Introduction
Heat stress may affect male reproductive functions.
The mammalian scrotal temperature is 2–8°C lower than the core body
temperature (1), and higher
temperatures may eliminate the spermatogonial germ cells in the
seminiferous tubules, resulting in decreased sperm density
(2,3). This may further induce testicular
tissue apoptosis and morphological changes affecting sperm
production processes, resulting in infertility (4–6). In
addition, increased scrotal temperatures resulting from
occupational exposure, lifestyle or cryptorchidism are notable
factors leading to male infertility (7). Limited research has been conducted on
the epididymis (8). Spermatozoa
are produced in the testis and acquire the maturation ability in
the epididymis, particularly in the caput epididymis, by
interacting with the epididymal fluids (9,10).
Although numerous studies have investigated the effect of heat
stress of the testis on spermatogenesis (11–13),
the association of heat stress and sperm maturation has yet to be
elucidated.
In the present study, a short heat stress was
performed on the rat epididymis to identify the affected sperm
maturation proteins. The epididymal fluid proteins identified may
provide useful information for the understanding of sperm
maturation, and provide clues for the screening of male
contraceptive agents.
Materials and methods
Sample preparation
Male Sprague-Dawley rats (n=16; weight, 300–400 g;
age, 8–10 weeks) were provided by the Experimental Animal Center of
Binzhou Medical College (Shandong, China). They were housed at
20±2°C with ad libitum access to food and water and a 12 h
light/dark cycle. Rats were randomly divided into two groups and
acclimatized to the experimental conditions for one week prior to
commencement of the experiments as follows: The normal control
group, rats were in the water bath for 60 min once prior to the
experiment at room temperature; the heat treatment group, rats
received heat shock pretreatment in a water bath once prior to the
experiment at 42°C for 60 min. The animals were then sacrificed by
intraperitoneal injection with ketamine (2.4 ml.kg; Fujian Gutian
Yuanhang Medical Company, Ningde, China). The study was approved by
the ethics committee of Yu Huang Ding Hospital (Yantai, China).
Protein extraction
Rat caput epididymides were collected for fluid
protein extraction. Briefly, the epididymides were coarsely minced
and gentle pressure was applied to release luminal fluid into
phosphate-buffered saline. Following centrifugation at 5,700 × g at
4°C for 10 min and microscopic examination (DM LB2; Leica
Microsystems, Wetzlar, Germany), the resultant sperm-free
supernatant was precipitated with four volumes of ice-cold acetone.
The precipitates were dissolved in 3 ml protein lysis buffer
(Sigma-Aldrich, St. Louis, MO, USA) consisting of 7 M urea, 2 M
thiourea, 4% (w/v) 3-[(3-cholamidopropyl)
dimethylammonio]-1-propanesulfonate and 65 mM dithiothreitol.
Protein concentration was determined using the Bradford assay
(Thermo Fisher Scientific, Inc., Waltham, MO, USA) and the samples
were stored at −80°C.
Two-dimensional gel electrophoresis
(2-DE)
The first dimension of the 2-DE was the separation
of proteins by isoelectric focusing using 18 cm nonlinear pH 3–10
immobilized pH gradient (IPG) strips (GE Healthcare Life Sciences,
Chalfont, UK). Subsequent to reduction and iodoacetamide
(Sigma-Aldrich), the IPG strips were run on a 12.5% (w/v) SDS-PAGE.
The gels were stained with Coomassie Brilliant Blue R-350 (GE
Healthcare Life Sciences), and were scanned by the Z320 scanner
(Founder Technology Group Co., Ltd., Beijing, China). The gel maps
were analyzed with Imagemaster 2D Platinum software, version 6.0
(GE Healthcare Life Sciences), and the electrophoresis experiments
were performed in triplicates. The gel spots were excised,
destained with 25 mM NH4HCO3/50% (v/v)
acetonitrile (both purchased from Yantai Sanhe Chemical Reagent
Company, Yanta, China), and digested by trypsin (Thermo Fisher
Scientific, Inc.) in 25 mM NH4HCO3 at 37°C
for 12 h. The resulting peptides were analyzed using the Applied
Biosystems Voyager-DE STR Biospectrometry Workstation (Thermo
Fisher Scientific, Inc.). The spectra data of the mass spectrometry
were searched against the NCBInr database for Rattus
norvegicus with Mascot (www.matrixscience.com/; Matrix Science Ltd., London,
UK). Providing that the protein score was >60 and the matched
peptides ≥4 in the peptide mass fingerprinting search, the protein
was confirmed as a successful identification. In the case that one
gel spot corresponded to >1 protein, then the protein with the
highest score was selected.
Bioinformatics
Proteins were distinguished functionally by a
step-by-step classification and each protein was placed in only one
category. Broad functions were classified according to the
annotations in the PIR database (pir.georgetown.edu) and gene ontology (GO) annotations
(www.geneontology.org), including
molecular function and biological process.
