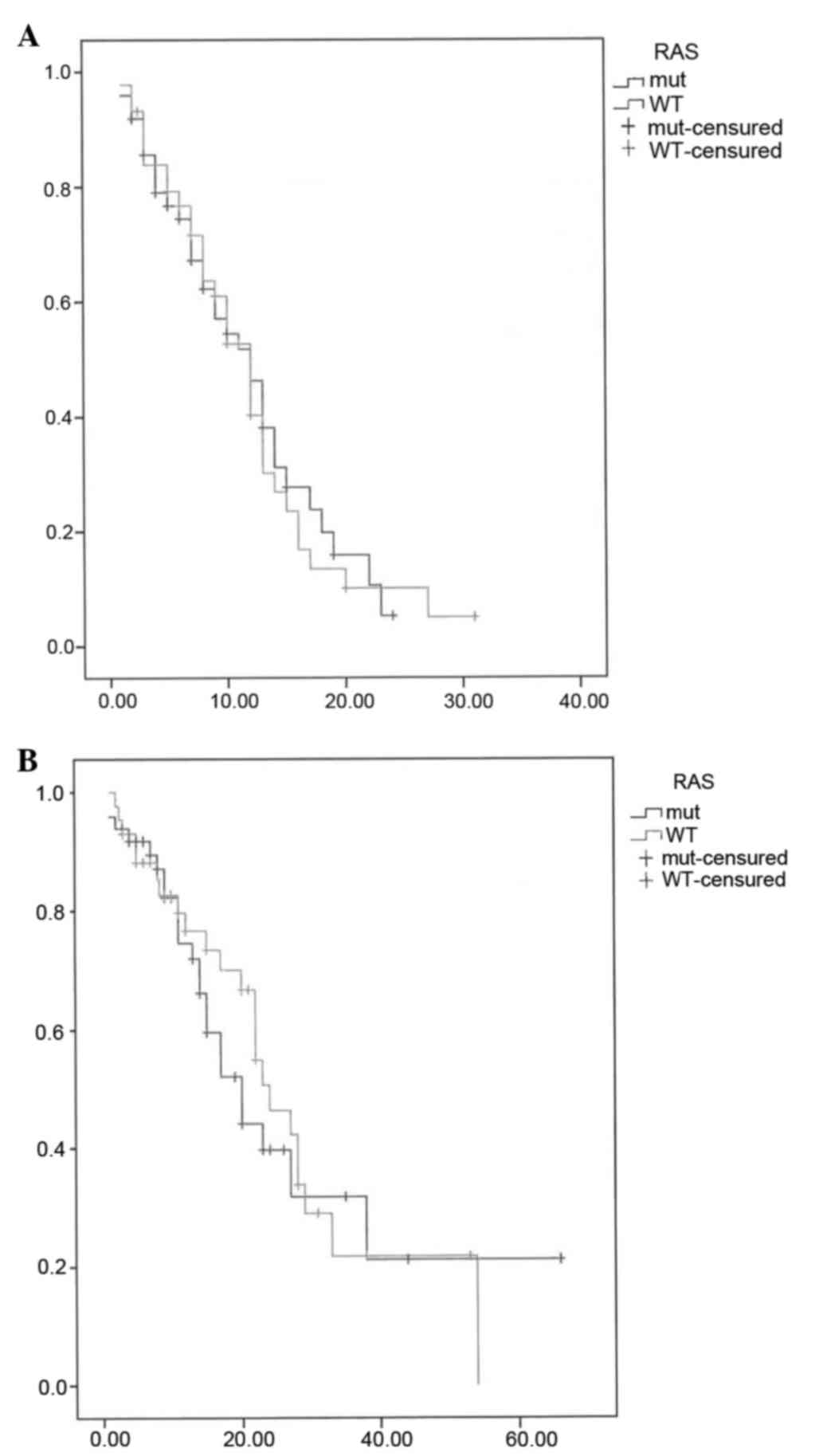
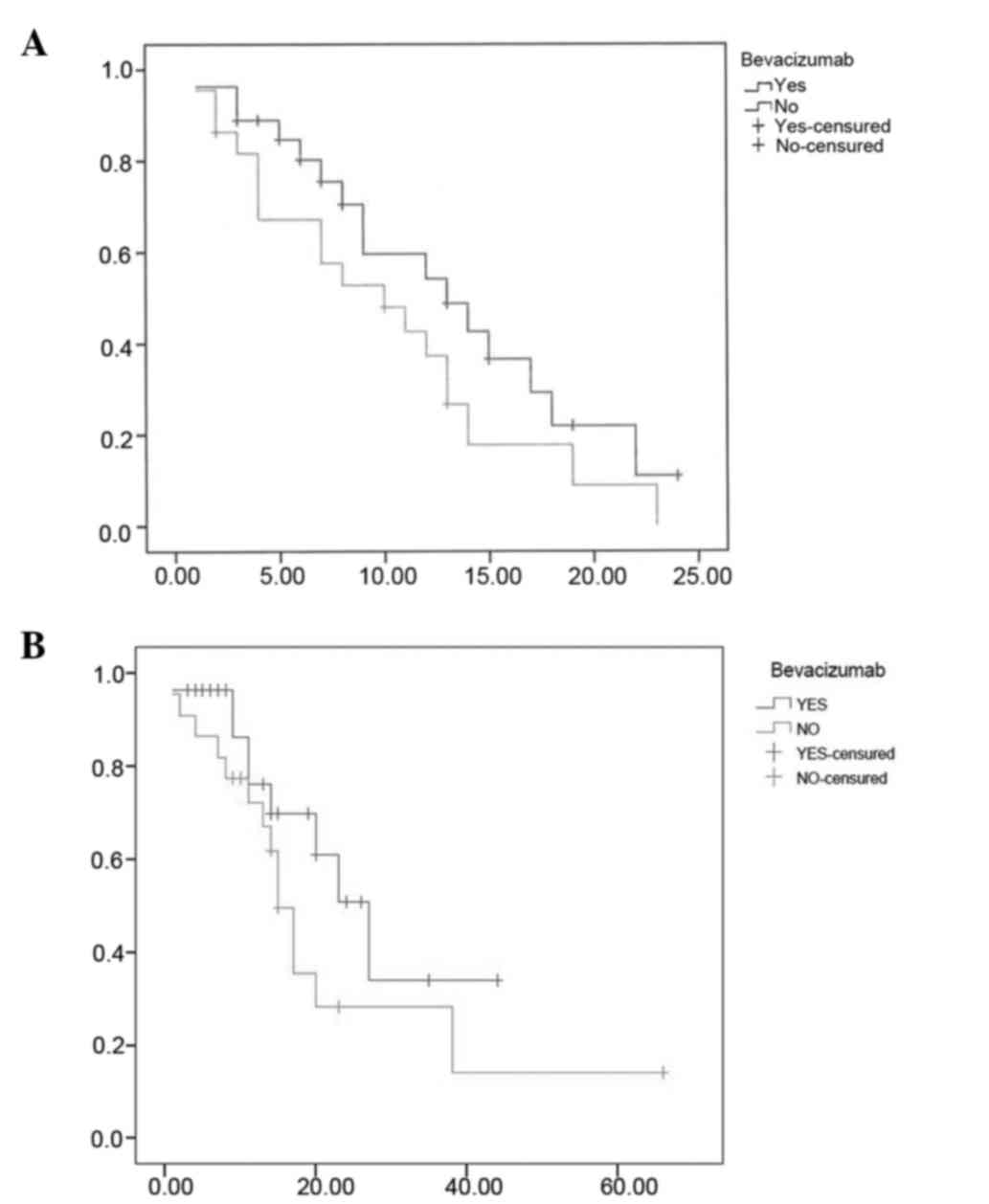
|
1
|
National Cancer Institute, . Surveillance,
Epidemiology and End Results Program. https://seer.cancer.gov/Accessed. May;2013.
|
|
2
|
Edge SB, Byrd DR, Compton CC, Fritz AG,
Greene FL and Trotti A: AJCC cancer staging manual. 7th. Springer;
New York, NY: pp. 1432010
|
|
3
|
Siegel R, Naishadham D and Jemal A: Cancer
statistics, 2012. CA Cancer J Clin. 62:10–29. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
4
|
Bleiberg H: Role of chemotherapy for
advanced colorrectal cancer. New oportunities. Semin Oncol.
23:(Suppl 3). 42–50. 1996.PubMed/NCBI
|
|
5
|
de Gramont A, Louvet C, André T,
Tournigand C and Krulik M: A review of GERCOD trials of bimonthly
leucovorin plus 5-fluorouracil 48-h continuous infusion in advanced
colorectal cancer: Evolution of a regimen. Groupe d'Etude et de
Recherche sur les Cancers de l'Ovaire et Digestifs (GERCOD). Eur J
Cancer. 34:619–626. 1998. View Article : Google Scholar : PubMed/NCBI
|
|
6
|
Capdevila J, Elez E, Peralta S, Macarulla
T, Ramos FJ and Tabernero J: Oxaliplatin-based chemotherapy in the
management of colorectal cancer. Expert Rev Anticancer Ther.
