Introduction
Pituitary tumor-transforming gene (PTTG) was first
identified in rat pituitary tumor by differential display PCR
(1). At present, three members of
the PTTG family have been identified in humans (2). Among those, the hPTTG1 has a human
securin function, while playing an important role in tumorigenesis
and tumor metastasis (3,4). The hPTTG1 was found to be
overexpressed in a variety of cancers, such as lung cancer, thyroid
tumor, as well as ovarian and esophageal cancer (5–8).
Normally, the hPTTG1 expression can only be detected in testis,
thymus, embryo and liver (4). The
abnormal expressions of hPTTG1 implied tumorigenesis or tumor
metastasis. However, the correlation between the expression of
hPTTG1 and gastric cancer has yet to be clarified.
Cancer has been the leading cause of mortality in
many countries (9,10), with gastric cancer being the second
leading cause of cancer-related mortality in both males and females
worldwide (11). Gastric cancer is
still widespread in China, as seen in a statistical report
demonstrating that China accounts for approximately 50% of the
global gastric cancer burden (11). It is widely accepted that early
discovery indicates a good prognosis in cancer therapy. Thus, it is
urgent to develop a facilitating technique to detect tumors, at
least tumor metastasis.
The hPTTG1 gene has transcriptional activity and
securin functions (12).
Overexpression of the hPTTG1 promotes lymph node metastasis in
human esophageal carcinoma (8).
Findings of previous studies revealed that the downregulation of
the expression of hPTTG1 resulted in the suppression of tumor
growth (13,14). We hypothesized that the abnormal
overexpression of hPTTG1 was involved in tumorigenesis and tumor
metastases in gastric cancer.
Lymph node metastasis is an important prognostic
factor in early gastric cancer, which indicates the risk of
recurrence (15). Thus it is
necessary to detect whether lymph node metastasis is present prior
to curative resection. The removal of lymph nodes should be carried
out during surgery. Therefore, a reliable method is needed to
accurately detect lymph node metastasis in primary gastric cancer.
However, the expression and correlation of hPTTG1 with
clinicopathological parameters in gastric cancer has not been
reported yet. Therefore, SP immunohistochemical technology was
applied to investigate the expression of hPTTG1 in gastric cancer
and adjacent normal tissues and explore its correlation with
clinicopathological features and significance.
The aim of the present study was to develop a
facilitation method to detect gastric cancer or lymph node
metastasis in gastric cancer. We found that the expression of
hPTTG1 was associated with gastric cancer and lymph node metastasis
in gastric cancer. We suggested that the immunohistochemistry
staining of hPTTG1 may be used as an effective tool to reveal its
clinicopathological parameters and significance in gastric
cancer.
Materials and methods
Patients and tissues
Gastric cancer and adjacent normal tissues were
collected at the time of surgery from patients with gastric cancer
between May, 2004 and May, 2008. The tumor samples were confirmed
by histopathological examination. Prior to surgery, the patients
did not receive radiation treatment, chemo-treatment or biological
therapy for the tumor. The adjacent normal tissues were obtained
from a site 5 cm away from the tumor and confirmed histologically.
These samples were obtained from 28 males and 17 females. The
average age was 59.5 years (range, 45–79). The
tumor-node-metastasis (TNM) stages were classified according to the
Union for International Cancer Control (UICC) 1987 of TNM
classification. Five cases of stage I, 10 cases of stage II, 20
cases of stage III and 10 cases of stage IV were involved in our
study. Of the 45 cases, 31 had a lymph node metastasis, and 14
cases were not detected at surgery. In the present study, there
were 16 cases with well-differentiated, 15 with
moderately-differentiated and 14 cases with poorly-differentiated
adenocarcinoma.
Immunohistochemistry
An SP immunohistochemical staining kit was purchased
from Beijing Zhongshan Bio-tech Co., Ltd. Immunohistochemistry was
performed according to the manufacturer's instructions. Briefly,
gastric cancer tissues and adjacent normal tissues were dissected
out, followed by fixing in Bouin's solution for 24 h. After an
extensive wash in 70% ethanol, the samples were dehydrated and
embedded in paraffin. The paraffin blocks were cut at 5 μm. The
deparaffinized sections were incubated with a 3% hydrogen peroxide
solution for 30 min. After being washed with PBS solution twice,
the slides with sample sections were incubated with 0.01 M citrate
solution at 95°C for 10 min to retrieve the epitope. After cooling
to room temperature, the glass slides were rinsed with PBS solution
twice. The slides were incubated with NGS-PBS (PBS containing 10%
normal goat serum) for 30 min, and then washed with PBS
solution.
