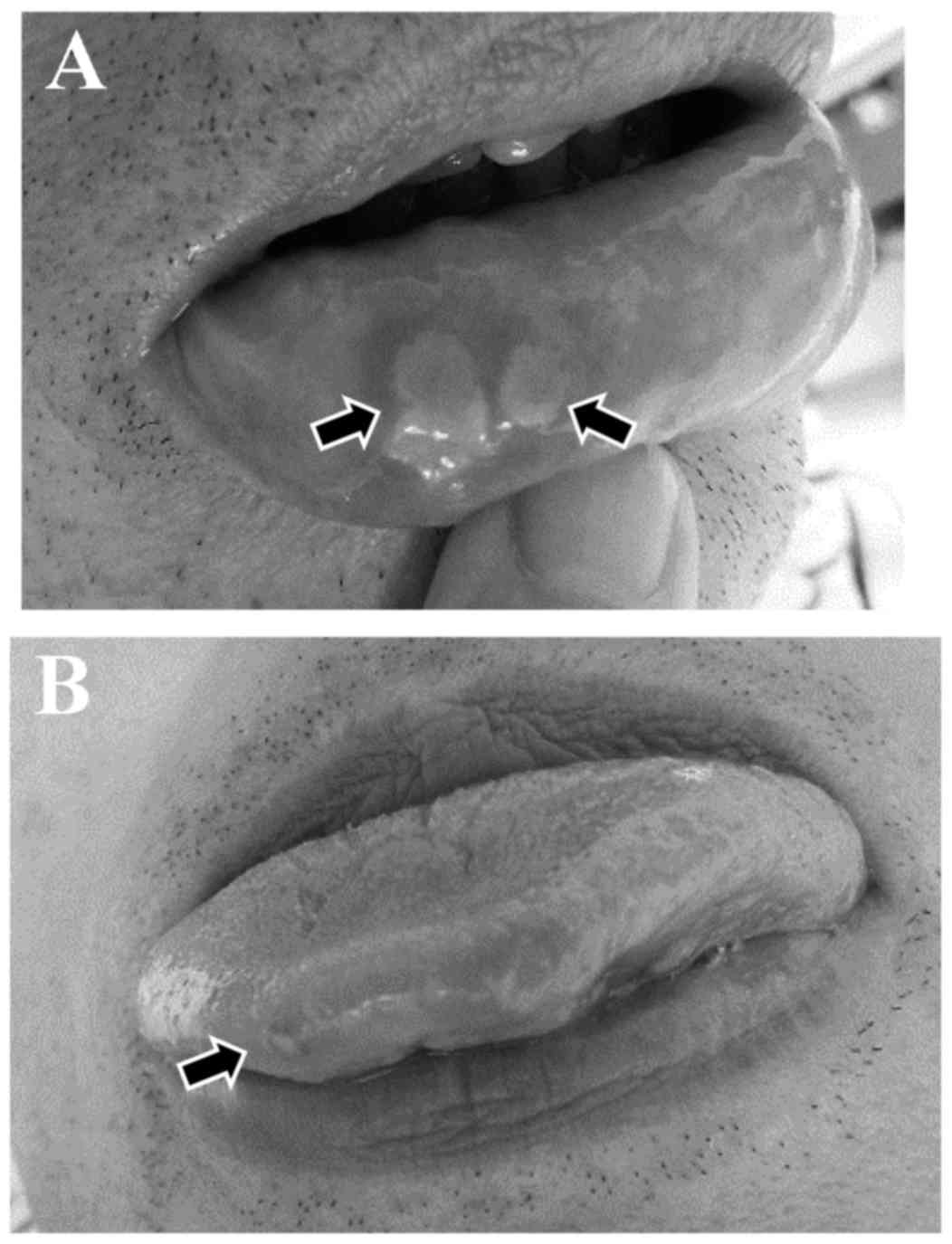
|
1
|
Califano R, Tariq N, Compton S, Fitzgerald
DA, Harwood CA, Lal R, Lester J, McPhelim J, Mulatero C,
Subramanian S, et al: Expert Consensus on the Management of Adverse
Events from EGFR Tyrosine Kinase Inhibitors in the UK. Drugs.
75:1335–1348. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
2
|
Sequist LV, Yang JC, Yamamoto N, O'Byrne
K, Hirsh V, Mok T, Geater SL, Orlov S, Tsai CM, Boyer M, et al:
Phase III study of afatinib or cisplatin plus pemetrexed in
patients with metastatic lung adenocarcinoma with EGFR mutations. J
Clin Oncol. 31:3327–3334. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
3
|
Ge ZD, Auchampach JA, Piper GM and Gross
GJ: Comparison of cardioprotective efficacy of two thromboxane A2
receptor antagonists. J Cardiovasc Pharmacol. 41:481–488. 2003.
View Article : Google Scholar : PubMed/NCBI
|
|
4
|
Rekka E, Chrysselis M, Siskou I and
Kourounakis A: Synthesis of new azulene derivatives and study of
their effect on lipid peroxidation and lipoxygenase activity. Chem
Pharm Bull. 50:904–907. 2002. View Article : Google Scholar : PubMed/NCBI
|
|
5
|
Akagi M, Matsui N, Mochizuki S and Tasaka
K: Inhibitory effect of egualen sodium: A new stable derivative of
azulene on histamine release from mast cell-like cells in the
stomach. Pharmacology. 63:203–209. 2001. View Article : Google Scholar : PubMed/NCBI
|
|
6
|
Kourounakis AP, Rekka EA and Kourounakis
PN: Antioxidant activity of guaiazulene and protection against
paracetamol hepatotoxicity in rats. J Pharm Pharmacol. 49:938–942.