8:1223–1236. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
7
|
McCormack PL and Keam SJ: Bevacizumab: A
review of its use in metastatic colorectal cancer. Drugs.
68:487–506. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
8
|
Blick SK and Scott LJ: Cetuximab: A review
of its use in squamous cell carcinoma of the head and neck and
metastatic colorectal cancer. Drugs. 67:2585–2607. 2007. View Article : Google Scholar : PubMed/NCBI
|
|
9
|
Amado RG, Wolf M, Peeters M, Van Cutsem E,
Siena S, Freeman DJ, Juan T, Sikorski R, Suggs S, Radinsky R, et
al: Wild-type KRAS is required for panitumumab efficacy in patients
with metastatic colorectal cancer. J Clin Oncol. 26:1626–1634.
2008. View Article : Google Scholar : PubMed/NCBI
|
|
10
|
Karapetis CS, Khambata-Ford S, Jonker DJ,
O'Callaghan CJ, Tu D, Tebbutt NC, Simes RJ, Chalchal H, Shapiro JD,
Robitaille S, et al: K-ras mutations and benefit from cetuximab in
advanced colorectal cancer. N Engl J Med. 359:1757–1765. 2008.
View Article : Google Scholar : PubMed/NCBI
|
|
11
|
Schubbert S, Shannon K and Bollag G:
Hyperactive Ras in developmental disorders and cancer. Nat Rev
Cancer. 7:295–308. 2007. View
Article : Google Scholar : PubMed/NCBI
|
|
12
|
Irahara N, Baba Y, Nosho K, Shima K, Yan
L, Dias-Santagata D, Iafrate AJ, Fuchs CS, Haigis KM and Ogino S:
NRAS mutations are rare in colorrectal cancer. Diagn Mol Pathol.
19:157–163. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
13
|
De Roock W, Claes B, Bernasconi D, De
Schutter J, Biesmans B, Fountzilas G, Kalogeras KT, Kotoula V,
Papamichael D, Laurent-Puig P, et al: Effects of KRAS, BRAF, NRAS,
and PIK3Ca mutations on the efficacy of cetuximab plus chemotherapy
in chemotherapy-refractory metastatic colorrectal cancer: A
retrospective consortium analisis. Lancet Oncol. 11:753–762. 2010.
View Article : Google Scholar : PubMed/NCBI
|
|
14
|
Chin L, Tam A, Pomerantz J, Wong M, Holash
J, Bardeesy N, Shen Q, O'Hagan R, Pantginis J, Zhou H, et al:
Essential role for oncogenic Ras in tumour maintenance. Nature.
400:468–472. 1999. View
Article : Google Scholar : PubMed/NCBI
|
|
15
|
Rak J, Mitsuhashi Y, Bayko L, Filmus J,
Shirasawa S, Sasazuki T and Kerbel RS: Mutant ras oncogenes
upregulate VEGF/VPF expression: Implications for induction and
inhibition of tumor angiogenesis. Cancer Res. 55:4575–4580.
1995.PubMed/NCBI
|
|
16
|
Watnick RS, Cheng YN, Rangarajan A, Ince
TA and Weinberg RA: Ras modulates Myc activity to repress
thrombospondin-1 expression and increase tumor angiogenesis. Cancer
Cell. 3:219–231. 2003. View Article : Google Scholar : PubMed/NCBI
|
|
17
|
Klump B, Nehls O, Okech T, Hsieh CJ, Gaco
V, Gittinger FS, Sarbia M, Borchard F, Greschniok A, Gruenagel HH,
et al: Molecular lesions in colorectal cancer: Impact on prognosis?
Original data and review of the literature. Int J Colorectal Dis.
19:23–42. 2004. View Article : Google Scholar : PubMed/NCBI
|
|
18
|
Roth AD, Tejpar S, Delorenzi M, Yan P,
Fiocca R, Klingbiel D, Dietrich D, Biesmans B, Bodoky G, Barone C,
et al: Prognostic role of KRAS and BRAF in stage II and III
resected colon cancer: Results of the translational study on the
PETACC-3, EORTC 40993, SAKK 60-00 trial. J Clin Oncol. 28:466–474.
