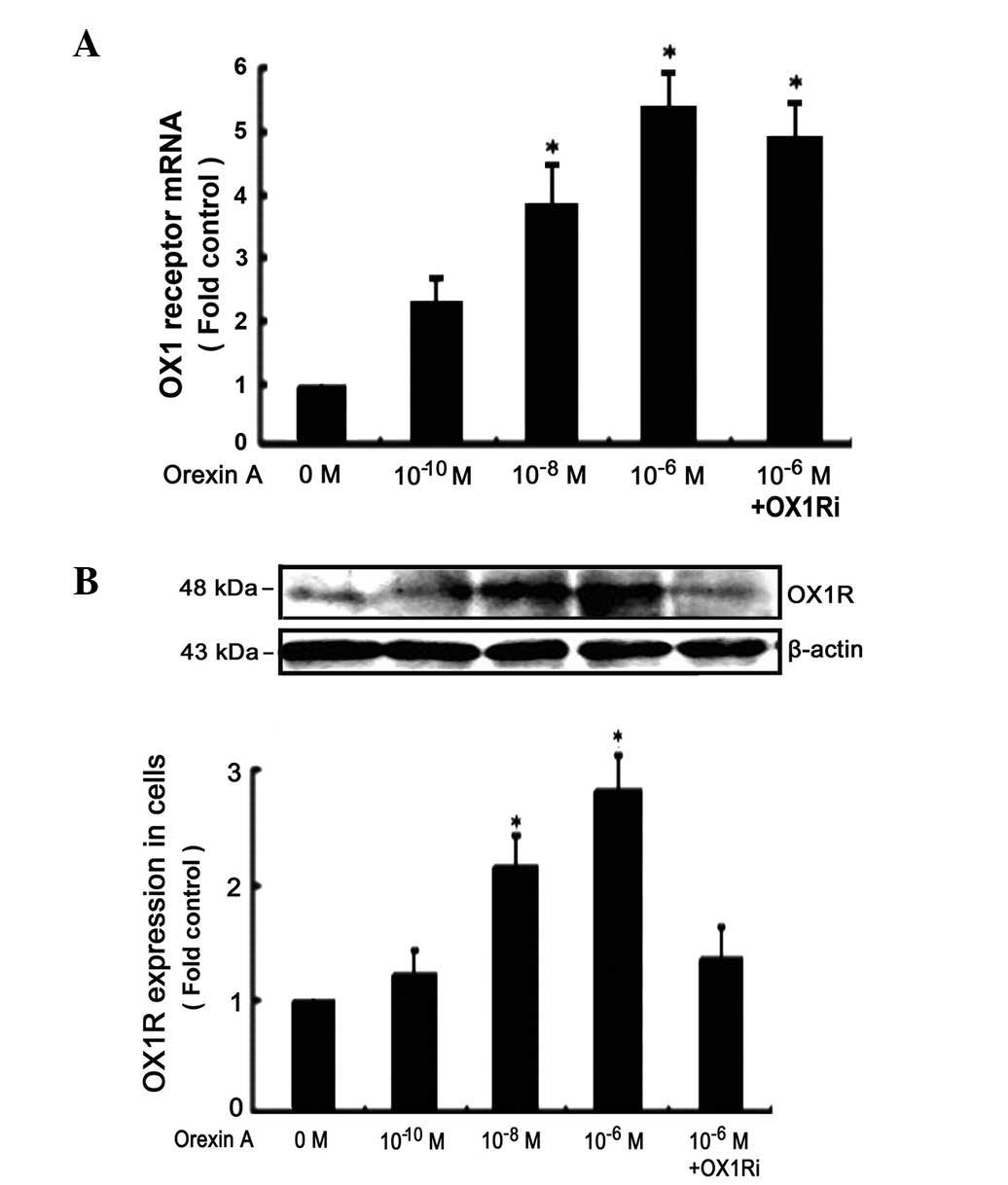
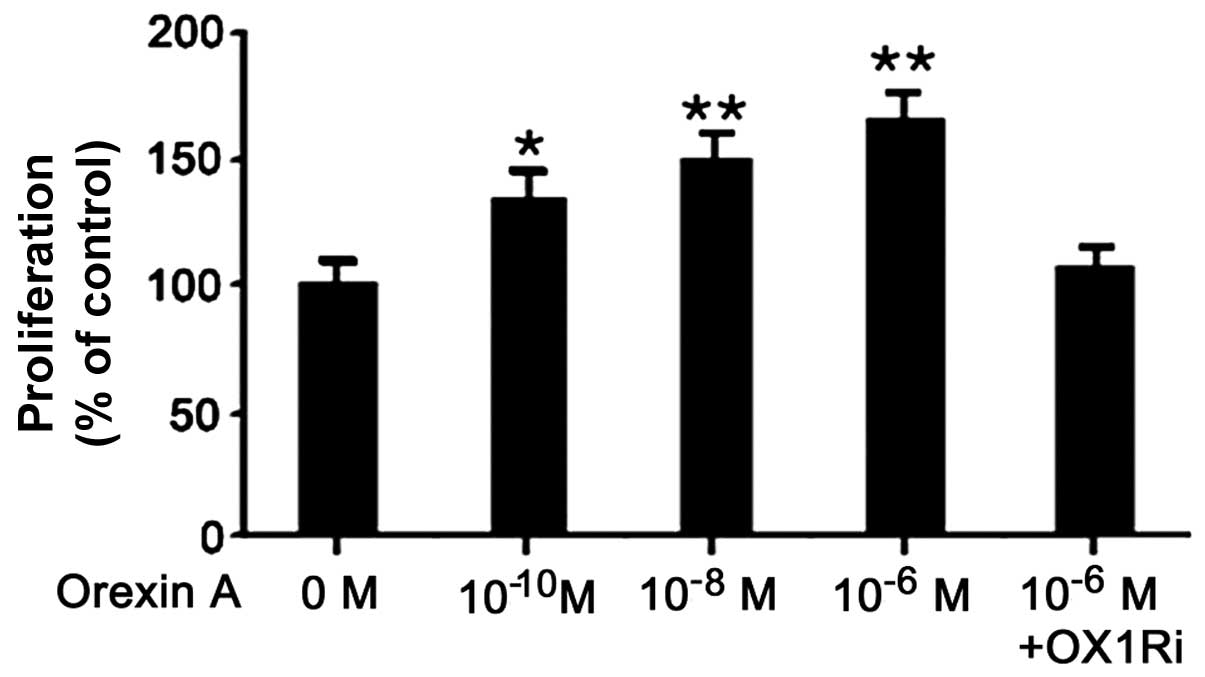
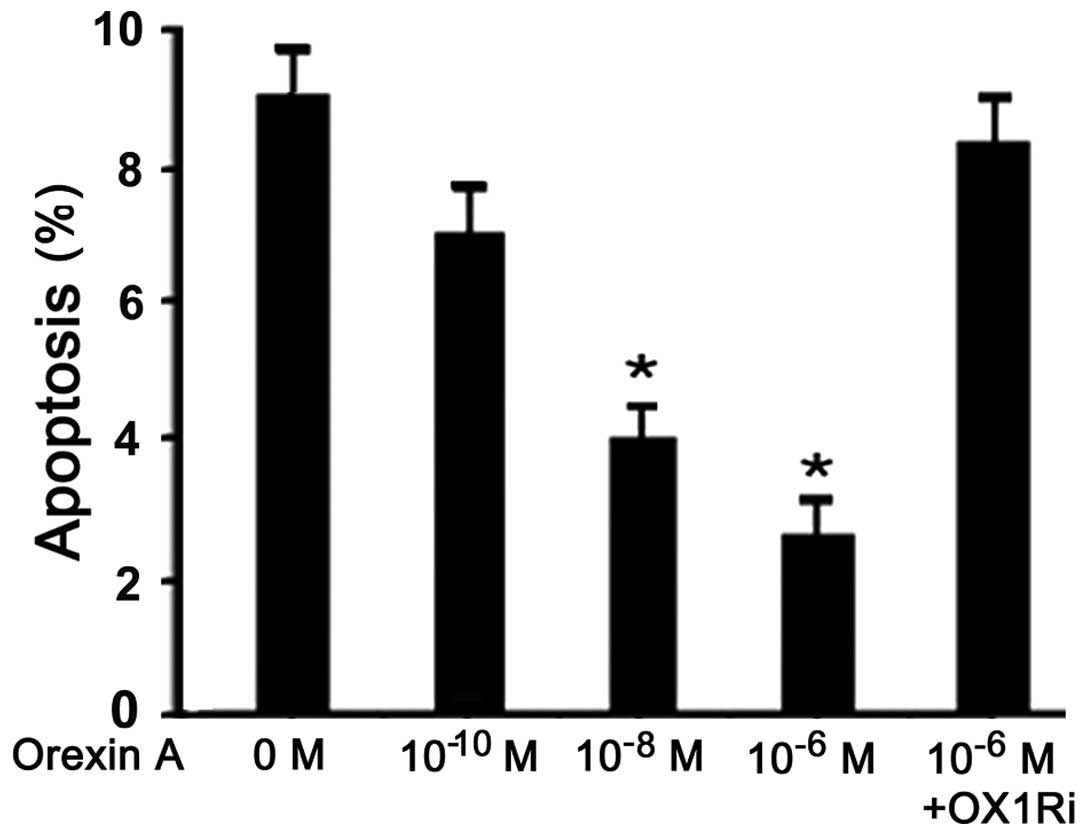
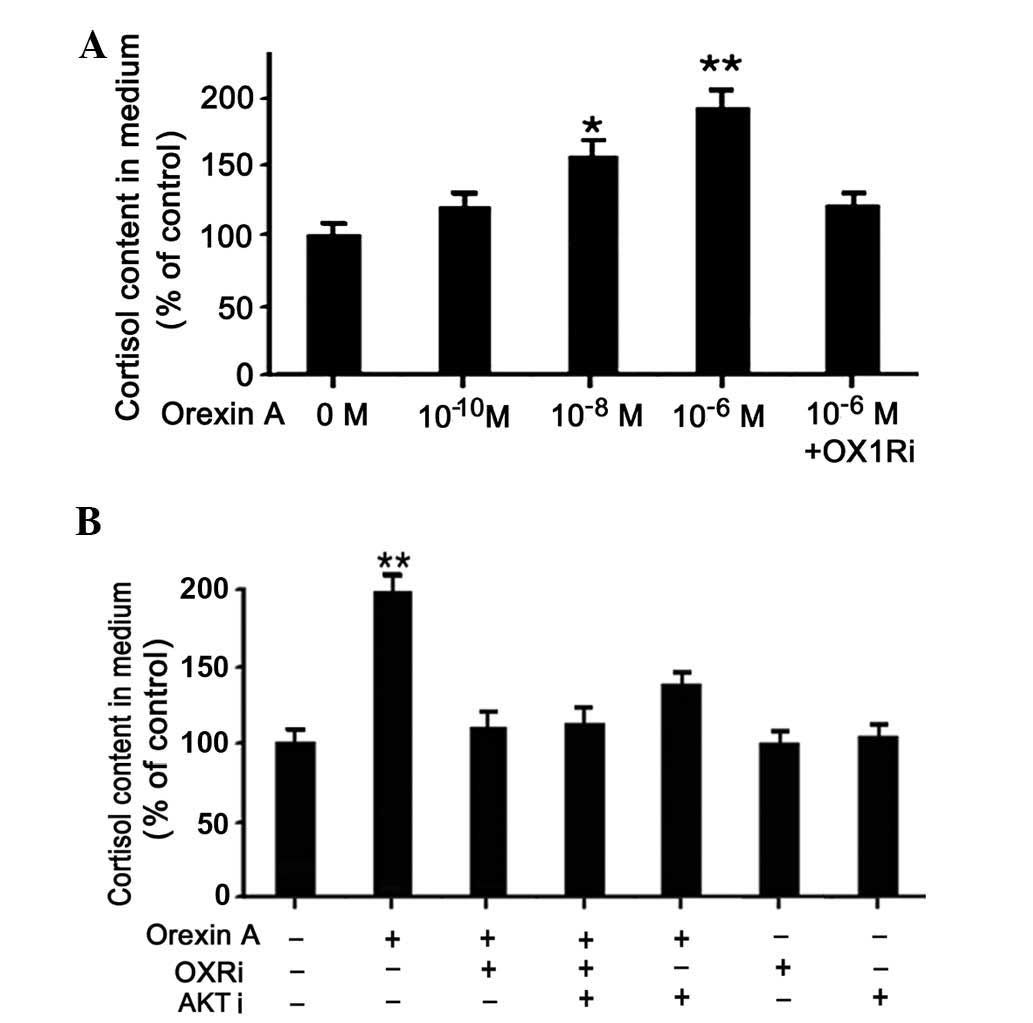
