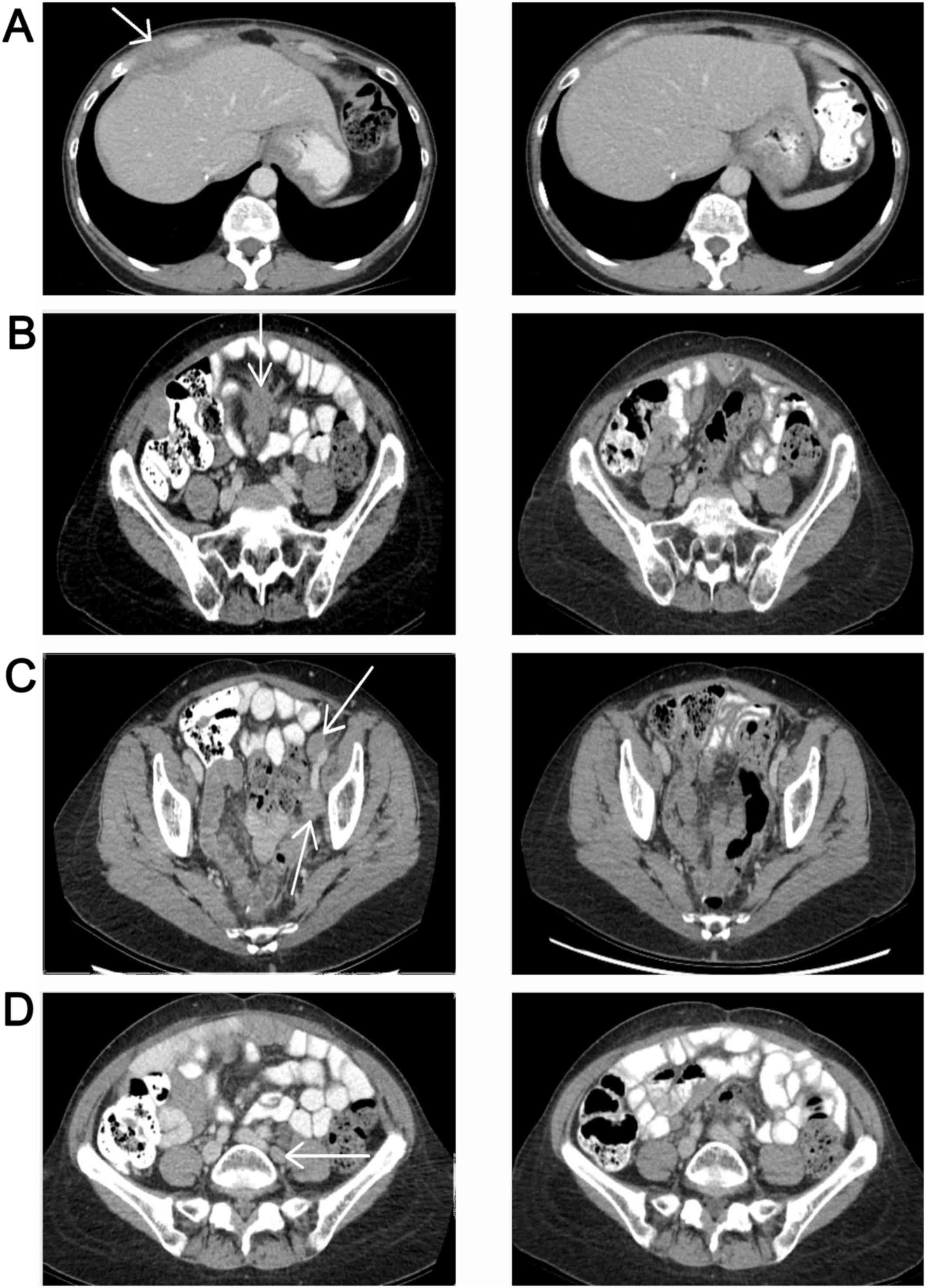
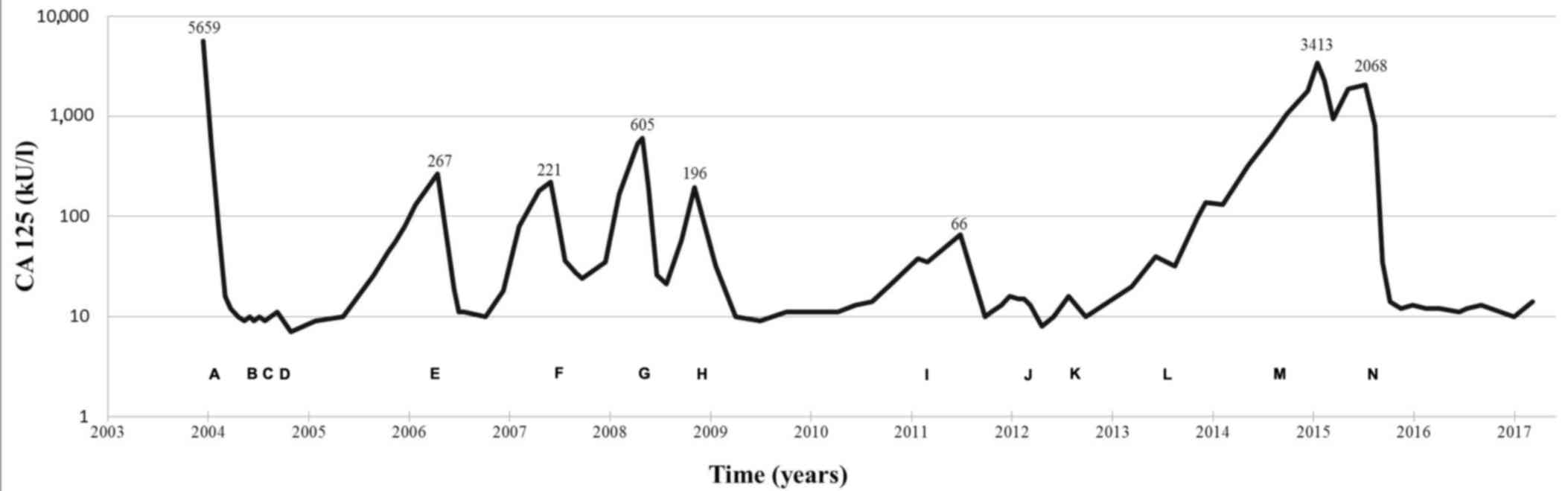
|
1
|
Bowtell DD, Böhm S, Ahmed AA, Aspuria PJ,
Bast RC Jr, Beral V, Berek JS, Birrer MJ, Blagden S, Bookman MA, et
al: Rethinking ovarian cancer II: Reducing mortality from
high-grade serous ovarian cancer. Nat Rev Cancer. 15:668–679. 2015.
View Article : Google Scholar : PubMed/NCBI
|
|
2
|
Liu G, Yang D, Sun Y, Shmulevich I, Xue F,
Sood AK and Zhang W: Differing clinical impact of BRCA1 and BRCA2
mutations in serous ovarian cancer. Pharmacogenomics. 13:1523–1535.
2012. View Article : Google Scholar : PubMed/NCBI
|
|
3
|
Ferrandina G, Corrado G, Mscilini F,
Malaguti P, Samaritani R, Distefano M, Masciullo V, Di Legge A,
Saveresse A and Scambia G: Metronomic oral cyclophosphamide (MOC)
in the salvage therapy of heavily treated recurrent ovarian cancer
patients: A retrospective, multicenter study. BMC Cancer.
14:9472014. View Article : Google Scholar : PubMed/NCBI
|
|
4
|
Du Bois A, Belau A, Wagner U, Pfisterer J,
Schmalfeldt B, Richter B, Staehle A, Jackisch C, Lueck HJ,
Schroeder W, et al: A phase II study of paclitaxel, carboplatin,
and gemcitabine in previously untreated patients with epithelial
ovarian cancer FIGO stage IC-IV (AGO-OVAR protocol OVAR-8). Gynecol
Oncol. 96:444–451. 2005. View Article : Google Scholar : PubMed/NCBI
|
|
5
|
Penel N, Adenis A and Bocci G:
Cyclophosphamide-based metronomic chemotherapy: 10 years of
experience, where do we stand and where are we going? Crit Rev
Oncol Hematol. 82:40–50. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
6
|
Kerbel RS and Kamen BA: The
anti-angiogenic basis of metronomic chemotherapy. Nat Rev Cancer.
4:423–436. 2004. View
Article : Google Scholar : PubMed/NCBI
|
|
7
|
Madondo MT, Quinn M and Plebanski M: Low
dose cyclophosphamide: Mechanisms of T cell modulation. Cancer
Treat Rev. 42:3–9. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
8
|
André N, Carré M and Pasquier E:
Metronomics: Towards personalized chemotherapy? Nat Rev Clin Oncol.
11:413–431. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
9
|
Scharovsky OG, Mainetti LE and Rozados VR:
Metronomic chemotherapy: Changing the paradigm that more is better.
