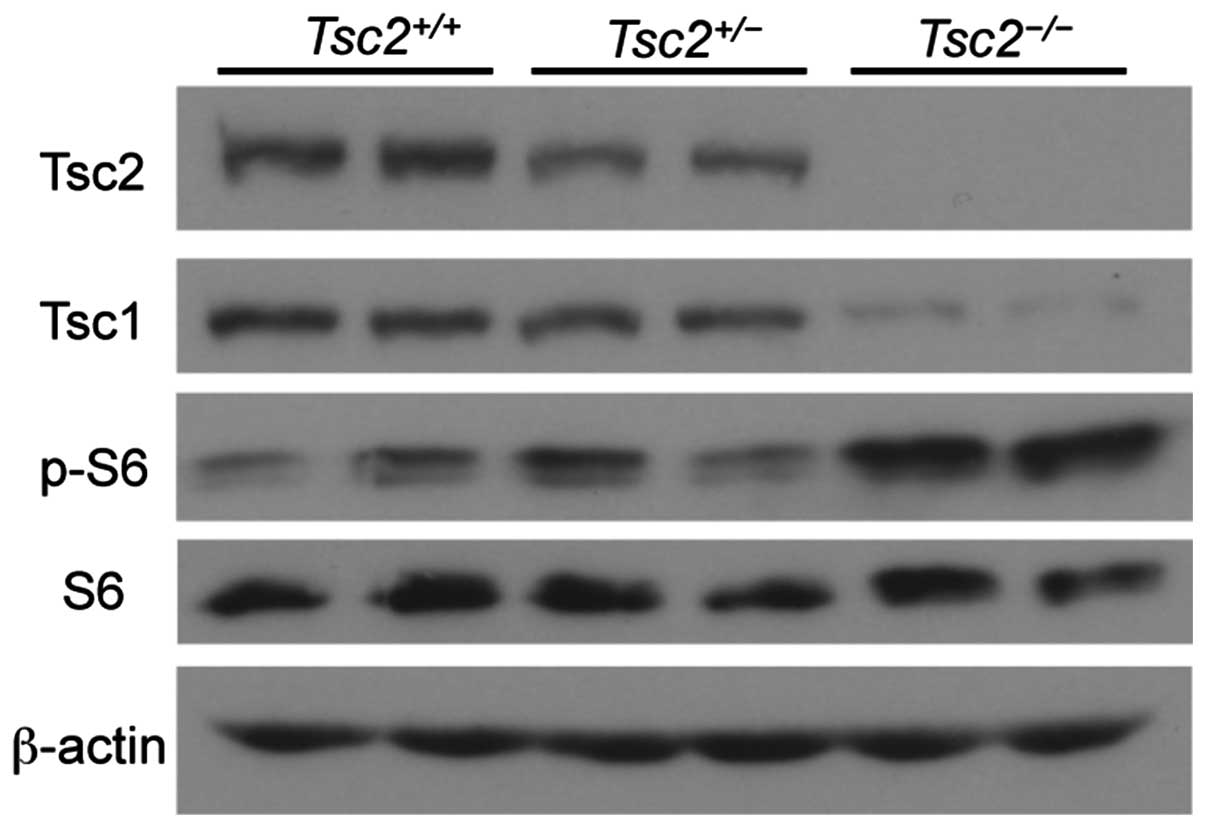
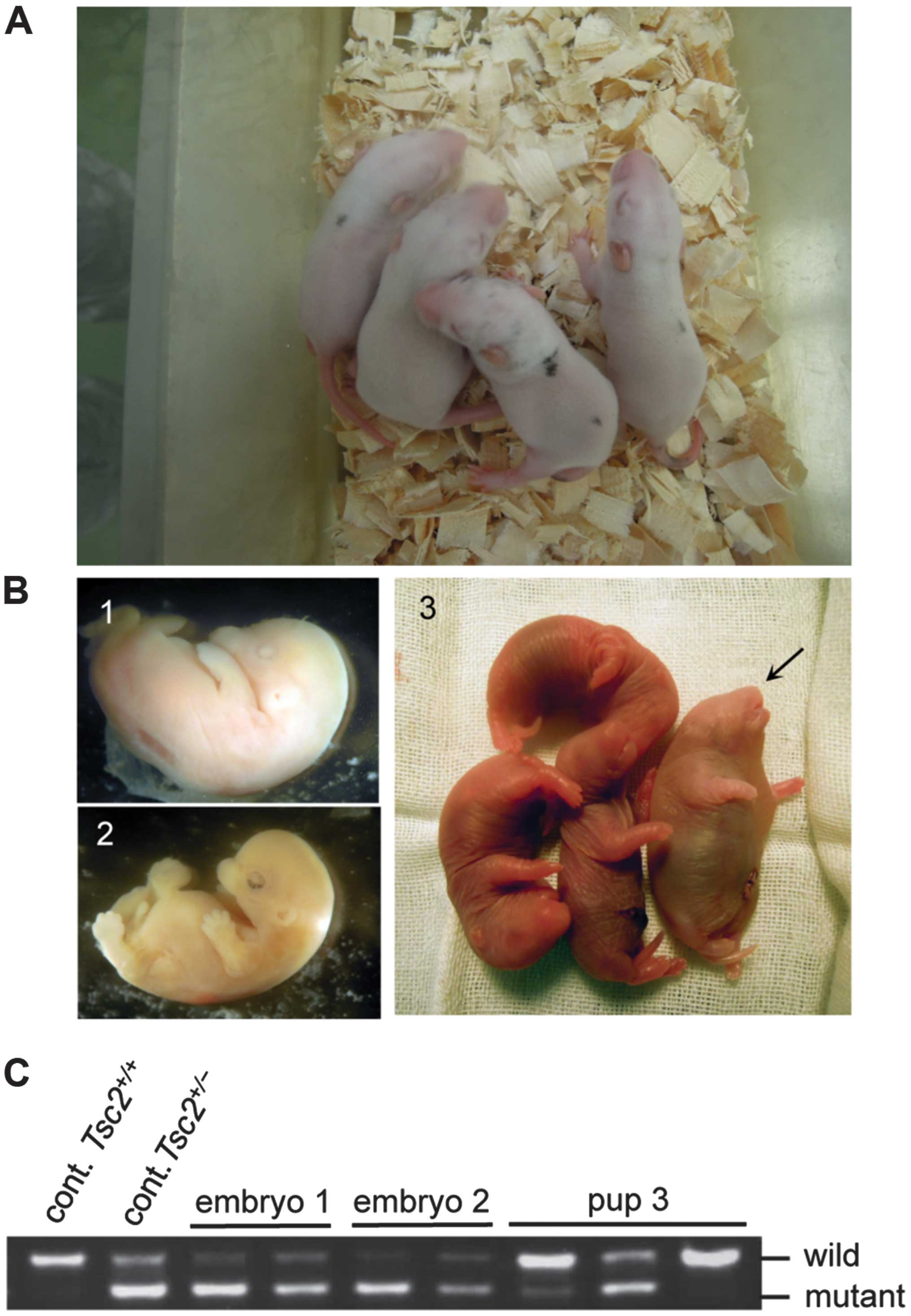
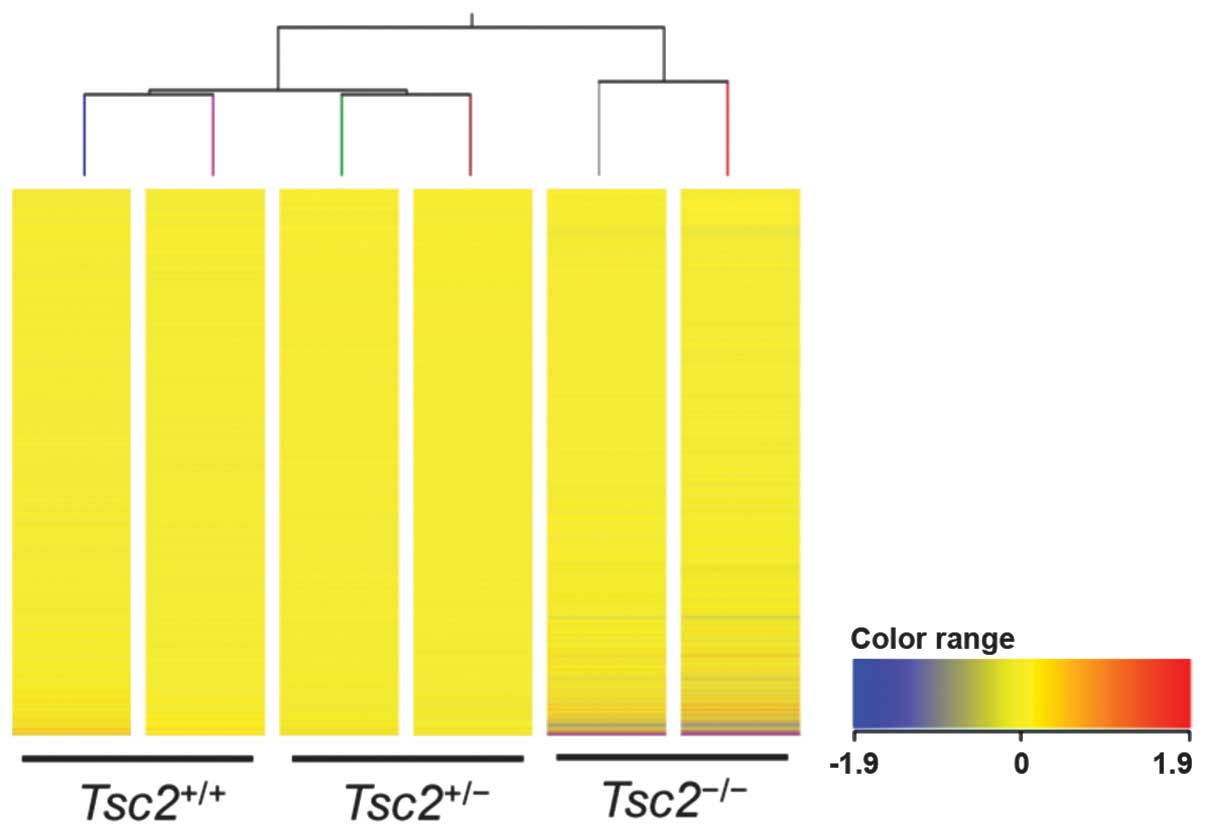
|
1
|
Kwiatkowski DJ, Whittemore VH and Thiele
EA: Tuberous Sclerosis Complex: Genes, Clinical Features, and
Therapeutics. Wiley-VCH Verlag GmbH & Co. KGaA; Weinheim: 2010,
View Article : Google Scholar
|
|
2
|
European Chromosome 16 Tuberous Sclerosis
Consortium. Identification and characterization of the tuberous
sclerosis gene on chromosome 16. Cell. 75:1305–1315. 1993.
View Article : Google Scholar : PubMed/NCBI
|
|
3
|
van Slegtenhorst M, de Hoogt R, Hermans C,