Immunohistochemical analysis
The rat caput epididymides were fixed in Bouin's
solution (Sigma-Aldrich) for 10 h at room temperature, and embedded
with paraffin (Yantai Sanhe Chemical Reagent Company). For antigen
retrieval, the 4-µm thick sections were cut and heated in a
microwave oven for 15 min. To eliminate the endogenous peroxidases,
3% (v/v) H2O2 was used to incubate the
sections for 10 min. Subsequent to antigen retrieval, blocking was
conducted with 3% bovine serum albumin for 30 min at 37°C. Then,
the samples were incubated with the following antibodies (dilution,
1:50) overnight at 4°C: Polyclonal anti-rabbit clusterin (sc-8354),
peroxiredoxin 6 (sc-134478) and superoxide dismutase-1 (sc-11407)
(all purchased from Santa Cruz Biotechnology, Inc., Dallas, TX,
USA). Sections were washed three times with Tris-buffered saline
(TBS) and then incubated with horseradish peroxidase
(HRP)-conjugated anti-rabbit IgG (dilution, 1:500; Zhongshan
Beijing Biotechnology Co., Ltd., Guangdong, China) for 1 h at 37°C.
A DAB kit (Zhongshan Beijing Biotechnology Co., Ltd.) was used to
reveal the binding sites, and then the sections were counterstained
with hematoxylin and eosin (HE; Abcam, Cambridge, MA, USA), and
mounted for bright-field microscopy with a DM LB2 microscope (Leica
Microsystems). Pre-immune rabbit IgG (sc-2027; Santa Cruz
Biotechnology, Inc.) was used instead of the primary antibody as
negative control.
Western blot analysis
Protein samples (50 µg) were separated by
12.5% (w/v) SDS-PAGE, and transferred to polyvinylidene fluoride
membranes. Subsequently, the membranes were blocked with 5% (w/v)
skimmed milk for 1 h and incubated with clusterin, peroxiredoxin 6
and superoxide dismutase-1 primary antibodies at room temperature
for 1 h. Membranes were washed three times with TBS-Tween 20 and
incubated with an HRP-conjugated anti-IgG for 1 h. A DAB kit
(Zhongshan Beijing Biotechnology Co., Ltd.) was used to visualize
the proteins. Western blot images were analyzed by densitometric
scanning and quantified using ImageQuant TL software (version 7.0;
GE Healthcare).
Tunel assay
DNA fragmentation was visualized by TUNEL assay
using the In Situ Cell Death Detection Kit, Fluorescein
(Roche Diagnostics GmbH, Mannheim, Germany), according to the
manufacturer's instructions. Sections were photographed by confocal
laser scanning microscopy using a LSM-510 META device (Carl Zeiss
AG, Oberkochen, Germany). The TUNEL positive cells (green staining)
were photographed and evaluated qualitatively using ImageQuant TL
7.0 software (GE Healthcare). Negative controls were processed with
TUNEL-Label Solution (Roche Diagnostics) without terminal
transferase instead of the TUNEL reaction mixture.
Statistical analysis
The unpaired t-test was used to assess the
differences among groups, using SPSS software, version 13.0 (SPSS,
Inc., Chicago, IL, USA). Data are presented as the mean ± standard
deviation. P<0.05 was considered to indicate a statistically
significant difference.