1997. View Article : Google Scholar : PubMed/NCBI
|
|
7
|
Yano S, Horie S, Wakabayashi S, Mochizuki
S, Tomiyama A and Watanabe K: Increasing effect of sodium
3-ethyl-7-isopropyl-1-azulenesulfonate 1/3 hydrate (KT1-32), a
novel antiulcer agent, on gastric mucosal blood flow in
anesthetized. Res Commun Chem Pathol Pharmacol. 70:253–256.
1990.PubMed/NCBI
|
|
8
|
Okabe S, Takeuchi K, Mori Y, Furukawa O
and Yamada Y: [Effects of KT1-32 on acute gastric lesions and
duodenal ulcers induced in rats]. Nippon Yakurigaku Zasshi.
88:467–476. 1986.(in Japanese). View Article : Google Scholar : PubMed/NCBI
|
|
9
|
Seki J, Mukai H and Sugiyama M: Studies on
the absorption of sodium guaiazulene-3-sulfonate. II. Absorption
mechanism from nasal and intestinal membrane. J Pharmacobiodyn.
8:337–343. 1985. View Article : Google Scholar : PubMed/NCBI
|
|
10
|
Hanzlik RP and Bhatia P: Metabolism of
azulene in rats. Xenobiotica. 11:779–783. 1981. View Article : Google Scholar : PubMed/NCBI
|
|
11
|
Matsuda C, Munemoto Y, Mishima H, Nagata
N, Oshiro M, Kataoka M, Sakamoto J, Aoyama T, Morita S and Kono T:
Double-blind, placebo-controlled, randomized phase II study of
TJ-14 (Hangeshashinto) for infusional fluorinated-pyrimidine-based
colorectal cancer chemotherapy-induced oral mucositis. Cancer
Chemother Pharmacol. 76:97–103. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
12
|
Yamashita T, Araki K, Tomifuji M, Kamide
D, Tanaka Y and Shiotani A: A traditional Japanese
medicine-Hangeshashinto (TJ-14)-alleviates chemoradiation-induced
mucositis and improves rates of treatment completion. Support Care
Cancer. 23:29–35. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
13
|
Aoyama T, Nishikawa K, Takiguchi N, Tanabe
K, Imano M, Fukushima R, Sakamoto J, Oba MS, Morita S, Kono T, et
al: Double-blind, placebo-controlled, randomized phase II study of
TJ-14 (hangeshashinto) for gastric cancer chemotherapy-induced oral
mucositis. Cancer Chemother Pharmacol. 73:1047–1054. 2014.
View Article : Google Scholar : PubMed/NCBI
|
|
14
|
Kono T, Kaneko A, Matsumoto C, Miyagi C,
Ohbuchi K, Mizuhara Y, Miyano K and Uezono Y: Multitargeted effects
of hangeshashinto for treatment of chemotherapy-induced oral
mucositis on inducible prostaglandin E2 production in human oral
keratinocytes. Integr Cancer Ther. 13:435–445. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
15
|
Okumi H and Koyama A: Kampo medicine for
palliative care in Japan. Biopsychosoc Med Jan. 8:62014. View Article : Google Scholar
|
|
16
|
Ohnishi S and Takeda H: Herbal medicines
for the treatment of cancer chemotherapy-induced side effects.
Front Pharmacol. Feb;10(6): 142015.https://doi.org/10.3389/fphar.2015.00014
|
|
17
|
Naidu MU, Ramana GV, Rani PU, Mohan IK,
Suman A and Roy P: Chemotherapy-induced and/or radiation
therapy-induced oral mucositis-complicating the treatment of
cancer. Neoplasia. 6:423–431. 2004. View Article : Google Scholar : PubMed/NCBI
|
|
18
|
Ke EE and Wu YL: Afatinib in the
first-line treatment of epidermal-growth-factor-receptor
mutation-positive non-small cell lung cancer: A review of the
clinical evidence. Ther Adv Respir Dis. 10:256–264. 2016.
View Article : Google Scholar : PubMed/NCBI
|
|
19
|
Bardy J, Molassiotis A, Ryder WD, Mais K,
Sykes A, Yap B, Lee L, Kaczmarski E and Slevin N: A double-blind,
placebo-controlled, randomised trial of active manuka honey and
standard oral care for radiation-induced oral mucositis. Br J Oral
Maxillofac Surg. 50:221–226. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
20
|
Ogata J, Minami K, Horishita T, Shiraishi
M, Okamoto T, Terada T and Sata T: Gargling with sodium azulene
sulfonate reduces the postoperative sore throat after intubation of
the trachea. Anesth Analg. 101:290–293. 2005. View Article : Google Scholar : PubMed/NCBI
|
|
21
|
Yen SH, Wang LW, Lin YH, Jen YM and Chung
YL: Phenylbutyrate mouthwash mitigates oral mucositis during
radiotherapy or chemoradiotherapy in patients with head-and-neck
cancer. Int J Radiat Oncol Biol Phys. 82:1463–1470. 2012.
View Article : Google Scholar : PubMed/NCBI
|