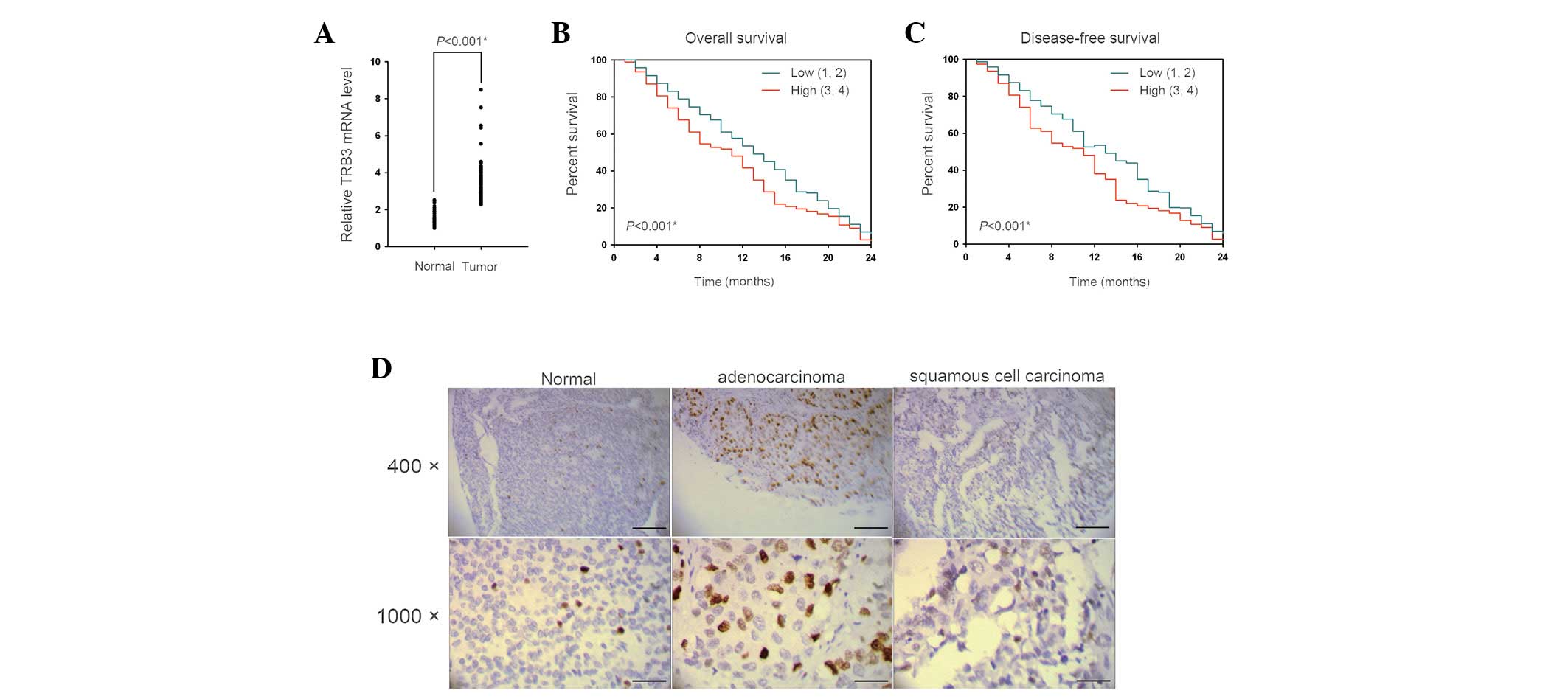
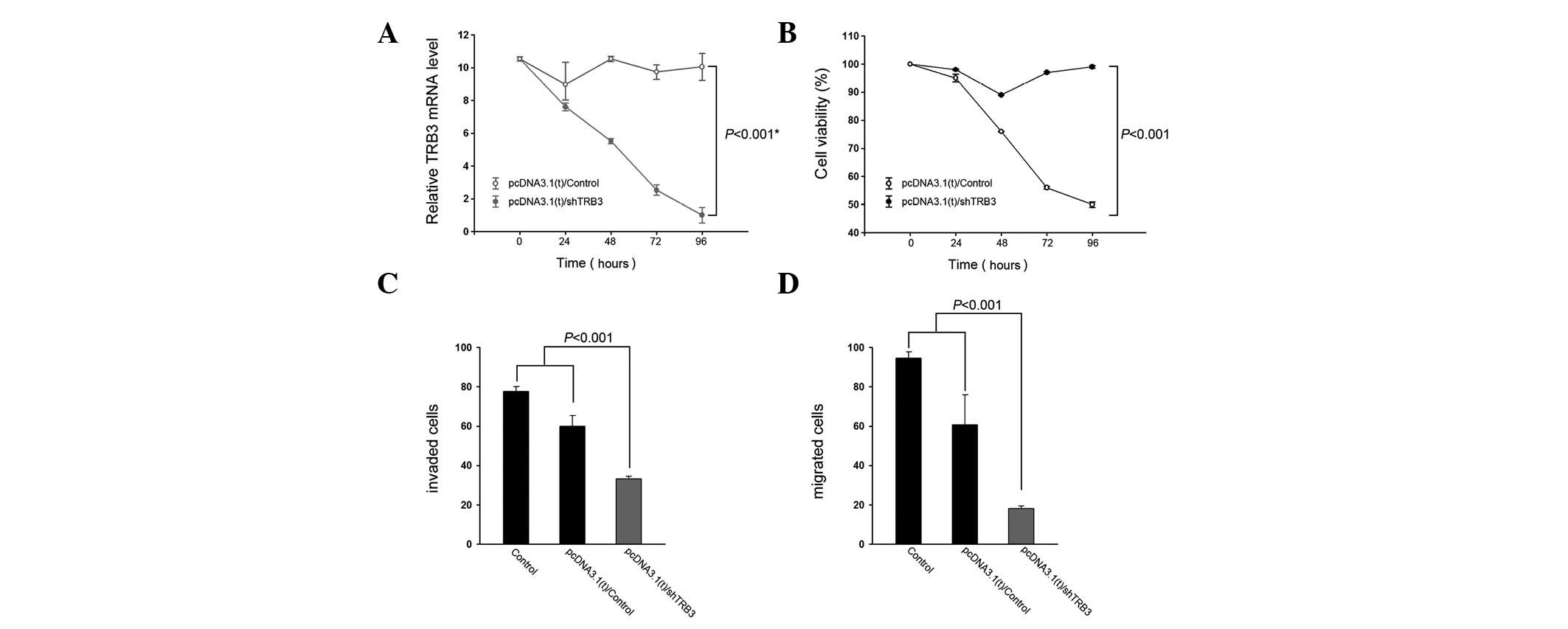
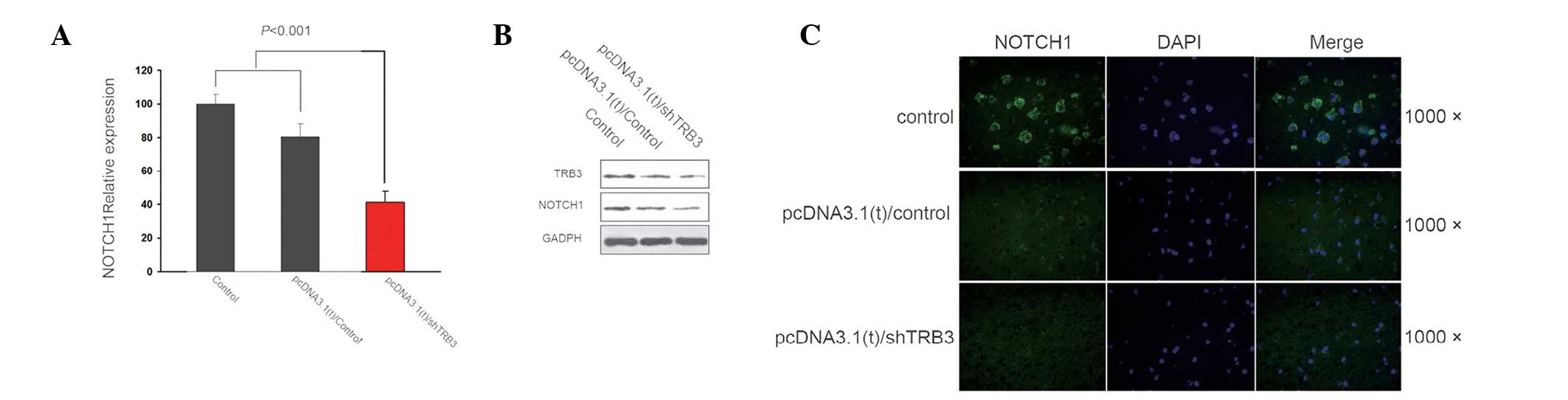
|
1
|
Boudeau J, Miranda-Saavedra D, Barton GJ
and Alessi DR: Emerging roles of pseudokinases. Trends Cell Biol.