et al: Identification of the tuberous sclerosis gene TSC1 on
chromosome 9q34. Science. 277:805–808. 1997. View Article : Google Scholar : PubMed/NCBI
|
|
4
|
Curatolo P, Bombardieri R and Jozwiak S:
Tuberous sclerosis. Lancet. 372:657–668. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
5
|
Laplante M and Sabatini DM: mTOR signaling
in growth control and disease. Cell. 149:274–293. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
6
|
Knudson AG Jr: Mutation and cancer:
Statistical study of retino-blastoma. Proc Natl Acad Sci USA.
68:820–823. 1971. View Article : Google Scholar
|
|
7
|
Henske EP, Scheithauer BW, Short MP,
Wollmann R, Nahmias J, Hornigold N, van Slegtenhorst M, Welsh CT
and Kwiatkowski DJ: Allelic loss is frequent in tuberous sclerosis
kidney lesions but rare in brain lesions. Am J Hum Genet.
59:400–406. 1996.PubMed/NCBI
|
|
8
|
Au KS, Hebert AA, Roach ES and Northrup H:
Complete inactivation of the TSC2 gene leads to formation of
hamartomas. Am J Hum Genet. 65:1790–1795. 1999. View Article : Google Scholar : PubMed/NCBI
|
|
9
|
Tucker T and Friedman JM: Pathogenesis of
hereditary tumors: Beyond the ‘two-hit’ hypothesis. Clin Genet.
