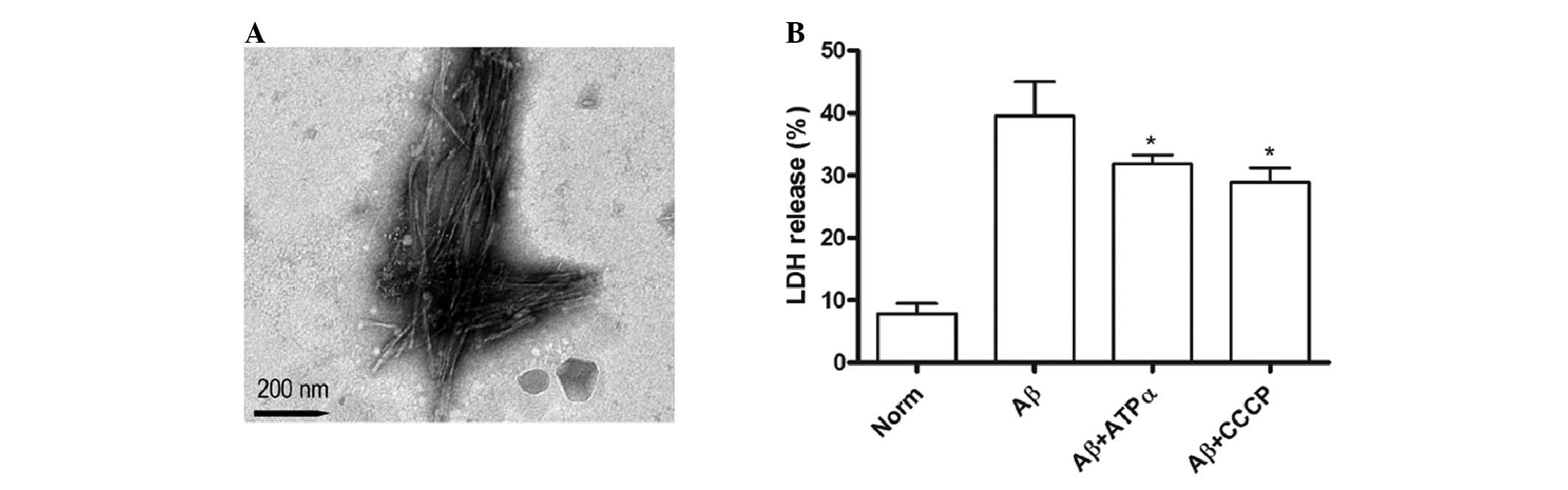
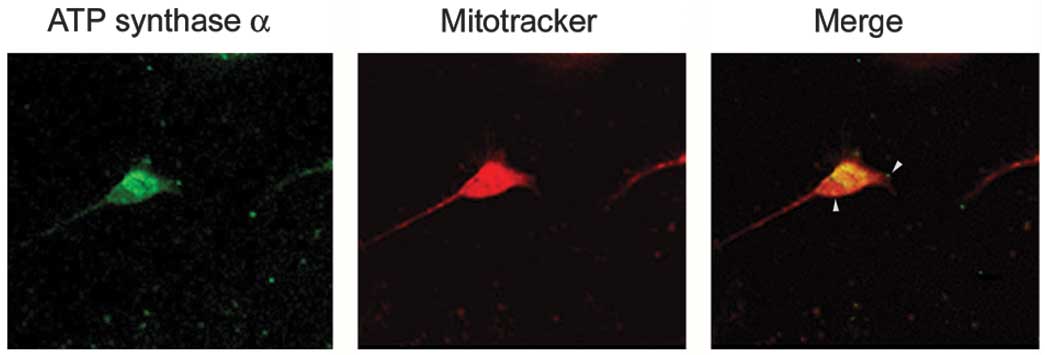
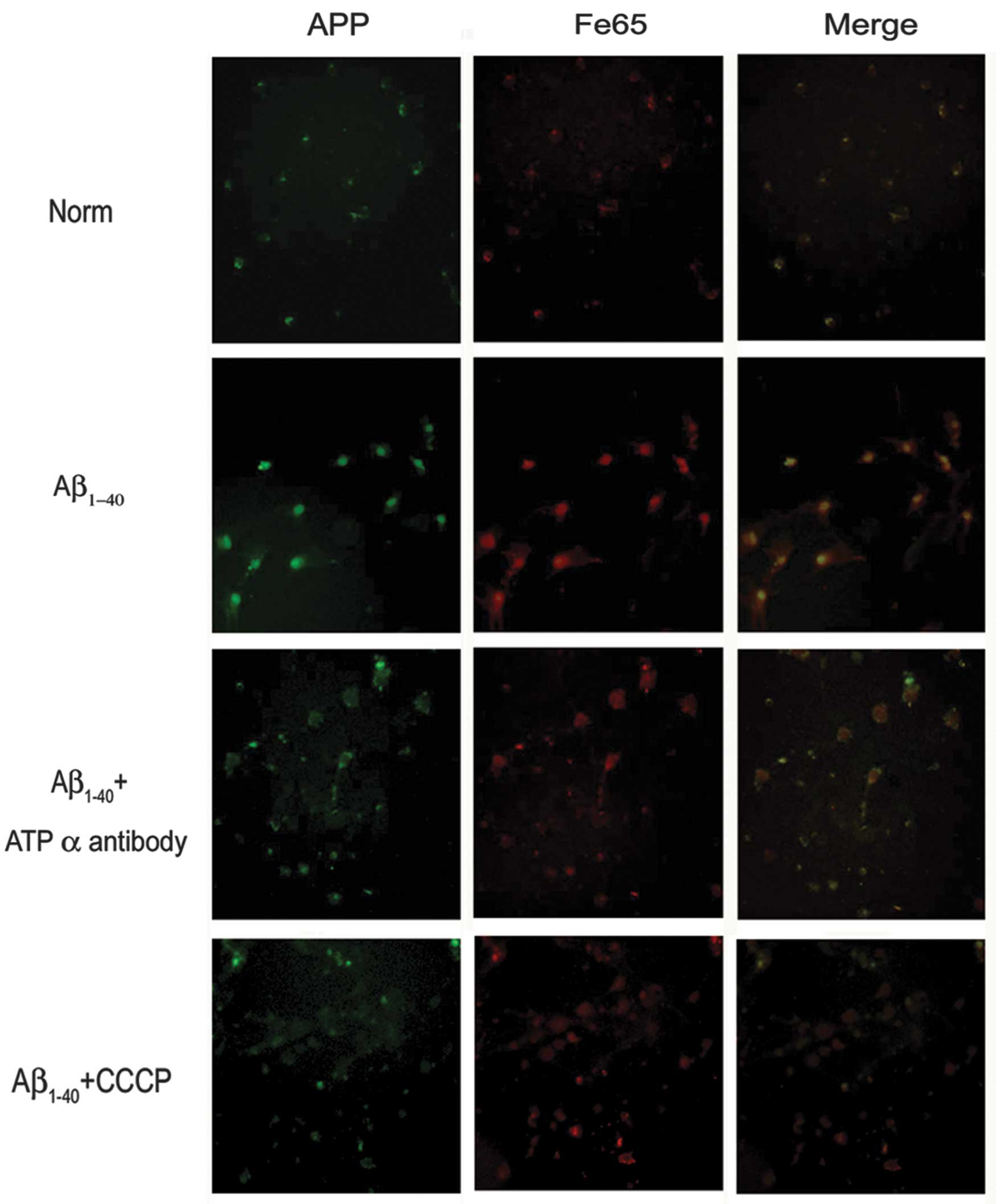
|
1
|
Kayed R, Head E, Thompson JL, et al:
Common structure of soluble amyloid oligomers implies common
mechanism of pathogenesis. Science. 300:486–489. 2003. View Article : Google Scholar : PubMed/NCBI
|
|
2
|
Fifre A, Sponne I, Koziel V, et al:
Microtubule-associated protein MAP1A, MAP1B, and MAP2 proteolysis
during soluble amyloid beta-peptide-induced neuronal apoptosis.
Synergistic involvement of calpain and caspase-3. J Biol Chem.
281:229–240. 2006. View Article : Google Scholar
|
|
3
|
Selkoe DJ: Soluble oligomers of the
amyloid beta-protein impair synaptic plasticity and behavior. Behav
Brain Res. 192:106–113. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
4
|
You H, Tsutsui S, Hameed S, et al: Aβ
neurotoxicity depends on interactions between copper ions, prion
protein, and N-methyl-D-aspartate receptors. Proc Natl Acad Sci
USA. 109:1737–1742. 2012.
|
|
5
|
Verdier Y, Zarándi M and Penke B: Amyloid
beta-peptide interactions with neuronal and glial cell plasma
membrane: binding sites and implications for Alzheimer’s disease. J
Pept Sci. 10:229–248. 2004.PubMed/NCBI
|
|
6
|
Kim BW, Choo HJ, Lee JW, Kim JH and Ko YG:
Extracellular ATP is generated by ATP synthase complex in adipocyte
lipid rafts. Exp Mol Med. 36:476–485. 2004. View Article : Google Scholar : PubMed/NCBI
|
|
7
|
Burrell HE, Wlodarski B, Foster BJ, et al:
Human keratinocytes release ATP and utilize three mechanisms for
nucleotide interconversion at the cell surface. J Biol Chem.
