Introduction
Sarcomas are a heterogeneous group of tumors that
are mesenchymal in origin and account for 1% of adult tumors and
15% of pediatric tumors (1,2). Approximately 80% of sarcomas originate
from soft tissue and 20% from bone. Similar to other types of solid
tumors, conventional therapy includes surgery, radiotherapy and
chemotherapy. The role of adjuvant or neoadjuvant chemotherapy for
sarcoma has not yet been fully elucidated, as large prospective
analyses have not demonstrated their unequivocal benefit (3,4);
however, a previous meta-analysis suggested that adjuvant
chemotherapy is associated with a moderate overall survival
advantage (5). A number of agents
are currently used for sarcoma therapy, including doxorubicin,
ifosfamide, dacarbazine, cisplatin, vincristine, cyclophosphamide
(CTX) and etoposide. Under most circumstances, the combination of
≥2 agents is commonly used in patients with sarcoma, as combination
chemotherapy appears to increase response rates; however, it is
associated with greater toxicity, with immunosuppression being one
of the most serious side effects, with no overall survival
advantage (6).
Yulangsan, also referred to as Longyanshen, is a
commonly used herb in the indigenous Zhuang community in Guangxi,
China, and is derived from the dried roots of Millettia pulchra
Kurz var-laxior (Dunn) Z. We. Locally, this herb is used for
patients recuperating after illness and those concerned about their
general health. In the published literature, it has been reported
that Yulangsan may enhance the function of the immune system
(7). Kong et al found that
extracts of Longyanshen (mainly the polysaccharide component)
enhance sensitivity to chemotherapy in drug-resistant cancer cell
lines (8), and improve CTX-induced
immune dysfunction in mice (9).
The present study was designed to investigate the
antitumor activity of YLSPS in mice bearing S180 sarcoma tumors. In
order to demonstrate the antitumor effect of YLSPS, as well as its
effects on the immunological organs, paralleled and combined
studies with CTX were simultaneously conducted under the same
experimental conditions.
Materials and methods
Chemicals
YLSPS was prepared as previously described (10). The root of Millettia pulchra
Kurz var. laxior (Dunn) Z. Wei was dried, and extracted
three times with boiling water. The polysaccharide in the filtrate
was precipitated fractionally with alcohol. The protein in the
product was removed and further purified using diethylaminoethyl
(DEAE) ion exchange cellulose (DEAE-52). Gas chromatography and
thin-layer chromatography analysis demonstrated that YLSPS was
composed of D-glucose and D-arabinose in a molar ratio of 90.79 and
9.21%, respectively, with an average molecular weight of ~14,301
Da. CTX injection was purchased from Shanxi Pude Pharmaceutical
Co., Ltd. (Datong, China).
Animals and sarcoma model
A total of 80 male and 80 female BALB/c mice (aged
4–6 weeks and weighing 20±2 g) were purchased from the Institute of
Animal Care Center of Guangxi Medical University. The mice were
acclimatized for 1 week prior to being used in the study. All
animals were cared for in accordance with the National Institutes
of Health's Guide for the Care and Use of Laboratory Animals. The
experimental protocols of the study were approved by the Animal
Care Committee at Guangxi Medical University (certificate no. SYKG
2013–0005).
Murine sarcoma 180 (S180) cells were injected into
the peritoneal cavity of the mice and proliferated to produce
ascites. The cell colonies were maintained by weekly
transplantation of the tumor cells from the ascitic fluid into the
peritoneal cavity of another mouse. S180 cells were isolated from
the ascitic fluid and suspended in saline at a density of
1×1010 cells/l. Subsequently, 2×106 cells (in
200 µl saline) were injected into the axillary fossa of the right
forelegs to prepare the tumor-bearing mice. Two days after S180
cell implantation, the mice were randomly divided into five groups
(10 mice per group), as follows: i) CTX group [0.02 g/kg,
intraperitoneal (i.p.) injection on days 1, 4, 7 and 10]; ii)
high-dose YLSPS group [0.6 g/kg/day, intragastric (i.g.)
administration; iii) intermediate-dose YLSPS group (0.3 g/kg/day,
i.g.); iv) low-dose YLSPS group (0.15 g/kg/day, i.g.); and v)
control group (saline, 0.2 ml/10 g/day, i.g.). The animals were
treated for 10 days and then sacrificed by cervical dislocation
under ethyl ether anesthesia on day 12.
