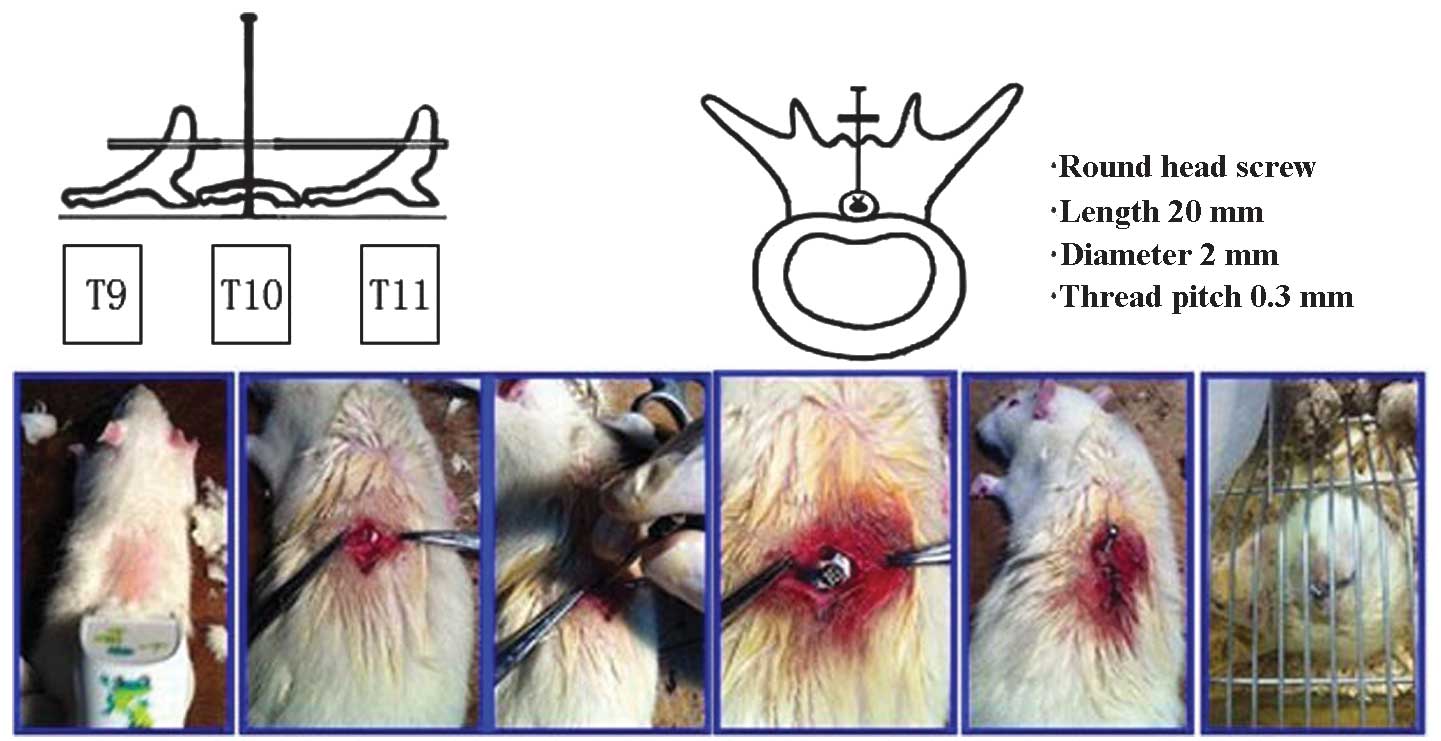
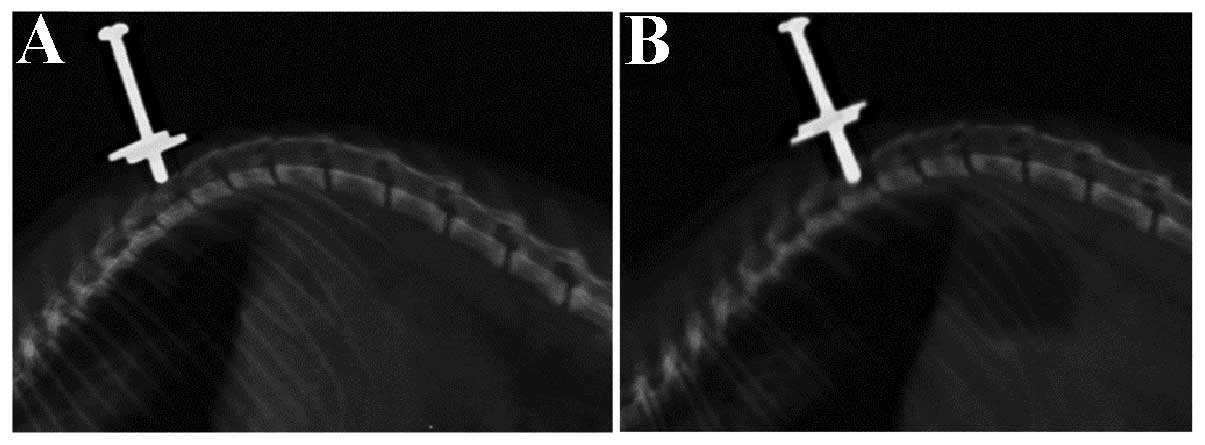
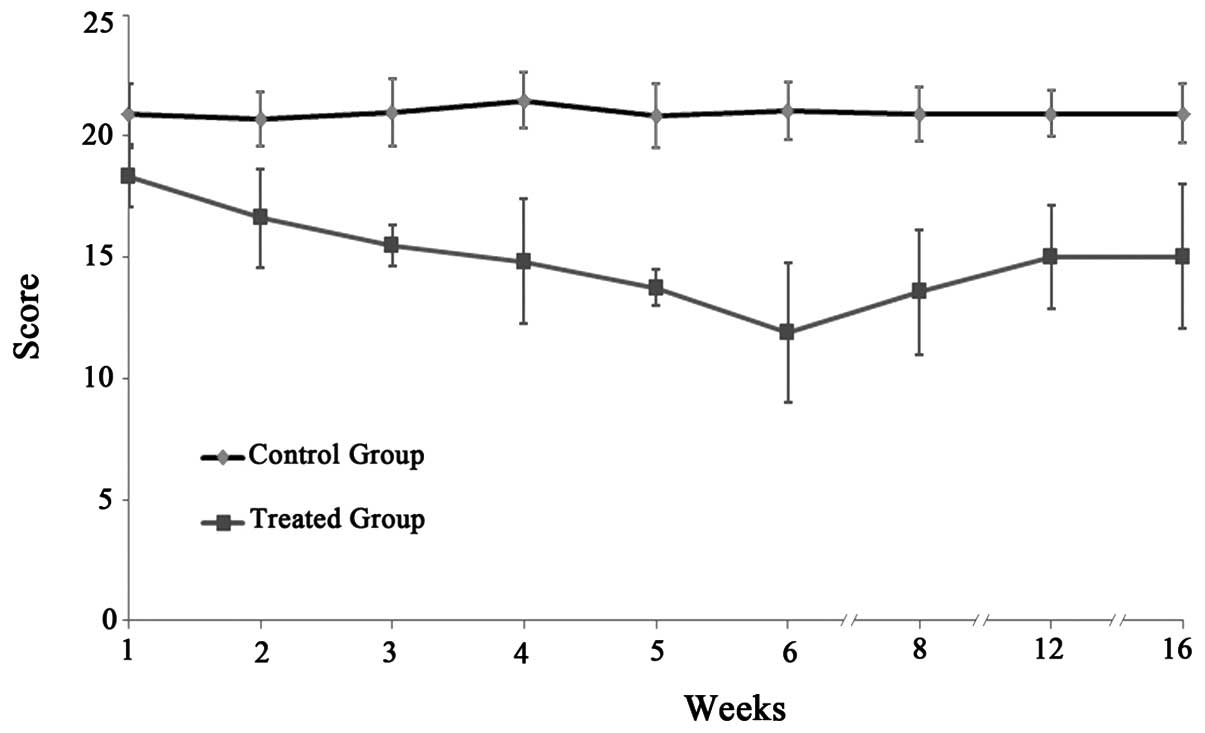
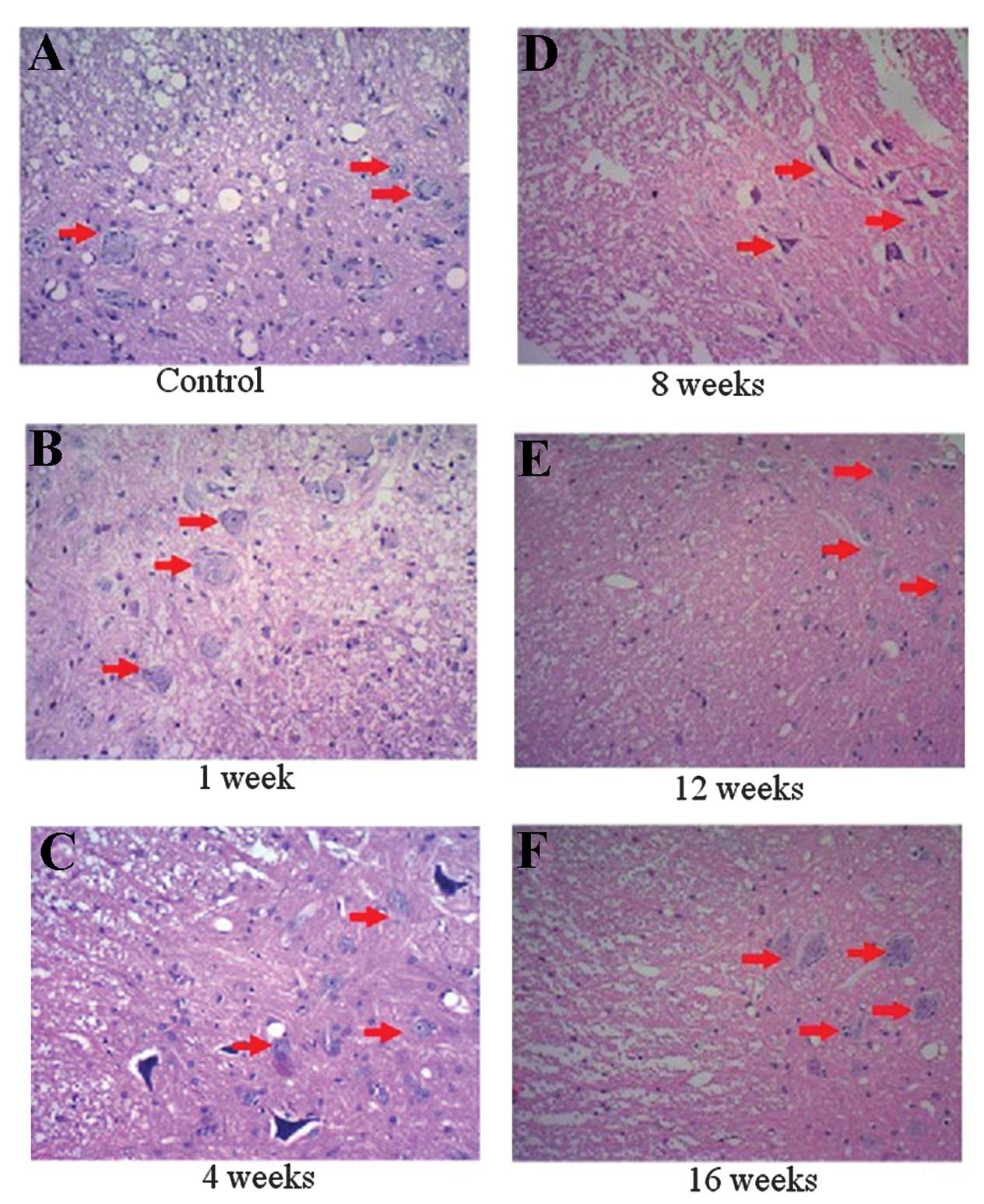
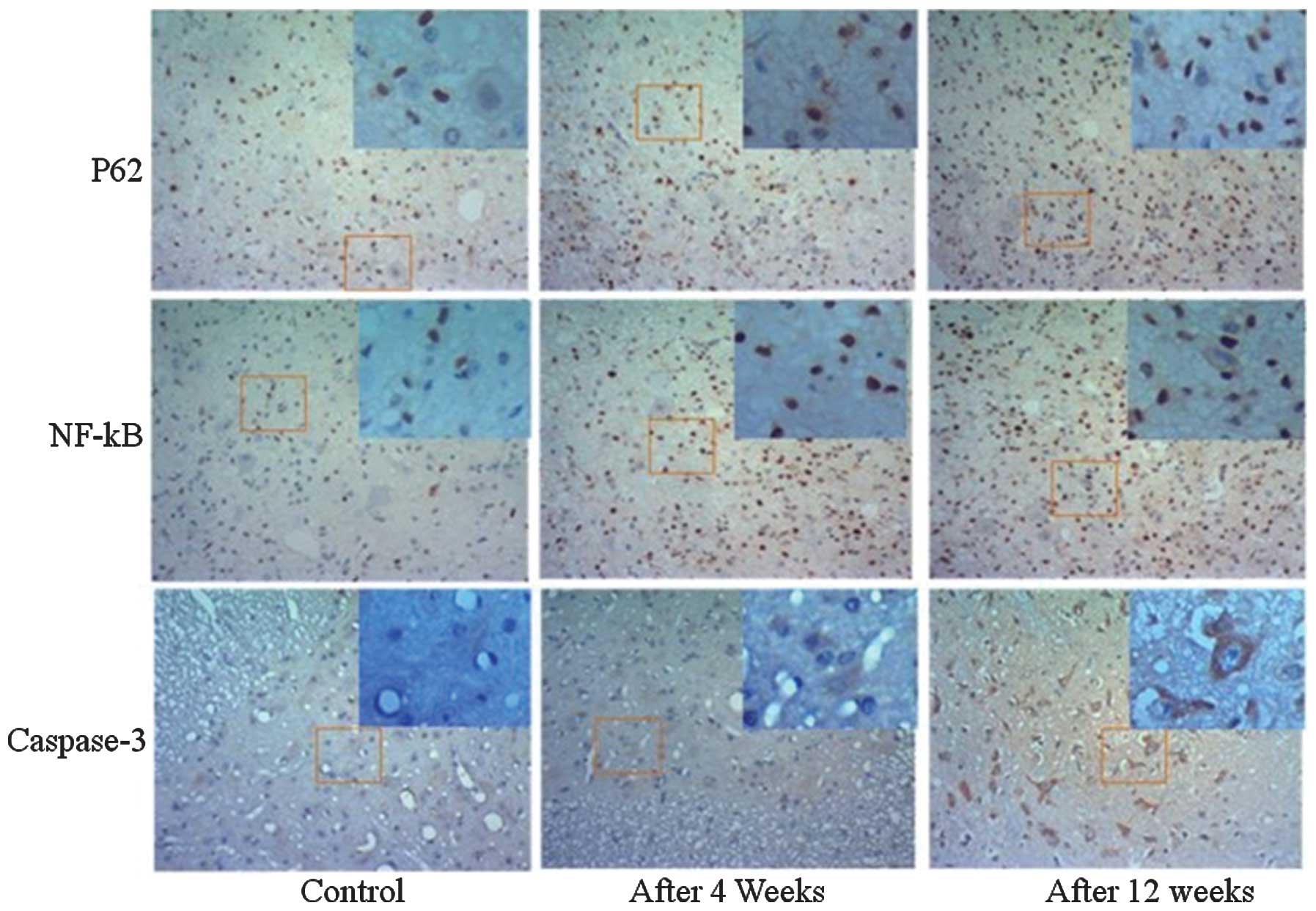
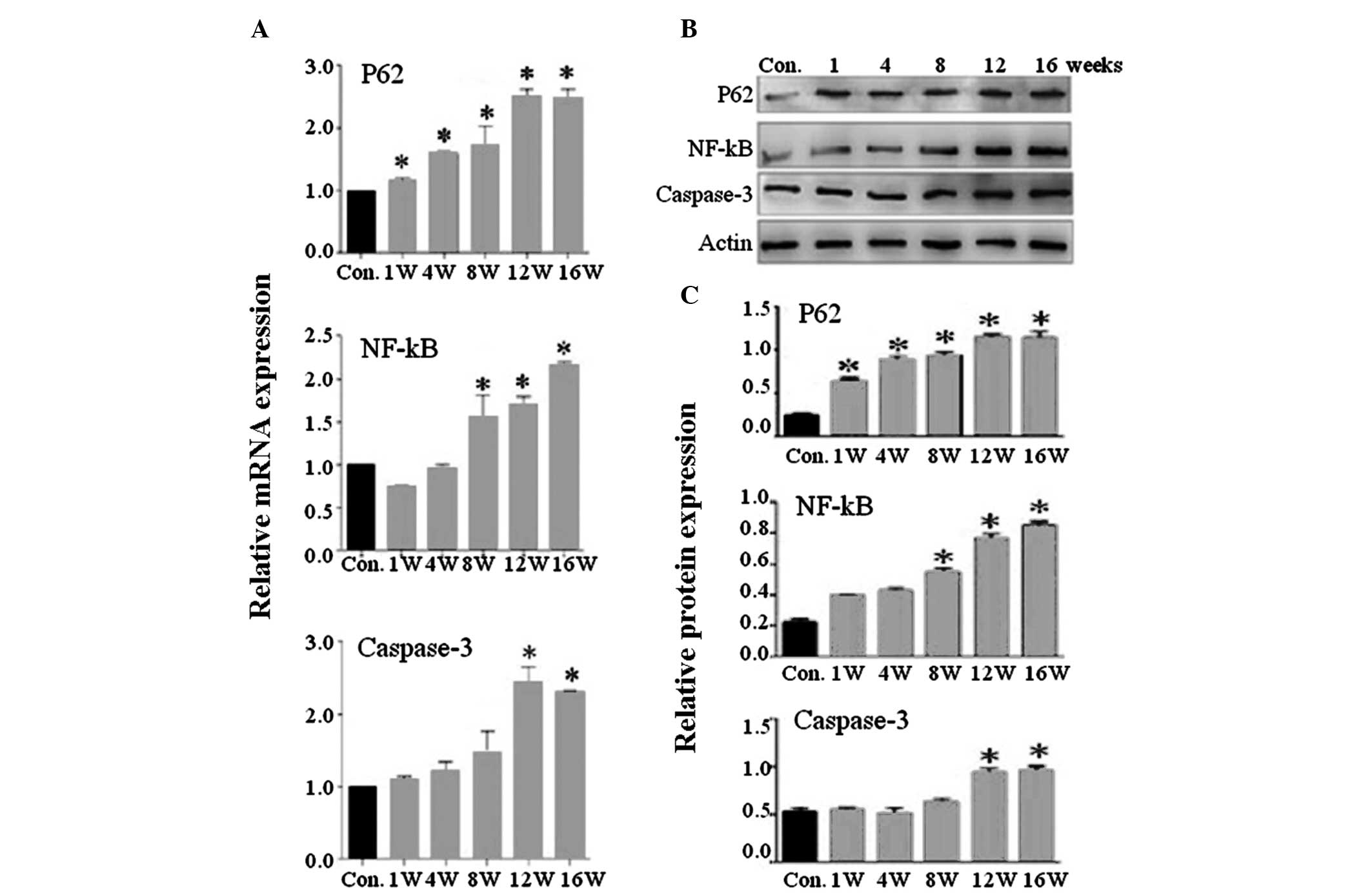
|
1
|
Hayashi H, Okada K, Hamada M, Tada K and