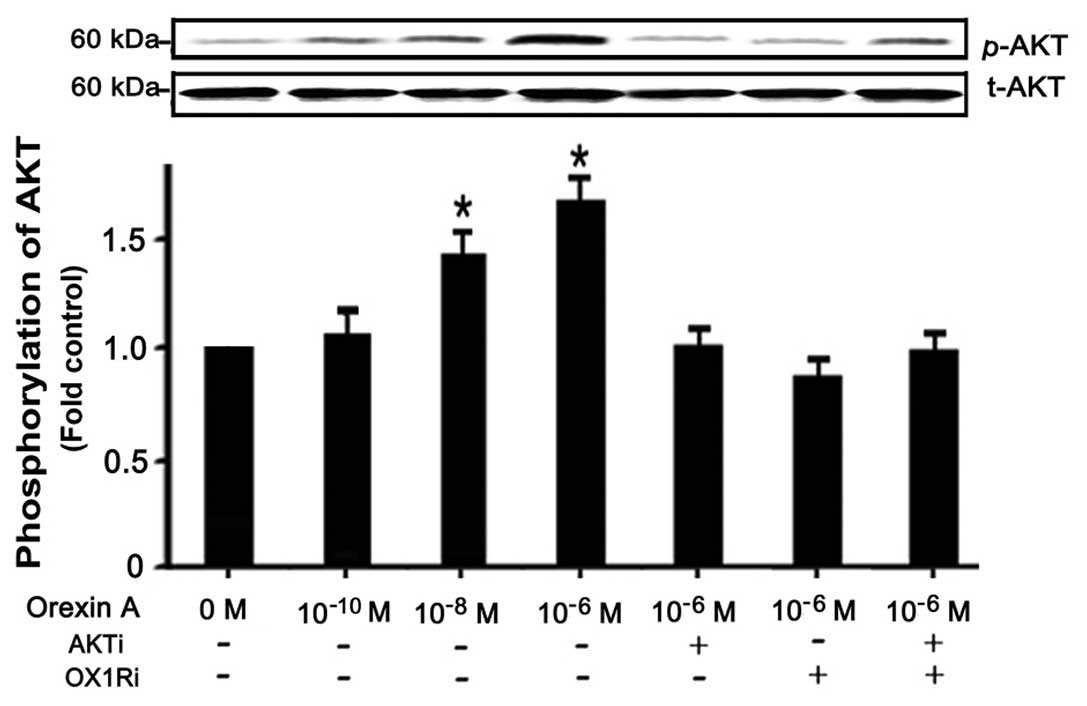
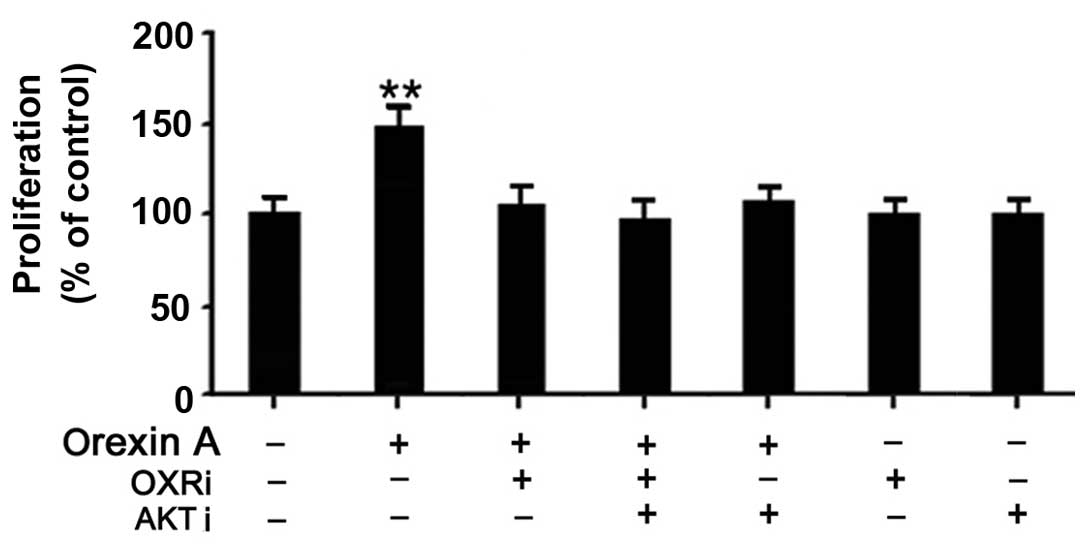
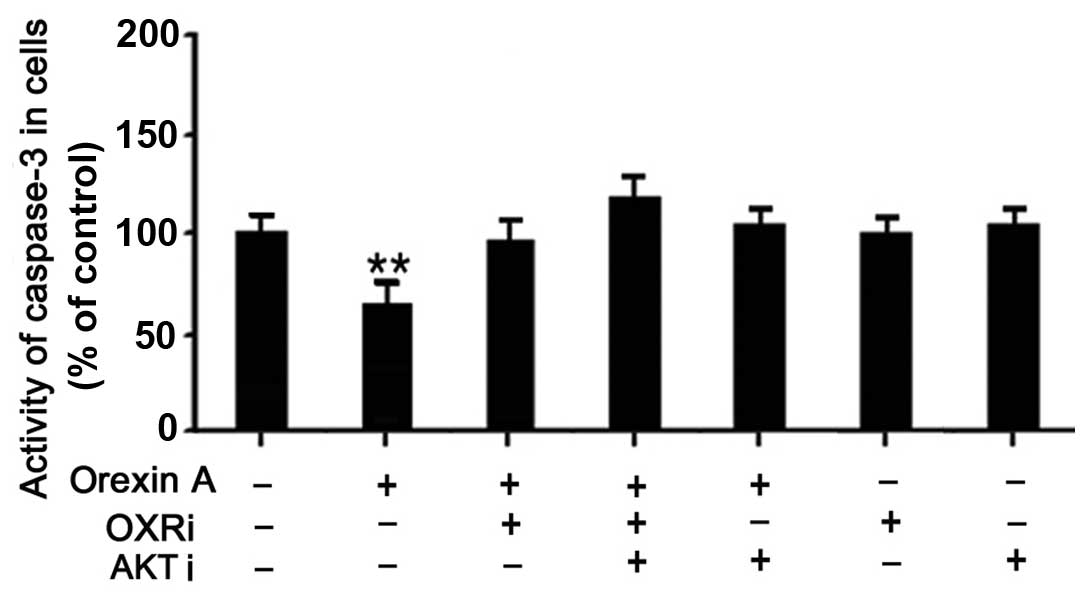
|
1
|
de Lecea L, Kilduff TS, Peyron C, Gao X,
Foye PE, Danielson PE, Fukuhara C, Battenberg EL, Gautvik VT,
Bartlett FS II, Frankel WN, et al: The hypocretins:
Hypothalamus-specific peptides with neuroexcitatory activity. Proc
Natl Acad Sci USA. 95:322–327. 1998. View Article : Google Scholar : PubMed/NCBI
|
|
2
|
Voisin T, Rouet-Benzineb P, Reuter N and
Laburthe M: Orexins and their receptors: Structural aspects and
role in peripheral tissues. Cell Mol Life Sci. 60:72–87. 2003.