Results
Morphological characteristics of rat
caput epididymis treated by short-term heat treatment
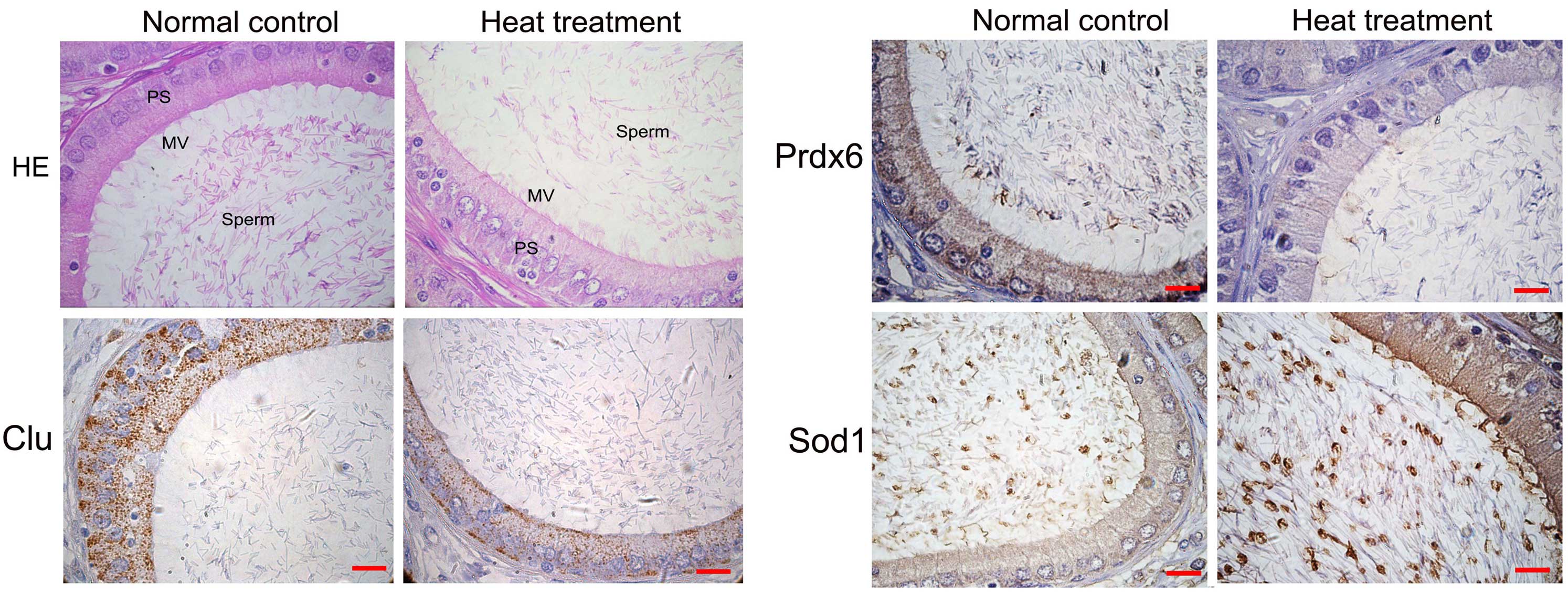
As demonstrated in Fig.
1, HE staining of rat caput epididymis indicated no significant
changes in cellular morphologies or lumen dimensions in the caput
epididymides of the heat treatment group compared with the normal
control group. However, nuclei cavitations were observed in the rat
caput epididymis of the heat treatment group (Fig. 1; HE staining).
Further cellular localizations of clusterin (Clu),
peroxiredoxin 6 (Prdx6) and superoxide dismutase (Sod1) were
investigated by immunohistochemistry. The results demonstrated that
Sod1, Prdx6 and Clu were mainly expressed by epididymal epithelial
cells (Fig. 1). Following heat
treatment, Prdx6 and Clu demonstrated decreased levels of
expression, and Sod1 indicated increased levels compared with the
normal control group (Fig. 1).
Identification of differentially
expressed proteins in rat caput epididymis with short-term heat
stress
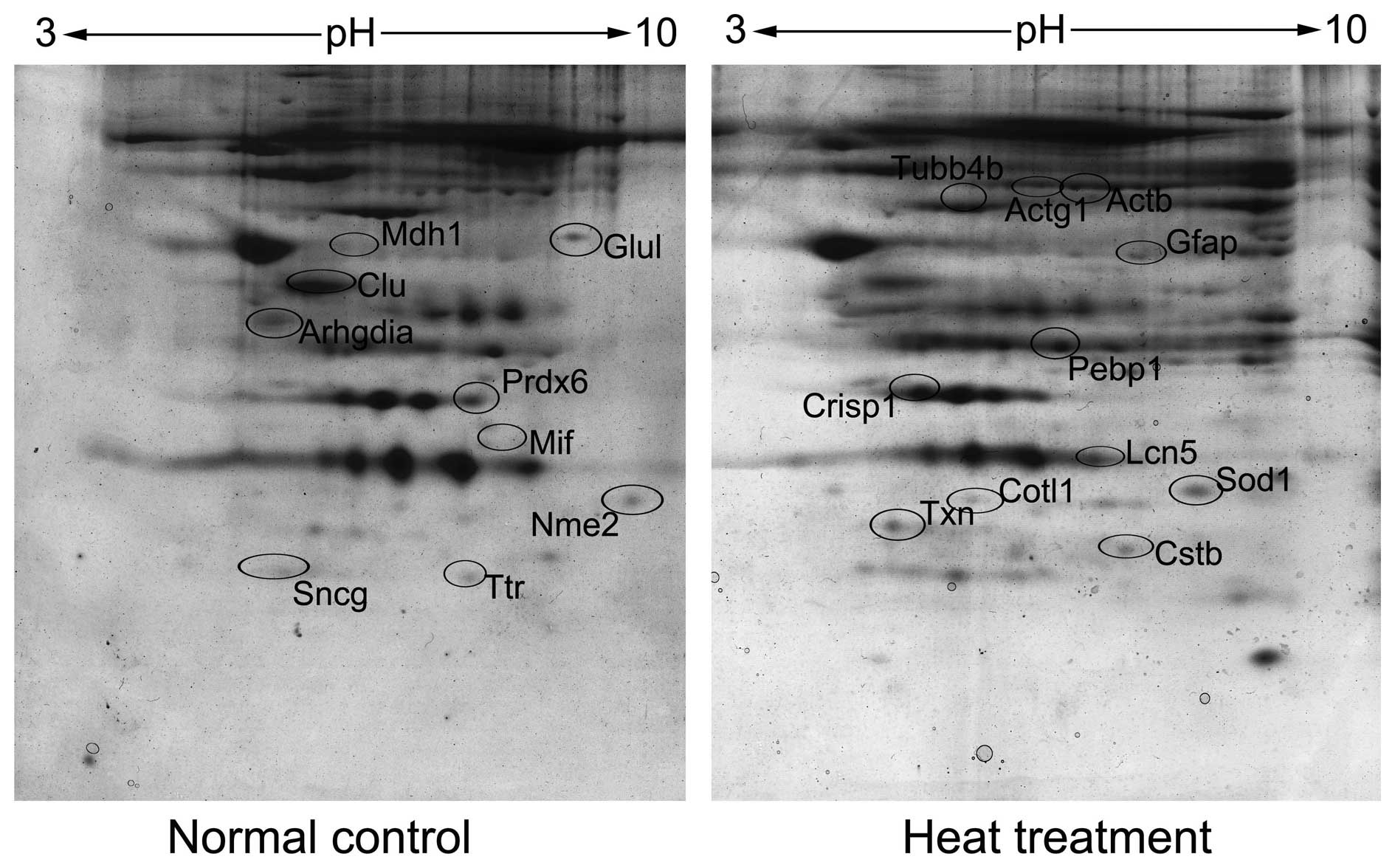
2-DE followed by mass identification was performed
to identify differentially expressed proteins. Triple gels were
repeatedly made and statistically compared by Imagemaster software.
A total of 29 protein spots demonstrated different density levels
between the heat treated and normal control groups (fold change,
>1.5). From the total number of spots, 21 unique proteins were
identified, including 11 proteins upregulated and 10 proteins
downregulated in the treatment group (Fig. 2; Table
I).
 | Table IDifferentially expressed proteins
between normal control and heat treatment of rat caput
epididymis. |
Table I
Differentially expressed proteins
between normal control and heat treatment of rat caput
epididymis.
| Spot no. | Swiss-Prot
accession | Molecular weight | Gene name | Score | Protein name |