Ueno R: Etiologic factors of myelopathy. A radiographic evaluation
of the aging changes in the cervical spine. Clin Orthop Relat Res.
200–209. 1987.PubMed/NCBI
|
|
2
|
Ogino H, Tada K, Okada K, et al: Canal
diameter, anteroposterior compression ratio, and spondylotic
myelopathy of the cervical spine. Spine (Phila Pa 1976). 8:1–15.
1983. View Article : Google Scholar : PubMed/NCBI
|
|
3
|
Edwards WC and LaRocca H: The
developmental segmental sagittal diameter of the cervical spinal
canal in patients with cervical spondylosis. Spine (Phila Pa 1976).
8:20–27. 1983. View Article : Google Scholar : PubMed/NCBI
|
|
4
|
Ito T, Oyanagi K, Takahashi H, Takahashi
HE and Ikuta F: Cervical spondylotic myelopathy. Clinicopathologic
study on the progression pattern and thin myelinated fibers of the
lesions of seven patients examined during complete autopsy. Spine
(Phila Pa 1976). 21:827–833. 1996. View Article : Google Scholar
|
|
5
|
Gooding MR, Wilson CB and Hoff JT:
Experimental cervical myelopathy. Effects of ischemia and
compression of the canine cervical spinal cord. J Neurosurg.