62:345–357. 2002. View Article : Google Scholar : PubMed/NCBI
|
|
10
|
Chan JA, Zhang H, Roberts PS, Jozwiak S,
Wieslawa G, Lewin-Kowalik J, Kotulska K and Kwiatkowski DJ:
Pathogenesis of tuberous sclerosis subependymal giant cell
astrocytomas: Biallelic inactivation of TSC1 or TSC2 leads to mTOR
activation. J Neuropathol Exp Neurol. 63:1236–1242. 2004.PubMed/NCBI
|
|
11
|
Niida Y, Stemmer-Rachamimov AO, Logrip M,
Tapon D, Perez R, Kwiatkowski DJ, Sims K, MacCollin M, Louis DN and
Ramesh V: Survey of somatic mutations in tuberous sclerosis complex
(TSC) hamartomas suggests different genetic mechanisms for
pathogenesis of TSC lesions. Am J Hum Genet. 69:493–503. 2001.
View Article : Google Scholar : PubMed/NCBI
|
|
12
|
Onda H, Lueck A, Marks PW, Warren HB and
Kwiatkowski DJ: Tsc2(+/−) mice develop tumors in multiple sites
that express gelsolin and are influenced by genetic background. J
Clin Invest. 104:687–695. 1999. View Article : Google Scholar : PubMed/NCBI
|
|
13
|
Kobayashi T, Minowa O, Kuno J, Mitani H,
Hino O and Noda T: Renal carcinogenesis, hepatic hemangiomatosis,
and embryonic lethality caused by a germ-line Tsc2 mutation in
mice. Cancer Res. 59:1206–1211. 1999.PubMed/NCBI
|
|
14
|
Kobayashi T, Minowa O, Sugitani Y, Takai
S, Mitani H, Kobayashi E, Noda T and Hino O: A germ-line Tsc1
mutation causes tumor development and embryonic lethality that are
similar, but not identical to, those caused by Tsc2 mutation in
mice. Proc Natl Acad Sci USA. 98:8762–8767. 2001. View Article : Google Scholar : PubMed/NCBI
|
|
15
|
Meikle L, Pollizzi K, Egnor A, Kramvis I,
Lane H, Sahin M and Kwiatkowski DJ: Response of a neuronal model of
tuberous sclerosis to mammalian target of rapamycin (mTOR)
inhibitors: Effects on mTORC1 and Akt signaling lead to improved
survival and function. J Neurosci. 28:5422–5432. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
16
|
Goorden SMI, van Woerden GM, van der Weerd
L, Cheadle JP and Elgersma Y: Cognitive deficits in
Tsc1+/− mice in the absence of cerebral lesions and
seizures. Ann Neurol. 62:648–655. 2007. View Article : Google Scholar : PubMed/NCBI
|
|
17
|
Ehninger D, Han S, Shilyansky C, Zhou Y,
Li W, Kwiatkowski DJ, Ramesh V and Silva AJ: Reversal of learning
deficits in a Tsc2+/− mouse model of tuberous sclerosis.
Nat Med. 14:843–848. 2008. View
Article : Google Scholar : PubMed/NCBI
|
|
18
|
Eker R and Mossige J: A dominant gene for
renal adenomas in the rat. Nature. 189:858–859. 1961. View Article : Google Scholar
|
|
19
|
Hino O, Mitani H and Knudson AG: Genetic
predisposition to transplacentally induced renal cell carcinomas in
the Eker rat. Cancer Res. 53:5856–5858. 1993.PubMed/NCBI
|
|
20
|
Kobayashi T, Hirayama Y, Kobayashi E, Kubo
Y and Hino O: A germline insertion in the tuberous sclerosis (Tsc2)
gene gives rise to the Eker rat model of dominantly inherited
cancer. Nat Genet. 9:70–74. 1995. View Article : Google Scholar : PubMed/NCBI
|
|
21
|
Rennebeck G, Kleymenova EV, Anderson R,
Yeung RS, Artzt K and Walker CL: Loss of function of the tuberous
sclerosis 2 tumor suppressor gene results in embryonic lethality
characterized by disrupted neuroepithelial growth and development.