Treatment/control |
|---|
| Antioxidant | | | | | | |
| F11 | O35244 | 24860 | Prdx6 | 835 | Peroxiredoxin-6 | 0.38 |
| A4 | P07632 | 16073 | Sod1 | 397 | Superoxide dismutase
(Cu-Zn) | 2.15 |
| G3 | P09606 | 42982 | Glul | 853 | Glutamine
synthetase | 0.43 |
| G16 | P11232 | 12008 | Txn | 294 | Thioredoxin | 2.32 |
| Chaperone | | | | | | |
| A1 | B0BNA5 | 16036 | Cotl1 | 73 | Coactosin-like
protein | 2.54 |
| Metabolism | | | | | | |
| C12 | O88989 | 36631 | Mdh1 | 82 | Malate dehydrogenase,
cytoplasmic | 0.33 |
| H6 | P19804 | 17386 | Nme2 | 553 | Nucleoside
diphosphate kinase B | 0.41 |
| A20 | P47819 | 49984 | Gfap | 91 | Glial fibrillary
acidic protein | 2.65 |
| P/P inhibitor | | | | | | |
| A17 | P01041 | 11303 | Cstb | 106 | Cystatin-B | 3.01 |
| G6 | Q5XI73 | 23450 | Arhgdia | 181 | Rho GDP-dissociation
inhibitor 1 | 0.36 |
| Reproduction | | | | | | |
| F2 | P06911 | 20828 | Lcn5 | 849 | Epididymal-specific
lipocalin-5 | 2.89 |
| A15 | P12020 | 28741 | Crisp1 | 459 | Cysteine-rich
secretory protein 1 | 2.64 |
| F13 | P05371 | 51970 | Clu | 430 | Clusterin | 0.28 |
| H2 | P30904 | 12640 | Mif | 127 | Macrophage
migration inhibitory factor | 0.34 |
| Structure | | | | | | |
| F24 | P60711 | 42052 | Actb | 599 | Actin, cytoplasmic
1 | 3.12 |
| E2 | P62986 | 8560 | Uba52 | 331 | Ubiquitin | 0.27 |
| F19 | P63259 | 42108 | Actg1 | 482 | Actin, cytoplasmic
2 | 2.57 |
| E13 | Q63544 | 12969 | Sncg | 140 | γ-Synuclein | 0.38 |
| B8 | Q6P9T8 | 50225 | Tubb4b | 425 | Tubulin β-4B
chain | 2.36 |
| Transporter | | | | | | |
| G24 | P02767 | 15824 | Ttr | 329 | Transthyretin | 0.47 |
| F8 | P31044 | 20902 | Pebp1 | 823 |
Phosphatidylethanolamine-binding protein
1 | 2.63 |
Broad functional analysis
All differentially expressed proteins were broadly
classified into seven functional groups according to GO and
literature annotations (Table I).