16:443–452. 2006. View Article : Google Scholar
|
|
2
|
Izrailit J, Berman HK, Datti A, Wrana JL
and Reedijk M: High throughput kinase inhibitor screens reveal TRB3
and MAPK-ERK/TGFβ pathways as fundamental Notch regulators in
breast cancer. Proc Natl Acad Sci USA. 110:1714–1719.
2013.PubMed/NCBI
|
|
3
|
Raja E: Cross-regulation between TGFβ/BMP
Signalling and the metabolic LKB1 pathway. Doctoral thesis. Uppsala
University; 2012
|
|
4
|
Bowers AJ, Scully S and Boylan JF: SKIP3,
a novel Drosophila tribbles ortholog, is overexpressed in
human tumors and is regulated by hypoxia. Oncogene. 22:2823–2835.
2003.PubMed/NCBI
|
|
5
|
Du K, Herzig S, Kulkarni RN and Montminy
M: TRB3: a tribbles homolog that inhibits Akt/PKB activation by
insulin in liver. Science. 300:1574–1577. 2003. View Article : Google Scholar : PubMed/NCBI
|
|
6
|
Wennemers M, Bussink J, Scheijen B, et al:
Tribbles homolog 3 denotes a poor prognosis in breast cancer and is
involved in hypoxia response. Breast Cancer Res. 13:R822011.
View Article : Google Scholar : PubMed/NCBI
|
|
7
|
Miyoshi N, Ishii H, Mimori K, et al:
Abnormal expression of TRIB3 in colorectal cancer: a novel marker
for prognosis. Br J Cancer. 101:1664–1670. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
8
|
Reedijk M, Odorcic S, Chang L, et al:
High-level coexpression of JAG1 and NOTCH1 is observed in human
breast cancer and is associated with poor overall survival. Cancer
Res. 65:8530–8537. 2005. View Article : Google Scholar : PubMed/NCBI
|
|
9
|
Lee CW, Raskett CM, Prudovsky I and
Altieri DC: Molecular dependence of estrogen receptor-negative
breast cancer on a notch-survivin signaling axis. Cancer Res.
68:5273–5281. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
10
|
Leong KG, Niessen K, Kulic I, et al:
Jagged1-mediated Notch activation induces epithelial-to-mesenchymal
transition through Slug-induced repression of E-cadherin. J Exp
Med. 204:2935–2948. 2007. View Article : Google Scholar : PubMed/NCBI
|
|
11
|
Rustighi A, Tiberi L, Soldano A, et al:
The prolyl-isomerase Pin1 is a Notch1 target that enhances Notch1
activation in cancer. Nat Cell Biol. 11:133–142. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
12
|
Shimizu M, Cohen B, Goldvasser P, Berman
H, Virtanen C and Reedijk M: Plasminogen activator uPA is a direct
transcriptional target of the JAG1-Notch receptor signaling pathway
in breast cancer. Cancer Res. 71:277–286. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
13
|
Honjo T: The shortest path from the
surface to the nucleus: RBP-J kappa/Su(H) transcription factor.
Genes Cells. 1:1–9. 1996. View Article : Google Scholar : PubMed/NCBI
|
|
14
|
Weijzen S, Rizzo P, Braid M, et al:
Activation of Notch-1 signaling maintains the neoplastic phenotype
in human Ras-transformed cells. Nat Med. 8:979–986. 2002.
View Article : Google Scholar : PubMed/NCBI
|
|
15
|
Pece S, Serresi M, Santolini E, et al:
Loss of negative regulation by Numb over Notch is relevant to human
breast carcinogenesis. J Cell Biol. 167:215–221. 2004. View Article : Google Scholar : PubMed/NCBI
|
|
16
|
Colaluca IN, Tosoni D, Nuciforo P, et al:
NUMB controls p53 tumour suppressor activity. Nature. 451:76–80.
2008. View Article : Google Scholar : PubMed/NCBI
|
|
17
|
Lee CW, Simin K, Liu Q, et al: A
functional Notch-survivin gene signature in basal breast cancer.
Breast Cancer Res. 10:R972008. View
Article : Google Scholar : PubMed/NCBI
|
|
18
|
Yamaguchi N, Oyama T, Ito E, et al: NOTCH3
signaling pathway plays crucial roles in the proliferation of
ErbB2-negative human breast cancer cells. Cancer Res. 68:1881–1888.
2008. View Article : Google Scholar : PubMed/NCBI
|
|
19
|
Dent R, Trudeau M, Pritchard KI, et al:
Triple-negative breast cancer: clinical features and patterns of
recurrence. Clin Cancer Res. 13:4429–4434. 2007. View Article : Google Scholar
|
|
20
|
Farnie G and Clarke RB: Mammary stem cells
and breast cancer - role of Notch signalling. Stem Cell Rev.
