PubMed/NCBI
|
|
17
|
Endo-Munoz L, Cumming A, Rickwood D, et
al: Loss of osteoclasts contributes to development of osteosarcoma
pulmonary metastases. Cancer Res. 70:7063–7072. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
18
|
Luo KW, Ko CH, Yue GG, et al: Anti-tumor
and anti-osteolysis effects of the metronomic use of zoledronic
acid in primary and metastatic breast cancer mouse models. Cancer
Lett. 339:42–48. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
19
|
Body JJ, Diel IJ, Bell R, et al: Oral
ibandronate improves bone pain and preserves quality of life in
patients with skeletal metastases due to breast cancer. Pain.
111:306–312. 2004. View Article : Google Scholar : PubMed/NCBI
|
|
20
|
Brufsky AM, Bosserman LD, Caradonna RR, et
al: Zoledronic acid effectively prevents aromatase
inhibitor-associated bone loss in postmenopausal women with early
breast cancer receiving adjuvant letrozole: Z-FAST study 36-month
follow-up results. Clin Breast Cancer. 9:77–85. 2009. View Article : Google Scholar
|
|
21
|
Bundred NJ, Campbell ID, Davidson N, et
al: Effective inhibition of aromatase inhibitor-associated bone
loss by zoledronic acid in postmenopausal women with early breast
cancer receiving adjuvant letrozole: ZO-FAST Study results. Cancer.
112:1001–1010. 2008. View Article : Google Scholar
|
|
22
|
Paterson AH, Powles TJ, Kanis JA,
McCloskey E, Hanson J and Ashley S: Double-blind controlled trial
of oral clodronate in patients with bone metastases from breast
cancer. J Clin Oncol. 11:59–65. 1993.PubMed/NCBI
|
|
23
|
Rosen LS, Gordon D, Kaminski M, et al:
Zoledronic acid versus pamidronate in the treatment of skeletal
metastases in patients with breast cancer or osteolytic lesions of
multiple myeloma: a phase III, double-blind, comparative trial.
Cancer J. 7:377–387. 2001.
|
|
24
|
Rosen LS, Gordon D, Tchekmedyian S, et al:
Zoledronic acid versus placebo in the treatment of skeletal
metastases in patients with lung cancer and other solid tumors: a
phase III, double-blind, randomized trial - the Zoledronic Acid
Lung Cancer and Other Solid Tumors Study Group. J Clin Oncol.
21:3150–3157. 2003. View Article : Google Scholar
|