Proc Natl Acad Sci USA. 95:15629–15634. 1998. View Article : Google Scholar : PubMed/NCBI
|
|
22
|
Mizuguchi M, Takashima S, Yamanouchi H,
Nakazato Y, Mitani H and Hino O: Novel cerebral lesions in the Eker
rat model of tuberous sclerosis: Cortical tuber and anaplastic
ganglioglioma. J Neuropathol Exp Neurol. 59:188–196.
2000.PubMed/NCBI
|
|
23
|
Yeung RS, Katsetos CD and Klein-Szanto A:
Subependymal astrocytic hamartomas in the Eker rat model of
tuberous sclerosis. Am J Pathol. 151:1477–1486. 1997.PubMed/NCBI
|
|
24
|
Takahashi DK, Dinday MT, Barbaro NM and
Baraban SC: Abnormal cortical cells and astrocytomas in the Eker
rat model of tuberous sclerosis complex. Epilepsia. 45:1525–1530.
2004. View Article : Google Scholar : PubMed/NCBI
|
|
25
|
Prabhakar S, Goto J, Zhang X, et al:
Stochastic model of Tsc1 lesions in mouse brain. PLoS One.
8:e642242013. View Article : Google Scholar : PubMed/NCBI
|
|
26
|
Kobayashi T, Adachi H, Mitani H, Hirayama
Y and Hino O: Toward chemotherapy for Tsc2-mutant renal tumor. Proc
Jpn Acad. 79:22–25. 2003. View Article : Google Scholar
|
|
27
|
Kenerson HL, Aicher LD, True LD and Yeung
RS: Activated mammalian target of rapamycin pathway in the
pathogenesis of tuberous sclerosis complex renal tumors. Cancer
Res. 62:5645–5650. 2002.PubMed/NCBI
|
|
28
|
Lee L, Sudentas P, Donohue B, et al:
Efficacy of a rapamycin analog (CCI-779) and IFN-γ in tuberous
sclerosis mouse models. Genes Chromosomes Cancer. 42:213–227. 2005.
View Article : Google Scholar
|
|
29
|
McCormack FX, Inoue Y, Moss J, et al:
National Institutes of Health Rare Lung Diseases Consortium; MILES
Trial Group: Efficacy and safety of sirolimus in
lymphangioleiomyomatosis. N Engl J Med. 364:1595–1606. 2011.
View Article : Google Scholar : PubMed/NCBI
|
|
30
|
Franz DN, Belousova E, Sparagana S, et al:
Efficacy and safety of everolimus for subependymal giant cell
astrocytomas associated with tuberous sclerosis complex (EXIST-1):
A multicentre, randomised, placebo-controlled phase 3 trial.
Lancet. 381:125–132. 2013. View Article : Google Scholar
|
|
31
|
Bissler JJ, Kingswood JC, Radzikowska E,
et al: Everolimus for angiomyolipoma associated with tuberous
sclerosis complex or sporadic lymphangioleiomyomatosis (EXIST-2): A
multicentre, randomised, double-blind, placebo-controlled trial.
Lancet. 381:817–824. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
32
|
Mizuguchi M: Abnormal giant cells in the
cerebral lesions of tuberous sclerosis complex. Congenit Anom
(Kyoto). 47:2–8. 2007. View Article : Google Scholar
|
|
33
|
Buehr M, Meek S, Blair K, Yang J, Ure J,
Silva J, McLay R, Hall J, Ying QL and Smith A: Capture of authentic
embryonic stem cells from rat blastocysts. Cell. 135:1287–1298.
2008. View Article : Google Scholar : PubMed/NCBI
|
|
34
|
Li P, Tong C, Mehrian-Shai R, et al:
Germline competent embryonic stem cells derived from rat
blastocysts. Cell. 135:1299–1310. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
35
|
Evans MJ and Kaufman MH: Establishment in
culture of pluripotential cells from mouse embryos. Nature.
292:154–156. 1981. View Article : Google Scholar : PubMed/NCBI
|
|
36
|
Martin GR: Isolation of a pluripotent cell
line from early mouse embryos cultured in medium conditioned by
teratocarcinoma stem cells. Proc Natl Acad Sci USA. 78:7634–7638.