Primary antibody, rabbit anti-human PTTG1 (Santa
Cruz Biotechnology Inc., Santa Cruz, CA, USA) at a dilution of
1:100 was dropped onto the slides. After incubation at 37°C for 1 h
the slides were washed with PBS solution twice. Subsequently, the
secondary antibody (biotin-conjugated goat anti-rabbit IgG, Beijing
Zhongshan Bio-tech Co., Ltd., China) at a dilution of 1:100 was
dropped onto the slides and incubated at 37°C for 30 min. After
washing with PBS solution twice, the slides were applied to
3,3′-diaminobenzidine (Dako Tech Co., Ltd,, Denmark) staining. The
positive slides were defined according to a previous study
(16). Briefly, the slides were
defined as i) (+) if from 10–25% of highest possible frequency
(HPF), showing a positive signal with a magnification of ×400; ii)
(++) if from 25–50% of HPF, showing positive signal with
magnification at ×400 and iii) (+++) if >50% of HPF, showing
positive signal with a magnification of ×400. Each slide was
examined by 2 pathologists, respectively. In case of a
disagreement, the slide was determined by a third pathologist.
Statistical analyses
Data were presented as the mean ± SD. Statistical
analysis was performed using SPSS 11.0.0 (SPSS Inc., Chicago).
Correlations were evaluated using the Chi-square test. p<0.05
was considered to indicate a statistically significant
difference.
Results
Expression of hPTTG1 in gastric cancer
and the adjacent normal tissues
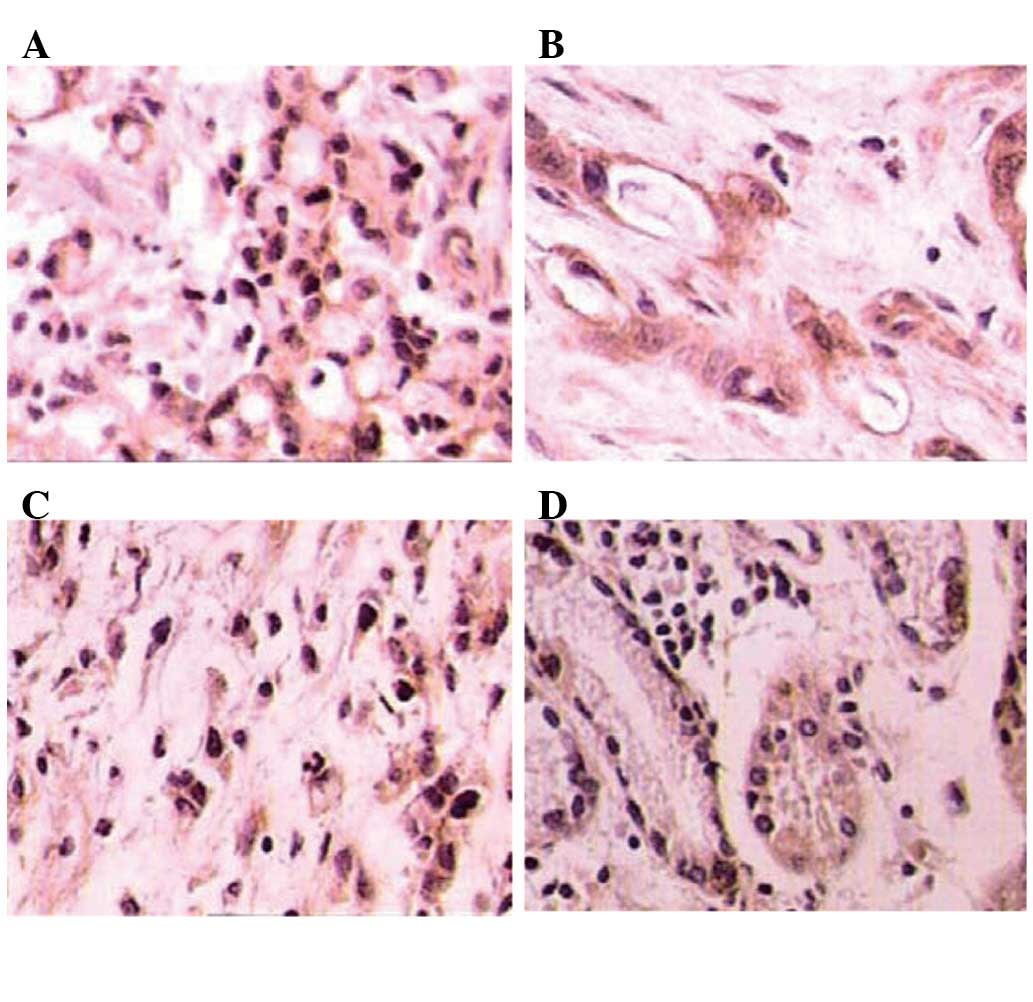
We detected the expression of hPTTG1 in different
cell types and tissues (Fig. 1). A
number of nuclei exhibited dark staining in signet-ring cell
carcinoma, poorly differentiated adenocarcinoma and adjacent normal
tissues, possibly indicating a poor prognosis. The statistical
analysis revealed that the expression of hPTTG1 was associated with
gastric cancer as compared to the adjacent normal tissues (Table I).