2010. View Article : Google Scholar : PubMed/NCBI
|
|
19
|
Samowitz WS, Curtin K, Schaffer D,
Robertson M, Leppert M and Slattery ML: Relationship of Ki-ras
mutations in colon cancers to tumor location, stage, and survival:
A population-based study. Cancer Epidemiol Biomarkers Prev.
9:1193–1197. 2000.PubMed/NCBI
|
|
20
|
Ince WL, Jubb AM, Holden SN, Holmgren EB,
Tobin P, Sridhar M, Hurwitz HI, Kabbinavar F, Novotny WF, Hillan KJ
and Koeppen H: Association of k-ras, b-raf, and p53 status with the
treatment effect of bevacizumab. J Natl Cancer Inst. 97:981–989.
2005. View Article : Google Scholar : PubMed/NCBI
|
|
21
|
Eisenhauer EA, Therasse P, Bogaerts J,
Schwartz LH, Sargent D, Ford R, Dancey J, Arbuck S, Gwyther S,
Mooney M, et al: New response evaluation criteria in solid tumours:
Revised RECIST guideline (version 1.1). Eur J Cancer. 45:228–247.
2009. View Article : Google Scholar : PubMed/NCBI
|
|
22
|
Doudillard JY, Oliner KS, Siena S,
Tabernero J, Burkes R, Barugel M, Humblet Y, Bodoky G, Cunningham
D, Jassem J, et al: Panitumumab-FOLFOX4 treatment and RAS mutations
in colorrectal cancer. N Engl J Med. 369:1023–1034. 2013.
View Article : Google Scholar : PubMed/NCBI
|
|
23
|
González-Aguilera JJ, Oliart S, Azcoita MM
and Fernández-Peralta AM: Simultaneous mutations in K-ras and TP53
are indicative of poor prognosis in sporadic colorectal cancer. Am
J Clin Oncol. 27:39–45. 2004. View Article : Google Scholar : PubMed/NCBI
|
|
24
|
Westra JL, Schaapveld M, Hollema H, de
Boer JP, Kraak MM, de Jong D, ter Elst A, Mulder NH, Buys CH,
Hofstra RM and Plukker JT: Determination of TP53 mutation is more
relevant than microsatellite instability status for the prediction
of disease-free survival in adjuvant-treated stage III colon cancer
patients. J Clin Oncol. 23:5635–5643. 2005. View Article : Google Scholar : PubMed/NCBI
|
|
25
|
Kim ST, Park KH, Kim JS, Shin SW and Kim
YH: Impact of KRAS mutation status on otucome in metastatic colon
cancer patients without anti-epidermal growth factor receptor
therapy. Cancer Res Treat. 45:55–62. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
26
|
Andreyev HJ, Norman AR, Cunningham D,
Oates JR and Clarke PA: Kirsten ras mutations in patients with
colorectal cancer: The multicenter ‘RASCAL’ study. J Natl Cancer
Inst. 90:675–684. 1998. View Article : Google Scholar : PubMed/NCBI
|
|
27
|
Andreyev HJ, Norman AR, Cunningham D,
Oates J, Dix BR, Iacopetta BJ, Young J, Walsh T, Ward R, Hawkins N,
et al: Kirsten ras mutations in patients with colorectal cancer:
The ‘RASCAL II’ study. Br J Cancer. 85:692–696. 2001. View Article : Google Scholar : PubMed/NCBI
|
|
28
|
Hurwitz H, Fehrenbacher L, Novotny W,
Cartwright T, Hainsworth J, Heim W, Berlin J, Baron A, Griffing S,
Holmgren E, et al: Bevacizumab plus irinotecan, fluorouracil, and
leucovorin for metastatic colorectal cancer. N Engl J Med.
350:2335–2342. 2004. View Article : Google Scholar : PubMed/NCBI
|
|
29
|
Price TJ, Hardingham JE, Lee CK,
Weickhardt A, Townsend AR, Wrin JW, Chua A, Shivasami A, Cummins
MM, Murone C and Tebbutt NC: Impact of KRAS and BRAF gene mutation
status on outcomes from the Phase III AGITG MAX Trial of
capecitabine alone or in Combination with bevacizumab and mitomycin
in advanced colorectal cancer. J Clin Oncol. 29:2675–2682. 2011.
View Article : Google Scholar : PubMed/NCBI
|
|
30
|
Kim ST, Park KH, Shin SW and Kim YH: Does
KRAS mutation status affect on the effect of VEGF therapy in
metastatic colon cancer patients? Cancer Res Treat. 46:48–54. 2014.
View Article : Google Scholar : PubMed/NCBI
|