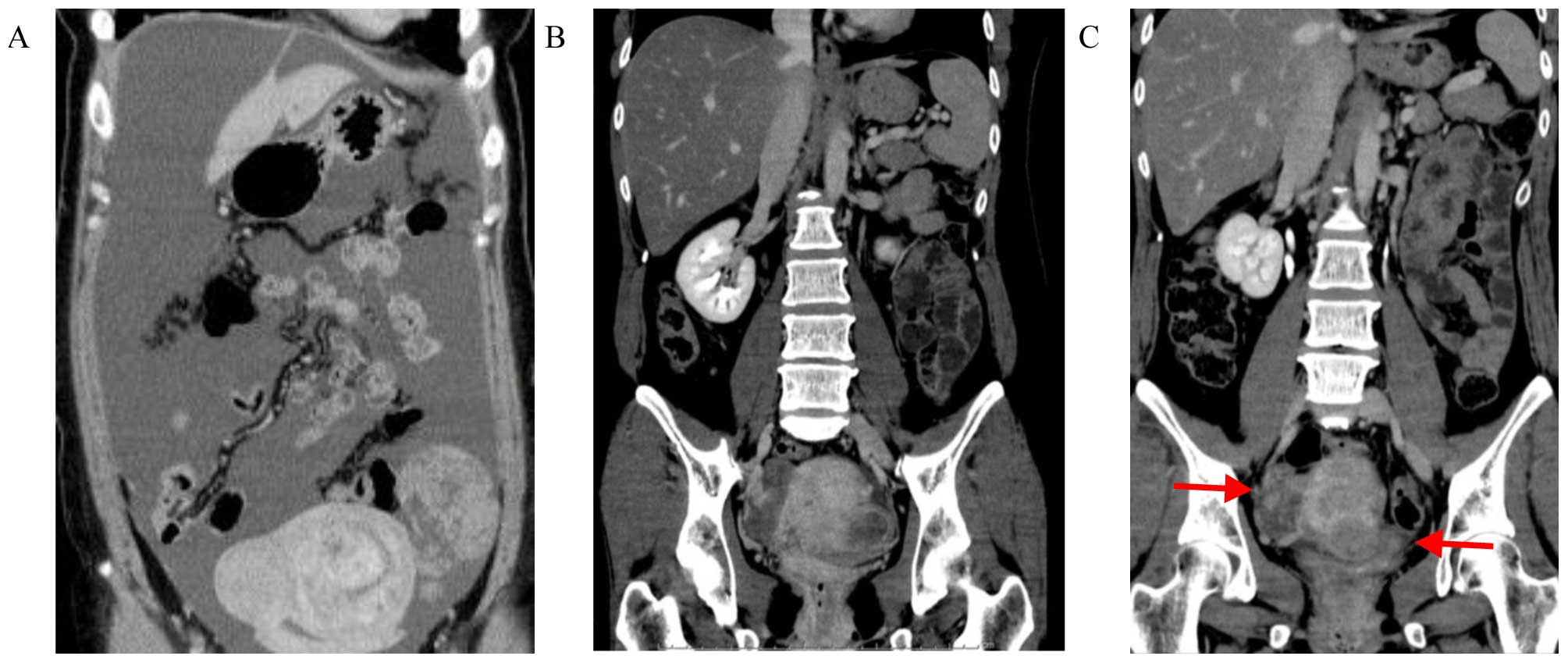
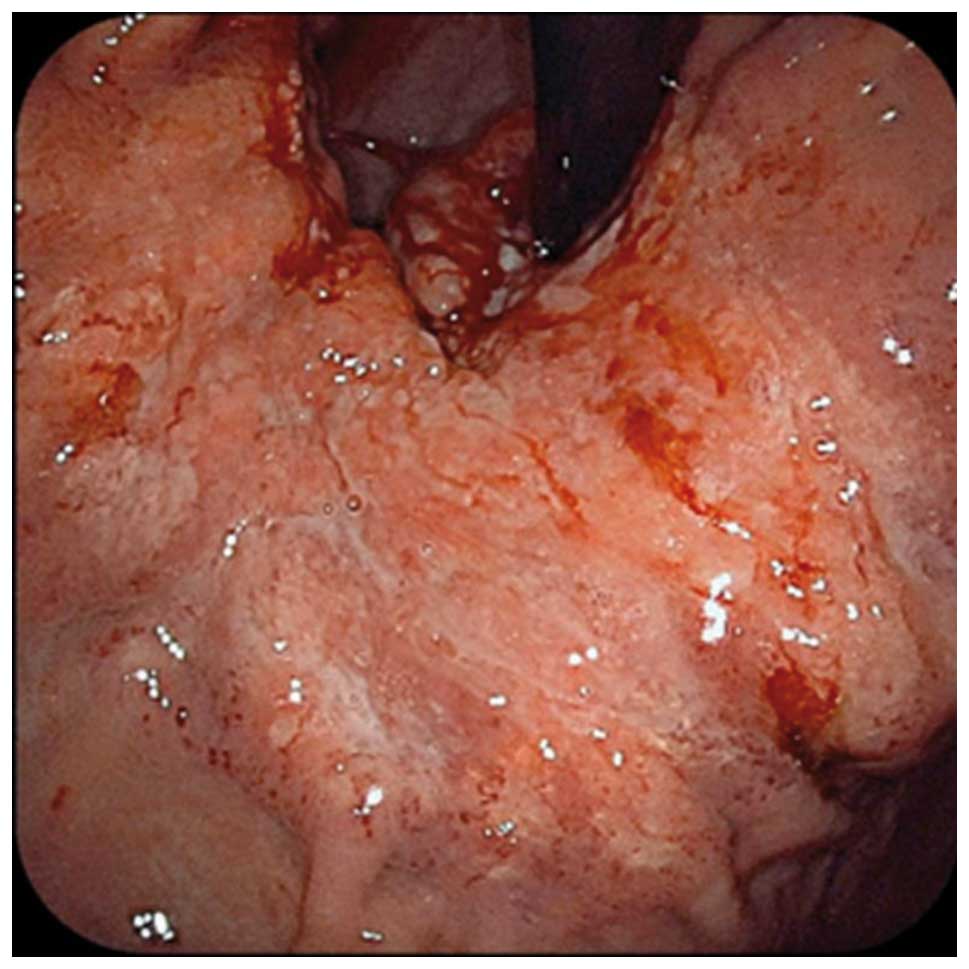
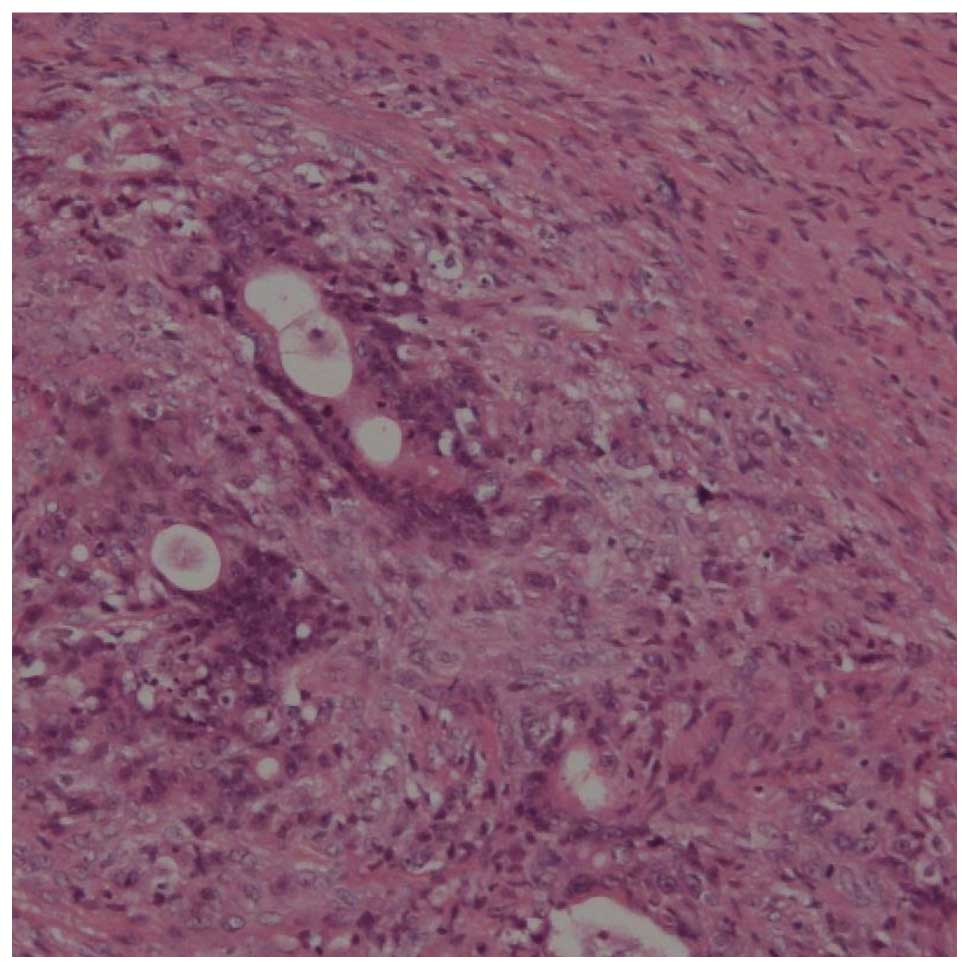
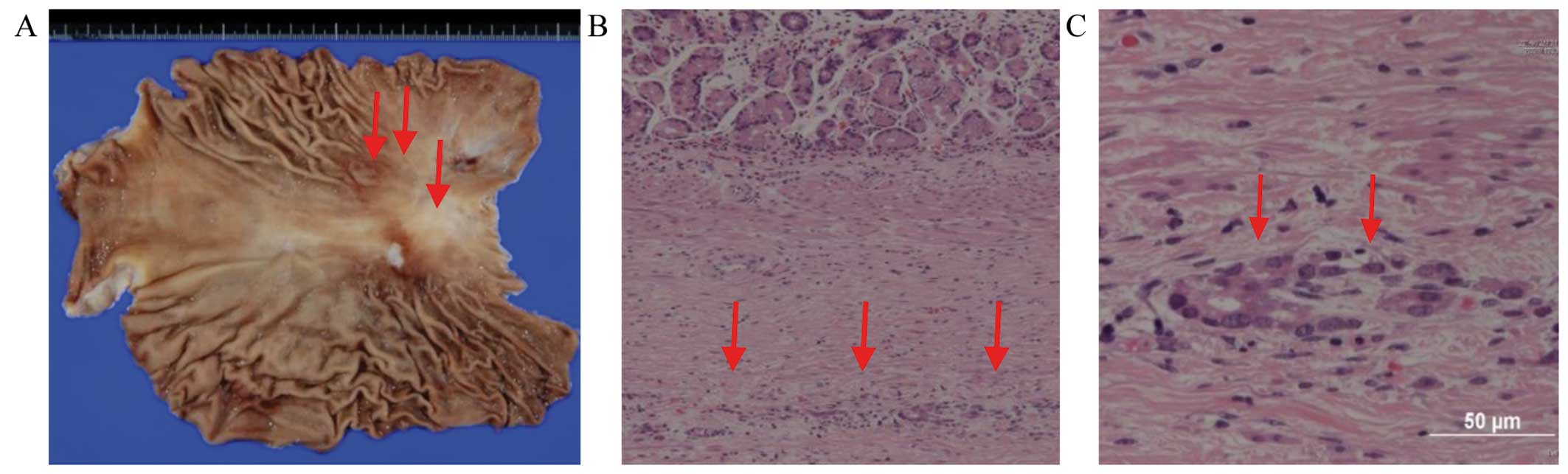
|
1
|
Sadeghi B, Arvieux C, Glehen O, Beaujard
AC, Rivoire M, Baulieux J, Fontaumard E, Brachet A, Caillot JL,
Faure JL, et al: Peritoneal carcinomatosis from non-gynecologic
malignancies: Results of the EVOCAPE 1 multicentric prospective
study. Cancer. 88:358–363. 2000. View Article : Google Scholar : PubMed/NCBI
|
|
2
|
Koizumi W, Narahara H, Hara T, Takagane A,
Akiya T, Takagi M, Miyashita K, Nishizaki T, Kobayashi O, Takiyama
W, et al: S-1 plus cisplatin versus S-1 alone for first-line
