Introduction
The vein of Galen is an intracerebral vein that
drains blood from the cerebral hemispheres and the basal ganglia
(1). It is formed by the fusion of
the internal cerebral veins and the basal veins of Rosenthal. Vein
of Galen malformation (VOGM) is an arteriovenous malformation with
a direct connection between the diencephalon and a dilated vein of
Galen (1). Steinheil first
discovered VOGM in 1895, accounting for 1% of pediatric congenital
malformations (2).
VOGMs occur when the prosencephalic vein of
Markowski, the fetal precursor of the vein of Galen, is
maldeveloped (3). At ~3 months of
gestation, the prosenchephalic vein of Markowski regresses and
forms the vein of Galen. However, in cases where there is an
arteriovenous malformation, the vein of Galen receives high blood
pressure, which it is not well adapted for, and, as a result,
dilates (3). This leads to increased
venous blood pressure and results in a whole array of symptoms,
most commonly increased cerebral pressure. Delayed neurodevelopment
and even the loss of brain tissue can result from VOGM. Another
common manifestation is heart failure (4). This is due to the shortened circuit of
venous return to the heart and the increased venous pressure, all
of which increase the preload on the neonatal heart, causing
high-output heart failure within the first few days of life
(5).
Currently, embolization is the main treatment method
for VOGM. Various studies have been conducted on embolization
techniques, such as transarterial and transvenous (6). Usually, there is a follow-up period of
6 months post-treatment. However, the rate of complications remains
high, with almost 41% of patients suffering from complications even
15 years following treatment (7).
The present study describes the case of a 6-month-old male infant
with VOGM and discusses the management techniques of VOGM and
evaluates prognosis.
Case report
Patient history
A 6-month-old male African American infant was
referred to the Department of Neurology, Nalanda Medical College
(Patna, India) clinic due to concerns regarding developmental
delays and an abnormal head circumference. The parents of the child
reported a progressive increase in head size since birth. Further
investigation revealed a systolic murmur and prominent scalp
veins.
Clinical examination
A physical examination confirmed macrocephaly,
bulging fontanelles and dilated scalp veins. A bruit was
auscultated over the anterior fontanelle, suggesting a vascular
abnormality. A neurological examination revealed hypotonia and
delayed developmental milestones.
Diagnostic workup
Neuroimaging was conducted to locate the suspected
vascular abnormality, and an immediate assessment was conducted
with a computed tomography (CT) scan to confirm its anatomical
position. The CT ruled out calcifications and bone structure
abnormalities, and helped to assess whether any complications had
already arisen, such as extensive hydrocephalus or a hemorrhage.
The patient underwent magnetic resonance imaging (MRI) with
contrast, which revealed a large arteriovenous malformation
involving the vein of Galen, causing the dilatation of the deep
venous system and hydrocephalus. The MRI provided high-resolution
images, particularly T1 and T2 weighted, and displayed soft tissue
contrast, which helped precisely distinguish the malformation from
surrounding structures. Subsequently, a magnetic resonance
angiography (MRA) was conducted to accurately map out the
angioarchitecture, including the feeding arteries, draining
arteries and the size of the vein of Galen aneurysm. This displayed
multiple abnormal arterial connections feeding into the dilated
vein of Galen, which when considering the presenting features of
hemodynamic impact on the heart and macrocephaly, is suggestive of
choroidal type of VOGM.