These proteins were mainly involved in functions of structure
(23.8%), reproduction (19%), antioxidants (19%), metabolism,
chaperones, proteases/inhibitors and transporters (Table I).
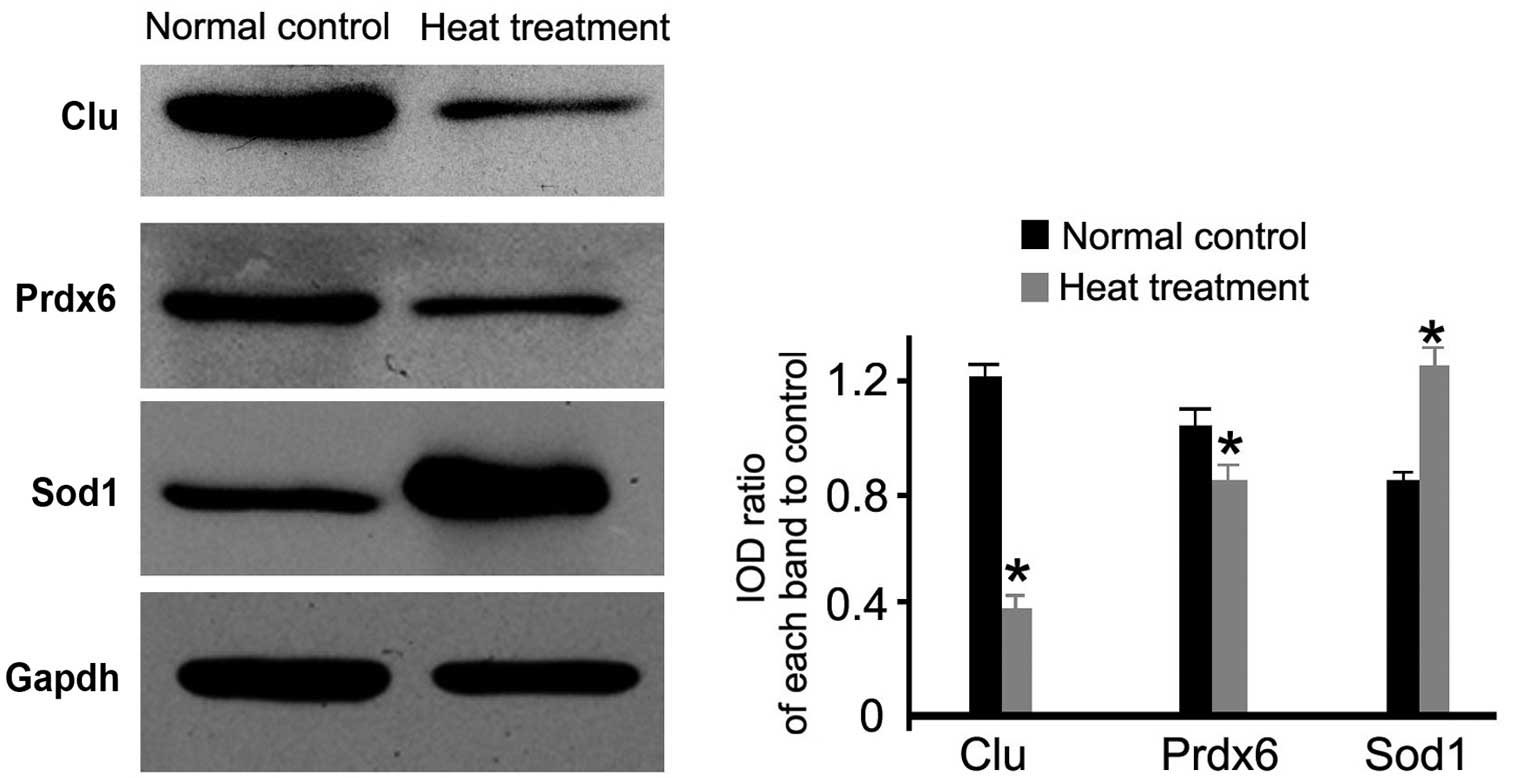
Western blot analysis
Antioxidant-associated (Sod1 and Prdx6) and a
reproduction-associated protein (Clu) were selected for validation
of their expression using western blotting. As demonstrated in
Fig. 3, the heat treatment group
exhibited significantly lower expression levels of Prdx6 and Clu
compared with the normal control group (P<0.05; Fig. 3). In addition, higher expression
levels of Sod1 were observed in the heat-treated group compared
with the normal control group (P<0.05; Fig. 3). The results were consistent with
the proteomic analysis.