Calculation of tumor inhibition,
spleen index and thymus index
The tumor inhibition rate was calculated as follows:
Inhibition (%)=(C-T)/C × 100%, where C and T represent the tumor
weights (in mg) of control and treated mice, respectively. Based on
the correlation between immune activity and the weights of the
spleen and thymus, the relative weights of the spleen and thymus
(in mg) with regards to the mouse body weight (10 g) were used to
obtain the spleen index (SI) and thymus index (TI), as previously
described (11,12).
Immunohistochemistry (IHC)
B-cell lymphoma 2 (Bcl-2), p53 and Bcl-2-associated
X (BAX) proteins were detected by IHC, as previously described
(13). Briefly, all specimens were
fixed in formalin, embedded in paraffin and cut into 4-µm sections
for IHC. Bcl-2, p53 and BAX were detected by rabbit anti-mouse
polyclonal antibodies (N-20; dilution, 1:50; Santa Cruz
Biotechnology, Santa Cruz, CA, USA) and IHC was performed using the
immunoperoxidase method as follows: Antigen retrieval was performed
using cell conditioning 1 antigen retrieval buffer (pH 8.5; Ventana
Medical Systems, Tucson, AZ, USA). The sections were incubated with
a primary antibody in phosphate-buffered saline (pH 7.4) with 1%
bovine serum albumin and stained on a BenchMark XT automated slide
stainer using a diaminobenzidine detection kit (ultraView Universal
DAB detection kit; Ventana Medical Systems). The positive reaction,
shown by brown color, was evaluated under a light microscope and
scored by two pathologists who were blinded to the origins of the
sections. The staining was scored on semi-quantitative scales as
follows: 0, no staining; 1, weak staining; 2, moderate staining;
and 3, strong staining.
Spleen lymphocyte preparation and cell
proliferation assessment
Spleens from S180 sarcoma-bearing mice treated with
CTX and/or YLSPS were aseptically removed for the preparation of
spleen lymphocytes. The removed spleens were homogenized in sterile
Hank's balanced salt solution (HBSS) and the homogenized spleen
tissue was passed through a mesh (200 mesh sieve) and washed twice
with HBSS. The erythrocytes were lysed with red blood cell lysis
buffer, while the remaining cells were suspended in RPMI-1640
medium with 10% fetal calf serum. Cells were counted with a
hemocytometer using the trypan blue exclusion technique (14). Cell viability exceeded 95% and cells
were resuspended with complete medium at a density of
1×1010 cells/l. ConA-stimulated cell proliferation was
detected with 3-(4,5-dimethylthiazol-2-yl)-2,5-diphenyltetrazolium
bromide (MTT). Briefly, cell suspensions (100 µl/well) were added
to 96-well culture plates followed by the addition of 100 µl ConA
(5 mg/l). The cells were then incubated at 37°C in a humidified, 5%
CO2 atmosphere for 68 h. Subsequently, 10 µl MTT (5
mg/ml) was added into each well followed by a 4-h additional
incubation. The cultures were centrifuged at 444 × g for 5 min and
the supernatant was carefully removed. After adding 200 µl dimethyl
sulfoxide, absorbance was assessed with a microplate reader at a
wavelength of 570 nm (A570; Jupiter ASYS, Montreal Biotech,
Kirkland, PQ, Canada).
Transmission electron microscopy
Mouse sarcoma samples were fixed with 4%
paraformaldehyde and 1% glutaraldehyde. The fixed tissues were
washed with phosphate buffer (pH 7.4) and post-fixed with osmium
tetroxide. These tissue samples were dehydrated through a series of
graded ethanol solutions and propylene oxide, and then embedded in
epoxy resin. Ultrathin sections (0.5 µm) of each sample were
stained with uranyl acetate and lead citrate and examined by
transmission electron microscopy (TEM; JEM-1200EX, JEOL, Tokyo,
Japan) with an accelerating voltage of 80 kV.
