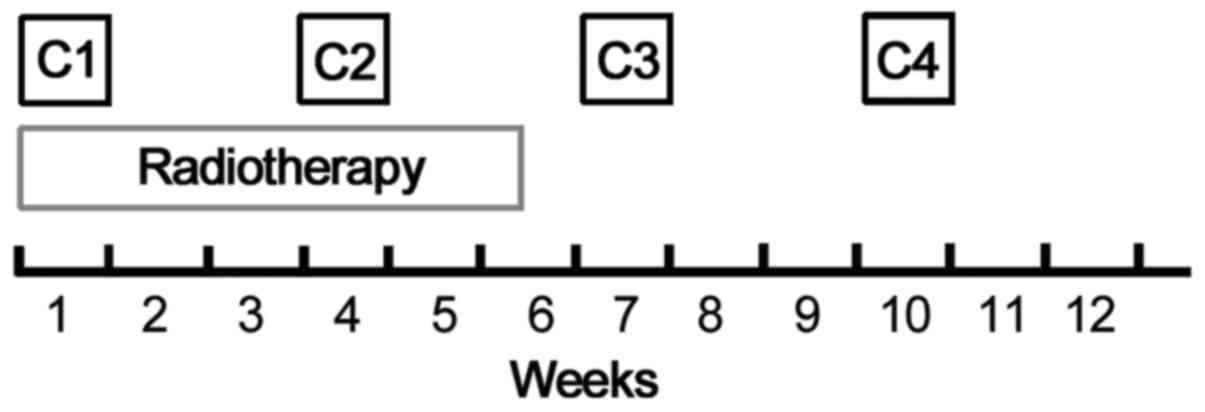
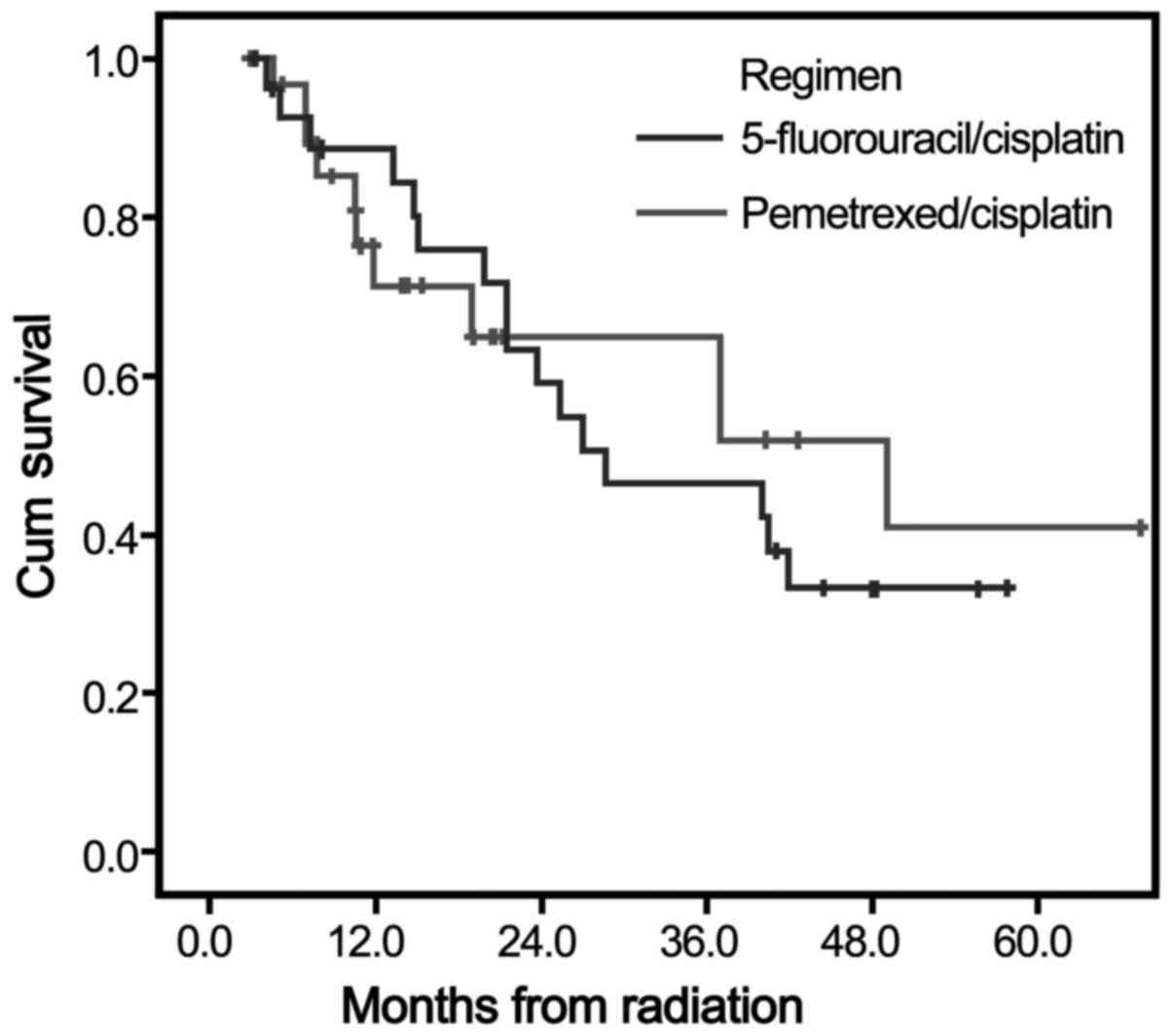
|
1
|
Cooper JS, Guo MD, Herskovic A, Macdonald
JS, Martenson JA Jr, Al-Sarraf M, Byhardt R, Russell AH, Beitler
JJ, Spencer S, et al: Chemoradiotherapy of locally advanced
esophageal cancer: Long-term follow-up of a prospective randomized
trial (RTOG 85-01). Radiation Therapy Oncology Group. JAMA.
281:1623–1627. 1999. View Article : Google Scholar : PubMed/NCBI
|
|
2
|
Minsky BD, Pajak TF, Ginsberg RJ, Pisansky
TM, Martenson J, Komaki R, Okawara G, Rosenthal SA and Kelsen DP:
INT 0123 (Radiation Therapy Oncology Group 94–05) phase III trial
of combined-modality therapy for esophageal cancer: High-dose
versus standard-dose radiation therapy. J Clin Oncol. 20:1167–1174.
2002. View Article : Google Scholar : PubMed/NCBI
|
|
3
|
Kato K, Muro K, Minashi K, Ohtsu A,
Ishikura S, Boku N, Takiuchi H, Komatsu Y, Miyata Y and Fukuda H:
Gastrointestinal Oncology Study Group of the Japan Clinical
Oncology Group (JCOG): Phase II study of chemoradiotherapy with
5-fluorouracil and cisplatin for Stage II–III esophageal squamous
