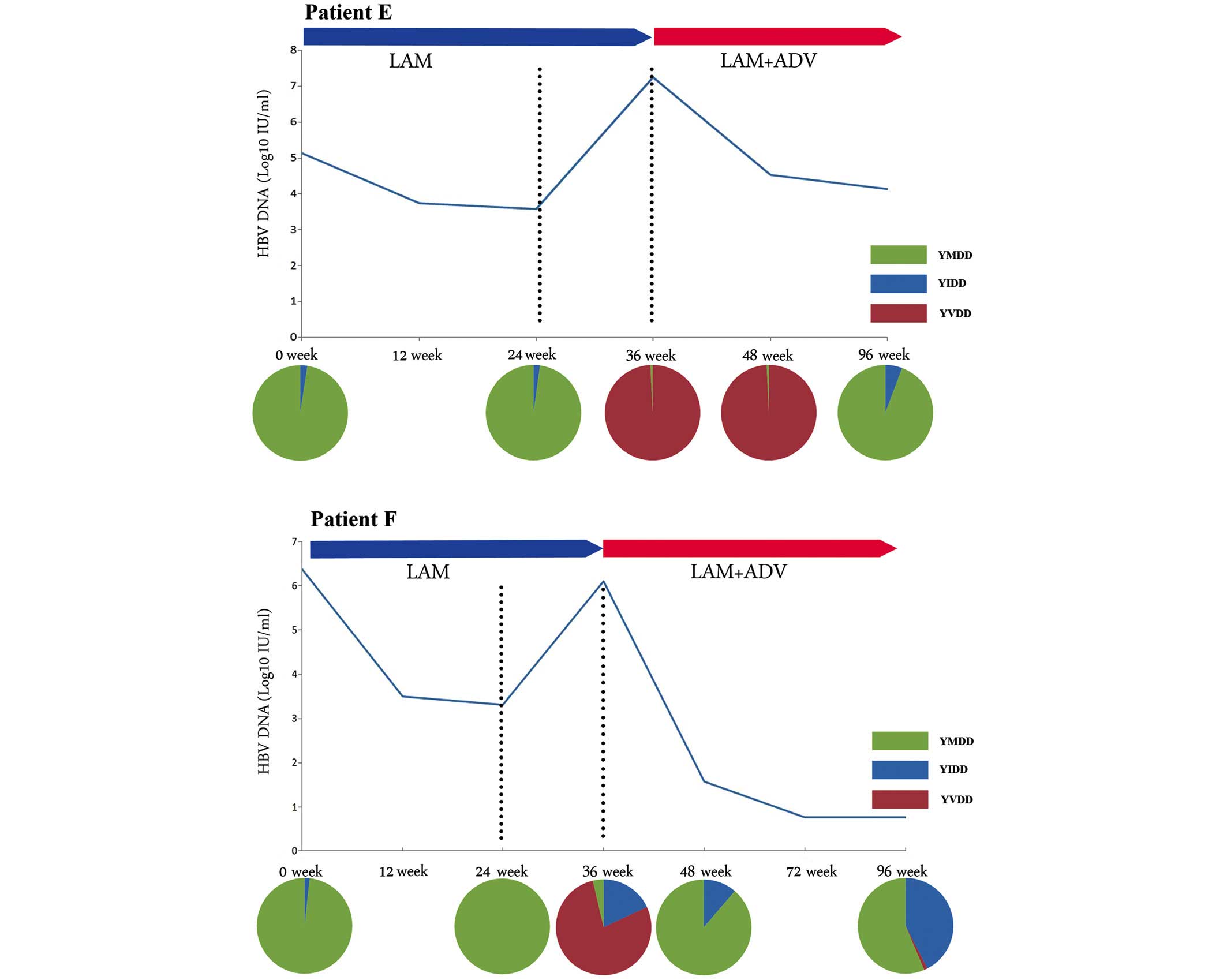
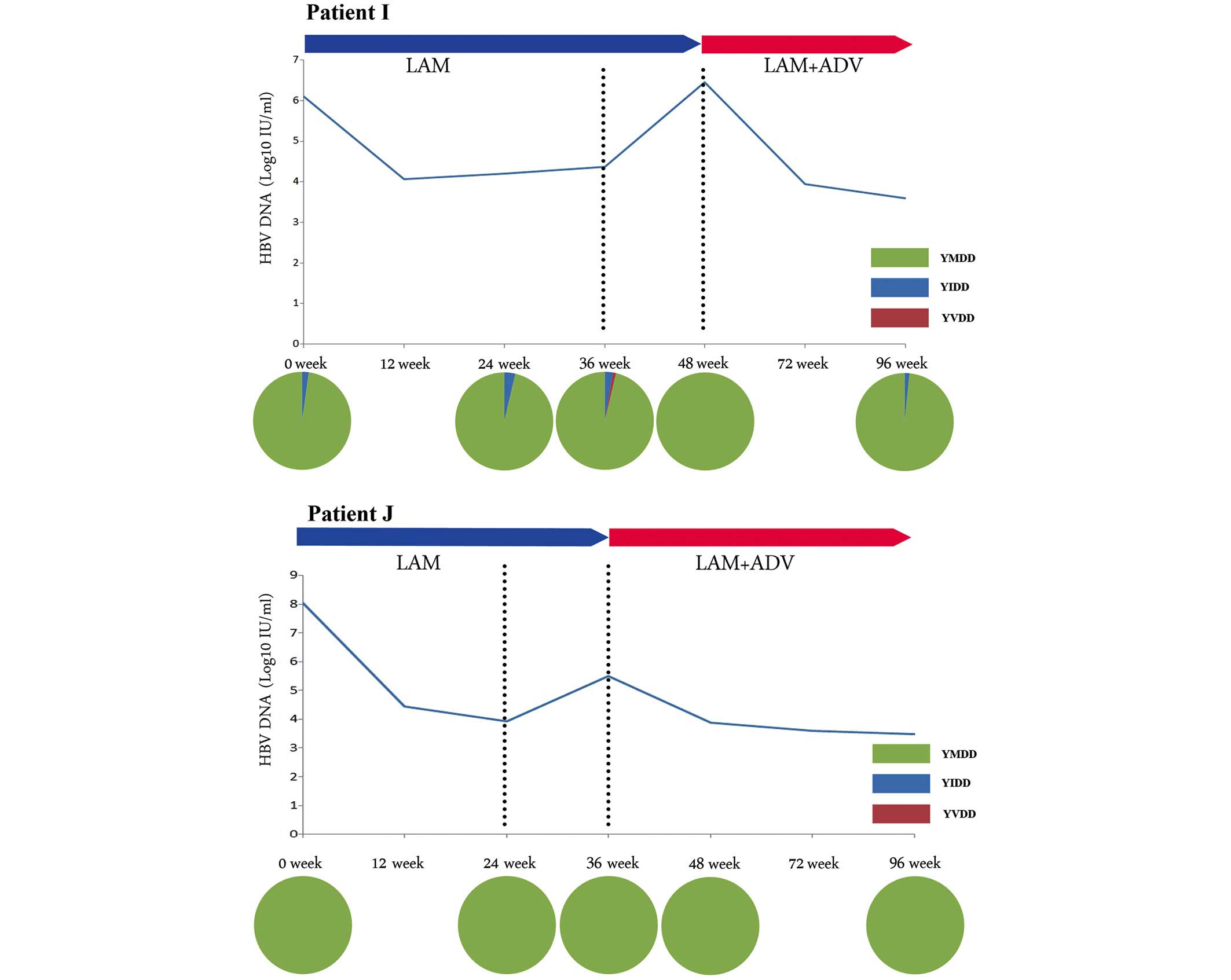
|
1
|
Lavanchy D: Hepatitis B virus
epidemiology, disease burden, treatment and current and emerging
prevention and control measures. J Viral Hepat. 11:97–107. 2004.
View Article : Google Scholar : PubMed/NCBI
|
|
2
|
Trépo C, Chan HL and Lok A: Hepatitis B
virus infection. Lancet. 384:2053–2063. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
3
|
Tujios SR and Lee WM: Update in the
management of chronic hepatitis B. Curr Opin Gastroenterol.