Hemodynamics were assessed, and a treatment plan,
the endovascular embolization, was devised based on the vascular
mapping. Finally, a brain arteriovenous malformation angiogram was
carried out to further visualize the vascular network and identify
the feeding and draining arteries. This is the gold standard for
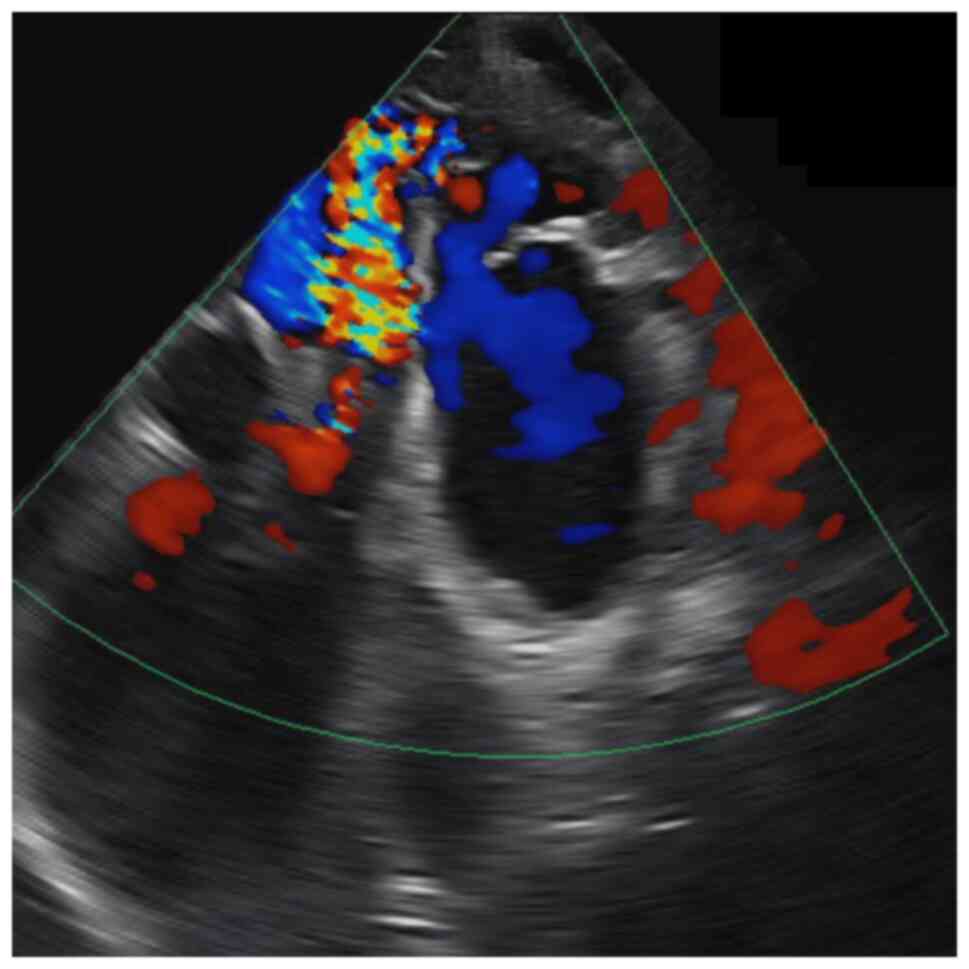
planning endovascular embolization. An additional echocardiography
was conducted (Fig. 1), to assess
the extent of heart involvement and whether the heart failure was
manageable. No typical heart failure was observed; however, there
was a high-flow cardiac murmur, which confirmed the hemodynamic
impact of the arteriovenous malformation.
Neuroimaging
The axial CT scan displayed hyperdense appearances
within the vascular channels, which are the abnormally dilated
venous structures in cerebral arteriovenous malformation, the VOGM.