cell carcinoma: JCOG trial (JCOG 9906). Int J Radiat Oncol Biol
Phys. 81:684–690. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
4
|
Zhao KL, Shi XH, Jiang GL, Yao WQ, Guo XM,
Wu GD and Zhu LX: Late course accelerated hyperfractionated
radiotherapy plus concurrent chemotherapy for squamous cell
carcinoma of the esophagus: A phase III randomized study. Int J
Radiat Oncol Biol Phys. 62:1014–1020. 2005. View Article : Google Scholar : PubMed/NCBI
|
|
5
|
Higuchi K, Koizumi W, Tanabe S, Sasaki T,
Katada C, Ishiyama H and Hayakawa K: A phase I trial of definitive
chemoradiotherapy with docetaxel, cisplatin, and 5-fluorouracil
(DCF-R) for advanced esophageal carcinoma: Kitasato digestive
disease & oncology group trial (KDOG 0501). Radiother Oncol.
87:398–404. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
6
|
Chiarion-Sileni V, Corti L, Ruol A,
Innocente R, Boso C, Del Bianco P, Pigozzo J, Mazzarotto R, Tomassi
O and Ancona E: Phase II trial of docetaxel, cisplatin and
fluorouracil followed by carboplatin and radiotherapy in locally
advanced oesophageal cancer. Br J Cancer. 96:432–438. 2007.