1981. View Article : Google Scholar : PubMed/NCBI
|
|
37
|
Thomson JA, Itskovitz-Eldor J, Shapiro SS,
Waknitz MA, Swiergiel JJ, Marshall VS and Jones JM: Embryonic stem
cell lines derived from human blastocysts. Science. 282:1145–1147.
1998. View Article : Google Scholar : PubMed/NCBI
|
|
38
|
Easley CA IV, Ben-Yehudah A, Redinger CJ,
Oliver SL, Varum ST, Eisinger VM, Carlisle DL, Donovan PJ and
Schatten GP: mTOR-mediated activation of p70 S6K induces
differentiation of pluripotent human embryonic stem cells. Cell
Reprogram. 12:263–273. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
39
|
Gan B, Sahin E, Jiang S, Sanchez-Aguilera
A, Scott KL, Chin L, Williams DA, Kwiatkowski DJ and DePinho RA:
mTORC1-dependent and -independent regulation of stem cell renewal,
differentiation, and mobilization. Proc Natl Acad Sci USA.
105:19384–19389. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
40
|
He J, Kang L, Wu T, et al: An elaborate
regulation of Mammalian target of rapamycin activity is required
for somatic cell reprogramming induced by defined transcription
factors. Stem Cells Dev. 21:2630–2641. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
41
|
Shiono M, Kobayashi T, Takahashi R, et al:
The G1556S-type tuberin variant suppresses tumor formation in
tuberous sclerosis 2 mutant (Eker) rats despite its deficiency in
mTOR inhibition. Oncogene. 27:6690–6697. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
42
|
Benvenuto G, Li S, Brown SJ, Braverman R,
Vass WC, Cheadle JP, Halley DJ, Sampson JR, Wienecke R and DeClue
JE: The tuberous sclerosis-1 (TSC1) gene product hamartin
suppresses cell growth and augments the expression of the TSC2
product tuberin by inhibiting its ubiquitination. Oncogene.
19:6306–6316. 2000. View Article : Google Scholar
|
|
43
|
Betschinger J, Nichols J, Dietmann S,
Corrin PD, Paddison PJ and Smith A: Exit from pluripotency is gated
by intracellular redistribution of the bHLH transcription factor
Tfe3. Cell. 153:335–347. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
44
|
Li S, Thangapazham RL, Wang JA, Rajesh S,
Kao TC, Sperling L, Moss J and Darling TN: Human TSC2-null
fibroblast-like cells induce hair follicle neogenesis and hamartoma
morphogenesis. Nat Commun. 2:2352011. View Article : Google Scholar : PubMed/NCBI
|
|
45
|
Ebert AD, Yu J, Rose FF Jr, Mattis VB,
Lorson CL, Thomson JA and Svendsen CN: Induced pluripotent stem
cells from a spinal muscular atrophy patient. Nature. 457:277–280.
2009. View Article : Google Scholar :
|
|
46
|
Lee G, Papapetrou EP, Kim H, et al:
Modelling pathogenesis and treatment of familial dysautonomia using
patient-specific iPSCs. Nature. 461:402–406. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
47
|
Yang YM, Gupta SK, Kim KJ, et al: A small
molecule screen in stem-cell-derived motor neurons identifies a
kinase inhibitor as a candidate therapeutic for ALS. Cell Stem
Cell. 12:713–726. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
48
|
Williams BO, Schmitt EM, Remington L,
Bronson RT, Albert DM, Weinberg RA and Jacks T: Extensive
contribution of Rb-deficient cells to adult chimeric mice with
limited histopathological consequences. EMBO J. 13:4251–4259.
1994.PubMed/NCBI
|
|
49
|
Kawamata M and Ochiya T: Two distinct
knockout approaches highlight a critical role for p53 in rat
development. Sci Rep. 2:9452012. View Article : Google Scholar : PubMed/NCBI
|
|
50
|
Kielman MF, Rindapää M, Gaspar C, van
Poppel N, Breukel C, van Leeuwen S, Taketo MM, Roberts S, Smits R
and Fodde R: Apc modulates embryonic stem-cell differentiation by
controlling the dosage of beta-catenin signaling. Nat Genet.
32:594–605. 2002. View
Article : Google Scholar : PubMed/NCBI
|