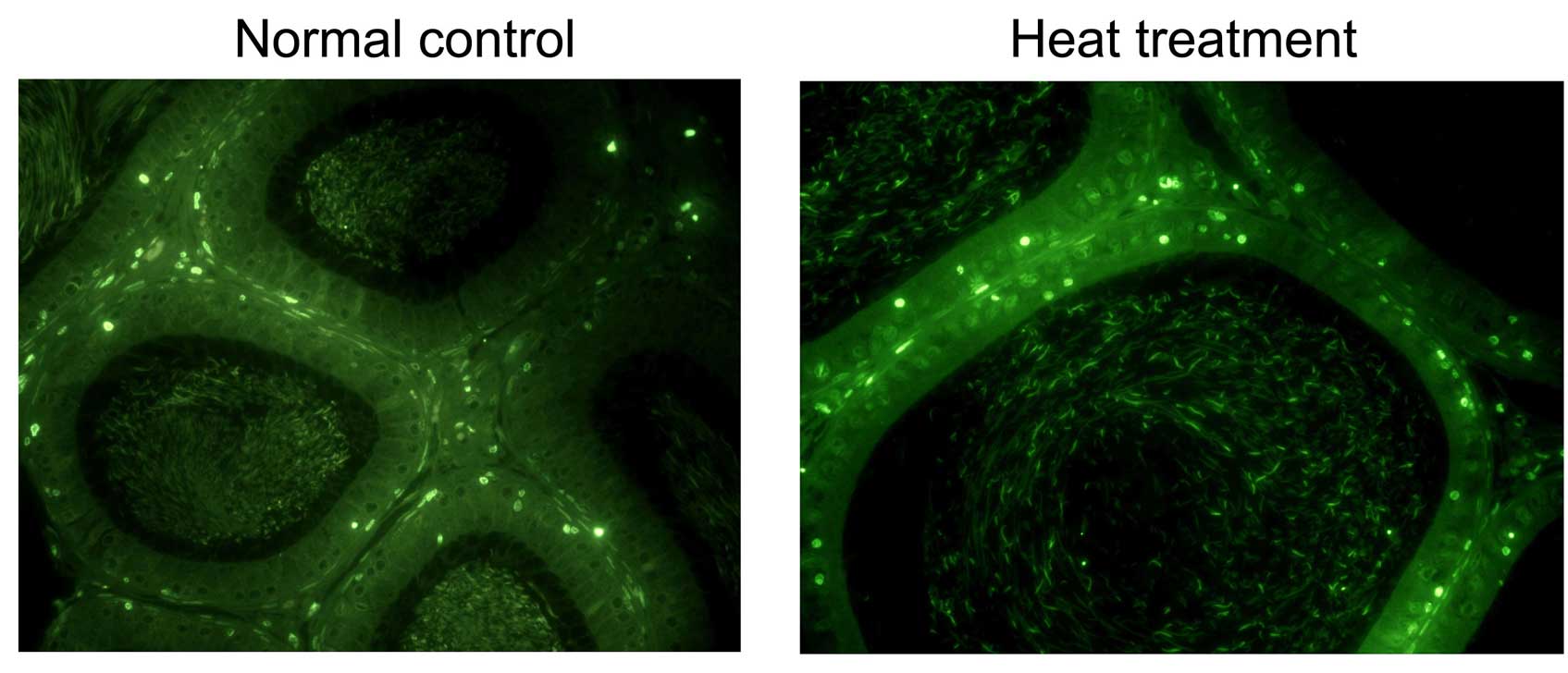
TUNEL assay
TUNEL assay was performed to determine the effect of
heat stress on the apoptotic status. Positive stained cells in the
heat-treated epididymis (68±5.2%) was significantly higher than
that in the control group (16±4.2%). As demonstrated in Fig. 4, positive staining results were
observed in epididymal epithelial cells from the heat-treated
rats.
Discussion
Increased temperature has an effect on the male
reproductive ability by declining male fertility (14). Heat stress may have a negative
effect on male fertility through oxidant damage by producing ROS
and further changing the intracellular signal transduction
(15). This effect may induce germ
cell loss and poor semen quality, however, its effect on sperm
maturation, particularly on the sperm maturation milieu, is not
clear (16,17). In the present study, a short-term
heat stress was performed on rats by exposure to high environmental
temperature (42°C) for 1 h, followed by identification of altered
key sperm milieu proteins by comparing a proteomic analysis of
caput epididymal fluids. To the best of our knowledge, this is the
first study attempting to identify sperm milieu proteins affected
by short-term heat stress, and may provide information for further
understanding of sperm maturation.