View Article : Google Scholar : PubMed/NCBI
|
|
7
|
Li BS, Gong HY, Huang W, Yi Y, Zhang ZC,
Li HS, Wang ZT and Yu JM: Phase I study of concurrent selective
lymph node late course accelerated hyper-fractionated radiotherapy
and pemetrexed, cisplatin for locally advanced esophageal squamous
cell carcinoma. Dis Esophagus. 24:251–257. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
8
|
Seiwert TY, Connell PP, Mauer AM, Hoffman
PC, George CM, Szeto L, Salgia R, Posther KE, Nguyen B, Haraf DJ
and Vokes EE: A phase I study of pemetrexed, carboplatin, and
concurrent radiotherapy in patients with locally advanced or
metastatic non-small cell lung or esophageal cancer. Clin Cancer
Res. 13:515–522. 2007. View Article : Google Scholar : PubMed/NCBI
|
|
9
|
Adjei AA: Pemetrexed (Alimta): A novel
multitargeted antifolate agent. Expert Rev Anticancer Ther.
3:145–156. 2003. View Article : Google Scholar : PubMed/NCBI
|
|
10
|
Jatoi A, Soori G, Foster NR, Hiatt BK,
Knost JA, Fitch TR, Callister MD, Nichols FC III, Husted TM and
Alberts SR: Phase II study of preoperative pemetrexed, carboplatin,
and radiation followed by surgery for locally advanced esophageal
cancer and gastroesophageal junction tumors. J Thorac Oncol.
5:1994–1998. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
11
|
Green FL, Page DL, Fleming ID, Fritzet AG,
Balch CM, Haller DG and Morrow M: AJCC cancer staging manual, 6th
edition. Ann Oncol. 14:345–346. 2003. View Article : Google Scholar
|
|
12
|
Li BS, Zhou T, Wang ZT, Li HS, Sun HF,
Zhang ZC, Lin HQ, Wei YM, Gong HY, Huang W, et al: Phase I study of
concurrent selective lymph node late course accelerated
hyper-fractionated radiotherapy and Capecitabine, Cisplatin for
locally advanced esophageal squamous cell carcinoma. Radiother
Oncol. 93:458–461. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
13
|
Therasse P, Eisenhauer EA and Verweij J:
RECIST revisited: A review of validation studies on tumour
assessment. Eur J Cancer. 42:1031–1039. 2006. View Article : Google Scholar : PubMed/NCBI
|
|
14
|
Trotti A, Colevas AD, Setser A, Rusch V,
Jaques D, Budach V, Langer C, Murphy B, Cumberlin R, Coleman CN and
Rubin P: CTCAE v3.0: Development of a comprehensive grading system
for the adverse effects of cancer treatment. Semin Radiat Oncol.
13:176–181. 2003. View Article : Google Scholar : PubMed/NCBI
|
|
15
|
Shi XH, Yao W and Liu T: Late course
accelerated fractionation in radiotherapy of esophageal carcinoma.
Radiother Oncol. 51:21–26. 1999. View Article : Google Scholar : PubMed/NCBI
|
|
16
|
Zhang YW, Chen L, Bai Y and Zheng X:
Long-term outcomes of late course accelerated hyper-fractionated
radiotherapy for localized esophageal carcinoma in Mainland China:
A meta-analysis. Dis Esophagus. 24:495–501. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
17
|
Liu CX, Li XY and Gao XS: Meta-analysis of
late course accelerated hyperfractionated radiotherapy combined
with FP chemotherapy for esophageal carcinoma. Chin J Cancer.
29:889–899. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
18
|
Zhou ZG, Gao XS, Qiao XY and Zhang P:
Literature analysis of radiotherapy for esophageal cancer in China.
Chin J Cancer. 29:873–881. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
19
|
Gilbert J, Murphy B, Dietrich MS, Henry E,
Jordan R, Counsell A, Wirth P, Yarbrough WG, Slebos RJ and Chung
CH: Phase 2 trial of oxaliplatin and pemetrexed as an induction
regimen in locally advanced head and neck cancer. Cancer.
118:1007–1013. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
20
|
Villaflor VM, Haraf D, Salama JK,
Kocherginsky M, Langerman A, Gomez-Abuin G, Beniwal P, Blair EA,
Stenson KM, Portugal L, et al: Phase II trial of pemetrexed-based
induction chemotherapy followed by concomitant chemoradiotherapy in
previously irradiated patients with squamous cell carcinoma of the
head and neck. Ann Oncol. 22:2501–2507. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
21
|
Berger AC, Farma J, Scott WJ, Freedman G,
Weiner L, Cheng JD, Wang H and Goldberg M: Complete response to
neoadjuvant chemoradiotherapy in esophageal carcinoma is associated
with significantly improved survival. J Clin Oncol. 23:4330–4337.
2005. View Article : Google Scholar : PubMed/NCBI
|