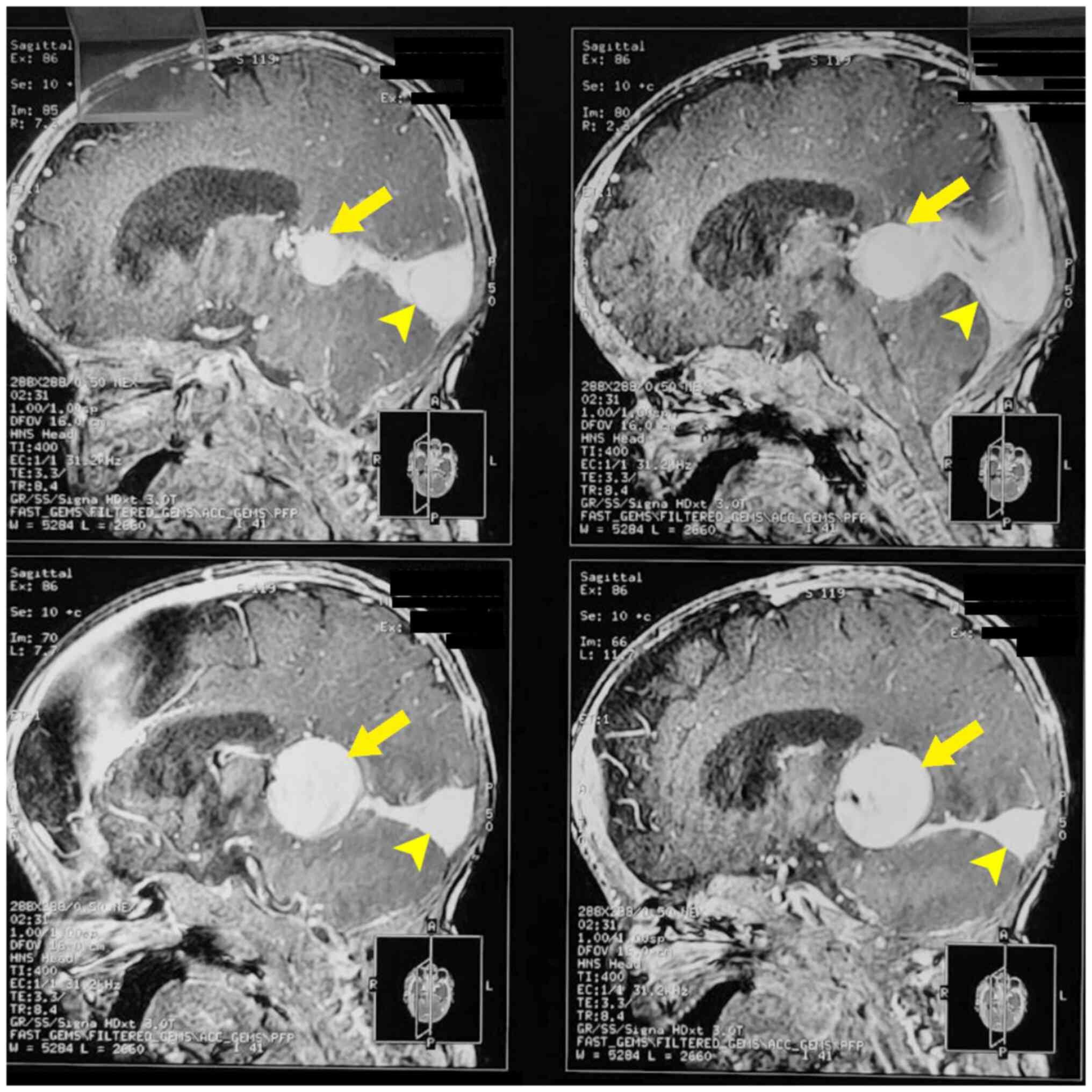
MRI scans carried out on the patient demonstrated a markedly
dilated median prosencephalic vein. The sagittal MRI T2-weighted
images are presented in Fig. 2. The
dilated galenic vein (yellow arrow; Fig.
2), also known as the median vein of prosencephalon, is located
midline in the cistern of the velum interpositum and drains into
the superior sagittal sinus (yellow arrowhead; Fig. 2).
An MRA was performed (image not available) and this
revealed multiple enlarged arterial branches from the anterior and
posterior cerebral arteries coalescing on the lateral margins of
the dilated VOGM.