 | Table IRelationship between hPTTG1 expression
and types of tissue. |
Table I
Relationship between hPTTG1 expression
and types of tissue.
| Types of tissue | n | −− | + | ++ | +++ | Positive rate
(%) | χ2 | P-value |
|---|
| Gastric cancer | 45 | 18 | 8 | 8 | 11 | 60.00 | 16.875 | 0.000 |
| Adjacent normal
tissues | 45 | 37 | 1 | 3 | 4 | 17.78 | | |
Correlation between hPTTG1 expression and
the clinicopathological variables of gastric cancer
The statistical analysis demonstrated that the
expression of hPTTG1 was associated with differentiation levels,
clinical classification and lymph node metastasis in gastric
cancer. This finding suggests that the overexpression of hPTTG1 may
indicate poor prognosis. Moreover, we found that the expression of
hPTTG1 did not correlate with gender, age or pathological types
(Table II).
 | Table IIRelationship between hPTTG1 expression
and clinicopathologic parameters in gastric cancer. |
Table II
Relationship between hPTTG1 expression
and clinicopathologic parameters in gastric cancer.
| Clinicopathologic
variables | n | −− | + | ++ | +++ | Positive rate
(%) | χ2 | P-value |
|---|
| Gender |
| Male | 28 | 12 | 4 | 4 | 8 | 57.14 | 1.079 | 0.299 |
| Female | 17 | 10 | 3 | 1 | 3 | 41.18 | | |
| Age, years |
| ≥60 | 33 | 13 | 5 | 4 | 11 | 60.61 | 0.019 | 0.891 |
| <60 | 12 | 5 | 2 | 3 | 2 | 58.33 | | |
| Pathological
types |
| Adenocarcinoma | 34 | 14 | 4 | 7 | 9 | 58.82 | 0.311 | 0.856 |
| Mucinous
adenocarcinoma | 8 | 4 | 0 | 2 | 2 | 50.00 | | |
| Signet-ring cell
carcinoma | 3 | 1 | 0 | 2 | 0 | 66.67 | | |
| Differentiation
levels |
| High | 16 | 13 | 2 | 1 | 0 | 18.75 | 13.582 | 0.001 |
| Moderate | 15 | 7 | 4 | 3 | 2 | 60.00 | | |
| Poor | 14 | 2 | 3 | 1 | 8 | 85.71 | | |
| Clinical
classification |
| TI+TII | 15 | 10 | 3 | 2 | 0 | 33.33 | 6.667 | 0.010 |
| TIII+TIV | 30 | 8 | 5 | 7 | 10 | 73.33 | | |
| Lymph node
metastasis |
| Presence | 31 | 8 | 5 | 6 | 12 | 74.19 | 6.075 | 0.014 |
| Absence | 14 | 9 | 3 | 2 | 0 | 35.71 | | |
Discussion
The abnormal expression of hPTTG1 is involved in
tumorigenesis and tumor metastasis (17). Our study showed that the
immunohistochemical analysis of hPTTG1 was suitable for detecting
different cell types and tissues in gastric cancer. We also showed
that the expression of hPTTG1 is correlated with gastric cancer as
compared to the adjacent normal tissues. This finding indicated
that the expression of hPTTG1 may be a useful reference to gastric
cancer detection.
Results from our study demonstrated that the
expression of hPTTG1 was associated with differentiation levels,
clinical classification and lymph node metastasis in primary
gastric cancer. Poorly differentiated cancer and higher clinical
classification indicated poor prognosis. Thus, this finding
suggests that the expression of hPTTG1 is associated with poor
prognosis. Lymph node metastasis is the most important prognostic
factor for patients with early gastric cancer. Since the presence
of lymph node metastasis indicates a poor prognosis on patients
with early gastric cancer, it is suggested that the first and
second tier lymph nodes be removed during surgery (15). The removal of lymph nodes was
reported to be associated with an extended period of survival
(18). Therefore, the detection of
lymph node metastasis is valuable for early gastric cancer
patients. We suggest that the expression of hPTTG1 be considered in
early gastric cancer therapy.
A statistical report of 2008 showed that
age-standardized incidence rates are approximately twice as high in
men as in women (11). The
expression of hPTTG1, however, did not correlate with gender.
Moreover, we found that the expression of hPTTG1 was also not
associated with age or pathological types.
Our results describe hPTTG1 expression as a
potential tool for the assessment of tumor aggressiveness. The
immunohistochemistry of hPTTG1 expression may be a convenient way
to large-scale analyses or routine pathological diagnosis. This
tool may have special value for early gastric cancer patients, for
the detection of lymph node metastasis and extention of their
period of survival.