43:9–17. 1975. View Article : Google Scholar : PubMed/NCBI
|
|
6
|
Hukuda S and Wilson CB: Experimental
cervical myelopathy: effects of compression and ischemia on the
canine cervical cord. J Neurosurg. 37:631–652. 1972. View Article : Google Scholar : PubMed/NCBI
|
|
7
|
Long HQ, Li GS, Hu Y, Wen CY and Xie WH:
HIF-1alpha/VEGF signaling pathway may play a dual role in secondary
pathogenesis of cervical myelopathy. Med Hypotheses. 79:82–84.
2012. View Article : Google Scholar : PubMed/NCBI
|
|
8
|
Fehlings MG and Skaf G: A review of the
pathophysiology of cervical spondylotic myelopathy with insights
for potential novel mechanisms drawn from traumatic spinal cord
injury. Spine (Phila Pa 1976). 23:2730–2737. 1998. View Article : Google Scholar : PubMed/NCBI
|
|
9
|
Yu WR, Baptiste DC, Liu T, Odrobina E,
Stanisz GJ and Fehlings MG: Molecular mechanisms of spinal cord
dysfunction and cell death in the spinal hyperostotic mouse:
implications for the pathophysiology of human cervical spondylotic
myelopathy. Neurobiol Dis. 33:149–163. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
10
|
Yu WR, Liu T, Kiehl TR and Fehlings MG:
Human neuropathological and animal model evidence supporting a role
for Fas-mediated apoptosis and inflammation in cervical spondylotic
myelopathy. Brain. 134:1277–1292. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
11
|
Kim DH, Vaccaro AR, Henderson FC and
Benzel EC: Molecular biology of cervical myelopathy and spinal cord
injury: role of oligodendrocyte apoptosis. Spine J. 3:510–519.
2003. View Article : Google Scholar : PubMed/NCBI
|
|
12
|
Karadimas SK, Gialeli CH, Klironomos G, et
al: The role of oligodendrocytes in the molecular pathobiology and
potential molecular treatment of cervical spondylotic myelopathy.
Curr Med Chem. 17:1048–1058. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
13
|
Hara T, Nakamura K, Matsui M, et al:
Suppression of basal autophagy in neural cells causes
neurodegenerative disease in mice. Nature. 441:885–889. 2006.
View Article : Google Scholar : PubMed/NCBI
|
|
14
|
Komatsu M, Waguri S, Chiba T, et al: Loss
of autophagy in the central nervous system causes neurodegeneration
in mice. Nature. 441:880–884. 2006. View Article : Google Scholar : PubMed/NCBI
|
|
15
|
Sarkar S, Ravikumar B, Floto RA and
Rubinsztein DC: Rapamycin and mTOR-independent autophagy inducers
ameliorate toxicity of polyglutamine-expanded huntingtin and
related proteinopathies. Cell Death Differ. 16:46–56. 2009.
View Article : Google Scholar : PubMed/NCBI
|
|
16
|
Pan T, Kondo S, Zhu W, Xie W, Jankovic J
and Le W: Neuroprotection of rapamycin in lactacystin-induced
neurodegeneration via autophagy enhancement. Neurobiol Dis.
32:16–25. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
17
|
Wong E and Cuervo AM: Autophagy gone awry
in neurodegenerative diseases. Nat Neurosci. 13:805–811. 2010.
View Article : Google Scholar : PubMed/NCBI
|
|
18
|
Keane RW, Kraydieh S, Lotocki G, et al:
Apoptotic and anti-apoptotic mechanisms following spinal cord
injury. J Neuropathol Exp Neurol. 60:422–429. 2001.PubMed/NCBI
|
|
19
|
Yoshino O, Matsuno H, Nakamura H, et al:
The role of Fas-mediated apoptosis after traumatic spinal cord
injury. Spine (Phila Pa 1976). 29:1394–1404. 2004. View Article : Google Scholar : PubMed/NCBI
|
|
20
|
Casha S, Yu WR and Fehlings MG: FAS
deficiency reduces apoptosis, spares axons and improves function
after spinal cord injury. Exp Neurol. 196:390–400. 2005. View Article : Google Scholar
|
|
21
|
Erlich S, Alexandrovich A, Shohami E and
Pinkas-Kramarski R: Rapamycin is a neuroprotective treatment for
traumatic brain injury. Neurobiol Dis. 26:86–93. 2007. View Article : Google Scholar : PubMed/NCBI
|
|
22
|
Sekiguchi A, Kanno H, Ozawa H, Yamaya S
and Itoi E: Rapamycin promotes autophagy and reduces neural tissue
damage and locomotor impairment after spinal cord injury in mice. J
Neurotrauma. 29:946–956. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
23
|
Duran A, Serrano M, Leitges M, et al: The
atypical PKC-interacting protein p62 is an important mediator of
RANK-activated osteoclastogenesis. Dev Cell. 6:303–309. 2004.