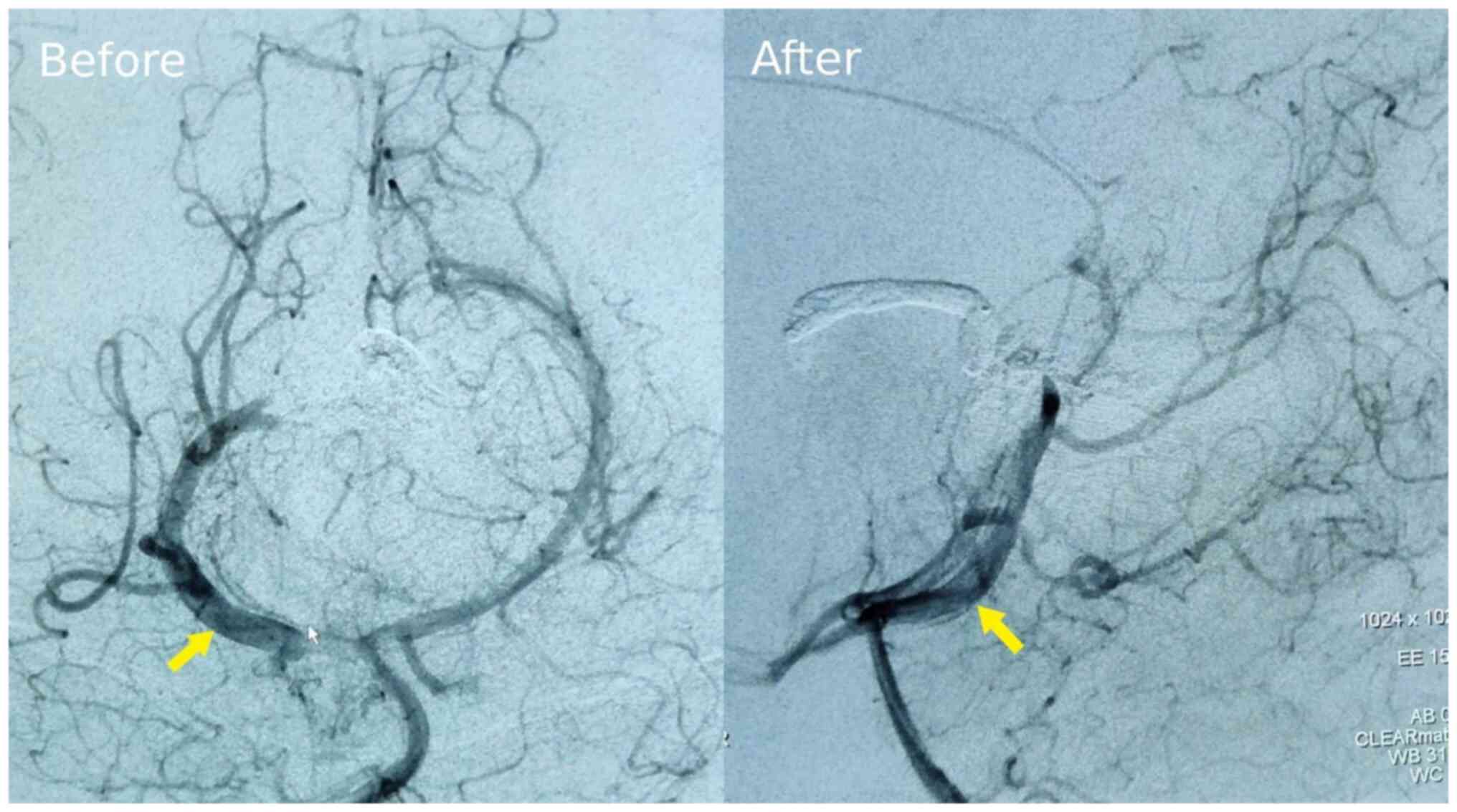
Of note, two brain artery malformation angiograms
were performed (Fig. 3). The image
on the left panel in Fig. 3, taken
at 6 months of age, displays the extensive network of abnormal
arterial connections feeding into the dilated median prosencephalic
vein. This is suggestive of the choroidal type of VOGM rather than
mural. The image on the right panel in Fig. 3, taken 2 weeks post-intervention,
demonstrates a reduction in the size and complexity of the
malformation, indicating successful embolization of the feeding
vessels. The yellow arrow points to the VOGM.
Diagnosis
The patient was diagnosed with choroidal VOGM with
associated hydrocephalus.