29:250–256. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
4
|
Ma H and Jia J: Why do I treat
HBeAg-positive chronic hepatitis B patients with a nucleoside
analogue. Liver Int. 33(Suppl 1): S133–S136. 2013. View Article : Google Scholar
|
|
5
|
Fung J, Lai CL, Seto WK and Yuen MF:
Nucleoside/nucleotide analogues in the treatment of chronic
hepatitis B. J Antimicrob Chemother. 66:2715–2725. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
6
|
Chotiyaputta W and Lok AS: Hepatitis B
virus variants. Nat Rev Gastroenterol Hepatol. 6:453–462. 2009.
View Article : Google Scholar : PubMed/NCBI
|
|
7
|
Rodriguez-Frias F, Buti M, Tabernero D and
Homs M: Quasispecies structure, cornerstone of hepatitis B virus
infection: Mass sequencing approach. World J Gastroenterol.
19:6995–7023. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
8
|
Capobianchi MR, Giombini E and Rozera G:
Next-generation sequencing technology in clinical virology. Clin
Microbiol Infect. 19:15–22. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
9
|
Abbate I, Rozera G, Tommasi C, Bruselles
A, Bartolini B, Chillemi G, Nicastri E, Narciso P, Ippolito G and
Capobianchi MR: Analysis of co-receptor usage of circulating viral
and proviral HIV genome quasispecies by ultra-deep pyrosequencing
in patients who are candidates for CCR5 antagonist treatment. Clin
Microbiol Infect. 17:725–731. 2011. View Article : Google Scholar
|
|
10
|
Armenia D, Vandenbroucke I, Fabeni L, Van
Marck H, Cento V, D'Arrigo R, Van Wesenbeeck L, Scopelliti F,
Micheli V, Bruzzone B, et al: Study of genotypic and phenotypic
HIV-1 dynamics of integrase mutations during raltegravir treatment:
A refined analysis by ultra-deep 454 pyrosequencing. J Infect Dis.
205:557–567. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
11
|
Trimoulet P, Pinson P, Papuchon J, Foucher
J, Vergniol J, Chermak F, Wittkop L, Castaing N, Merrouche W,
Reigadas S, et al: Dynamic and rapid changes in viral quasispecies
by UDPS in chronic hepatitis C patients receiving telaprevir-based
therapy. Antivir Ther. 18:723–727. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
12
|
Applegate TL, Gaudieri S, Plauzolles A,
Chopra A, Grebely J, Lucas M, Hellard M, Luciani F, Dore GJ and
Matthews GV: Naturally occurring dominant drug resistance mutations
occur infrequently in the setting of recently acquired hepatitis C.
Antivir Ther. 20:199–208. 2015. View
Article : Google Scholar :
|
|
13
|
Cortes KC, Zagordi O, Perlejewski K,
Laskus T, Maroszek K, Bukowska-Ośko I, Pawełczyk A, Płoski R, Berak
H, Horban A and Radkowski M: Deep sequencing of hepatitis C virus
hypervariable region 1 reveals no correlation between genetic
heterogeneity and antiviral treatment outcome. BMC Infect Dis.
14:3892014. View Article : Google Scholar : PubMed/NCBI
|
|
14
|
Wang H, Ji YY, Yao GB, Ma XY, Xie Q, Pang
HY, Wu SM, Li J, Chen CW, Xu XW and Gu EL: Two years efficiency of
lamivudine and adefovir dipivoxil combined therapy in chronic
hepatitis B patients. Eur Rev Med Pharmacol Sci. 17:636–643.
2013.PubMed/NCBI
|
|
15
|
European Association For The Study Of The
Liver: EASL clinical practice guidelines: Management of chronic
hepatitis B virus infection. J Hepatol. 57:167–185. 2012.
View Article : Google Scholar : PubMed/NCBI
|
|
16
|
Wu J, Liu W, He L, Huang F, Chen J, Cui P,
Shen Y, Zhao J, Wang W, Zhang Y, et al: Sputum microbiota
associated with new, recurrent and treatment failure tuberculosis.