treatment of advanced gastric cancer (SPIRITS trial): A phase III
trial. Lancet Oncol. 9:215–221. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
3
|
Yamaguchi H, Kitayama J, Ishigami H, Emoto
S, Yamashita H and Watanabe T: A phase 2 trial of intravenous and
intraperitoneal paclitaxel combined with S-1 for treatment of
gastric cancer with macroscopic peritoneal metastasis. Cancer.
119:3354–3358. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
4
|
Kurokawa Y, Sugimoto N, Miwa H, Tsuda M,
Nishina S, Okuda H, Imamura H, Gamoh M, Sakai D, Shimokawa T, et
al: Phase II study of trastuzumab in combination with S-1 plus
cisplatin in HER2-positive gastric cancer (HERBIS-1). Br J Cancer.
110:1163–1168. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
5
|
Japanese Gastric Cancer Association, .
Japanese classification of gastric carcinoma: 3rd English edition.
Gastric Cancer. 14:101–112. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
6
|
Yamada Y, Shirao K, Ohtsu A, Boku N, Hyodo
I, Saitoh H, Miyata Y and Taguchi T: Phase II trial of paclitaxel
by three-hour infusion for advanced gastric cancer with short
premedication for prophylaxis against paclitaxel-associated
hypersensitivity reactions. Ann Oncol. 12:1133–1137. 2001.