Management
A multidisciplinary team consisting of
neurosurgeons, interventional neuroradiologists and pediatric
cardiologists assessed the case and evaluated possible treatment
plans. Currently, the gold-standard treatment for VOGM is
endovascular embolization, and since the patient was 6 months old,
which is the optimum age for carrying out the procedure, the
decision was finalized. A surgical strategy was devised with the
aid of the MRA and arteriovenous malformation angiogram. An
endovascular embolization was performed, using a combination of
transarterial and transvenous routes, known as the kissing
microcatheter technique, to occlude abnormal vessels feeding into
the malformation, reducing blood flow and improving symptoms. In
addition, a ventriculoperitoneal shunt was placed to alleviate
hydrocephalus and control intracranial pressure. Following the
procedure, another arteriovenous malformation angiogram was
conducted to assess the situation and determine the success of the
intervention. Finally, regular follow-up appointments and imaging
studies were scheduled every 2 months for the subsequent 6 months
to monitor the patient's neurological development and assess the
effectiveness of the intervention.
Outcome
The post-operative recovery was marked by an
improvement in the neurodevelopmental milestones and a reduction in
the head circumference of the child. Follow-up imaging demonstrated
the successful embolization of the malformation with decreased
arteriovenous shunting. The bi-monthly follow-up appointments
demonstrated that the patient tolerated the procedure well and is
showing significant clinical improvement in his appetite, speech,
and cognitive functions. He did not experience any seizure
activity, and his follow-up echocardiology revealed an improvement
with no residual high-flow murmur.
Discussion
VOGM presents with various manifestations, all of
which are detrimental to fetal growth and neurodevelopment.
Although the onset of pathophysiology is in utero, numerous
signs and symptoms arise after birth when the protective
low-resistance circulation generated by the placenta is removed
(8). One of the more significant
manifestations is high-output cardiac failure (5). This is a result of the increased venous
pressure and venous return to the heart, causing right-sided heart
failure. The increased preload to the heart can display different
degrees of damage depending on the size of the fistula in the VOGM.
Small shunts are associated with an improved prognosis and usually
present at a later stage in the neonate's development, with
tachycardia and cardiomegaly (4).
Larger shunts are associated with a worse prognosis and present at
a much earlier stage with heart failure. The main manifestation of
VOGM is hydrocephalus, usually remedied by installing a
ventriculoperitoneal shunt to alleviate the pressure (8). Galen's dilating vein in an aneurysmal
manner leads to aqueduct compression and subsequent abnormal CSF
flow and venous congestion. This manifests as seizures and
developmental abnormalities, and in severe cases, it can lead to
‘melting brain’ syndrome (9). This
results from venous hypertension, and blood flow is mainly directed
to the fistula, causing ischemic damage and loss of brain tissue,
commonly called ‘melting brain’ syndrome (10).
There are two main types of VOGM: The choroidal and
mural types (11). The choroidal
type is where multiple fistulas feed into the VOGM, and it is more
severe as it has a higher risk of causing heart failure, as well as
other symptoms, such as macrocephaly and dilated orbital veins
(12). The case described herein was
classified as choroidal VOGM, as multiple fistulas were feeding
into the vein of Galen aneurysm and the patient presented with
classical features of heart involvement and macrocephaly. By
contrast, the mural type usually presents with one fistula in the
wall of the vein of Galen and is less severe, as it restricts more
blood flow, but at the cost of greater dilatation (13). In such cases, there is a lower risk
of heart failure; however, such cases usually present with
macrocephaly, hydrocephaly, seizures and commonly, with
developmental delays.
An ultrasound is usually the first investigation
carried out when suspecting fetal central nervous system
abnormalities; however, it is not as sensitive as a fetal MRI.
Currently, the classic presentation of VOGM would be found
prenatally in the third trimester with a fetal MRI or CT scan if
needed (14). This is more
advantageous as it displays the anatomy of the brain and provides a
clearer image of the damaged and abnormal structures (15). MRA/MRV angiography is the gold
standard for gauging a better view of the angioarchitecture, which
is necessary to plan the endovascular procedure to treat the
malformation (16). Angiography
plays a crucial role in the diagnosis and evaluation of the stage
of the disease (17). Yuval et
al (18) identified various
prognostic features that help better predict the course of the
disease, and the two most important are the number of feeding
arteries and the volume of venous drainage. Of note, more than five
arteries feeding into the VOGM can be considered an indirect
indication of potential massive shunting, which will likely lead to
severe congestive heart failure (CHF). Furthermore, the less
obstructed the venous drainage, the greater the volume overload and
returning blood pressure of the heart, which increases the risk of
developing CHF postnatally. Fetal echocardiography is also the gold
standard for gauging the extent of cardiological involvement, which
could indicate the nature of the VOGM (14).