Statistical analysis
Numerical data were expressed as mean ± standard
error. Statistical differences between the means for the different
groups were evaluated with SPSS 13.0 software (SPSS Inc., Chicago,
IL, USA) using Student's t-test, with the level of significance set
at P<0.05.
Results
Antitumor effect of YLSPS in mice
bearing S180 solid sarcoma tumors
The tumor growth was significantly inhibited by
YLSPS administration at a low dose (39.7%), intermediate dose
(41.3%) and high dose (52.6%), compared with control mice. The
dose-dependent pattern of YLSPS-induced inhibition indicated the
therapeutic effect of YLSPS on S180 sarcoma tumors. To provide an
antitumor effect comparable with commonly used anticancer drugs,
CTX was also used at a standard dose (0.02 g/kg) in separate
positive-control mice, resulting in a tumor inhibition rate of
71.1%.
YLSPS- and CTX-induced changes of the
SI and TI in S180 sarcoma-bearing mice
Immunosuppression is considered to be a major side
effect of anticancer agents. As such, the changes in the SI and TI
induced by YLSPS and CTX treatment were compared. YLSPS increased
the SI as well as the TI at all three dose levels. With increasing
YLSPS dose (0.15, 0.30 and 0.60 g/kg/day), the SI values were
90.89±16.13, 92.93±7.26 and 104.52±8.22 mg/10 g, respectively, with
related TI values of 29.49±7.11, 33.81±6.02 and 38.39±5.50 mg/10 g,
respectively. The SI and TI values were 81.90±6.95 and 26.90±4.94
mg/10 g in the control group. By contrast, CTX significantly
decreased the SI and TI compared with the YLSPS and control groups
(28.45±7.65 and 8.76±2.59 mg/10 g, respectively).
Expression of apoptosis-related
proteins in the sarcoma tissue of S180-bearing mice
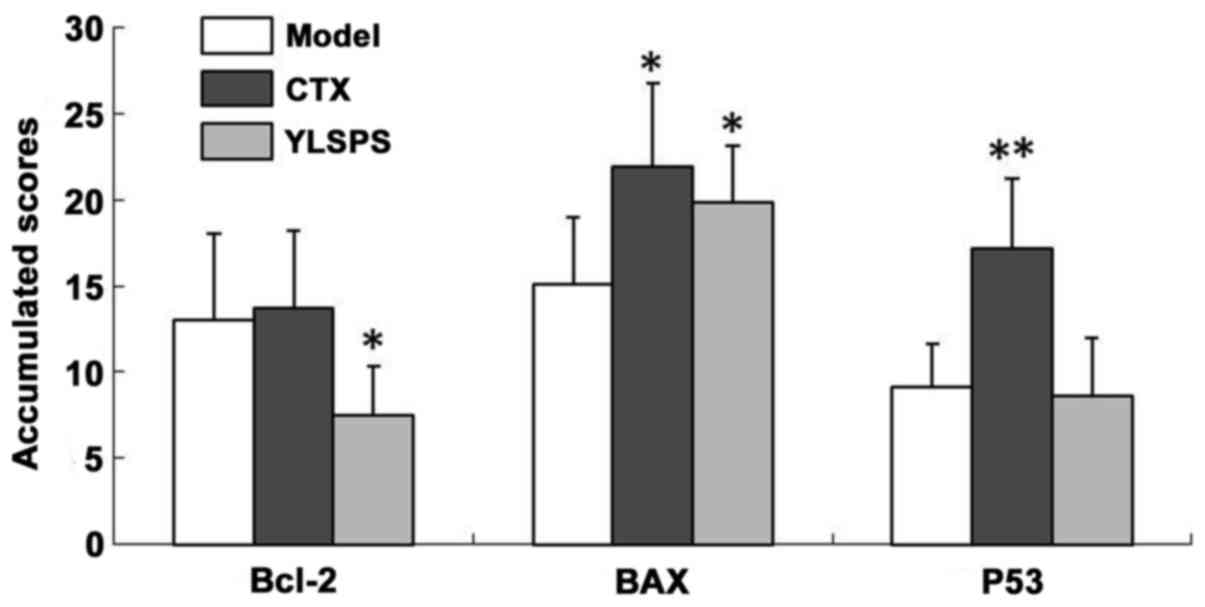
The changes in p53, Bcl-2 and BAX expression
following administration of YLSPS or CTX are summarized in Fig. 1. The accumulated scores represent the
relative expression of the studied proteins in the sarcoma tissues
of S180-bearing mice.