PLoS One. 8:e834452013. View Article : Google Scholar : PubMed/NCBI
|
|
17
|
Hamady M and Knight R: Microbial community
profiling for human microbiome projects: Tools, techniques, and
challenges. Genome Res. 19:1141–1152. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
18
|
Zoulim F and Locarnini S: Hepatitis B
virus resistance to nucleos(t)ide analogues. Gastroenterology.
137:1593–1608.e1. –e2. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
19
|
Rhee SY, Margeridon-Thermet S, Nguyen MH,
Liu TF, Kagan RM, Beggel B, Verheyen J, Kaiser R and Shafer RW:
Hepatitis B virus reverse transcriptase sequence variant database
for sequence analysis and mutation discovery. Antiviral Res.
88:269–275. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
20
|
Chan HL and Jia J: Chronic hepatitis B in
Asia-new insights from the past decade. J Gastroenterol Hepatol.
26(Suppl 1): 131–137. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
21
|
Wargo AR and Kurath G: Viral fitness:
Definitions, measurement, and current insights. Curr Opin Virol.
2:538–545. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
22
|
Gong L, Han Y, Chen L, Liu F, Hao P, Sheng
J, Li XH, Yu DM, Gong QM, Tian F, et al: Comparison of
next-generation sequencing and clone-based sequencing in analysis
of hepatitis B virus reverse transcriptase quasispecies
heterogeneity. J Clin Microbiol. 51:4087–4094. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
23
|
Ramírez C, Gregori J, Buti M, Tabernero D,
Camós S, Casillas R, Quer J, Esteban R, Homs M and Rodriguez-Frías
F: A comparative study of ultra-deep pyrosequencing and cloning to
quantitatively analyze the viral quasispecies using hepatitis B
virus infection as a model. Antiviral Res. 98:273–283. 2013.
View Article : Google Scholar : PubMed/NCBI
|
|
24
|
Rodriguez C, Chevaliez S, Bensadoun P and
Pawlotsky JM: Characterization of the dynamics of hepatitis B virus
resistance to adefovir by ultra-deep pyrosequencing. Hepatology.
58:890–901. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
25
|
Hongthanakorn C, Chotiyaputta W,
Oberhelman K, Fontana RJ, Marrero JA, Licari T and Lok AS:
Virological breakthrough and resistance in patients with chronic
hepatitis B receiving nucleos(t)ide analogues in clinical practice.
Hepatology. 53:1854–1863. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
26
|
Menendez-Arias L, Alvarez M and Pacheco B:
Nucleoside/nucleotide analog inhibitors of hepatitis B virus
polymerase: mechanism of action and resistance. Curr Opin Virol.
8:1–9. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
27
|
Keeffe EB, Zeuzem S, Koff RS, Dieterich
DT, Esteban-Mur R, Gane EJ, Jacobson IM, Lim SG, Naoumov N,
Marcellin P, et al: Report of an international workshop: Roadmap
for management of patients receiving oral therapy for chronic
hepatitis B. Clin Gastroenterol Hepatol. 5:890–897. 2007.
View Article : Google Scholar : PubMed/NCBI
|
|
28
|
Lo AO, Wong VW, Wong GL, Chan HY, Cheung
CM and Chan HL: Efficacy of entecavir switch therapy in chronic
hepatitis B patients with incomplete virological response to
telbivudine. Antivir Ther. 18:671–679. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
29
|
Piratvisuth T, Komolmit P, Tanwandee T,
Sukeepaisarnjaroen W, Chan HL, Pessôa MG, Fassio E, Ono SK, Bessone
F, Daruich J, et al: 52-week efficacy and safety of telbivudine
with conditional tenofovir intensification at week 24 in
HBeAg-positive chronic hepatitis B. PLoS One. 8:e542792013.
View Article : Google Scholar : PubMed/NCBI
|
|
30
|
Sun J, Xie Q, Tan D, Ning Q, Niu J, Bai X,
Fan R, Chen S, Cheng J, Yu Y, et al: The 104-week efficacy and
safety of telbivudine-based optimization strategy in chronic
hepatitis B patients: A randomized, controlled study. Hepatology.
59:1283–1292. 2014. View Article : Google Scholar : PubMed/NCBI
|