Endovascular embolization is currently the only
well-established treatment for VOGM (8). Prior to the development of endovascular
embolization, the mortality rate of patients suffering from VOGM
was almost 100% (4). Since the
introduction of endovascular embolization, the prognosis of
patients with VOGM has markedly improved over time, with the
mortality decreasing from 17 to 12% and post-embolization
complications decreasing from 45 to 35% in the 1980s and 2000s,
respectively (19). The main
complications reported for post-embolization were hematomas (37%),
cerebral ischemia (6%) and hydrocephalus (3%). Yan et al
(19) reported that good clinical
outcome percentages increased substantially from 49 to 70% across
the same period of time. A previous systematic review of
endovascular embolization performed for 667 patients with VOGM
between 1987 and 2014 demonstrated that 23-70% of the neonates were
neurologically normal (20). In an
adjacent 15-year study, those who did not receive endovascular
embolization demonstrated a poor prognosis (21). A recent study by Lasjaunias et
al on 233 patients with VOGM receiving endovascular
embolization reported 10.6% mortality, and 74% were neurologically
normal. The complications were mainly delayed development and
psychomotor impairments (10).
Endovascular embolization is carried out at ~6
months of age unless there is an emergency situation that would
require earlier endovascular embolization, such as congestive heart
failure, that is refractory to medication. The aim is to ensure
there are no developmental delays caused by cerebral venous
hypertension and that the heart failure is manageable and not
terminal (8). The agents used to
embolize the fistulas are N-butyl-cyanoacrylate or onyx (22). Another more recent agent used is
detachable micro-coils; however, these are associated with a higher
risk of rupture and longer procedure durations. There are two
routes with which VOGM can be accessed for endovascular
embolization: Transarterial and transvenous, both of which achieve
heart failure control in different situations. The transarterial
route is more suitable for a small number of arterial feeders in
the VOGM, whilst the transvenous one is more suitable for VOGM with
many small arterial pedicles feeding into the fistula (3). The transvenous route is less favorable,
as it has the associated risk of impairing deep venous drainage and
subsequent aneurysm perforation (8).
Currently, a combination of transarterial and transvenous routes,
known as the kissing microcatheter technique has demonstrated
promising results (6). Near complete
angiographically confirmed closure of the VOGM in 79% of patients,
and 69% reported normal outcomes post combination endovascular
embolization routes.
The patient described herein presented with
classical features of VVOGM that align with previous case reports
on VOGM, which was later confirmed with MRI scans and
echocardiography. The post-operative recovery was marked by an
improvement in the neurodevelopmental milestones and a reduction in
the head circumference of the child. Follow-up imaging demonstrated
the successful embolization of the malformation with decreased
arteriovenous shunting. This is a novel case as the interventional
radiology specialty approach demonstrates the optimum diagnostic
and preplanning investigations for cases of vascular origin. The
learning point to appreciate is the need for interdisciplinary
collaboration, particularly between interventional radiologists and
neurosurgeons. Furthermore, the benefit of conducting a
comprehensive radiological study of the case to optimize planning
for a complex surgery. In addition, the kissing microcatheter
endovascular embolization technique that was performed highlights
the importance of considering this innovative surgical option in
complex vascular cases. The limitation of this approach is the
duration of these procedures that, in emergency cases, would be
overlooked for more definitive management rather than undertaking
an investigative approach. However, it is important to note that in
the majority of cases of VOGM, if hemodynamic impact is minimal and
manageable, then waiting until the patient is 6 months old is the
convention as it is the optimal age for surgical intervention.
Therefore, there is sufficient time to carry out a comprehensive
neuroimaging investigation.