Ultrastructural changes of sarcoma
cells
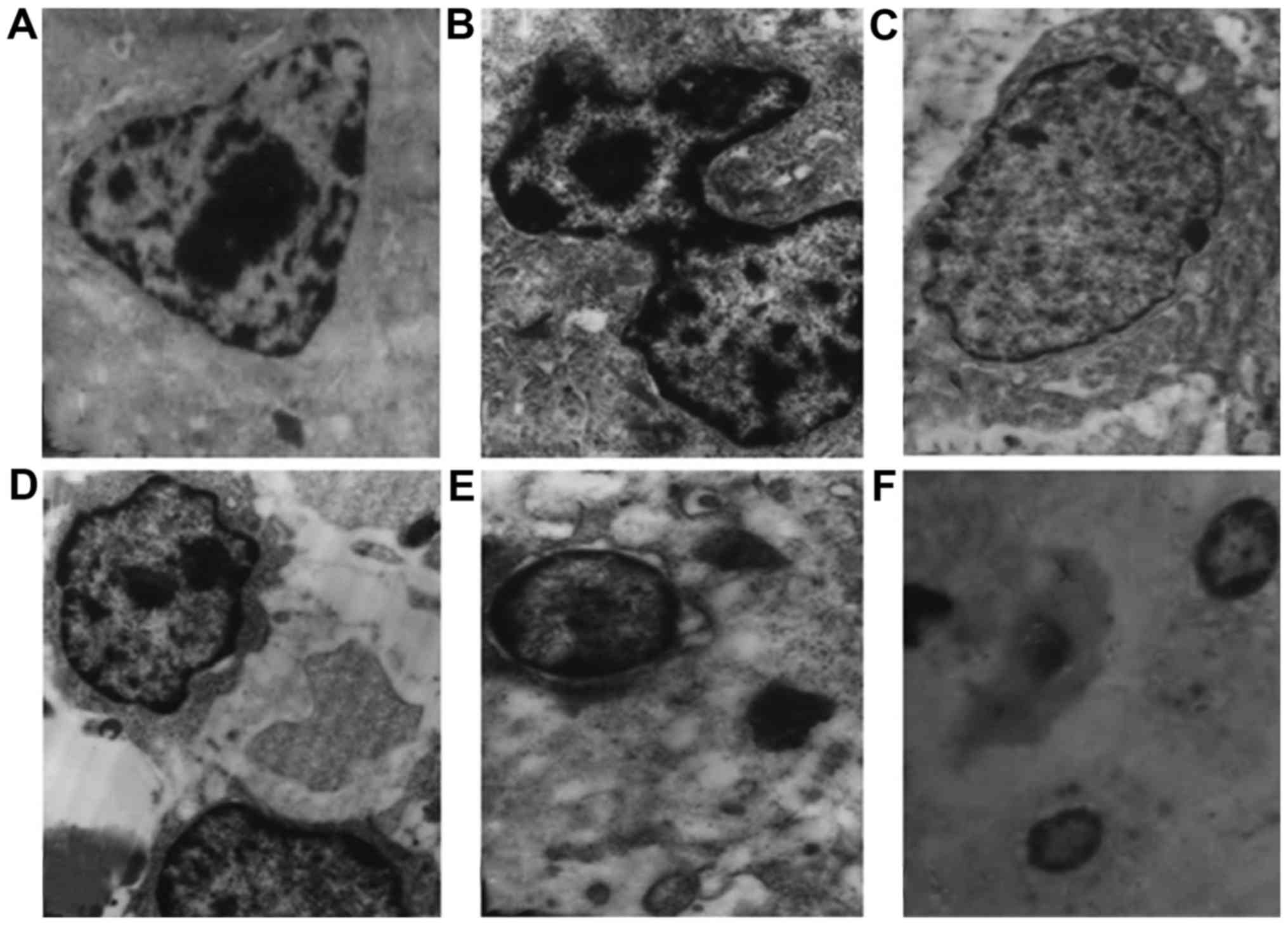
The majority of the cells in the sarcoma tissue were
cerioid-shaped, with increased internuclear distance and atypical
nuclei (Fig. 2A). S180 cells
exhibited nuclear membrane integrity, typical organelles and
mitotic activity (Fig. 2B).