View Article : Google Scholar : PubMed/NCBI
|
|
3
|
Korczynski W, Ceregrzyn M, Matyjek R, Kato
I, Kuwahara A, Wolinski J and Zabielski R: Central and local
(enteric) action of orexins. Physiol Pharmacol. 57(Suppl 6): 17–42.
2006.
|
|
4
|
Sakurai T: Orexins and orexin receptors:
Implication in feeding behavior. Regul Pept. 85:25–30. 1999.
View Article : Google Scholar : PubMed/NCBI
|
|
5
|
Sakurai T, Amemiya A, Ishii M, Matsuzaki
I, Chemelli RM, Tanaka H, Williams SC, Richardson JA, Kozlowski GP,
Wilson S, Arch JR, et al: Orexins and orexin receptors: a family of
hypothalamic neuropeptides and G protein-coupled eceptors that
regulate feeding behavior. Cell. 92:573–585. 1998. View Article : Google Scholar : PubMed/NCBI
|
|
6
|
Kukkonen JP, Holmqvist T, Ammoun S and
Åkerman KE: Functions of the orexinergic/hypocretinergic system. Am
J Physiol Cell Physiol. 283:C1567–C1591. 2002. View Article : Google Scholar : PubMed/NCBI
|
|
7
|
Randeva HS, Karteris E, Grammatopoulos D
and Hillhouse EW: Expression of orexin-A and functional orexin type
2 receptors in the human adult adrenals: Implications for adrenal
function and energy homeostasis. J Clin Endocrinol Metab.
86:4808–4813. 2001. View Article : Google Scholar : PubMed/NCBI
|
|
8
|
Xu TR, Ward RJ, Pediani JD and Milligan G:
The orexin OX1 receptor exists predominantly as a homodimer in the
basal state: Potential regulation of receptor organization by both
agonist and antagonist ligands. Biochem J. 439:171–183. 2011.
View Article : Google Scholar : PubMed/NCBI
|
|
9
|
Kim MK, Park HJ, Kim SR, Choi YK, Shin HK,
Jeon JH, Jang HO, Yun I, Bae SK and Bae MK: Angiogenic role of
orexin-A via the activation of extracellular signal-regulated
kinase in endothelial cells. Biochem Biophys Res Commun. 403:59–65.
2010. View Article : Google Scholar : PubMed/NCBI
|
|
10
|
Ramanjaneya M, Conner AC, Chen J, Kumar P,
Brown JE, Jöhren O, Lehnert H, Stanfield PR and Randeva HS:
Orexin-stimulated MAP kinase cascades are activated through
multiple G-protein signalling pathways in human H295R
adrenocortical cells: diverse roles for orexins A and B. J
Endocrinol. 202:249–261. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
11
|
Voisin T, El Firar A, Fasseu M,
Rouyer-Fessard C, Descatoire V, Walker F, Paradis V, Bedossa P,
Henin D, Lehy T and Laburthe M: Aberrant expression of OX1
receptors for orexins in colon cancers and liver metastases: An
openable gate to apoptosis. Cancer Res. 71:3341–3351. 2011.
View Article : Google Scholar : PubMed/NCBI
|
|
12
|
Rouet-Benzineb P, Rouyer-Fessard C, Jarry
A, Avondo V, Pouzet C, Yanagisawa M, Laboisse C, Laburthe M and
Voisin T: Orexins acting at native OX (1) receptor in colon cancer
and neuroblastoma cells or at recombinant OX (1) receptor suppress
cell growth by inducing apoptosis. J Biol Chem. 279:45875–45886.