A limitation of the present study was the lack of an
ability to generalize, as this is a case report meant to
demonstrate a case of optimal management of VOGM with specific
focus on interventional radiology and the kissing catheter
endovascular embolization technique. Furthermore, there is no
possibility to establish better treatment efficacy, as cases with
VOGM are critical, with no room for trialing, as the patients are
at an age of critical neurological development.
In conclusion, intracranial VOGM poses a complex
challenge requiring a multidisciplinary approach for optimal
management. In conjunction with interventional radiology,
endovascular embolization has proven to markedly improve outcomes
in affected infants. The present case report demonstrates the novel
and efficient kissing microcatheter endovascular embolization
technique and that suggests that this is a surgical option that
could be considered more often when devising management plans.
Furthermore, long-term follow-up is crucial to monitor potential
complications and ensure ongoing neurodevelopmental progress.
Acknowledgements
Not applicable.
Funding
Funding: No funding was received.
Availability of data and materials
The datasets used and/or analyzed during the current
study are available from the corresponding author on reasonable
request.
Authors' contributions
ZAE, RR, MAT, MZS, PKY, JAH and HM contributed to
the conception, design, data collection, analysis, and writing of
the present case report. RR was responsible for the treatment and
management of the patient. RR and ZAE confirm the authenticity of
all the raw data. All authors have read and approved the final
manuscript.
Ethics approval and consent to
participate
Ethical approval was obtained from the Nalanda
Medical College Ethics Committee. The parents of the patient,
provided informed consent to participate in the study.
Patient consent for publication
Written consent for publication was obtained from
parents of the patient involved in the present case report for the
publication of the patient's data and any related images.
Competing interests
The authors declare that they have no competing
interests.
References
|
1
|
Safadi AO and Tadi P: Anatomy, head and
neck, cerebral venous system. PubMed. Published 2022. https://www.ncbi.nlm.nih.gov/books/NBK560496/.
|
|
2
|
Recinos PF, Rahmathulla G, Pearl M,
Recinos VR, Jallo GI, Gailloud P and Ahn ES: Vein of galen
malformations: Epidemiology, clinical presentations, management.
Neurosurg Clin N Am. 23:165–177. 2012.PubMed/NCBI View Article : Google Scholar
|
|
3
|
Puvabanditsin S, Mehta R, Palomares K,
Gengel N, Da Silva CF, Roychowdhury S, Gupta G, Kashyap A and
Sorrentino D: Vein of Galen malformation in a neonate: A case
report and review of endovascular management. World J Clin Pediatr.
6:103–109. 2017.PubMed/NCBI View Article : Google Scholar
|
|
4
|
Hoang S, Choudhri O, Edwards M and Guzman
R: Vein of Galen malformation. Neurosurg Focus.
27(E8)2009.PubMed/NCBI View Article : Google Scholar
|
|
5
|
Spazzapan P, Milosevic Z and Velnar T:
Vein of galen aneurismal malformations- clinical characteristics,
treatment and presentation: Three cases report. World J Clin Cases.
7:855–862. 2019.PubMed/NCBI View Article : Google Scholar
|
|
6
|
Meila D, Hannak R, Feldkamp A,
Schlunz-Hendann M, Mangold A, Jacobs C, Papke K and Brassel F: Vein
of galen aneurysmal malformation: Combined transvenous and
transarterial method using a ‘kissing microcatheter technique’.
Neuroradiology. 54:51–59. 2012.PubMed/NCBI View Article : Google Scholar
|
|
7
|
Nuñez FB and Dohna-Schwake C:
Epidemiology, diagnostics, and management of vein of galen
malformation. Pediatr Neurol. 119:50–55. 2021.PubMed/NCBI View Article : Google Scholar
|
|
8
|
Bhattarai K, Patel M, Garcia M and Litra
F: Vein of galen aneurysmal malformation: A case report and
literature review. Cureus. 15(e51305)2023.PubMed/NCBI View Article : Google Scholar
|
|
9
|
Chow M, Cooke DL, Fullerton HJ, Amans MR,
Narvid J, Dowd CF, Higashida RT, Halbach VV and Hetts SW:
Radiological and clinical features of vein of galen malformations.