Following treatment with a low dose of YLSPS, the cell size
decreased, with formation of mitochondrial vacuoles (Fig. 2C). S180-bearing mice treated with an
intermediate dose of YLSPS exhibited an increased number of
apoptotic cells with overt karyopyknosis and disordered cell
structure (Fig. 2D). After treatment
with a high dose of YLSPS (Fig. 2E),
there was an increase in cell debris in the intercellular space
accompanied by nuclear fragmentation and the formation of apoptotic
bodies. Furthermore, in addition to organelle fragmentation and
apoptotic body formation, the cell membrane structure completely
dissolved following treatment with CTX (Fig. 2F).
Effects of combined YLSPS and CTX
treatment on tumor growth and immune function
The additive effect of YLSPS and CTX is shown by the
q-value: q=EAB/[EA+(1-EA) EB], where EA and EB are the tumor
inhibition rates of drug A and drug B, respectively, and EAB is the
tumor inhibition rate of drugs A and B used in combination. In this
case, q=0.85–1.25 indicates an additive effect of drugs A and B,
q>1.25 indicates enhancement (or a synergistic effect) of the
two drugs, and q<0.85 indicates that the two drugs are
antagonistic (15). The tumor
inhibition rate was 64.8% for the CTX treatment group, whereas for
the high, medium and low doses of YLPS together with CTX, the tumor
inhibition rates were 74.8, 76.1 and 67.4%, respectively, with
related q-values of 0.905, 0.984 and 0.887, respectively. All the
q-values were in the range of 0.85 to 1.25, which indicated that
different doses of YLSPS may enhance the CTX antitumor effect on
mouse S180 tumors in an additive manner.
To investigate the mechanisms through which YLSPS
potentiates the antitumor effect of CTX, the SI, TI and peripheral
blood leukocyte count were assessed following administration of
various drug combinations. As shown in Table I, the administration of CTX
significantly inhibited tumor growth, but its antitumor activity
was accompanied by a decrease in SI, TI and peripheral blood
leukocytes. However, the combination of CTX with YLSPS at the three
tested doses achieved further tumor growth inhibition, but improved
the SI, TI and peripheral blood leukocyte count compared with CTX
alone. The results of spleen lymphocyte proliferation and tumor
necrosis factor α (TNF-α) production analyses in S180-bearing mice
were in accordance with the YLSPS-induced SI and TI changes
following CTX treatment (Table
II).
 | Table I.Effects of YLSPS alone or in
combination with CTX on the SI, TI and peripheral blood leukocyte
count. |
Table I.