2004. View Article : Google Scholar : PubMed/NCBI
|
|
13
|
Laburthe M and Voisin T: The orexin
receptor OX1R in colon cancer: a promising therapeutic target and a
new paradigm in G protein-coupled receptor signalling through
ITIMs. Br J Pharmacol. 165:1678–1687. 2012. View Article : Google Scholar :
|
|
14
|
Kagerer SM and Johren O: Interactions of
orexins/hypocretins with adrenocortical functions. Acta Physiol
(Oxf). 198:361–432. 2010. View Article : Google Scholar
|
|
15
|
Martelli AM, Tabellini G, Bressanin D,
Ognibene A, Goto K, Cocco L and Evangelisti C: The emerging
multiple roles of nuclear Akt. Biochim Biophys Acta.
1823:2168–2178. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
16
|
Zhou BP, Liao Y, Xia W, Zou Y, Spohn B and
Hung MC: HER-2/neu induces p53 ubiquitination via AKT-mediated MDM2
phosphorylation. Nat Cell Biol. 3:973–982. 2001. View Article : Google Scholar : PubMed/NCBI
|
|
17
|
Datta SR, Dudek H, Tao X, Masters S, Fu H,
Gotoh Y and Greenberg ME: AKT phosphorylation of BAD couples
survival signals to the cell-intrinsic death machinery. Cell.
91:231–241. 1997. View Article : Google Scholar : PubMed/NCBI
|
|
18
|
Brunet A, Bonni A, Zigmond MJ, Lin MZ, Juo
P, Hu LS, Anderson MJ, Arden KC, Blenis J and Greenberg ME: AKT
promotes cell survival by phosphorylating and inhibiting a Forkhead
transcription factor. Cell. 96:857–868. 1999. View Article : Google Scholar : PubMed/NCBI
|
|
19
|
Burgering BM and Kops GJ: Cell cycle and
death control: long live Forkheads. Trends Biochem Sci. 27:352–360.
2002. View Article : Google Scholar : PubMed/NCBI
|
|
20
|
Fu L, Kim YA, Wang X, Wu X, Yue P, Lonial
S, Khuri FR and Sun SY: Perifosine inhibits mammalian target of
rapamycin signaling through facilitating degradation of major
components in the mTOR axis and induces autophagy. Cancer Res.
69:8967–8976. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
21
|
Hay N and Sonenberg N: Upstream and
downstream of mTOR. Genes Dev. 18:1926–1945. 2004. View Article : Google Scholar : PubMed/NCBI
|
|
22
|
Richardson CJ, Schalm SS and Blenis J:
PI3-kinase and TOR: PIKTORing cell growth. Semin Cell Dev Biol.
15:147–159. 2004. View Article : Google Scholar : PubMed/NCBI
|
|
23
|
Steelman LS, Chappell WH, Abrams SL, Kempf
RC, Long J, Laidler P, Mijatovic S, Maksimovic-Ivanic D, Stivala F,
Mazzarino MC, Donia M, et al: Roles of the Raf/MEK/ERK and
PI3K/PTEN/AKT/mTOR pathways in controlling growth and sensitivity
to therapy-implications for cancer and aging. Aging (Albany NY).
3:192–222. 2011.
|
|
24
|
Hsieh AC, Truitt ML and Ruggero D:
Oncogenic AKTivation of translation as a therapeutic target. Br J
Cancer. 105:329–336. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
25
|
Sale EM and Sale GJ: Protein kinase B:
signalling roles and therapeutic targeting. Cell Mol Life Sci.