J Neurointerv Surg. 7:443–448. 2015.PubMed/NCBI View Article : Google Scholar
|
|
10
|
Lasjaunias PL, Chng SM, Sachet M, Alvarez
H, Rodesch G and Garcia-Monaco R: The management of vein of galen
aneurysmal malformations. Neurosurgery. 59 (5 Suppl 3):S184–S194.
2006.PubMed/NCBI View Article : Google Scholar
|
|
11
|
Lasjaunias P, Ter Brugge K and Berenstein
A: Vein of Galen aneurismal malformation. In: Surgical
Neuroangiography 3. Clinical and Interventional Aspects in
Children. Vol 3. 2nd edition. Springer, Berlin, pp105-226,
2006.
|
|
12
|
Mortazavi MM, Griessenauer CJ, Foreman P,
Shahripour RB, Shoja MM, Rozzelle CJ, Tubbs RS, Fisher WS III and
Fukushima T: Vein of galen aneurysmal malformations: Critical
analysis of the literature with proposal of a new classification
system. J Neurosurg Pediatr. 12:293–306. 2013.PubMed/NCBI View Article : Google Scholar
|
|
13
|
O'Brien MS and Schechter MM: Arteriovenous
malformations involving the galenic system. Am J Roentgenol Radium
Ther Nucl Med. 110:50–55. 1970.PubMed/NCBI View Article : Google Scholar
|
|
14
|
Wagner MW, Vaught AJ, Poretti A, Blakemore
KJ and Huisman TA: Vein of galen aneurysmal malformation:
Prognostic markers depicted on fetal MRI. Neuroradiol. 28:72–75.
2014.PubMed/NCBI View Article : Google Scholar
|
|
15
|
Kośla K, Majos M, Polguj M,
Antosik-Biernacka A, Stefańczyk L and Majos A: Prenatal diagnosis
of a vein of galen aneurysmal malformation with MR imaging-report
of two cases. Pol J Radiol. 78:88–92. 2013.PubMed/NCBI View Article : Google Scholar
|
|
16
|
Brunelle F: Brain vascular malformations
in the fetus: Diagnosis and prognosis. Childs Nerv Syst.
19:524–528. 2003.PubMed/NCBI View Article : Google Scholar
|
|
17
|
Deloison B, Chalouhi GE, Sonigo P, Zerah
M, Millischer AE, Dumez Y, Brunelle F, Ville Y and Salomon LJ:
Hidden mortality of prenatally diagnosed vein of galen aneurysmal
malformation: Retrospective study and review of the literature.
Ultrasound Obstet Gynecol. 40:652–658. 2012.PubMed/NCBI View Article : Google Scholar
|
|
18
|
Yuval Y, Lerner A, Lipitz S, Rotstein Z,
Hegesh J and Achiron R: Prenatal diagnosis of vein of Galen
aneurysmal malformation: Report of two cases with proposal for
prognostic indices. Prenat Diagn. 17:972–977. 1997.PubMed/NCBI
|
|
19
|
Yan J, Wen J, Gopaul R, Zhang CY and Xiao
SW: Outcome and complications of endovascular embolization for vein
of galen malformations: A systematic review and meta-analysis. J
Neurosurg. 123:872–890. 2015.PubMed/NCBI View Article : Google Scholar
|
|
20
|
Karadeniz L, Coban A, Sencer S, Has R,
Ince Z and Can G: Vein of galen aneurysmal malformation: Prenatal
diagnosis and early endovascular management. J Chin Med Assoc.
74:134–137. 2011.PubMed/NCBI View Article : Google Scholar
|
|
21
|
Frawley GP: Clinical course and medical
management of neonates with severe cardiac failure related to vein
of galen malformation. Arch Dis Child Fetal Neonatal Ed.
87:F144–F149. 2002.PubMed/NCBI View Article : Google Scholar
|
|
22
|
Gailloud P, O'Riordan DP, Burger I,
Levrier O, Jallo G, Tamargo RJ, Murphy KJ and Lehmann CU: Diagnosis
and management of vein of galen aneurysmal malformations. J
Perinatol. 25:542–551. 2005.PubMed/NCBI View Article : Google Scholar
|