Effects of YLSPS alone or in
combination with CTX on the SI, TI and peripheral blood leukocyte
count.
| Groups | Dose (g/kg/day) | n | SI (mg/10 g) | TI (mg/10 g) | WBC (×10−9
cells/l) | Tumor weight (g) | Inhibition (%) |
|---|
| Control |
| 10 |
71.89±5.56 |
26.15±4.40 |
16.88±3.64 |
1.29±0.34 |
|
| CTX | 0.02 | 9 |
23.16±3.87a |
10.83±2.50a |
6.87±1.76a |
0.27±0.12a | 79.4 |
| YLSPS+ CTX | 0.15+0.02 | 10 |
26.18±7.47a |
11.63±3.91a |
7.10±2.63a |
0.22±0.05a | 82.8 |
|
| 0.30+0.02 | 10 |
31.93±7.34a,c |
14.24±3.91a,b |
11.00±3.20a,c |
0.18±0.05a,b | 86.3 |
|
| 0.60+0.02 | 10 |
37.42±5.86a,c |
16.99±4.33a,c |
10.81±2.84a,c |
0.18±0.04a,b | 86.4 |
 | Table II.Effects of YLSPS on ConA-induced
proliferation of spleen lymphocytes and TNF-α production in
S180-bearing mice treated with CTX. |
Table II.
Effects of YLSPS on ConA-induced
proliferation of spleen lymphocytes and TNF-α production in
S180-bearing mice treated with CTX.
| Groups | Dose (g/kg) | Tumor weight (g) | Inhibition (%) | A570 | TNF-α (pg/ml) |
|---|
| Control | – |
1.794±0.463 | – |
0.286±0.039 |
904.9±70.0 |
| CTX | 0.02 |
0.566±0.090b | 68.5 |
0.171±0.018b |
209.3±46.9b |
| YLSPS+ CTX | 0.15+0.02 |
0.448±0.108b | 75.1 |
0.178±0.038b |
205.9±43.0b |
|
| 0.30+0.02 |
0.371±0.131b,c | 79.3 |
0.218±0.038a,c |
333.0±61.3b,d |
|
| 0.60+0.02 |
0.393±0.085b,c | 78.1 |
0.213±0.028b,c |
362.8±70.4b,d |
Discussion
Sarcomas are tumors of mesenchymal origin that
comprise ~1% of human cancers. Soft tissue sarcomas are a
relatively rare and heterogeneous group of malignancies that are
characterized by mesodermal differentiation (16). The international incidence is
estimated to be ~1.8–5 per 100,000 people annually (17). There are at least 50 different
subtypes of soft tissue sarcoma, with new ones described at an ever
increasing frequency (2). Sarcomas
present a challenge with regards to their treatment due to their
rarity, biological heterogeneity and the need for multimodality
therapy. Current conventional cancer therapies (surgery,
chemotherapy and radiotherapy) are, to a significant extent,
symptomatic and passive in nature. The majority of cancer patients
(including those with sarcoma) succumb to recurrence, metastasis,
or therapy-related life-threatening complications (18,19), in
which chemotherapy-induced immunosuppression plays a major role.
Certain polysaccharides extracted from various Chinese herbs have
been demonstrated to possess antitumor properties in recent
studies, including the polysaccharides from Ganoderma
lucidum (20), Ganoderma
atrum (21) and ginseng neutral
(22). One of the most obvious
advantages of herbal-extracted polysaccharides is their low
incidence of side effects and potent anticancer activity. In the
present study, the antitumor effect of YLSPS was investigated in
combination with CTX, using a model of S180 sarcoma-bearing
mice.
YLSPS significantly inhibited the growth of S180
tumors at three dosages; however, the rate of inhibition with YLSPS
was lower compared with that of CTX. However, CTX exerted obvious
damage to the immune organs. The changes in the apoptosis-related
protein expression levels, i.e., the decrease in Bcl-2 and the
increase in BAX, suggested the involvement of S180 cell apoptosis
induced by administration of YLSPS (Fig.
1). This hypothesis was supported by the apoptotic appearance
of cells observed under TEM, i.e., condensation of the chromatin at
the margins of the nuclei, and disintegration of the nucleolus and
the cytoplasmic vacuoles (Fig. 2).
An additive antitumor effect was obtained when YLSPS was combined
with CTX in S180 sarcoma-bearing mice. Interestingly, in this
combined therapy, YLSPS ameliorated CTX-induced changes in the SI,
TI and peripheral leukocyte count (Table
I). This additive effect of YLSPS on CTX was confirmed by
consideration of the q-values.