65:113–127. 2008. View Article : Google Scholar
|
|
26
|
Blandino-Rosano M, Chen AY, Scheys JO,
Alejandro EU, Gould AP, Taranukha T, Elghazi L, Cras-Méneur C and
Bernal-Mizrachi E: mTORC1 signaling and regulation of pancreatic
β-cell mass. Cell Cycle. 11:1892–1902. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
27
|
Cheng GZ, Zhang W and Wang LH: Regulation
of cancer cell survival, migration and invasion by twist: AKT2
comes to interplay. Cancer Res. 68:957–960. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
28
|
Buttrick GJ and Wakefield JG: PI3-K and
GSK-3: AKT-ing together with microtubules. Cell Cycle. 7:2621–2625.
2008. View Article : Google Scholar : PubMed/NCBI
|
|
29
|
Meuillet EJ: Novel inhibitors of AKT:
assessment of a different approach targeting the pleckstrin
homology domain. Curr Med Chem. 18:2727–2742. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
30
|
Bartholomeusz C and Gonzalez-Angulo AM:
Targeting the PI3K signaling pathway in cancer therapy. Expert Opin
Ther Targets. 16:121–130. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
31
|
Ramanjaneya M, Conner AC, Chen J, Kumar P,
Brown JE, Jöhren O, Lehnert H, Stanfield PR and Randeva HS:
Orexin-stimulated MAP kinase cascades are activated through
multiple G-protein signalling pathways in human H295R
adrenocortical cells:diverse roles for orexins A and B. J
Endocrinol. 202:249–261. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
32
|
Ramanjaneya M, Conner AC, Chen J,
Stanfield PR and Randeva HS: Orexins stimulate steroidogenic acute
regulatory protein expression through multiple signaling pathways
in human adrenal H295R cells. Endocrinology. 149:4106–4115. 2008.
View Article : Google Scholar : PubMed/NCBI
|
|
33
|
Rainey WE, Saner K and Schimmer BP:
Adrenocortical cell lines. Mol Cell Endocrinol. 228:23–38. 2004.
View Article : Google Scholar : PubMed/NCBI
|
|
34
|
Ammoun S, Holmqvist T, Shariatmadari R,
Oonk HB, Detheux M, Parmentier M, Åkerman KE and Kukkonen JP:
Distinct recognition of OX1 and OX2 receptors by orexin peptides. J
Pharmacol Exp Ther. 305:507–514. 2003. View Article : Google Scholar : PubMed/NCBI
|
|
35
|
Kim MS, Yoon CY, Jang PG, Park YJ, Shin
CS, Park HS, Ryu JW, Pak YK, Park JY, Lee KU, Kim SY, et al: The
mitogenic and antiapoptotic actions of ghrelin in 3T3-L1
adipocytes. Mol Endocrinol. 18:2291–2301. 2004. View Article : Google Scholar : PubMed/NCBI
|
|
36
|
Li Y, Jiang Y, Wan Y, Zhang L, Tang W, Ma
J, Wu S and Cheng W: Medroxyprogestogen enhances apoptosis of
SKOV-3 cells via inhibition of the PI3K/AKT signaling pathway. J
Biomed Res. 27:43–50. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
37
|
Rajput S, Kumar BN, Sarkar S, Das S, Azab
B, Santhekadur PK, Das SK, Emdad L, Sarkar D, Fisher PB and Mandal
M: Targeted apoptotic effects of thymoquinone and tamoxifen on XIAP
mediated AKT regulation in breast cssancer. PLoS One. 8:e613422013.
View Article : Google Scholar
|
|
38
|
Skrzypski M, Kaczmarek P, Le TT,
Wojciechowicz T, Pruszyńska-Oszmalek E, Szczepankiewicz D, Sassek
M, Arafat A, Wiedenmann B, Nowak KW and Strowski MZ: Effects of
orexin A on proliferation, survival, apoptosis and differentiation
of 3T3-L1 preadipocytes into mature adipocytes. FEBS Lett.
586:4157–4164. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
39
|
Cryns V and Yuan J: Protease to die for.
Genes Dev. 12:1551–1570. 1998. View Article : Google Scholar : PubMed/NCBI
|