3:169–175. 2007. View Article : Google Scholar : PubMed/NCBI
|
|
21
|
Yin X, Wolford CC, Chang YS, et al: ATF3,
an adaptive-response gene, enhances TGF{beta} signaling and
cancer-initiating cell features in breast cancer cells. J Cell Sci.
123:3558–3565. 2010.PubMed/NCBI
|
|
22
|
Kalluri R and Weinberg RA: The basics of
epithelial-mesenchymal transition. J Clin Invest. 119:1420–1428.
2009. View
Article : Google Scholar : PubMed/NCBI
|
|
23
|
Gavert N and Ben-Ze’ev A:
Epithelial-mesenchymal transition and the invasive potential of
tumors. Trends Mol Med. 14:199–209. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
24
|
Timmerman LA, Grego-Bessa J, Raya A, et
al: Notch promotes epithelial-mesenchymal transition during cardiac
development and oncogenic transformation. Genes Dev. 18:99–115.
2004. View Article : Google Scholar
|
|
25
|
Xie L, Law BK, Chytil AM, Brown KA, Aakre
ME and Moses HL: Activation of the Erk pathway is required for
TGF-beta1-induced EMT in vitro. Neoplasia. 6:603–610. 2004.
View Article : Google Scholar : PubMed/NCBI
|
|
26
|
Sethi N, Dai X, Winter CG and Kang Y:
Tumor-derived JAGGED1 promotes osteolytic bone metastasis of breast
cancer by engaging notch signaling in bone cells. Cancer cell.
19:192–205. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
27
|
Grosshans J and Wieschaus E: A genetic
link between morphogenesis and cell division during formation of
the ventral furrow in Drosophila. Cell. 101:523–531. 2000.
View Article : Google Scholar : PubMed/NCBI
|
|
28
|
Seher TC and Leptin M: Tribbles, a
cell-cycle brake that coordinates proliferation and morphogenesis
during Drosophila gastrulation. Curr Biol. 10:623–629. 2000.
View Article : Google Scholar : PubMed/NCBI
|
|
29
|
Sieber C, Kopf J, Hiepen C and Knaus P:
Recent advances in BMP receptor signaling. Cytokine Growth Factor
Rev. 20:343–355. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
30
|
Hua F, Mu R, Liu J, et al: TRB3 interacts
with SMAD3 promoting tumor cell migration and invasion. J Cell Sci.
124:3235–3246. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
31
|
Matsushima R, Harada N, Webster NJ,
Tsutsumi YM and Nakaya Y: Effect of TRB3 on insulin and
nutrient-stimulated hepatic p70 S6 kinase activity. J Biol Chem.
281:29719–29729. 2006. View Article : Google Scholar : PubMed/NCBI
|
|
32
|
Izrailit J, Berman HK, Datti A, Wrana JL
and Reedijk M: High throughput kinase inhibitor screens reveal TRB3
and MAPK-ERK/TGFβ pathways as fundamental Notch regulators in
breast cancer. Proc Natl Acad Sci USA. 110:1714–1719.
2013.PubMed/NCBI
|
|
33
|
Xu J, Lv S, Qin Y, et al: TRB3 interacts
with CtIP and is overexpressed in certain cancers. Biochim Biophys
Acta. 1770:273–278. 2007. View Article : Google Scholar : PubMed/NCBI
|
|
34
|
Schwarzer R, Dames S, Tondera D, Klippel A
and Kaufmann J: TRB3 is a PI 3-kinase dependent indicator for
nutrient starvation. Cell Signal. 18:899–909. 2006. View Article : Google Scholar : PubMed/NCBI
|
|
35
|
Koo SH, Satoh H, Herzig S, et al: PGC-1
promotes insulin resistance in liver through PPAR-alpha-dependent
induction of TRB-3. Nat Med. 10:530–534. 2004. View Article : Google Scholar : PubMed/NCBI
|
|
36
|
Ohoka N, Yoshii S, Hattori T, Onozaki K
and Hayashi H: TRB3, a novel ER stress-inducible gene, is induced
via ATF4-CHOP pathway and is involved in cell death. EMBO J.
24:1243–1255. 2005. View Article : Google Scholar : PubMed/NCBI
|
|
37
|
Valastyan S and Weinberg RA: Tumor
metastasis: molecular insights and evolving paradigms. Cell.
147:275–292. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
38
|
Weilbaecher KN, Guise TA and McCauley LK:
Cancer to bone: a fatal attraction. Nat Rev Cancer. 11:411–425.
2011. View
Article : Google Scholar : PubMed/NCBI
|