Curr Oncol. 16:7–15. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
10
|
Samaritani R, Corrado G, Vizza E and
Sbiroli C: Cyclophosphamide ‘metronomic’ chemotherapy for
palliative treatment of a young patient with advanced epithelial
ovarian cancer. BMC Cancer. 7:652007. View Article : Google Scholar : PubMed/NCBI
|
|
11
|
Kummar S, Oza AM, Fleming GF, Sullivan DM,
Gandara DR, Naughton MJ, Villalona-Calero MA, Morgan RJ Jr, Szabo
PM, Youn A, et al: Randomized trial of oral cyclophosphamide and
veliparib in high-grade serous ovarian, primary peritoneal, or
fallopian tube cancers, or BRCA-mutant ovarian cancer. Clin Cancer
Res. 21:1574–1582. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
12
|
Perroud HA, Scharovsky OG, Rozados VR and
Alasino CM: Clinical response in patients with ovarian cancer
treated with metronomic chemotherapy. Ecancermedicalscience.
11:7232017. View Article : Google Scholar : PubMed/NCBI
|
|
13
|
Ledermann J, Harter P, Gourley C,
Friedlander M, Vergote I, Rustign G, Scott C, Meier W,
Shapira-Frommer R, et al: Olaparib maintenance therapy in
platinum-sensitive relapsed ovarian cancer. Engl J Med.
366:1382–1392. 2012. View Article : Google Scholar
|
|
14
|
Gordon AN, Fleagle JT, Guthrie D, Parkin
DE, Gore ME and Lacave AJ: Recurrent epithelial ovarian carcinoma:
A randomized phase III study of pegylated liposomal doxorubicin
versus topotecan. J Clin Oncol. 19:3312–3322. 2001. View Article : Google Scholar : PubMed/NCBI
|
|
15
|
Pfisterer J, Plante M, Vergote I, du Bois
A, Hirte H, Lacave AJ, Wagner U, Stähle A, Stuart G, Kimmig R, et
al: Gemcitabine plus carboplatin compared with carboplatin in
patients with platinum-sensitive recurrent ovarian cancer: An
intergroup trial of the AGO-OVAR, the NCIC CTG, and the EORTC GCG.
J Clin Oncol. 24:4699–4707. 2006. View Article : Google Scholar : PubMed/NCBI
|
|
16
|
Aghajanian C, Blank SV, Goff BA, Judson
PL, Teneriello MG, Husain A, Sovak MY, Yi J and Nycum LR: OCEANS: A
randomized, double-blind, placebo-controlled phase III trial of
chemotherapy with or without bevacizumab in patients with
platinum-sensitive recurrent epithelial ovarian, primary
peritoneal, or fallopian tube cancer. J Clin Oncol. 30:2039–2045.
2012. View Article : Google Scholar : PubMed/NCBI
|
|
17
|
Cancer Genome Atlas Research Network:
Integrated genomic analyses of ovarian carcinoma. Nature.
474:609–615. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
18
|
Bast RC Jr and Mills GB: Personalizing
therapy for ovarian cancer: BRCAness and beyond. J Clin Oncol.
28:3545–3548. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
19
|
Mirza MR, Monk BJ, Herrstedt J, Oza AM,
Mahner S, Redondo A, Fabbro M, Ledermann JA, Lorusso D, Vergote I,
et al: Niraparib maintenance therapy in platinum-sensitive,
recurrent ovarian cancer. N Engl J Med. 375:2154–2164. 2016.
View Article : Google Scholar : PubMed/NCBI
|
|
20
|
El-Husseiny K, Motawei H and Ali MS:
Continuous low-dose oral cyclophosphamide and methotrexate as
maintenance therapy in patients with advanced ovarian carcinoma
after complete clinical response to platinum and paclitaxel
chemotherapy. Int J Gynecol Cancer. 26:437–442. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
21
|
Ferrandina G, Corrado G, Mascilini F,
Malaguti P, Samaritani R, Distefano M, Masciullo V, Di Legge A,
Saverese A, et al: Metronomic oral cyclophosphamide (MOC) in the
salvage therapy of heavily treated recurrent ovarian cancer
patients: a retrospective, multicenter study. BMC Cancer.
14:9472014. View Article : Google Scholar : PubMed/NCBI
|
|
22
|
Lee YK, Park NH and Lee H:
Clinicopathological values of NBS1 and DNA damage response genes in
epithelial ovarian cancers. Exp Mol Med. 47:e1952015. View Article : Google Scholar : PubMed/NCBI
|