View Article : Google Scholar : PubMed/NCBI
|
|
24
|
Rodriguez A, Durán A, Selloum M, et al:
Mature-onset obesity and insulin resistance in mice deficient in
the signaling adapter p62. Cell Metab. 3:211–222. 2006. View Article : Google Scholar : PubMed/NCBI
|
|
25
|
Duran A, Linares JF, Galvez AS, et al: The
signaling adaptor p62 is an important NF-kappaB mediator in
tumorigenesis. Cancer Cell. 13:343–354. 2008. View Article : Google Scholar
|
|
26
|
Kuusisto E, Salminen A and Alafuzoff I:
Ubiquitin-binding protein p62 is present in neuronal and glial
inclusions in human tauopathies and synucleinopathies. Neuroreport.
12:2085–2090. 2001. View Article : Google Scholar : PubMed/NCBI
|
|
27
|
Zatloukal K, Stumptner C, Fuchsbichler A,
et al: p62 is a common component of cytoplasmic inclusions in
protein aggregation diseases. Am J Pathol. 160:255–263. 2002.
View Article : Google Scholar : PubMed/NCBI
|
|
28
|
Nagaoka U, Kim K, Jana NR, et al:
Increased expression of p62 in expanded polyglutamine-expressing
cells and its association with polyglutamine inclusions. J
Neurochem. 91:57–68. 2004. View Article : Google Scholar : PubMed/NCBI
|
|
29
|
Komatsu M, Waguri S, Koike M, et al:
Homeostatic levels of p62 control cytoplasmic inclusion body
formation in autophagy-deficient mice. Cell. 131:1149–1163. 2007.
View Article : Google Scholar : PubMed/NCBI
|
|
30
|
Jin Z, Li Y, Pitti R, et al: Cullin3-based
polyubiquitination and p62-dependent aggregation of caspase-8
mediate extrinsic apoptosis signaling. Cell. 137:721–735. 2009.
View Article : Google Scholar : PubMed/NCBI
|
|
31
|
Sanz L, Diaz-Meco MT, Nakano H and Moscat
J: The atypical PKC-interacting protein p62 channels NF-kappaB
activation by the IL-1-TRAF6 pathway. EMBO J. 19:1576–1586. 2000.
View Article : Google Scholar : PubMed/NCBI
|
|
32
|
Moscat J, Diaz-Meco MT, Albert A and
Campuzano S: Cell signaling and function organized by PB1 domain
interactions. Mol Cell. 23:631–640. 2006. View Article : Google Scholar : PubMed/NCBI
|
|
33
|
Moscat J and Diaz-Meco MT: p62 at the
crossroads of autophagy, apoptosis, and cancer. Cell.
137:1001–1004. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
34
|
Xu P, Gong WM, Li Y, et al: Destructive
pathological changes in the rat spinal cord due to chronic
mechanical compression. Laboratory investigation. J Neurosurg
Spine. 8:279–285. 2008. View Article : Google Scholar
|
|
35
|
Basso DM, Beattie MS and Bresnahan JC: A
sensitive and reliable locomotor rating scale for open field
testing in rats. J Neurotrauma. 12:1–21. 1995. View Article : Google Scholar : PubMed/NCBI
|
|
36
|
Kuusisto E, Kauppinen T and Alafuzoff I:
Use of p62/SQSTM1 antibodies for neuropathological diagnosis.
Neuropathol Appl Neurobiol. 34:169–180. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
37
|
Karin M and Lin A: NF-kappaB at the
crossroads of life and death. Nat Immunol. 3:221–227. 2002.
View Article : Google Scholar : PubMed/NCBI
|
|
38
|
Sheikh MS and Huang Y: Death receptor
activation complexes: it takes two to activate TNF receptor 1. Cell
Cycle. 2:550–552. 2003. View Article : Google Scholar : PubMed/NCBI
|