View Article : Google Scholar : PubMed/NCBI
|
|
7
|
Markman M, Rowinsky E, Hakes T, Reichman
B, Jones W, Lewis JL Jr, Rubin S, Curtin J, Barakat R, Phillips M,
et al: Phase I trial of intraperitoneal taxol: A Gynecoloic
Oncology Group study. J Clin Oncol. 10:1485–1491. 1992.PubMed/NCBI
|
|
8
|
Gelderblom H, Verweij J, van Zomeren DM,
Buijs D, Ouwens L, Nooter K, Stoter G and Sparreboom A: Influence
of Cremophor El on the bioavailability of intraperitoneal
paclitaxel. Clin Cancer Res. 8:1237–1241. 2002.PubMed/NCBI
|
|
9
|
Armstrong DK, Bundy B, Wenzel L, Huang HQ,
Baergen R, Lele S, Copeland LJ, Walker JL and Burger RA:
Gynecologic Oncology Group: Intraperitoneal cisplatin and
paclitaxel in ovarian cancer. N Engl J Med. 354:34–43. 2006.
View Article : Google Scholar : PubMed/NCBI
|
|
10
|
Boku N, Yamamoto S, Fukuda H, Shirao K,
Doi T, Sawaki A, Koizumi W, Saito H, Yamaguchi K, Takiuchi H, et
al: Gastrointestinal Oncology Study Group of the Japan Clinical
Oncology Group: Fluorouracil versus combination of irinotecan plus
cisplatin versus S-1 in metastatic gastric cancer: A randomised
phase 3 study. Lancet Oncol. 10:1063–1069. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
11
|
Bang YJ, Van Cutsem E, Feyereislova A,
Chung HC, Shen L, Sawaki A, Lordick F, Ohtsu A, Omuro Y, Satoh T,
et al: ToGA Trial Investigators: Trastuzumab in combination with
chemotherapy versus chemotherapy alone for treatment of
HER2-positive advanced gastric or gastro-oesophageal junction
cancer (ToGA): A phase 3, open-label, randomised controlled trial.
Lancet. 376:687–697. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
12
|
Power DG and Kemeny NE: Chemotherapy for
the conversion of unresectable colorectal cancer liver metastases
to resection. Crit Rev Oncol Hematol. 79:251–264. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
13
|
Yoshida K, Yamaguchi K, Okumura N,
Tanahashi T and Kodera Y: Is conversion therapy possible in stage
IV gastric cancer: The proposal of new biological categories of
classification. Gastric Cancer. 19:329–338. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
14
|
Kitayama J, Ishigami H, Yamaguchi H,
Yamashita H, Emoto S, Kaisaki S and Watanabe T: Salvage gastrectomy
after intravenous and intraperitoneal paclitaxel (PTX)
administration with oral S-1 for peritoneal dissemination of
advanced gastric cancer with malignant ascites. Ann Surg Oncol.
21:539–546. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
15
|
Kim KH, Lee KW, Baek SK, Chang HJ, Kim YJ,
Park DJ, Kim JH, Kim HH and Lee JS: Survival benefit of gastrectomy
± metastasectomy in patients with metastatic gastric cancer
receiving chemotherapy. Gastric Cancer. 14:130–138. 2011.
View Article : Google Scholar : PubMed/NCBI
|
|
16
|
Peng W, Hua RX, Jiang R, Ren C, Jia YN, Li
J and Guo WJ: Surgical treatment for patients with Krukenberg tumor
of stomach origin: Clinical outcome and prognostic factors
analysis. PLoS One. 8:e682272013. View Article : Google Scholar : PubMed/NCBI
|
|
17
|
Eveno C, Goéré D, Dartigues P, Honoré C,
Dumont F, Tzanis D, Benhaim L, Malka D and Elias D: Ovarian
metastasis is associated with retroperitoneal lymph node relapses
in women treated for colorectal peritoneal carcinomatosis. Ann Surg
Oncol. 20:491–496. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
18
|
Soma D, Kitayama J, Konno T, Ishihara K,
Yamada J, Kamei T, Ishigami H, Kaisaki S and Nagawa H:
Intraperitoneal administration of paclitaxel solubilized with
poly(2-methacryloxyethyl phosphorylcholine-co n-butyl methacrylate)
for peritoneal dissemination of gastric cancer. Cancer Sci.
100:1979–1985. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
19
|
Kamei T, Kitayama J, Yamaguchi H, Soma D,
Emoto S, Konno T, Ishihara K, Ishigami H, Kaisaki S and Nagawa H:
Spatial distribution of intraperitoneally administrated paclitaxel
nanoparticles solubilized with poly (2-methacryloxyethyl
phosphorylcholine-co n-butyl methacrylate) in peritoneal metastatic
nodules. Cancer Sci. 102:200–205. 2011. View Article : Google Scholar : PubMed/NCBI
|