Acknowledgements
This study was supported by a grant from the
National Special Funds for the Health Research Foundation of China
(no. 200802112) and the National Natural Science Foundation of
China (no. 81071991).
References
|
1
|
Pei L and Melmed S: Isolation and
characterization of a pituitary tumor-transforming gene (PTTG). Mol
Endocrinol. 11:433–441. 1997. View Article : Google Scholar : PubMed/NCBI
|
|
2
|
Zhang X, Horwitz GA, Prezant TR, Valentini
A, Nakashima M, Bronstein MD and Melmed S: Structure, expression,
and function of human pituitary tumor-transforming gene (PTTG). Mol
Endocrinol. 13:156–166. 1999. View Article : Google Scholar : PubMed/NCBI
|
|
3
|
Chen L, Puri R, Lefkowitz EJ and Kakar SS:
Identification of the human pituitary tumor transforming gene
(hPTTG) family: molecular structure, expression, and chromosomal
localization. Gene. 248:41–50. 2000. View Article : Google Scholar : PubMed/NCBI
|
|
4
|
Hamid T and Kakar SS: PTTG and cancer.
Histol Histopathol. 18:245–251. 2003.
|
|
5
|
Shah PP, Fong MY and Kakar SS: PTTG
induces EMT through integrin alpha(V)beta(3)-focal adhesion kinase
signaling in lung cancer cells. Oncogene. Nov 14–2011.(E-pub ahead
of print).
|
|
6
|
Chintharlapalli S, Papineni S, Lee SO, Lei
P, Jin UH, Sherman SI, Santarpia L and Safe S: Inhibition of
pituitary tumor-transforming gene-1 in thyroid cancer cells by
drugs that decrease specificity proteins. Mol Carcinogen.
50:655–667. 2011. View
Article : Google Scholar : PubMed/NCBI
|
|
7
|
El-Naggar SM, Malik MT and Kakar SS: Small
interfering RNA against PTTG: A novel therapy for ovarian cancer.
Int J Oncol. 31:137–143. 2007.PubMed/NCBI
|
|
8
|
Yan S, Zhou CQ, Lou XM, Xiao ZF, Zhu HX,
Wang QF, Wang YH, Lu N, He S, Zhan QM, et al: PTTG overexpression
promotes lymph node metastasis in human esophageal squamous cell
carcinoma. Cancer Res. 69:3283–3290. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
9
|
Jung KW, Park S, Kong HJ, Won YJ, Boo YK,
Shin HR, Park EC and Lee JS: Cancer Statistics in Korea: incidence,
mortality and survival in 2006–2007. J Korean Med Sci.
25:1113–1121. 2010.
|
|
10
|
Boyle P and Ferlay J: Cancer incidence and
mortality in Europe, 2004. Ann Oncol. 16:481–488. 2005. View Article : Google Scholar : PubMed/NCBI
|
|
11
|
Ferlay J, Shin HR, Bray F, Forman D,
Mathers C and Parkin DM: Estimates of worldwide burden of cancer in
2008: GLOBOCAN 2008. Int J Cancer. 127:2893–2917. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
12
|
Tong Y and Eigler T: Transcriptional
targets for pituitary tumor-transforming gene-1. J Mol Endocrinol.
43:179–185. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
13
|
Panguluri SK, Yeakel C and Kakar SS: PTTG:
an important target gene for ovarian cancer therapy. J Ovarian Res.
1:62008. View Article : Google Scholar : PubMed/NCBI
|
|
14
|
Kakar SS and Malik MT: Suppression of lung
cancer with siRNA targeting PTTG. Int J Oncol. 29:387–395.
2006.PubMed/NCBI
|
|
15
|
Okamura T, Tsujitani S, Korenaga D,
Haraguchi M, Baba H, Hiramoto Y and Sugimachi K: Lymphadenectomy
for cure in patients with early gastric cancer and lymph node
metastasis. Am J Surg. 155:476–480. 1988. View Article : Google Scholar : PubMed/NCBI
|
|
16
|
Lu CD, Altieri DC and Tanigawa N:
Expression of a novel antiapoptosis gene, survivin, correlated with
tumor cell apoptosis and p53 accumulation in gastric carcinomas.
Cancer Res. 58:1808–1812. 1998.PubMed/NCBI
|
|
17
|
Salehi F, Kovacs K, Scheithauer BW, Lloyd
RV and Cusimano M: Pituitary tumor-transforming gene in endocrine
and other neoplasms: a review and update. Endocr Relat Cancer.
15:721–743. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
18
|
Sano T, Sasako M, Kinoshita T and Maruyama
K: Recurrence of early gastric cancer. Follow-up of 1475 patients
and review of the Japanese literature. Cancer. 72:3174–3178. 1993.
View Article : Google Scholar : PubMed/NCBI
|