Caput epididymal fluids provide a microenvironment
for sperm maturation for active epithelial secretion and absorption
activity (18). This
microenvironment serves as the first step for the sperm to obtain
its maturation ability. The present study hypothesized that heat
stress may affect caput epididymal epithelial protein expression
patterns, further altering caput epididymal fluid proteins that are
mainly associated with sperm maturation.
The present study may provide understanding for
heat-stress-induced reproductive dysfunction in males. The
morphological analysis of the present study demonstrated no
significant differences between the treated and normal control
groups of rat caput epididymides. This implies that short-term heat
stress may not affect epithelial structures. To further explore the
effect on the main activities of caput epididymis on protein
synthesis and secretion, a comparative proteomic study was
performed to identify differentially expressed fluid proteins. A
total of 29 proteins were identified to have varying expression
levels between the treated and normal control groups. A broad
functional classification categorized these proteins into seven
functional groups, and the majority were associated with
antioxidant and reproductive functions.
Antioxidant enzymes in the epididymal lumen protect
spermatozoa from oxidative damage. Optimal antioxidant levels serve
crucial roles in sperm function (particularly sperm motility)
(19). The results of the present
study demonstrated that Prdx6 and Glul, two proteins with
protective roles in sperm, had lower expression levels in the
epididymal fluids following heat stress, suggesting that an altered
protection balance may occur in epididymal fluids induced by heat
stress. Another two ROS-associated proteins, Sod1 and Txn,
presented with upregulated expression levels, potentially
implicating an epididymis response partly compensating for
spermatozoa oxidative damage following heat stress. These proteins
were associated with certain aspects of sperm maturation (20), and the association of their
function with heat stress was further investigated. Another
promising function group was the reproduction-associated proteins.
These proteins were established sperm proteins, such as Clu, a
heterodimeric glycoprotein produced by a variety of tissues and
present in numerous biological fluids (21). In addition, Clu is a major
component of epididymal fluids, and mainly secreted in the caput
epididymis. Previous studies have demonstrated that Clu is involved
in cell adhesion, apoptosis and transformation functions, and sperm
maturation (18,22,23).
In the present study, Clu levels were observed to be significantly
lower following heat stress, suggesting that heat stress may have a
direct effect on the epididymal fluid component, and directly or
indirectly alter sperm maturation. The above proteins should be
further validated and investigated to identify their regulation and
roles in sperm maturation.
In the present study, western blot analysis
validated the expression levels of proteins in epididymal fluids,
and immunohistochemical analysis identified the expression of
proteins in the epididymal epithelia. The results indicated the
same altered expression in epididymal epithelia and fluids,
suggesting that heat stress induced protein expression changes in
epididymal fluids.
In conclusion, by utilizing heat stress exposure on
rat epididymis, a set of differentially expressed caput epididymal
fluid proteins were identified, whose activities were associated
with sperm maturation. The results of the present study, in
combination with data from our previous work, will be used as the
basis for future studies on biological functions of the epididymis
and sperm maturation.
Acknowledgments
The current study was supported by the National
Natural Science Foundation of China (grant no. 81300533) and
Shandong Provincial Natural Science Foundation, China (grant nos.
ZR2013HQ002 and ZR2014HQ068).
References
|
1
|
Banks S, King SA, Irvine DS and Saunders
PT: Impact of a mild scrotal heat stress on DNA integrity in murine
spermatozoa. Reproduction. 129:505–514. 2005. View Article : Google Scholar : PubMed/NCBI
|
|
2
|
Wang P, Li Y, Hu XC, Cai XL, Hou LP, Wang
YF, Hu JH, Li QW, Suo LJ, Fan ZG and Zhang B: Cryoprotective
effects of low-density lipoproteins, trehalose and soybean lecithin
on murine spermatogonial stem cells. Zygote. 22:158–163. 2014.
View Article : Google Scholar
|
|
3
|
Rasooli A, Taha Jalali M, Nouri M,
Mohammadian B and Barati F: Effects of chronic heat stress on
testicular structures, serum testosterone and cortisol
concentrations in developing lambs. Anim Reprod Sci. 117:55–59.
2010. View Article : Google Scholar
|
|
4
|
Li YS, Piao YG, Nagaoka K, Watanabe G,
Taya K and Li CM: Preventive effect of tert-butylhydroquinone on
scrotal heat-induced damage in mouse testes. Genet Mol Res.
12:5433–5441. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
5
|
Pei Y, Wu Y, Cao J and Qin Y: Effects of
chronic heat stress on the reproductive capacity of male Rex
rabbits. Livest Sci. 146:13–21. 2012. View Article : Google Scholar
|
|
6
|
Takahashi M: Heat stress on reproductive
function and fertility in mammals. Reprod Med Biol. 11:37–47. 2012.