The beneficial effect of YLSPS on CTX with regards
to the treatment of sarcoma in S180-bearing mice may be considered
to have two aspects. First, YLSPS itself exerted an antitumor
action, most likely through the induction of S180 cell apoptosis,
although this activity was weaker in comparison to that of CTX
alone. Second, YLSPS attenuated CTX-induced toxicity in the immune
system, which was shown by the changes in the SI and TI (Table I), as well as the CoA-induced
proliferation of splenic lymphocytes and TNF-α production in
S180-bearing mice (Table II).
In conclusion, YLSPS inhibited sarcoma growth in
S180-bearing mice. The antitumor effect of YLSPS likely resulted
through the induction of apoptosis in S180 sarcoma cells. YLSPS
also attenuated CTX-induced immune system cytotoxicity when used in
combination, thereby potentiating the tumor suppression effect of
CTX, while limiting side effects. These results provide additional
information on combination therapy for the treatment of
sarcoma.
Acknowledgements
The authors are grateful to James Dai for his
assistance in preparing the manuscript. The present study was
supported by the Guangxi Science and Technology Development Project
(grant no. 14124003-8), and the Guangxi Natural Science Foundation
(grant nos. 2013GXNSFAA019253 and 2015GXNSFAA139163).
References
|
1
|
Lewin J, Puri A, Quek R, Ngan R, Alcasabas
AP, Wood D and Thomas D: Management of sarcoma in the Asia-Pacific
region: Resource-stratified guidelines. Lancet Oncol. 14:e562–e570.
2013. View Article : Google Scholar : PubMed/NCBI
|
|
2
|
Forscher C, Mita M and Figlin R: Targeted
therapy for sarcomas. Biologics. 8:91–105. 2014.PubMed/NCBI
|
|
3
|
Italiano A, Penel N, Robin YM, Bui B, Le
Cesne A, Piperno-Neumann S, Tubiana-Hulin M, Bompas E, Chevreau C,
Isambert N, et al: Neo/adjuvant chemotherapy does not improve
outcome in resected primary synovial sarcoma: A study of the French
Sarcoma Group. Ann Oncol. 20:425–430. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
4
|
Woll PJ, Reichardt P, Le Cesne A, Bonvalot
S, Azzarelli A, Hoekstra HJ, Leahy M, Van Coevorden F, Verweij J,
Hogendoorn PC, et al: Adjuvant chemotherapy with doxorubicin,
ifosfamide, and lenograstim for resected soft-tissue sarcoma (EORTC
62931): A multicentre randomised controlled trial. Lancet Oncol.
13:1045–1054. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
5
|
Pervaiz N, Colterjohn N, Farrokhyar F,
Tozer R, Figueredo A and Ghert M: A systematic meta-analysis of
randomized controlled trials of adjuvant chemotherapy for localized
resectable soft-tissue sarcoma. Cancer. 113:573–581. 2008.
View Article : Google Scholar : PubMed/NCBI
|
|
6
|
Bramwell VH, Anderson D and Charette ML:
Sarcoma Disease Site Group: Doxorubicin-based chemotherapy for the
palliative treatment of adult patients with locally advanced or
metastatic soft tissue sarcoma. Cochrane Database Syst Rev:
CD003293. 2003. View Article : Google Scholar
|
|
7
|
Region DoPHoGZA: Guangxi Chinese Materia
Medica Standards. Guangxi Kexue Jishu Publishing House, Guangxi
Zhuang Autonomous Region. 1990.(In Chinese).
|
|
8
|
Kong X, Jiang W and Lin Z: Reversal effect
of extract from Longyanshen on the resistance of human cancer cells
MCF-7/ADR and KB/MIT. J Guangxi Med Univ. 20:495–496. 2003.(In
Chinese).
|
|
9
|
Kong X, Jiang W and Lin Z: Effect of
extract of Longyanshen (EL) on immunodepression induced by
cyclophosphamide in mice. China Pharmacy. 15:335–336. 2004.(In
Chinese).