280:29667–29676. 2005. View Article : Google Scholar : PubMed/NCBI
|
|
8
|
Champagne E, Martinez LO, Collet X and
Barbaras R: Ecto-F1Fo ATP synthase/F1 ATPase: metabolic and
immunological functions. Curr Opin Lipidol. 17:279–284. 2006.
View Article : Google Scholar : PubMed/NCBI
|
|
9
|
Mowery YM and Pizzo SV: Targeting cell
surface F1F0 ATP synthase in cancer therapy. Cancer Biol Ther.
7:1836–1838. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
10
|
Vantourout P, Radojkovic C, Lichtenstein
L, Pons V, Champagne E and Martinez LO: Ecto-F1-ATPase:
a moonlighting protein complex and an unexpected apoA–I receptor.
World J Gastroenterol. 16:5925–5935. 2010.
|
|
11
|
Xing SL, Yan J, Yu ZH and Zhu CQ: Neuronal
cell surface ATP synthase mediates synthesis of extracellular ATP
and regulation of intracellular pH. Cell Biol Int. 35:81–86.
2011.PubMed/NCBI
|
|
12
|
Xing SL, Chen B, Shen DZ and Zhu CQ:
β-amyloid peptide binds and regulates ectopic ATP synthase α-chain
on neural surface. Int J Neurosci. 122:290–297. 2012.
|
|
13
|
Magdesian MH, Carvalho MM, Mendes FA, et
al: Amyloid-beta binds to the extracellular cysteine-rich domain of
Frizzled and inhibits Wnt/beta-catenin signaling. J Biol Chem.
283:9359–9368. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
14
|
Shaked GM, Kummer MP, Lu DC, Galvan V,
Bredesen DE and Koo EH: Abeta induces cell death by direct
interaction with its cognate extracellular domain on APP (APP
597–624). FASEB J. 20:1254–1256. 2006.PubMed/NCBI
|
|
15
|
Fabre AC, Vantourout P, Champagne E, et
al: Cell surface adenylate kinase activity regulates the
F(1)-ATPase/P2Y (13)-mediated HDL endocytosis pathway on human
hepatocytes. Cell Mol Life Sci. 63:2829–2837. 2006. View Article : Google Scholar
|
|
16
|
Martinez LO, Jacquet S, Esteve JP, et al:
Ectopic beta-chain of ATP synthase is an apolipoprotein A–I
receptor in hepatic HDL endocytosis. Nature. 421:75–79. 2003.
|
|
17
|
Sergeant N, Wattez A, Galván-valencia M,
et al: Association of ATP synthase alpha-chain with neurofibrillary
degeneration in Alzheimer’s disease. Neuroscience. 117:293–303.
2003.PubMed/NCBI
|
|
18
|
Schmidt C, Lepsverdize E, Chi SL, et al:
Amyloid precursor protein and amyloid beta-peptide bind to ATP
synthase and regulate its activity at the surface of neural cells.
Mol Psychiatry. 13:953–969. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
19
|
Guénette SY, Chen J, Jondro PD and Tanzi
RE: Association of a novel human FE65-like protein with the
cytoplasmic domain of the beta-amyloid precursor protein. Proc Natl
Acad Sci USA. 93:10832–10837. 1996.PubMed/NCBI
|
|
20
|
Zambrano N, Buxbaum JD, Minopoli G, et al:
Interaction of the phosphotyrosine interaction/phosphotyrosine
binding-related domains of Fe65 with wild-type and mutant
Alzheimer’s beta-amyloid precursor proteins. J Biol Chem.
272:6399–6405. 1997.PubMed/NCBI
|
|
21
|
Ando K, Iijima KI, Elliott JI, Kirino Y
and Suzuki T: Phosphorylation-dependent regulation of the
interaction of amyloid precursor protein with Fe65 affects the
production of beta-amyloid. J Biol Chem. 276:40353–40361. 2001.
View Article : Google Scholar : PubMed/NCBI
|
|
22
|
Xie Z, Dong Y, Maeda U, Xia W and Tanzi
RE: RNA interference silencing of the adaptor molecules ShcC and
Fe65 differentially affect amyloid precursor protein processing and
Abeta generation. J Biol Chem. 282:4318–4325. 2007. View Article : Google Scholar : PubMed/NCBI
|
|
23
|
Fiore F, Zambrano N, Minopoli G, Donini V,
Duilio A and Russo T: The regions of the Fe65 protein homologous to
the phosphotyrosine interaction/phosphotyrosine binding domain of
Shc bind the intracellular domain of the Alzheimer’s amyloid
precursor protein. J Biol Chem. 270:30853–30856. 1995.PubMed/NCBI
|