View Article : Google Scholar
|
|
7
|
Kim B, Park K and Rhee K: Heat stress
response of male germ cells. Cell Mol Life Sci. 70:2623–2636. 2013.
View Article : Google Scholar
|
|
8
|
Hou Y, Wang X, Lei Z, Ping J, Liu J, Ma Z,
Zhang Z, Jia C, Jin M, Li X, et al: Heat-stress-induced metabolic
changes and altered male reproductive function. J Proteome Res.
14:1495–503. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
9
|
Downs SM: Nutrient pathways regulating the
nuclear maturation of mammalian oocytes. Reprod Fertil Dev. Mar
24–2015.Epub ahead of print. View
Article : Google Scholar : PubMed/NCBI
|
|
10
|
Gatti JL, Castella S, Dacheux F, Ecroyd H,
Métayer S, Thimon V and Dacheux JL: Post-testicular sperm
environment and fertility. Anim Reprod Sci. 82–83:321–339. 2004.
View Article : Google Scholar
|
|
11
|
Shen H, Liao K, Zhang W, Wu H, Shen B and
Xu Z: Differential expression of peroxiredoxin 6, annexin A5 and
ubiquitin carboxyl-terminal hydrolase isozyme L1 in testis of rat
fetuses after maternal exposure to di-n-butyl phthalate. Reprod
Toxicol. 39:76–84. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
12
|
Fu-Jun L and Xiao-Fang S: Comparative
analysis of human reproductive proteomes identifies candidate
proteins of sperm maturation. Mol Biol Rep. 39:10257–10263. 2012.
View Article : Google Scholar : PubMed/NCBI
|
|
13
|
Xun W, Shi L, Cao T, Zhao C, Yu P, Wang D,
Hou G and Zhou H: Dual functions in response to heat stress and
spermatogenesis: Characterization of expression profile of small
heat shock proteins 9 and 10 in goat testis. Biomed Res Int.
2015:6862392015. View Article : Google Scholar : PubMed/NCBI
|
|
14
|
Paul C, Teng S and Saunders PT: A single,
mild, transient scrotal heat stress causes hypoxia and oxidative
stress in mouse testes, which induces germ cell death. Biol Reprod.
80:913–919. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
15
|
Sanocka D and Kurpisz M: Reactive oxygen
species and sperm cells. Reprod Biol Endocrinol. 2:122004.
View Article : Google Scholar : PubMed/NCBI
|
|
16
|
Saez F, Frenette G and Sullivan R:
Epididymosomes and prostasomes: Their roles in posttesticular
maturation of the sperm cells. J Androl. 24:149–154. 2003.
View Article : Google Scholar : PubMed/NCBI
|
|
17
|
Dacheux JL and Dacheux F: New insights
into epididymal function in relation to sperm maturation.
Reproduction. 147:R27–R42. 2014. View Article : Google Scholar
|
|
18
|
Maldera JA, Vasen G, Ernesto JI,
Weigel-Muñoz M, Cohen DJ and Cuasnicu PS: Evidence for the
involvement of zinc in the association of CRISP1 with rat sperm
during epididymal maturation. Biol Reprod. 85:503–510. 2011.
View Article : Google Scholar : PubMed/NCBI
|
|
19
|
Park K, Jeon S, Song YJ and Yi LS:
Proteomic analysis of boar spermatozoa and quantity changes of
superoxide dismutase 1, glutathione peroxidase, and peroxiredoxin 5
during epididymal maturation. Anim Reprod Sci. 135:53–61. 2012.
View Article : Google Scholar : PubMed/NCBI
|
|
20
|
Dacheux JL, Belleannée C, Guyonnet B,
Labas V, Teixeira-Gomes AP, Ecroyd H, Druart X, Gatti JL and
Dacheux F: The contribution of proteomics to understanding
epididymal maturation of mammalian spermatozoa. Syst Biol Reprod
Med. 58:197–210. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
21
|
Gbormittah FO, Bones J, Hincapie M, Tousi
F, Hancock WS and Iliopoulos O: Clusterin glycopeptide variant
characterization reveals significant site-specific glycan changes
in the plasma of clear cell renal cell carcinoma. J Proteome Res.
14:2425–2436. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
22
|
Labas V, Spina L, Belleannee C,
Teixeira-Gomes AP, Gargaros A, Dacheux F and Dacheux JL: Analysis
of epididymal sperm maturation by MALDI profiling and top-down mass
spectrometry. J Proteomics. 113:226–243. 2015. View Article : Google Scholar
|
|
23
|
Baker MA, Weinberg A, Hetherington L,
Villaverde AI and Velkov T: Analysis of protein thiol changes
occurring during rat sperm epididymal maturation. Biol Reprod.
92:112015. View Article : Google Scholar
|