|
|
10
|
Lin X, Huang Z, Chen X, Rong Y, Zhang S,
Jiao Y, Huang Q and Huang R: Protective effect of Millettia pulchra
polysaccharide on cognitive impairment induced by D-galactose in
mice. Carbohydr Polym. 101:533–543. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
11
|
Wiens GD, Vallejo RL, Leeds TD, Palti Y,
Hadidi S, Liu S, Evenhuis JP, Welch TJ and Rexroad CE III:
Assessment of genetic correlation between bacterial cold water
disease resistance and spleen index in a domesticated population of
rainbow trout: Identification of QTL on chromosome Omy19. PLoS One.
8:e757492013. View Article : Google Scholar : PubMed/NCBI
|
|
12
|
Fang JJ, Zhu ZY, Dong H, Zheng GQ, Teng AG
and Liu AJ: Effect of spleen lymphocytes on the splenomegaly in
hepatocellular carcinoma-bearing mice. Biomed Environ Sci.
27:17–26. 2014.PubMed/NCBI
|
|
13
|
Decastel M, Ossondo M, Andrea AM,
Tressieres B, Veronique-Baudin J, Deloumeaux J, Lubeth M and
Smith-Ravin J: Colorectal cancer in patients seen at the teaching
hospitals of Guadeloupe and Martinique: Discrepancies, similarities
in clinicopathological features, and p53 status. BMC Clin Pathol.
14:122014. View Article : Google Scholar : PubMed/NCBI
|
|
14
|
Alonso-Castro AJ, Ortiz-Sánchez E,
Domínguez F, Arana-Argáez V, Juárez-Vázquez Mdel C, Chávez M,
Carranza-Álvarez C, Gaspar-Ramírez O, Espinosa-Reyes G,
López-Toledo G, et al: Antitumor and immunomodulatory effects of
Justicia spicigera Schltdl (Acanthaceae). J Ethnopharmacol.
141:888–894. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
15
|
Liu J, Su F and Sun J: The synergistic
effect and mechanism of Agaricus blazei polysaccharides compound
and Cytoxan. Chin J Clin Rehabil. 10:118–121. 2006.(In
Chinese).
|
|
16
|
Cormier JN and Pollock RE: Soft tissue
sarcomas. CA Cancer J Clin. 54:94–109. 2004. View Article : Google Scholar : PubMed/NCBI
|
|
17
|
Wibmer C, Leithner A, Zielonke N, Sperl M
and Windhager R: Increasing incidence rates of soft tissue
sarcomas? A population-based epidemiologic study and literature
review. Ann Oncol. 21:1106–1111. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
18
|
Dai LJ, Moniri MR, Zeng ZR, Zhou JX, Rayat
J and Warnock GL: Potential implications of mesenchymal stem cells
in cancer therapy. Cancer Lett. 305:8–20. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
19
|
Sun XY, Nong J, Qin K, Warnock GL and Dai
LJ: Mesenchymal stem cell-mediated cancer therapy: A dual-targeted
strategy of personalized medicine. World J Stem Cells. 3:96–103.
2011. View Article : Google Scholar : PubMed/NCBI
|
|
20
|
Wang PY, Zhu XL and Lin ZB: Antitumor and
immunomodulatory effects of polysaccharides from broken-spore of
ganoderma lucidum. Front Pharmacol. 3:1352012. View Article : Google Scholar : PubMed/NCBI
|
|
21
|
Zhang S, Nie S, Huang D, Huang J, Wang Y
and Xie M: Polysaccharide from Ganoderma atrum evokes antitumor
activity via Toll-like receptor 4-mediated NF-κB and
mitogen-activated protein kinase signaling pathways. J Agric Food
Chem. 61:3676–3682. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
22
|
Ni W, Zhang X, Wang B, Chen Y, Han H, Fan
Y, Zhou Y and Tai G: Antitumor activities and immunomodulatory
effects of ginseng neutral polysaccharides in combination with
5-fluorouracil. J Med Food. 13:270–277. 2010. View Article : Google Scholar : PubMed/NCBI
|